Previous Issues Volume 1, Issue 2 - 2016
Human Hair as a Long Term Biological Indicator for Assessing Strontium Nutritional Status
Berislav Momcilovic1, Juraj Prejac2, Andrey Anatolyevic Skalny3, Ninoslav Mimica4
1Institute for Research and Development of the Sustainable Ecosystems (IRES), Srebrnjak 59, 10000 Zagreb, Croatia.
2University Hospital Centre Zagreb, Department of Oncology, Kispaticeva 12, HR-10000 Zagreb, Croatia.
3ANO Center for Biotic Medicine, Zemlyanoi val 46-48, 103604 Moscow, Russia.
4University Psychiatric Hospital Vrapce, School of Medicine, University of Zagreb Bolnicka cesta 32, HR-10090 Zagreb, Croatia.
Corresponding Author:Berislav Momcilovic, Institute for Research and Development of the Sustainable Ecosystems, (IRES) Srebrnjak 59, 10000 Zagreb, Croatia,Tel: +385 1 2430288; E-Mail:[email protected]
Received Date: 07 Jul 2016 Accepted Date: 19 Aug 2016 Published Date: 29 Aug 2016
Copyright © 2016 Momcilovic B
Citation:Momcilovic B, Prejac J, Skalny AA and Mimica N. (2016). Human Hair as a Long Term Biological Indicator for Assessing Strontium Nutritional Status. Mathews J Nutr Diet. 1(2): 007.
ABSTRACT
Strontium (Sr) is a bone seeking trace element that closely parallels calcium metabolism. At the present, there is no available long-term biological indicator for assessing the human Sr nutritional status. In this article, we have presented a novel concept on how to assess the human strontium nutritional status by studying its frequency distribution in the hair and whole blood. In this prospective, observational, cross-sectional, and exploratory epidemiological study, we analyzed hair Sr (SrH) in 311 apparently healthy adult Croatians (123 men, 188 women); among them, whole blood strontium (SrWB) was detected in 142 women and 89 men. Hair and WB samples were analyzed for strontium by the inductively coupled mass plasma spectrometry (ICP MS). The natural frequency distribution of hair strontium obeyed the power law so that the data were analyzed by fitting the logistic bioassay sigmoid curve (ogive) of the median derivatives. Women have accumulated more SrH than men (median men 0.867 µg-g-1 vs. median women 3.120 µg-g-1). The normal physiological response range covered by the linear part of the sigmoid curve was 0.418 - 2.950 µg-g-1 for men and 0.846 - 7.260 µg-g-1 for women. Values below that linear range were considered to denote low Sr nutritional status, whereas values above that range would indicate excessive dietary strontium exposure. The long term biological indicator of hair strontium (SrH) is incommensurable with that of the short term biological indicator of the whole blood strontium (SrWB). On the average, women have about three times more strontium in their hair than men, regardless of the age. Analysis of the hair strontium frequency distribution is a valuable long term biological indicator in assessing the population strontium nutritional status.
KEYWORDS
Strontium Nutritional Status; Hair; Long Term Biological Indicator; Women; Men; Age.
INTRODUCTION
Strontium is an osteotrophic (bone-seeking) trace element that is normally present in our diet and our bodies [1]. The pioneers of trace element analysis considered strontium to be an essential trace element for humans , but the conclusive evidence on strontium essentiality for humans has yet to be provided [2, 3]. Strontium was in the limelight in the 50's and 60's of the last century when it was discovered that the metabolism of strontium was closely related to the metabolism of calcium in bone tissue [4]. Indeed, 99% of typical body burden of 320 mg Sr is found in the bone [5]. The atmospheric fallout of fission products after nuclear testing included a long-living bone seeker of 90Sr which has a physical half-life of ~ 27 y and a biological T/2 ~ 10 y [6]. Radioactive 90Sr is a powerful beta radiation source and, when lodged in the bones, would act as a strong radio-toxic carcinogen [7]. Moreover, strontium, like calcium, could be transferred from mother to fetus/newborn via the mother's milk and get deposed in their mineralized tissue with possibly deleterious consequences [8, 9]. Later on, the interest in strontium was renewed when Skoryna demonstrated the pharmacological effect of both the stable and 89Sr radiopharmaceutical isotope Metasteron® to alleviate the pain of mammary and prostate carcinoma bone metastases [10, 11]. Today, the most recent research has shown that strontium ranelate [Protelos®, Osseor®] acts as an effective nutritive agent, capable of slowing down mineral bone loss due to old age. Indeed, these strontium compounds are preventing and delaying the onset and progression of osteoporosis, including both femur and vertebral osteoporosis [12-18]. How much such a treatment can affect the strontium body burden and the metabolism of other trace elements in the skeleton remains to be elucidated. Since strontium is not as yet recognized to be an essential trace element, there are no recommended dietary allowances (RDA's) for strontium, and there is also no available long term biological indicator tissue for assessing the strontium nutritional status of the body; the US Environmental Protection Agency has derived an oral recommended dose for strontium of 0.6 mg per kg body weight per day [19]. Today, hair trace element analysis is accepted as a long-term biological indicator for assessing trace element nutritional status and the level of their environmental exposure [20-23]. The aim of this study was to demonstrate how strontium hair analysis (SrH) may serve as a long term biological indicator tissue for assessing the strontium nutritional status of the human body by analyzing hair strontium frequency distribution properties with a median derivative method. Thus far, we have shown that our innovative approach yielded a reliable assessment of the nutritional status of the essential trace elements iodine and selenium, and in evaluating the exposure to the non-essential elements of silver and aluminium [24-27]. The observed data on hair strontium analysis were compared with that for whole blood strontium (SrWB), i.e., a common short term biological indicator tissue for the assessment of numerous bioelements and other different clinical parameters.
SUBJECTS AND METHODS
This prospective, observational, cross-sectional, and exploratory epidemiological study was approved by the Ethical Committee of the Institute for Research and Development of the Sustainable Eco Systems (IRES), Zagreb, Croatia. The study was conducted in strict adherence to the Declaration of Helsinki on Human Subject Research and to the complementary Croatian national bylaws and regulations [28]. Every subject gave his/her written consent to participate in the study and filled out a short questionnaire on his/her health status and medical history (data not shown) [29]. Data on hair shampooing were also recorded to control for the possible external strontium contamination, a highly unlikely event in general Croatian population. Hair strontium (SrH) was analyzed in a random sample of 311 apparently healthy adults (123 Men, 188 Women), the median age of women and men was 47 and 50 years, respectively. Our population consisted of subjects from the general Croatian population who were concerned about their health status; the majority of them were living in Zagreb, the capital region of Croatia. All the subjects were fed their usual home prepared, mixed mid-European diet and reported no adverse medical conditions. None of our subjects was occupationally or otherwise exposed to some strontium source. Hair strontium and whole blood strontium was analyzed with the ICP MS (Elan 9000, Perkin Elmer, USA) at the Center for Biotic Medicine (CBM). Moscow, Russia, The CBM is an ISO Europe certified commercial laboratory for analysing bioelements (macro elements, trace elemnts, and ultratrace elements) in different biological matrices [30].
HAIR STRONTIUM (SRH) ANALYSIS
In brief, hair analysis was performed following the International Atomic Energy Agency recommendations and other validated analytical methods and procedures [31, 32]. A strand of hair 5-7 cm long and weighting less than one gram would be cut with titanium-coated scissors over the anatomically welldefined bone bump at the back of the head (lat. protubarantia occipitalis externa). The individual hair samples were further minced into strands less than 1 cm long prior to chemical analysis, stirred 10 min in an ethylether/acetone (3:1, w/w), rinsed three times with redistilled H2O, dried at 85°C for one hour to constant weight, immersed one hour in 5% EDTA, rinsed again in redistilled H2O, dried at 85°C for twelve hours, wet digested in HNO3/H2O2 in a plastic tube, sonicated and microwaved. We used certified GBW0910b Human Hair Reference Material (Shanghai Institute for Nuclear Research, Academia Sinica, Shanghai 201849, China.
WHOLE BLOOD STRONTIUM (SRH) ANALYSIS
Whole blood was drawn by vein puncture at the cubital vein. and collected into the green-cup Vacuette collecting tubes (#454082 LotA13030M7m Greineer Bio On International AG, Kremsmunster, Austria), which were randomly assigned for the ICP-MS analysis. Whole blood samples of 0.5 ml were digested in a microwave oven with 0.1 ml of HNO3 at 175°C. Blood standards were liophylised Seronorm TM Trace Elements Whole Blood Reference Standards Level 1 (OK 0036), Level 2 (MR 9067), and Level 3 (Ok 0337) for strontium in the whole blood (SERO AS, Bilingstadt, Norway). Five ml of redistilled H2O were added to every reference standard and stirred gently at a room temperature for two hours to equilibrate. One ml of such equilibrated standard was pipetted in 25 ml quartz glass vials, dried at 105°C for 24 hours. The microwaved samples were dissolved in 5 ml of redistilled H2O with 0.1 ml of H2O2 added. The detection limits for Sr in the hair and whole blood were 0.0009 and 0.00009 µg-g-1, respectively; strontium was detected in all the 311hair samples and in all the 212 whole blood samples. All chemicals were proanalysis grade (Khimmed Sintez, Moscow, Russia). Current CBM admissible hair and whole blood strontium ranges (µg-g-1) are set 0.00 - 6.0 for both men and women, respectively. Values above that range are considered to indicate excessive strontium intake. Strontium belongs to the plead of 143 elements sharing the same mass number (number of isotopes/element): 3 As, 9 Se, 12 Br, 14 Kr, 20 Rb, 18 Sr, 21 Y, 12 Zr, 14 Nb, 8 Mo, 7 Tc, 5 Ru, e.g. there are three arsenic isotopes sharing the same mass number with some 18 strontium isotopes, etc. [33].
MEDIAN DERIVATIVES
To scrutinize the hair strontium concentration frequency distribution, we used the median derivative model to fit the sigmoid logistic regression analysis function (power function) for men and women separately (Appendix 1) [25, 34]: A2 + (A1 - A2)/[1+(x/x0)p] Where A1 is the initial value (lower horizontal asymptote, A2 is final value (upper horizontal asymptote), x0 is center (point of inflection, in our case it is the median M0 detected, p is power (the parameter that affects the slope of the area about the inflection point) (Appendix 1). The Qtiplot Data Analysis and Scientific Visualisation programs were used for this analysis (www.softproindependent.com/qtiplot.html). The samples below the detection limits were not excluded from the analysis but were treated as having zero strontium concentration.
RESULTS
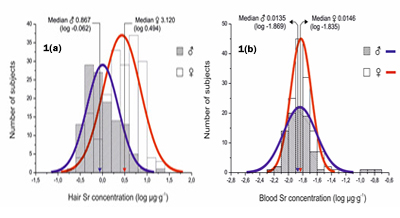
Strontium was detected in all 311 of analyzed hair samples (188 women and 123 men) whereas SrWB was detected in all 231 analyzed samples (142 women and 89 men). After the data were log transformed, the previous skewed and kurtous strontium data distribution was changed into standard Gaussian frequency distribution curve for both the hair (Figure 1a.) and whole blood (Figure 1b). Apparently, women accumulate and retain three time more strontium in their hair than men. The respective SrH medians were for ? = 0.867 and for ? = 3.120 µg-g-1. There were no statistically significant differences between men and women in SrWB.
Figure 1a: Normal hair strontium frequency distribution in 123 men (blue) and 188 women (red) (log µg-g-1). Figure 1b: Normal whole blood strontium frequency distribution in 89 men (blue) and 142 women (red) (log µg-g-1).
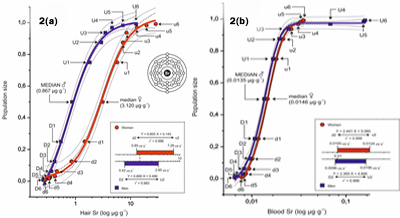
Median derivatives (Appendix 2) were used to fit the power function sigmoid curve and they are shown in Appendix 2 (Figure 2a Hair, Figure 2b Whole Blood). The data on the upward and downward arm of the median derivatives are shown separately for men in blue and women in red. Following the principles of bioassay analysis, we have defined the hair linear segments (downward and upward): ?d2-u2 and ?D1-U1 as strontium physiologically normal range [30]. Normal hair Sr concentrations of Croatian women range from 0.85-7.26 µg-g-1 (median 3.12 µg-g-1), and that for Croatian men range from 0.42-2.95 µg-g-1 (median 0.867 µg-g-1). The respective low linear region of the sigmoid power function curve, i.e., the segments d2-d6 for women and D1-D6 for men, was defined as a low Sr nutritional status range.
Figure 2a: The power function sigmoid curve of hair strontium median derivatives. The difference between the SrH median derivatives of men n = 123 (blue) and women n = 188 (red). D, U men downward (D) and upward (U) median derivatives; d, u women downward (d) and upward (u) median derivatives. Logistic function Y = A2 + (A1-A2)/(1 + (X/X0)p), - - - 0.95 confidence limit, ••• 0.95 prediction limit. Men: Y = 1.010 + (-0.207 - 1.010)/(1 + (X/0.754)1.644), r2 = 0.997; Women: Y = 1.033 + (0.010 - 1.033)/(1 + (X/3.045)1.773), r2 = 0.996. Box: Strontium linear saturation range for ? and ? (log conc.). See Appendix 1 for the model and Appendix 2 for median derivatives input values. Figure 2b: The power function sigmoid curve of whole blood strontium median derivatives. The difference between the SrWB median derivatives of men n=89 (blue) and women n=142 (red). D, U men downward (D) and upward (U) median derivatives; d, u women downward (d) and upward (u) median derivatives. -- Logistic function Y = A2 + (A1-A2)/(1 + (X/X0)p), - - - 0.95 confidence limit, ••• 0.95 prediction limit. Men: Y = 0.976 + (-0.008 - 0.976)/(1 + (X/0.013)5.514), r2 = 0.999; Women: Y = 1.000 + (-0.053 - 1.000)/(1 + (X/0.014)4.742), r2 = 0.999. Box: Strontium linear saturation range for ? and ? (log conc). See Appendix 1 for the model and Appendix 2 for median derivatives input values.
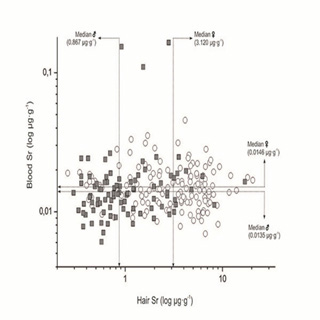
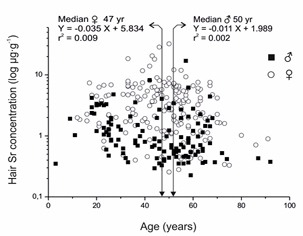
Similarly, the respective upper linear region of the sigmoid power function curve above the respective segments u3 for women (range u3-u6) and segment U2 for men (range U2-U6), was defined as an excessively high SrH region. Evidently, women retain more strontium than men. There was no correlation between the SrH and SrWB (Figure 3) and there was no effect of age upon the observed gender difference of SrH (Figure 4). Indeed, all the data shown in the Figure's 2a, 3, and 4 demonstrated the higher accumulation of strontium in the hair of women than that of men.
Figure 3: Incommensurability of the hair strontium (long term biological indicator) and whole blood strontium (short term biological indicator). ? Women ? men
Figure 4: Age does not affect the gender dependent difference in strontium distribution in men and women.
DISCUSSION
Our results on hair and whole blood strontium in the linear (adequate, physiological) data range are in good agreement with those obtained by Goulle et al. [35, 36] and Wolowiec et al. [23]. They are considerably lower than that reported by Caroli et al. [20] who, however, have referred on arithmetic means of the highly skewed data distribution (N.B.: The data would be comparable if we did the analysis in such a way). This study has demonstrated that the strontium nutritional status may be assessed by hair strontium analysis and that hair strontium deposition is distinctly different between men and women, i.e., women tend to accumulate more strontium in their hair than men. It is pertinent to note here that the major osteotrophic elements, i.e., calcium and magnesium, followed the same gender dependent power Law pattern of hair deposition [37]. All the three elements, i.e., calcium, magnesium, and strontium belong to the same 2a column of the Periodic system of elements [38, 39], and they all were substantially higher in the hair of women than that of men. Presumably, the sex dependent hormonal status difference affects strontium hair follicule deposition, their metabolic turnover, throughout and bio mineralization [40]. The sex dependent preference of Ca, Mg, and Sr deposition observed by these researchers, combined with the identical sporadic observations by other researchers for the respective hair Ca, Mg, and Sr, strongly supports Sr status as an essential bioelement, or at the least, a conditionally essential bioelement where essentiality will depend upon the given inter-relational circumstances[41- 43]. It should be noted that, in difference to the dietary intake data, hair strontium, as well as other trace elements, reflect the individual (personalized) absorption. Indeed, it is well known that different persons may absorb different amounts of trace elements from the same dietary source due to their individual biological variability [20]. We should be aware that one cm of human hair is a reliable metabolic time log that reflects strontium over a period of an entire month [44, 45]. To assess the nutritional status of strontium, by comparing the short term biological indicator of SrWB with the long term biological indicator of SrH, revealed that these two bioindicators are incommensurabile. Indeed, there is an irrevocable difference in the time base between the short term (SrWB) and long term (SrH) bioindicators; the absorbed strontium will be equilibrated between the blood and various body compartments within a period of days, whereas strontium would be uni directionally and irreversibly deposited in the hair [46]. If, for a moment, we put aside a fundamental difference between the equilibration process of SrWB with the surrounding tissues and that of the irreversible unidirectional growth of SrH, a simple calculation would show that one cm of hair is time-equivalent of a thirty days of cumulative urine collection, i.e., 30 liters of urine. N.B.: Urine is another short term biological indicator like the SrWB, which is thus far preferentially used in trace element nutritional studies. What we are saying in this study is not how much strontium needs to be in the diet, but how much strontium should be found in the hair to claim that strontium nutritional status is adequate for a certain individual subject. Indeed, by using SrH median derivatives frequency distribution analysis, we can, for the first time, accurately assess the individual strontium nutritional status and even differentiate the degree of how much is adequate (linear/physiological range of the ogive). Since hair is an easily available biological material that can be collected in a non-invasive way and that hair is easily transported and stored preceding the analysis in some well-equipped analytical laboratory, nutritionists and clinicians have a powerful new tool for assessing strontium and other trace element status in health and disease. More studies like this, by different researchers and for different elements, are needed before the hair multi-element profile analysis could find its right place in personalized medicine and in public health nutrition in particular. Strontium was the secret ingredient of the Roman gladiators' potion used to promote bone fracture healing [47]. N.B.: Our short vignette about secret gladiators' potion for promoting bone healing is intriguing since strontium was discovered in 1787, some 15-17 centuries AFTER it was knowingly used by the Roman gladiators [48]. Ancient knowledge lost and rediscovered?
ACKNOWLEDGEMENTS
We would like to thank Prof. David F Marshall, Professor Emeritus of English, for his help with English language and Ms. Alison Ranisate, librarian for her help with the literature references, both from UND, GF, ND, USA.
image 1
We studied the frequency distribution of hair strontium (HSr) median and its derivatives to assess the strontium nutritional status. First we assess the median (M0) hair strontium concentration of our subject population. By definition, one half of the studied population was above the median (upward median branch, U0), and the other half was below the median (downward median branch, D0). Hence, the population size (PS) for M0 is the sum of the respective upward and downward median branches around the central inflection "hinge" M0, i.e., PS = U0 + D0 = 0.5 + 0.5 = 1.0. Both the respective upward and downward median branches can be further divided in the same "median of median" way into a series of sequential median derivatives (U0,1,2,3 …n-1, n and D0,1,2,3,..n-1, n). For every median derivative of the population, the actual hair strontium concentration can be identified. Thus, instead of mechanically throwing the preconceived percentile grid upon the observed data, we inferred the median derivative grid out from the data set itself [49, 50].
Appendix 2: Median derivatives to fit the power function sigmoid (see Appendix). Hair Median Derivative Concentrations (MDC).
HAIR STRONTIUM
| Men (n = 123) | Women (n = 188) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Median (M0) = 0.867 µg-g-1 Sr | Median (M0) = 3.120 µg-g-1 Sr | ||||||||||
| MDC | n | Sr | MDC | n | Sr | MDC | n | Sr | MDC | n | Sr |
| D1 | 62 | 0.544 | U1 | 62 | 1.820 | d1 | 94 | 1.467 | u1 | 94 | 5.710 |
| D2 | 31 | 0.418 | U2 | 31 | 2.950 | d2 | 47 | 0.846 | u2 | 47 | 7.260 |
| D3 | 16 | 0.363 | U3 | 16 | 3.489 | d3 | 24 | 0.459 | u3 | 24 | 8.457 |
| D4 | 8 | 0.351 | U4 | 8 | 4.611 | d4 | 12 | 0.425 | u4 | 12 | 13.030 |
| D5 | 4 | 0.312 | U5 | 4 | 7.233 | d5 | 6 | 0.335 | u5 | 6 | 19.515 |
| D6 | 2 | 0.260 | U6 | 2 | 12.621 | d6 | 3 | 0.281 | u6 | 3 | 28.610 |
WHOLE BLOOD STRONTIUM
| Men (n = 89) | Women (n = 142) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Median (M0) = 0.0146 µg-g-1 Sr | Median (M0) = 0.0135 µg-g-1 Sr | ||||||||||
| MDC | n | Sr | MDC | n | Sr | MDC | n | Sr | MDC | n | Sr |
| D1 | 45 | 0.0107 | U1 | 45 | 0.0168 | d1 | 71 | 0.0117 | u1 | 71 | 0.0182 |
| D2 | 23 | 0.0096 | U2 | 23 | 0.0164 | d2 | 36 | 0.0101 | u2 | 36 | 0.0214 |
| D3 | 12 | 0.0083 | U3 | 12 | 0.0245 | d3 | 18 | 0.0093 | u3 | 18 | 0.0261 |
| D4 | 6 | 0.0080 | U4 | 6 | 0.0682 | d4 | 9 | 0.0086 | u4 | 9 | 0.0311 |
| D5 | 3 | 0.0071 | U5 | 3 | 0.1540 | d5 | 5 | 0.0081 | u5 | 5 | 0.0332 |
| D6 | 2 | 0.0066 | U6 | 2 | 0.1595 | d6 | 3 | 0.0079 | u6 | 3 | 0.0353 |
Capital letters (D1 - D6, U1 - U6) men, small letters (d1 - d6, u1 - u6) women.
REFERENCES
- Boivin G and Cournot G. (2001). Manifestations osseuses et articulaires des intoxications metalliques et metalloidiques. Encyclopedie Medico-Chirurgicale. 14-025-R-10, 8.
- Schroeder HA, Tipton IH and Nason AP. (1972). Trace metals in men: strontium and barium. J Chronic Dis. 25(9), 491-517.
- Seifert M. Strontium. In: Merian E, Anke M, Ihnat M, et al. Elements and their Compounds in the Environment. 2nd Edition. Wiley-VCH, Weinheim. 619-626
- Comar CL, Wasserman RH, Ullberg S and Andrews GA. (1957). Strontium metabolism and strontium-calcium discrimination in man. Proc Soc Exp Biol Med. 95(2), 386-391.
- Beliles RP. (1994). The metals, in Patty's Indudtrial Hygiene and Toxicology (Clayton GD and Clayton FE, eds.), John Eiley & Sons, New York, NY, USA. 1879-2352.
- The Merck Index. 13th ed., Merck & Co. Inc., Whitehause Station, NJ, USA
- Engstrom A, Bjornerstedt B, Clemedson CJ, Nelson A, et al. (1958). Bone and racdiostrontium. John Eiley & Sons, New York, NY, USA. 10(3), 357-358
- Comar CL and Wasserman RH. (1964). Strontium. In Mineral Metabolism. An advanced treatise , Vol.2, Part A, (Comar XL and Bfronnr F, eds.), Acadedmic Press, New York and London, 523-571.
- Momcilovic B, Durakovic A and Kostial K. (1971). Infulence of lactation on 85Sr and 47Ca mobilization from the skeleton. Arh Hig Rada Toksikol. 22, 235-243.
- Skoryna SC. (1981). Effects of oral supplementation with stable strontium. Can Med Assoc J. 125(7), 703-712.
- Amersham. (1989). Metastron in prostatic cancer. Aylesbury Buckinghamshire, UK: Amersham Intl.
- Boonen S, Body J-J, Boutsen Y, Devogelaer J-P, et al. (2005). Evidence-based guidelines for the treatment of postmenopausal osteoporosis: a consensus document of the Belgian Bone Club. Osteoporosis Int. 16(3), 239-254
- Reginster JY, Deroisy R, Dougados M, Jupsin I, et al. (2002). Prevention of early postmenopausal bone loss by strontium ranelate: the randomized, two-year, double-masked, doseranging, placebo-controlled PREVOS trial. Osteoporosis Int.13(12), 925-931.
- Marie PJ. (2003). Optimizing bone metabolism in osteoporosis: insight into pharmacoogic profile of strontium ranelate.Osteoporosis Int 14(suppl3), s9-s12.
- Stevenson M, Davis S, Lloyd-Jones M, Beverley C, et al. (2007). The clinical effectiveness and cost- effectiveness of strontium ranelate for the prevention of osteoporotic fragility fractures in postmenopausal women. Health Technol Assess. 11(4), 1-134.v
- Reginster JY, Deroisy R and Jupsin I. (2003). Strontium ranelate: a new paradigm in the treatment of osteoporosis. Drugs Today (Barc). 39(2), 89-101.
- Reginster JY, Seeman E, De Vernejoul MC, Adami S, et al. (2005). Strontium ranelate reduces the risk of nonvertebral fractures in postmenopausal women with osteoporosis: treatment of peripheral osteoporosis (TROPOS) study. J Clin Endocrinol Metab. 90(5), 2816-2822.
- Blake GM and Fogelman I. (2006). Strontium ranelate: a novel treatment for postmenopausal osteoporosis: a review of safety and efficacy. Clin Interv Aging. 1(4), 367-375.
- US Environmental Protection Agency. Integrated risk information system (IRIS).
- Carolli S, Almonti A, Coni E, Petrucci E, et al. (1994). The assessment of reference values for elements in human biological tissues and fluids. A systematic review. Critical Reviews in Analytical Chemistry. 24, 363-398.
- Druyan ME, Bass D, Puchyr R, Urek K, et al. (1998). Determination of reference ranges for elements in human scalp hair. Biol Trace Elem Res. 62(3), 185-197.
- Esteban M and Castano A. (2009). Non-invasive matrices in human biomonitoring: a review. Environ Intl. 35(2), 438-449.
- Wolowiec P, Michalak I, Chojnacka K, Miukulewicz M, et al. (2013). Hair analysis in health assessment. Clin Chim Acta. 419, 139-171.
- Momcilovic B, Prejac J, Visnjevic V, Skalnaya MG, et al. (2014). Hair iodine for human iodine status assessment. Thyroid. 24(6), 1018-1026.
- Momcilovic B, Prejac J, Visnjevic V, Brundic S, et al. (2016). High hair selenium mother to fetus transfer after the Brazil nuts consumption. J Trace Elem Med Biol. 33, 110-113.
- Momcilovic B, Prejac J, Visnjevic V, Mimica N, et al. (2012). Environmental human silver exposure. Toxicol Environ Chem. 94, 1238-1246.
- Momcilovic B, Prejac J, Skalny AA, Serebryansky EP, et al. (2016). Assessing human aluminum environmental exposureby analyzing frequency distribution properties of hair and whole blood aluminum concentrations. Matthews J Nutrition Dietetics. 1(1), 1-11.
- Browne M. Charter of rights is adopted in the UN. The New York Times.
- Oppenheim AN. (2004). Questionnaire - design, interviewing, and attitude measurement. London: Continuum.
- Momcilovic B, Prejac J, Brundic S, Skalny AV, et al. (2010). An essay on human and elements, multielement profiles, and depression. Transl Neurosci. 1(4), 322-334.
- IAEA. (1980). Elemental Analysis of biological materisals. IAEA-TEC. DOC 197. Vienna: International Atomic Energy Agency.
- Burges C. (2000). Valid analytical methods and procedures. Cambridge: The Royal Society of Chemistry.
- Momcilovic B, Prejac J, Momcilovic R, Ivicic N, et al. (2008). On the same element isotope mass number (Pleiad) and the clusters of elements bearing the same mass numbers in the Periodic system - the "chesshyja" (fish skin) model. Trace Elem Med (Moscow). 9(3-4), 5-20.
- Momcilovic B, Prejac J, Visnjevic V, Drmic S, et al. (2012). The muscle immobility of depression - The weightlessness within. Psychology. 3, 825-33.
- Harpley FW, Stewart GA and Young PA. (1973). Principles of biological assay. In: Delanuois (section ed,) International Encyclopedia of Pharmacology and Therapeutics, Oxford: Pergamon Press.
- Goulle J-P, Mahieu L, Castermant J, Neveu N, et al. (2005). Meatal and metalloid multi-elementary ICP-MS validation in wwhole blood, plasma, urine and hair. Reference values. Forensic Science International. 153(1), 39-44.
- Nacher JC and Akutsu T. (2007). Recent progress in the analysis of power law features in complex cellular networks. Cell Biochem Biophys. 49(1), 37-47.
- Momcilovic B, Prejac J, Visnjevic V, Skalnaya MG. Drmic S, et al. (2013). Gender dependent differences in hair calcium, potassium, magnesium, sodium, phosphorus, and zinc. 5th International Symposium of the Federation of European Societies on Trace Elements and Minerals (FESTEM 5). Avignon, France, May 22-24, Poster 1-22.
- Momcilovic B. (2007). Novel visualization of the periodic system of elements. The orthogonal dimension. Trace Elem Med (Moscow). 8(4), 1-6.
- Mann S. (2001). Biomineralization. Principles and concepts in bioinorganic materials chemistry'. Oxford and New York: Oxford University Press.
- Eatough DJ, Chrfistensen JJ, Izatt RM, Haartley C, et al. (1974). Level of selected trace elements in human hair. The First Human Hair Symposium (Brown AC ed.) Medcom Press, New York, NY 377.
- Ashurbekov TR. (1974). Eleemental composition of human hair in relation to sex, preganancy, and presence of climacteric. Sud Med Ekspert. 17, 23
- Othman L and Spyron NM. (1978). Elemental concentration in hair and nail from a selected population group in the Machakos district of Kenya. IAEA-SM-227/33 Proceedings of the Symposium on Nuclear Activation Technques in Life Sciences. IAEA, Vieanna. 53.
- Rook A and Dawber R. (1982). Diseases of hair and scalp. Oxford: Blackwell Sci Publ. UK.
- Robins and Clarence R. (2012). Chemical and physical behavior of human hair, 5th ed. Berlin: Springer.
- Momcilovic B, Prejac J, Visnjevic V, Skalnaya MG, et al. ( 2012). Incomensurability of human hair and whole blood iodine. Trace Elem Med (Moscow). 13(1), 20-24.
- Losch S, Moghaddam N, Grossschmidt K, Risser DU, et al. (2014). Stable isotope and trace element studies on gladiators and contemporary Romans from Ephesus (Turkey, 2nd and 3rd Ct. AD) - implications for differences in diet. PLoS ONE. 9(10), e110489.
- Emsley J and Strontium. (2001). In Naatures building blocks, Oxford University Press, Oxford, UK. 405-408.
- Smylevich L and Dougherty ER. (2012). Probabilistic Boolean network. Philadephia: Society for industrial and Applied Mathematics.
- Momcilovic B, Prejac J, Visnjevic V, Mimica N, et al. (2013). Hair silver median derivatives for the assessment of the environemental silver exposure, overexposure aand toxicity. Trace Eleemednts in Medicine (Moscow). 13(3), 27-36.