Information Links
Related Conferences
Previous Issues Volume 7, Issue 1 - 2024
The Efficacy and Safety of Vitamin D Supplementation on the Incidence Rate of Pre-Eclampsia: A Systematic Review
Rehab Owayn Alanazi1,*, Fiona McCullough2
1Northern Border University, Arar, Saudi Arabia
2Division of Food, Nutrition and Dietetics, School of Biosciences, University of Nottingham, Nottingham, United Kingdom
*Corresponding author: Rehab Owayn Alanazi, Northern Border University, Arar, Saudi Arabia, Phone: 966531500347, E-mail: [email protected].
Received Date: February 11, 2024
Published Date: March 04, 2024
Citation: Alanazi RO, et al. (2024). The Efficacy and Safety of Vitamin D Supplementation on the Incidence Rate of Pre-Eclampsia: A Systematic Review. Mathews J Nutr Diet. 7(1):33.
Copyrights: Alanazi RO, et al. © (2024).
ABSTRACT
Background: The aetiology of pre-eclampsia (PE) remains poorly understood. An increase in inflammatory mediators produced by the placenta may be a potential causative mechanism. However, studies regarding the efficacy of Vitamin D (Vit D) supplementation in reducing PE risk are inconclusive. Objectives: Evaluation of the effect of Vit D supplementation on the incidence of PE, and determination of the optimal supplementation strategy. Methodology: Four databases were searched, according to the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) of papers (2013 to 2023). Eligible studies were randomised controlled trials (RCTs) that examined the effects of Vit D supplementation on the incidence of PE and maternal and infant outcomes referring to birth weight, preterm delivery, Caesarean (C-) section and 25(OH)-D levels. Results: In total, six RCTs with a total sample size of 762 pregnant women were included. Prenatal Vit D supplementation was associated with a non-significant decrease in PE risk, birth weight (BW), preterm delivery, C-section and 25(OH)-D levels (46.11±7421 nmol/l vs 80±51.53 nmol/l. P=0.143). No major side effects were reported. Conclusion: Vit D supplementation during pregnancy is a potential safe intervention strategy for preventing pre-eclampsia. More RCTs are needed to determine the optimal strategy (population-specific dose recommendations and timing).
Keywords: Pre-eclampsia, Vitamin D, Pregnancy, Birth Weight, Preterm Delivery, Caesarean Section.
LIST OF ABBREVIATIONS
PE: Pre-eclampsia; Vit D: Vitamin D; C-section: Caesarean Section; PD: Pre-term Delivery; BW: Birth Weight; IG: Intervention Group; CG: Control Group; Vit D3: Cholecalciferol; RCT: Randomised Controlled Trial; 25OHD: 25-Hydroxyvitamin D.
INTRODUCTION
In recent years, vitamin D has attracted increasing scientific attention (Yang et al., 2019). Vit D is a fat-soluble seco-sterol vitamin synthesised in the skin through exposure to sunlight or acquired through dietary sources such as cod, olive oil, salmon, and mushrooms [1].
Traditionally, Vit D is known to affect bone metabolism and mineral balance, as well as an anti-proliferative and immunomodulatory effects unrelated to its skeletal regulator activity, in addition to its possible impacts on Metabolic and Cardiovascular Outcomes [2,3].
Insufficiency and deficiency of maternal Vit D have been recognised as public health concerns for many years. Based on a systematic review conducted in 2016, a substantial number of pregnant women and newborns were found to suffer from Vit D deficiency across five global regions [4]. To date, there is no precise definition of Vit D deficiency during pregnancy as determined by 25-hydroxyvitamin D concentrations, with no universal Vit D prophylactic dosage [2].
Although Vit D concentrations in blood during pregnancy have been associated with pregnancy complicated, such as preterm birth and Pre-Eclampsia [5], a limited attention has been focused on the role of Vit D in pregnancy complications.
Pre-eclampsia
Pre-eclampsia (PE) is a disorder characterised by pregnancy-induced high blood pressure (blood pressure equal to or less than 140 mmHg systolic or 90 mmHg diastolic) in two separate instances [6].
Globally, PE is diagnosed in an estimated 4 million women, resulting in the deaths of more than 70,000 women and 500,000 infants [7].
There are numerous risk factors related to PE [8]. The aetiology of PE remains poorly understood [9]. Mechanisms involve multiple processes, including immunological processes [10]. There is a strong correlation between PE and genetic variations associated with inflammation, thrombophilia, oxidative stress, and the renin- angiotensin system [11].
The management of PE is currently focused on targeting different aspects of PE pathogenesis [12]. However, according to the National Health Service (NHS), the only definitive PE treatment is placenta delivery, which may induce preterm labour and cause several newborn complications [13].
Most risk factors for PE are uncontrollable; however dietary factors have been found to protect against PE [14] suggesting that diet may be a factor that mothers can influence.
A systematic review investigated the association between Vit D potential role of in the causation and prevention of PE. They reported a relationship between Vit D level and PE; however, the included studies did not demonstrate a statistically significant independent association between Vit D and the risk of PE [15].
Another systematic review and meta-analysis by Fogacci et al. [16] examined Vit D plus probiotic supplementation on the risk of PE, both concluding that Vit D supplementation was useful in preventing PE occurrence, but also indicating the importance of further reviews as well as studies to examine the impact of Vit D specifically.
Since no previous review was identified that examined the effectiveness of Vit D (without being combined with multiple nutrients), this paper has assessed the efficacy and safety of Vit D supplementation on PE incidence, investigated the ideal timing to introduce the supplements and its effect on maternal and infant outcomes (including birth weight, term, preterm delivery and C-section).
METHODOLOGY
The review followed the Reporting Elements for Systematic Reviews and Meta- Analyses (PRISMA 2015) guidelines [17].
Two reviewers independently conducted a comprehensive and systematic search of the scientific literature through electronic medical databases Web of Science and Embase (Ovid), to find relevant studies regarding the efficacy and safety of Vit D supplementations in reducing the PE incidence rate from June 2013 until July 2023. The PICO framework was used to perform the search strategy, as illustrated in Table 1, by employing the following key terms:
- "Pre-Eclampsia"[Mesh] [1] OR "Preeclampsia"
- "Vitamin D"[Mesh]) OR "Cholecalciferol"[Mesh]) OR "1,25-dihydroxyvitamin D" [Supplementary Concept])
- "Pregnant Women"[Mesh] OR "Pregnancy"[Mesh]
- "Premature Birth"[Mesh] OR "Infant, Premature"[Mesh]) AND "Infant, Low Birth Weight"[Mesh] AND "Safety"[Mesh])
- "Randomized Controlled Trial" [Publication Type]) OR "Controlled Clinical Trial" [Publication Type] OR "Clinical Trials as Topic"[Mesh]
- #1 and #2 OR #2 and #3
- 7- #5 and #6
Table 1. PICOS framework for article selection
|
Participants |
Pregnant Women |
|
Intervention |
Vitamin D supplementation |
|
Comparison |
Control group |
|
Outcomes |
Primary: reduce the incidence rate of preeclampsia and determine the ideal timing strategy |
|
Secondary: infant health outcomes including birth weight, preterm birth and delivery, and safety of the supplements |
|
|
Study design |
Randomised Control Trials (RCTs). |
|
Open-label RCTs |
Selection criteria
Relevant articles published in the last ten years between 2013 and 2023 were deemed eligible for this review. The search was restricted to full-access English- language articles. Table 2 presents the inclusion and exclusion criteria along with their rationale.
Table 2. The inclusion and exclusion criteria with rationale
|
Inclusion criteria |
Exclusion Criteria |
Rationale |
|
|
Publication Date |
2013 - 2023 |
<2013 |
To ensure an up-to-date review |
|
Language |
English |
Non-English language |
To prevent incorrect data interpretation |
|
Journal Publication |
Articles with full access Peer-reviewed journals |
Articles with restricted access |
To guarantee sufficient analysis and interpretation of the data |
|
Location |
Universal |
- |
Worldwide, preeclampsia is the most common cause of maternal and neonatal mortality [18] |
|
Study Design |
RCTs (blinded or not blinded) Open-label RCTs with control group |
Not RCTs such as observational studies or open-label trials that lacked a control group |
RCTs are the gold standard tool to assess the efficacy of a dietary intervention |
|
Intervention |
Vit D supplements in comparison to a control group |
Vit D combination with other dietary interventions |
To evaluate the effect of Vit D exclusively |
|
Target Population |
Pregnant women |
- |
To explore Vit D effectiveness in reducing the occurrence of PE and how it would affect other maternal and neonatal health outcomes |
|
Primary Outcomes |
Reduce the incidence rate of PE |
Not assessing the main outcome |
To accomplish the purpose of the current review |
In this review the available data didn’t allow meta-analysis to be conducted, however, an independent t-test was performed using SPSS software (version 29). Categorical data was analysed by frequencies and percentages. A P-value < 0.05 was defined as statistically significant.
Quality assessment and risk of bias
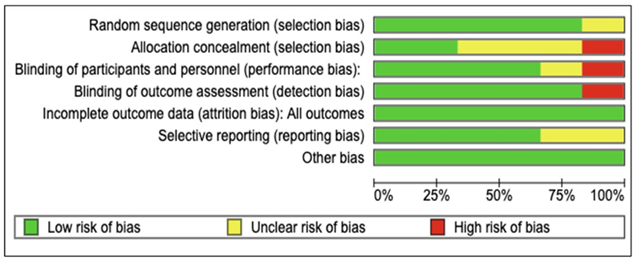
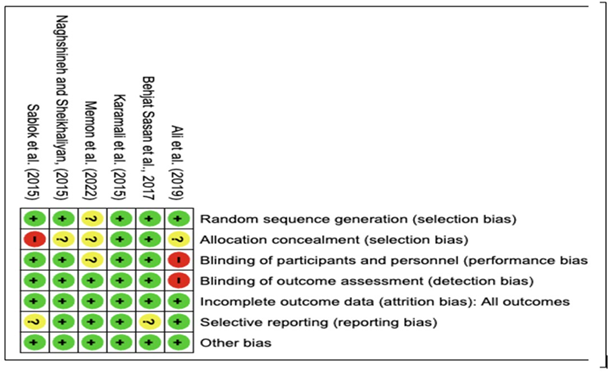
The assessment of the methodological quality of the selected RCTs was conducted according to the risk of bias criteria outlined in the Cochrane Collaboration's tool for evaluating the risk of bias in randomised trials [19]. Figure 2 shows the summary of risk-of-bias of the included studies. Due to the limited number of the available papers, no exclusion was made based on the quality assessment process.
RESULTS
The diagram in Figure 1 depicts the systematic process of excluding irrelevant studies, leading to the inclusion of only six Randomised Controlled Trials (RCTs) in the review.
Figure 1. PRISMA literature flow chart
Risk of bias in the selected studies
Figure 2A. The risk of bias for the selected studies.
Figure 2B. The risk of bias summary of the include studies.
Figure 2. Cochrane quality assessment of the included studies.
The characteristics of the included studies are summarized in Table 3. In total, 762 pregnant women with a mean age of 26.73 years were included in this review, of which, 54.3% were included in the IG and 45.66% were in the CG. All the included studies were performed in various locations at a single center and were published between 2015 and 2022.
Characteristics of the interventions
The intervention duration ranged from 12 to 23 weeks, and in Ali et al. [20] data was reported for another 12 weeks considered as a follow-up period. Two trials [21,22] began supplementing between the 20th and 32nd weeks of gestation, after 24 weeks of pregnancy [23], in the third trimester [24], first trimester [25], and early second trimester [20]. The supplements in all studies were cholecalciferol delivered in different doses and methods. The dose of 50,000 IU once every 2 weeks was used in two studies [21,25] 25 000 IU once every 2 weeks in Memon et al. [23], and 600 IU per day in Naghshineh and Sheikhaliyan [24], Ali et al. [20], used 4000 IU once a day, whereas in Sablok et al. [22], the dose ranged between 60,000 IU and 120,000 IU based on the mother’s Vit D level.
Three studies used a placebo as a comparator [21,24,25], two compared Vit D with a CG who received no supplements [22,23] and Ali et al. [20] compared it with a group who received multivitamins.
Outcomes of the selected studies
All six RCTs assessed the impact of Vit D on the incidence rate of PE by measuring the change in blood pressure and proteinuria before and after the intervention. A statistically significant difference in the incidence rate was reported in three studies [20,23,25] (p= 0.049; p=0.019; p=0.035, respectively). Three studies indicated a non-significant change p >0.05; [21,22,24]. Figure 3 shows the mean difference between the intervention and control groups (8.47± 8.070 vs 15.59±9.522, P=0.193).
Figure 3. The mean PE rate in the control and intervention groups. Data are presented as percentages.
Five studies assessed the change in maternal and infant health outcomes after the intervention. Four studies evaluated the mean change in birthweight, percentage of preterm labour and C-section rate [20-24] and one study only evaluated the percentage of women who delivered by C-section [25] .
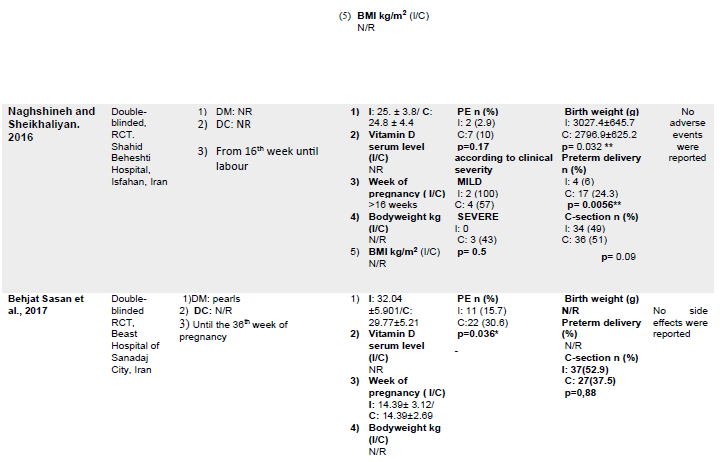
Furthermore, Naghshineh and Sheikhaliyan [24] reported significant differences between the IG and CG in birth weight (p = 0.04) and preterm delivery (p = 0.0056).
Sablolk et al. [22] reported that 8% of pregnant women in the IG had preterm labour compared with 21% of women in the CG (p = 0.032), with a mean change in birthweight of 2600±410 g from baseline (p = 0.032). However, no statistically significant differences were observed among the remaining studies [20,21,25].
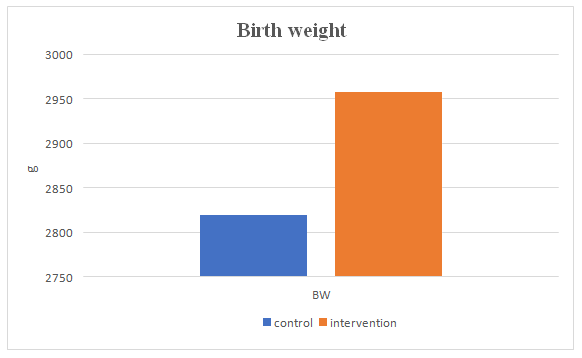
Figures 4-B, 4-C and 4-D demonstrate the mean change in IG compared to CG in birth weight, preterm delivery and C-section as follows: body weight (2957.75±296.679 vs 2819.47±313.248 g, P= 0.545); preterm delivery (7.07 % vs 15.92%, P=0.165); C-section (37.55 vs 42.80%, P=0.464).
Figure 4-B: The mean birth weight in the control and intervention groups. Data presented as mean grams.
Figure 4. The mean change in C: preterm delivery (P=0.545) and D: C-Section (P=0.156) in the control and intervention groups. Data is presented as percentages.
Change in 25(OH)-D levels
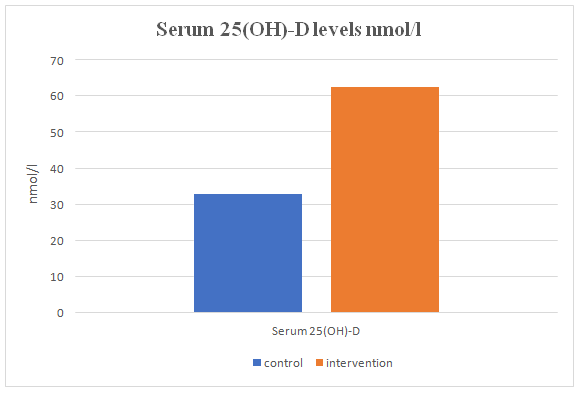
Three studies evaluated the change in blood 25(OH)-D levels after and before the intervention. Karamali et al. [21] and Ali et al. [20] reported a significant increase in 25(OH)-D serum (p<0.001; p<0.0001, respectively), whereas Sablolk et al. [22] found that 25(OH)-D levels were lower in the CG with a mean change of 46.11±7421 nmol/l vs 80±51.53 nmol/l in the Vit D group. Figure 4-E indicates that there is a difference between the means of the CG and the IG for serum 25(OH)-D levels, however, it did not reach statistical significance.
Figure 4-E. The mean serum 25(OH)-D levels in the control and intervention groups. Data is presented as means.
Safety of supplements
Two studies assessed the safety of the supplements [20,25], observing no adverse effects related to Vit D. Furthermore, among all the included studies, no trial reported the occurrence of hypervitaminosis D.
Table 3. The characteristics and the main outcomes of the included studies
DISCUSSION AND CONCLUSION
This systematic review investigated the efficacy of Vit D supplementation in reducing or preventing PE and how the timing of introducing supplements influences the findings, in addition to its impacts on both maternal and infant health outcomes. The six RCTs included, with a total sample of 444 pregnant women, indicated the potential beneficial effect of using Vit D supplementation as a preventive strategy to reduce the risk of PE and maternal and infant outcomes, especially when introduced in the early stages of pregnancy.
Regarding the aim to establish any reduction or prevention of PE, as it is a major contributor to adverse health outcomes for both mothers and neonates, three studies showed a significant reduction in the incidence of PE compared to the CG [20,23,25], whereas the remaining three studies reported no significant change in the incidence rate [21,22,24].
The findings of this review highlighted that Vit D supplementation reduced the PE incidence compared to the CG. Nonetheless, it is important to note that the observed difference was not statistically significant for combined data from both groups of all studies (intervention 8.47± 8.070 vs. control 15.59±9.522, P=0.193). These findings are consistent with those in a Cochrane review that found a single or ongoing dose of Vit D has a possible advantage in reducing the risk of PE [26].
In the context of determining an optimal dosing schedule, such as identifying the appropriate trimester to initiate treatment, it seems possible that these results are affected due to the time of initiation of the supplements. When supplements were offered late between the 20th and 32nd weeks [21,22], and in the third trimester [24], there was no evidence supporting the efficacy of VIt D supplementation in mitigating the risk of PE, although various dosages and methods were used. In contrast, when the supplements were administered in the first and early second trimesters [20,25] a significant reduction in the incidence rate was observed. This is in line with the study by Manasova et al. [27], which showed that introducing additional Vit D in the first trimester led to a significant reduction in PE rates. However, more than 90% of the population of Ali et al. [20] was of the same nationality, so the findings of the study may not be generalisable. Interestingly, Memon et al. [23] found a significant decrease in PE risk although they started supplementation after 24 weeks. This inconsistency in the results may be attributable to the heterogeneity of those studies, which differ in terms of ethnicity, environment, geography (northern or southern), baseline Vit D levels, and dosages (starting from 4000 UI to even 120 000 UI).
However, the RCT conducted by Xiaomang and Yanling [28] reported that a 4000 IU dose of Vit D administrated in the first trimester significantly reduced the PE risk among 54 at-risk women. This result was validated in the meta-analysis by Fogacci et al. [16] which reported that supplementation before 20 weeks of pregnancy, regardless of dosage and duration, was of considerably greater benefit for pregnant women.
There is no explanation regarding the precise mechanism by which Vit D exerts a positive effect on PE. It is assumed that the higher incidence of PE in pregnant women who did not receive Vit D supplementation is attributed to specific physiological and pathological responses associated with Vit D. A recent review by Poniedziałek-Czajkowska and Mierzyski [29] accords with our earlier observations, which demonstrated the possible use of Vit D in treating the underlying pathogenic factors associated with PE development. They concluded that in the presence of Vit D deficiency, an imbalance between pro- and anti-angiogenic factors during placenta formation can result in PE development. On the other side, sufficient Vit D consumption plays a role in both immune regulation and vascular function. This includes blood pressure regulation, through Vit D’s effects on endothelial cells and smooth muscle cells to reduce risk. Vit D deficiency may result in vascular smooth muscle cell proliferation, endothelial cell dysfunction, and heightened inflammation [30], via multiple and shared pathways mediated by intracrine and/or paracrine mechanisms [31].
Five studies reported changes in maternal and infant health outcomes; four assessed the change in birth weight, preterm delivery, and C-section, and one recorded the rate of C-section. Overall, Vit D supplementation showed a trend towards a favourable change in those health outcomes, although the difference did not reach statistical significance.
Our findings are consistent with another review which found no significant link between Vit D supplementation and birth weight, preterm and C-section delivery [5]. In contrast, considering the birth weight, a study restricting fortified dairy consumption, a proxy of Vit D intake, resulted in lower birth weight in educated, non- smoking, and healthy mothers [32].
Moreover, a meta-analysis of 24 RCTs reported that prenatal Vit D supplementation substantially increased the BW of newborns (MD, 75.38 g; 95% CI, 22.88 to 127.88 g) [33]. Furthermore, a meta-analysis conducted by Roth et al. [34] indicated that Vit D administration significantly increased the average BW. However, it is important to note that the results were not consistently supported in sensitivity and subgroup analyses, indicating some limitations in their findings.
In terms of preterm and C-section delivery, the observed decline in preterm birth rates among those receiving Vit D supplementation in the present review correlates with a quantifiable decline in the need for caesarean sections. These results suggest that promoting favourable pregnancy outcomes may reduce the need for medical interventions such as caesarean deliveries caused by foetal distress which are mutually beneficial for the mother [35].
An RCT involving 54 Vit D deficient women administered two doses of cholecalciferol, 4000 IU until the 16th week of pregnancy, then 2000 IU until the end of pregnancy. Preterm birth was twofold more prevalent (36% vs. 17.2%; p=0.01) and C-section was 1.7 times more prevalent (48% vs. 27.6%; p=0.05) in the group that did not receive additional Vit D [27]. A possible explanation for this is that the Vit D receptor in the skeletal muscle and insufficient Vit D levels are associated with muscular weakness, as well as inadequate muscle function and strength [36].
The present review indicated the importance of sufficiently increasing Vit D levels to avoid negative perinatal outcomes. There is a concern that women who experience preterm delivery may have lower concentrations of 25(OH)D due to a reduced duration of gestation, which limits the opportunity for Vit D supplementation to enhance their Vit D status [37]. In an RCT by Hollis et al. [38] they found that those who took a daily dose of 400 IU of Vit D, as well as those who took higher doses, reached a state of balance within two months of beginning supplementation.
Furthermore, Wagner et al. [38] observed that maternal Vit D status near the delivery date was strongly correlated with preterm birth. They indicated that despite a baseline deficiency, Vit D supplementation can affect the risk of preterm delivery.
This may be explained by the fact that Vit D influences well-established physiological pathways in the pathogenesis of preterm birth, such as immunomodulation, inflammation, and transcription of genes implicated in placental function [37].
Vit D supplementation exhibited a difference between the means of the CG and the IG on 25(OH)D levels (32±14.492 vs. 62.40 62.40±24.119), however, the observed difference was not statistically significant (P=0143). All the included studies used cholecalciferol (D3) which may have a substantial impact on the findings. As supported in a study comparing Vit D3 and D2 supplements for improving serum 25(OH)D levels, total 25(OH)D increased more significantly with D3 supplementation [40]. Maintaining adequate 25(OH)D levels in the circulation is the key to optimal dietary Vit D status. Since hepatic Vit D hydroxylation might be a saturated process, baseline 25(OH)D concentrations may affect Vit D response [41]. In addition, Vit D increases 25(OH)D concentrations less in individuals with higher BMIs. As observed by Sablock et al. [22], the deposition of Vit D in adipose tissue decreases its bioavailability [42].
However, the precise amount of Vit D or its metabolites required to achieve optimal blood levels remains uncertain. It has been suggested that a 4000 IU Vit D supplement increases circulating 25(OH)D levels to a level that completely normalises Vit D metabolism in pregnant women regardless of race [43]. This is in agreement with the findings of Ali et al. [20], who showed that a dose of 4000 IU significantly increased 25(OH)D from 16.3 ± 5 nmol/mL to 72.3 ± 30.9 nmol/mL (p > 0.0001). Nevertheless, only 50% of the group was able to attain the normal level with a dosage of 4000 IU, while 6.5% of the other half remained deficient.
Furthermore, in Karamali et al. [21], a dose of 50,000 IU significantly increased 25(OH)-D levels in Vit D deficient pregnant women. Similarly, a study reported that a dosage of 50,000 IU/week of Vit D3 significantly increased 25(OH)-D levels compared to CG 25(OH)-D levels (33.7 ng/mL vs. 14.6 ng/mL, P < 0.050) [44]. In Sablok et al. [22], only 52% of women with baseline levels <25 nmol/l achieved levels >50 nmol/l at delivery with a dose of 480,000 IU. Moreover, Hollis and Wagnar [45], indicated that daily Vit D supplementation as opposed to administering a bolus, has greater overall effectiveness and benefits. They attributed this to the short Vit D half-life in circulation and indicated that weekly or longer administration of Vit D results in substantial variations in circulating Vit D levels sustaining steady levels of 25(OH)D.
Despite the use of varied dosages, supplements were generally well tolerated, and no adverse events were reported. Similarly, a study involving pregnant women who received a dose of 50,000 IU every two weeks experienced no adverse effects [46]. Also, Mir et al. [47] found that a daily dose of 2,000 IU or a monthly dose of 60,000 IU is safe and effective for expectant women. These findings indicate the safety of Vit D even in considerable doses; however, caution is required for avoiding undesirable impacts of supplements such as confusion, vomiting, arrhythmia, and nephrocalcinosis.
Although this is the first systematic review to evaluate data from RCTs examining the efficacy of Vit D alone in reducing the incidence rate of PE among pregnant women, some limitations must be acknowledged. First, based on the limited number of studies included in the review, it can be inferred that ethnicity might potentially exert an influence on the generalizability of the findings. Second, only three studies considered maternal baseline 25-OH-D and most did not measure the preconception 25-OH level and dietary intake including calcium intake, which may confound the association between Vit D deficiency and PE risk in mothers. Finally, only one study involved a follow-up which limited our knowledge of the long-term effects of the supplements.
This systematic review provides numerous recommendations for future research and practice. First, determining pregnant women's Vit D needs and recommended daily dose considering Vit D status at baseline, different body types, and lifestyles in addition to the optimal timing for starting Vit D supplementation. Secondly, investigating the minimum clinically effective dosage and the safety of different dosages. Additionally, the most effective supplementation regimen (daily weekly, or bolus dose) and investigating the efficacy of Vit D supplementation alone or in combination with other nutrients. Finally, more studies are needed to investigate the functional role of Vit D to substantiate recommendations and public health policies.
In conclusion, based on the high to moderate quality of the available evidence, the results revealed that Vit D supplementation is generally safe and has potential benefits in reducing PE incidence and the optimal time to commence supplementation is early in pregnancy, between the first and second trimesters.
Nevertheless, the impact of the supplementation was not significant on birth weight, preterm delivery, C-section and 25(OH)-D levels. However, it was not possible to recommend the optimal dosages due to the variable dose ranges employed. Finally, well-designed, prospective, randomised trials of supplementation with large sample sizes that control for nutritional exposure are necessary.
AUTHORS CONTRIBUTIONS
Rehab and Fiona. The authors confirm responsibility for the following: study conception and design, data collection, analysis and interpretation of results, and manuscript preparation. All authors reviewed the results and approved the final version of the manuscript.
CONFLICT OF INTEREST
The authors declare that there are no conflicts of interest related to this study.
REFERENCES
- Hossein-nezhad A, Holick MF. (2013 ). Vitamin D for health: a global perspective. Mayo Clin Proc. 88(7):720-755.
- Kaushal M, Magon N. (2013). Vitamin D in pregnancy: A metabolic outlook. Indian J Endocrinol Metab. 17(1):76-82.
- Delrue C, Speeckaert R, Delanghe JR, Speeckaert MM. (2023). Vitamin D Deficiency: An Underestimated Factor in Sepsis? Int J Mol Sci. 24(3):2924.
- Gao F, Guo F, Zhang Y, Yuan Y, Chen D, Bai G. (2021). Vitamin A, D, and E Levels and Reference Ranges for Pregnant Women: A Cross-Sectional Study 2017-2019. Front Nutr. 8:628902.
- Palacios C, Kostiuk LK, Peña-Rosas JP. (2019). Vitamin D supplementation for women during pregnancy. Cochrane Database Syst Rev. 7(7):CD008873.
- Macedo TCC, Montagna E, Trevisan CM, Zaia V, de Oliveira R, Barbosa CP, et al. (2020). Prevalence of preeclampsia and eclampsia in adolescent pregnancy: A systematic review and meta-analysis of 291,247 adolescents worldwide since 1969. Eur J Obstet Gynecol Reprod Biol. 248:177-186.
- Dimitriadis E, Rolnik DL, Zhou W, Estrada-Gutierrez G, Koga K, Francisco RPV,et al. (2023). Pre-Eclampsia. Nat Rev Dis Primers. 9(1):8.
- Ngene NC, Moodley J. (2018). Role of angiogenic factors in the pathogenesis and management of pre-eclampsia. Int J Gynaecol Obstet. 141(1):5-13.
- Chaiworapongsa T, Chaemsaithong P, Yeo L, Romero R. (2014). Pre-eclampsia part 1: current understanding of its pathophysiology. Nat Rev Nephrol. 10(8):466-480.
- Akbari S, Khodadadi B, Ahmadi SAY, Abbaszadeh S, Shahsavar F. (2018). Association of vitamin D level and vitamin D deficiency with risk of preeclampsia: A systematic review and updated meta-analysis. Taiwan J Obstet Gynecol. 57(2):241-247.
- Williams PJ, Broughton Pipkin F. (2011). The genetics of pre-eclampsia and other hypertensive disorders of pregnancy. Best Pract Res Clin Obstet Gynaecol. 25(4):405-417.
- Duley L, Meher S, Jones L. (2013). Drugs for treatment of very high blood pressure during pregnancy. Cochrane Database Syst Rev. 2013(7):CD001449.
- Meazaw MW, Chojenta C, Muluneh MD, Loxton D. (2020). Systematic and meta-analysis of factors associated with preeclampsia and eclampsia in sub-Saharan Africa. PLoS One. 15(8):e0237600.
- Purswani JM, Gala P, Dwarkanath P, Larkin HM, Kurpad A, Mehta S. (2017). The role of vitamin D in pre-eclampsia: a systematic review. BMC Pregnancy Childbirth. 17(1):231.
- Fogacci S, Fogacci F, Banach M, Michos ED, Hernandez AV, Lip GYH, et al. (2020). Vitamin D supplementation and incident preeclampsia: A systematic review and meta-analysis of randomized clinical trials. Clin Nutr. 39(6):1742-1752.
- Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. (2015). Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst Rev. 4(1):1.
- Yang Y, Le Ray I, Zhu J, Zhang J, Hua J, Reilly M. (2021). Preeclampsia Prevalence, Risk Factors, and Pregnancy Outcomes in Sweden and China. JAMA Netw Open. 4(5):e218401.
- Higgins JP, Altman DG, Gøtzsche PC, Jüni P, Moher D, Oxman AD, et al. (2011). The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ. 343:d5928.
- Ali AM, Alobaid A, Malhis TN, Khattab AF. (2019). Effect of vitamin D3 supplementation in pregnancy on risk of pre-eclampsia - Randomized controlled trial. Clin Nutr. 38(2):557-563.
- Karamali M, Beihaghi E, Mohammadi AA, Asemi Z. (2015). Effects of High-Dose Vitamin D Supplementation on Metabolic Status and Pregnancy Outcomes in Pregnant Women at Risk for Pre-Eclampsia. Horm Metab Res. 47(12):867-872.
- Sablok A, Batra A, Thariani K, Batra A, Bharti R, Aggarwal AR, et al. (2015). Supplementation of vitamin D in pregnancy and its correlation with feto-maternal outcome. Clin Endocrinol (Oxf). 83(4):536-541.
- Memon HA, Rafique M, Khalid S, Perveen R, Imtiaz R, Naqvi N. (2022). Role of vitamin Dforthepreventionofpre-eclampsiainpregnantwomen:A randomizedcontrolledtrial. Pakistan Journal of Medical and Health Sciences. 16(1):1086-1088.
- Naghshineh E, Sheikhaliyan S. (2016). Effect of vitamin D supplementation in the reduce risk of preeclampsia in nulliparous women. Adv Biomed Res. 5:7.
- Behjat Sasan S, Zandvakili F, Soufizadeh N, Baybordi E. (2017). The Effects of Vitamin D Supplement on Prevention of Recurrence of Preeclampsia in Pregnant Women with a History of Preeclampsia. Obstet Gynecol Int. 2017:8249264.
- De-Regil LM, Palacios C, Lombardo LK, Peña-Rosas JP. (2016). Vitamin D supplementation for women during pregnancy. Cochrane Database Syst Rev. (1):CD008873.
- Manasova GS, Kuzmin NV, Didenkul NV, Derishov SV, Badiuk NS. (2021). Supplementation of Vitamin D in Pregnant Women with 25 (OH) DDeficiency andRiskofPreeclampsiaDevelopmentImproves PerinatalOutcomes. Pharmacology OnLine3:1107-1116.
- Xiaomang J, Yanling W. (2020). Effect of vitamin D3 supplementation during pregnancy on high risk factors - a randomized controlled trial. J Perinat Med. 49(4):480-484.
- Poniedziałek-Czajkowska E, Mierzyński R. (2021). Could Vitamin D Be Effective in Prevention of Preeclampsia? Nutrients. 13(11):3854.
- de la Guía-Galipienso F, Martínez-Ferran M, Vallecillo N, Lavie CJ, Sanchis-Gomar F, Pareja-Galeano H. (2021). Vitamin D and cardiovascular health. Clin Nutr. 40(5):2946-2957.
- Washington K, Ghosh S, Reeves IV. (2018). A review: Molecular concepts and common pathways involving vitamin D in the pathophysiology of preeclampsia. Open J Obstet Gynecol. 8(3):198-229.
- Mannion CA, Gray-Donald K, Koski KG. (2006). Association of low intake of milk and vitamin D during pregnancy with decreased birth weight. CMAJ. 174(9):1273-1277.
- Bi WG, Nuyt AM, Weiler H, Leduc L, Santamaria C, Wei SQ. (2018). Association Between Vitamin D Supplementation During Pregnancy and Offspring Growth, Morbidity, and Mortality: A Systematic Review and Meta-analysis. JAMA Pediatr. 172(7):635-645.
- Roth DE, Leung M, Mesfin E, Qamar H, Watterworth J, Papp E. (2017). Vitamin D supplementation during pregnancy: state of the evidence from a systematic review of randomised trials. BMJ. 359:j5237.
- Amiri M, Rostami M, Sheidaei A, Fallahzadeh A, Ramezani Tehrani F. (2023). Mode of delivery and maternal vitamin D deficiency: an optimized intelligent Bayesian network algorithm analysis of a stratified randomized controlled field trial. Sci Rep. 13(1):8682.
- Ceglia L, Harris SS. (2013). Vitamin D and its role in skeletal muscle. Calcif Tissue Int. 92(2):151-162.
- Bodnar LM, Klebanoff MA, Gernand AD, Platt RW, Parks WT, Catov JM, et al. (2014). Maternal vitamin D status and spontaneous preterm birth by placental histology in the US Collaborative Perinatal Project. Am J Epidemiol. 179(2):168-176.
- Hollis BW, Johnson D, Hulsey TC, Ebeling M, Wagner CL. (2011). Vitamin D supplementation during pregnancy: double-blind, randomized clinical trial of safety and effectiveness. J Bone Miner Res. 26(10):2341-2357.
- Wagner CL, Baggerly C, McDonnell SL, Baggerly L, Hamilton SA, Winkler J, et al. (2015). Post-hoc comparison of vitamin D status at three timepoints during pregnancy demonstrates lower risk of preterm birth with higher vitamin D closer to delivery. J Steroid Biochem Mol Biol. 148:256-260.
- Shieh A, Chun RF, Ma C, Witzel S, Meyer B, Rafison B, et al. (2016). Effects of High-Dose Vitamin D2 Versus D3 on Total and Free 25-Hydroxyvitamin D and Markers of Calcium Balance. J Clin Endocrinol Metab. 101(8):3070-3078.
- Ramasamy I. (2020). Vitamin D Metabolism and Guidelines for Vitamin D Supplementation. Clin Biochem Rev. 41(3):103-126.
- Wortsman J, Matsuoka LY, Chen TC, Lu Z, Holick MF. (2000). Decreased bioavailability of vitamin D in obesity. Am J Clin Nutr. 72(3):690-693.
- Hollis BW, Wagner CL. (2017). New insights into the vitamin D requirements during pregnancy. Bone Res. 5:17030.
- Etemadifar M, Janghorbani M. (2015). Efficacy of high-dose vitamin D3 supplementation in vitamin D deficient pregnant women with multiple sclerosis: Preliminary findings of a randomized-controlled trial. Iran J Neurol. 14(2):67-73.
- Hollis BW, Wagner CL. (2013). Clinical review: The role of the parent compound vitamin D with respect to metabolism and function: Why clinical dose intervals can affect clinical outcomes. J Clin Endocrinol Metab. 98(12):4619-4628.
- Soheilykhah S, Mojibian M, Moghadam MJ, Shojaoddiny-Ardekani A. (2013). The effect of different doses of vitamin D supplementation on insulin resistance during pregnancy. Gynecol Endocrinol. 29(4):396-399.
- Mir SA, Masoodi SR, Shafi S, Hameed I, Dar MA, Bashir MI, et al. (2016). Efficacy and safety of Vitamin D supplementation during pregnancy: A randomized trial of two different levels of dosing on maternal and neonatal Vitamin D outcome. Indian J Endocrinol Metab. 20(3):337-342.
[1]Medical Subject Headings (MeSH) is a standardized and regulated vocabulary dictionary employed to index papers within database
.png)
.png)
.png)