Previous Issues Volume 1, Issue 2 - 2016
Changes in Plasma Amino Acid Levels After the Administration of Glucose or Sucrose to Healthy Young and Aged Males
Ogawa M1, Takao T1, Ishii Y1, Shimizu F1, Takada A2
1Faculty of Human Life and Environmental Sciences, Showa Women's University, Tokyo, Japan.
2International Projects on Food and Health (NPO), Tokyo, Japan.
Corresponding Author:Takada A, International Projects on Food and Health (NPO), Sumida-ku Ishiwara 1-30-6-802, Tokyo 130-0011, Japan, Tel: 81338291849; E-Mail:[email protected]
Received Date: 14 Sep 2016 Accepted Date: 06 Oct 2016 Published Date: 10 Oct 2016
Copyright © 2016 Takada A
Citation:Takada A, Ogawa M, Takao T, Ishii Y, et al. (2016). Changes in Plasma Amino Acid Levels After the Administration of Glucose or Sucrose to Healthy Young and Aged Males. Mathews J Nutr Diet. 1(2): 008.
ABSTRACT
Background: No research has been reported about changes in plasma levels of amino acids after the administration of glucose or sucrose to young and aged men. Objective: We want to know whether essential and non-essential amino acids are influenced by the administration of glucose or sucrose in the same way. Results: Plasma levels of amino acids were measured after the administration of 50 grams of glucose or sucrose to young (18-22 years old) and aged (=50 years old) male adults. Plasma levels of total amino acids decreased after the administration of glucose. Decrease in the total amino acid levels was significant in aged men after the administration of sucrose. A significant decrease in plasma levels of total non-essential amino acids was observed at 120 min after the administration of glucose but not sucrose in both aged and young men. Both glucose and sucrose administrations resulted in a significant decrease in the plasma levels of the total essential amino acid levels and branched amino acids in young and aged men. Conclusion: These results suggest that plasma amino acids, particularly essential amino acids, may be transported from the blood soon after the administration of sugar (glucose or sucrose) to the tissues, such as muscles, possibly due to an increase in the insulin levels.
KEYWORDS
Amino Acid; Essential Amino Acid; Non-Essential Amino Acids; Glucose; Sucrose.
INTRODUCTION
Recently, a high mortality risk of low carbohydrate diet was reported [1]. Dangers of low carbohydrate diet with respect to impaired muscle function were discussed in Japanese media, although no scientific papers were published in this issue. In fact, Robert Atkins who advocated low carbohydrate diet died by a serious head injury when he toppled down [2]. It is proposed that muscle loses power due to energy loss because of decreased uptake of glucose. There are some papers indicating that insulin regulates carbohydrate, lipid, protein, and amino acid metabolism [3]. Insulin inhibits proteolysis and associated release of amino acids and stimulates amino acid uptake and protein synthesis in skeletal muscle [4, 5]. High insulin levels were shown to stimulate skeletal muscle protein synthesis [6]. As to individual amino acids, the levels of alanine, phenylalanine, valine, leucine, isoleucine, and tyrosine were shown to increase and the levels of histidine and glutamine decreased in hyperglycemia [7]. There has been no research investigation concerning the effects of glucose or sucrose administration on plasma levels of various amino acids in young and aged male adults. Fernstrom, J.D. and Wurtman, R.J. indicated that when plasma tryptophan concentrations were raised by receiving tryptophan in foods or injection of insulin, the brain serotonin and tryptophan concentration increased [8, 9]. They indicated that carbohydrate ingestion increased insulin secretion which raised plasma tryptophan and lowered the concentration of the competing amino acids such as branched neutral amino acids in rats [9]. Carbohydrate ingestion was shown to decrease plasma free amino acid levels, and glucose intake resulted in a decrease in large neutral amino acids such as methionine, phenylalanine, tyrosine, and tryptophan [10, 11]. These results suggested that an increase in plasma levels of glucose or insulin may increase the transport of some amino acids using various transporters, thus decreasing the concentration of such amino acids.
METHODS
Male acquaintances older than 50 years old (n = 36, age; 62.4 ± 9.6) and male college students (n = 36, age; 20.8 ± 1.6) were requested to participate in the experiments. We carefully assessed their health, and allowed those who do not smoke, with no sign of health problems such as diabetes and hypertension, as well as no history of serious disease, to participate. We obtained informed consent from the participants prior to conducting the protocol, which had been approved by the Ethical Committee of Showa Women'sUniversity (15-02) and the non-profit organization (NPO) "International projects on food and health" (15-01). Participants were provided with a questionnaire relating to self-administered diets they may have followed in the past. From these completed questionnaires, we calculated their past intakes of energy, carbohydrate, fat, and protein. We use BDHQ ( brief-type self-administered diet history questionaire) and asked participants to answer about the history of foods uptakes since one month before.
MEASUREMENTS OF BLOOD VARIABLES
After fasting overnight, participants were randomly assigned to groups. Depending on their group, each participant received a 550-mL solution containing 50 g of glucose or sucrose (or 500 mL water as a control). Either 50 g of glucose or sucrose was added and dissolved in each bottle containing 500 mL of water. Participants were requested not to eat anything after 09:00 PM on the previous night and not eat anything for breakfast the following morning. Between 9:00 AM and 10:00 AM, blood was sampled using a syringe, and participants were given either glucose or sucrose solution or water as a control. We measured blood glucose using a finger stick (TERUMO kit) before and 120 min after the administration of glucose or sucrose. Furthermore, other plasma factors were measured after plasma was separated from blood. Ethylenediaminetetraacetic acid (EDTA) was used as an anticoagulant. Plasma was obtained by centrifuging the blood samples, and the amino acid and insulin levels were measured for backgrounds of these participants. The samples were analyzed by SRL, Inc. (Tokyo Japan) using the UF-Amino Station®, which is a liquid chromatography- mass spectrometry system with an automated pre-column derivatization for simultaneous determination of amino acids (Shimadzu Corporation, Kyoto Japan). The original concept of this system was developed by Ajinomoto Co., Inc. (Tokyo Japan) as an automated method of analyzing major free amino acids in human plasma in the field of clinical chemistry [12,13]. The human plasma samples were cryopreserved with EDTA- 2Na before the analysis. The thawed samples were deproteinized with acetonitrile followed by the amino acid analysis. Pre-column derivatization in the UF-Amino Station was automatically performed using an automated sample injector with the regent APDSTAG® (Wako Pure Chemical Industries, Ltd., Osaka Japan). Target free amino acids as derivatized compounds were separated under a reversed phase ultra-high performance liquid chromatography condition and determined by the liquid chromatograph mass spectrometer. Insulin was measured by the CLEIA (chemiluminescent immunoassay) method.
ETHICS
This work was approved by the ethical committees of Showa Women's University and the NPO "International projects on food and health" and was conducted in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments.
STATISTICS
The results are presented as means ± SEM. Statistical significance of the differences between groups was calculated according by one-way ANOVA. When ANOVA indicated a significant difference (P < 0.05), the mean values of the treatment were compared using Tukey's least significant difference test at P < 0.05.
RESULTS
Table 1: Basic parameters of participants.
| Subjects | Aged(n=44) | Young(n=36) | Significant difference |
|---|---|---|---|
| Age(years) | 62.4±9.6 | 20.8±1.6 | ** |
| Length(m) | 1.68±0.07 | 1.72±0.06 | * |
| Weight(kg) | 68.8±10.9 | 65.5±10.2 | |
| BMI | 24.3±3.2 | 22.2±3.3 | * |
| Enagy intake(kcal/day) | 2115.1±460.2 | 1988.8±591.8 | |
| Protein intake(g/day) | 66.6±28.8 | 69.3±25.1 | |
| Lipid intake(g/day) | 49.1±22.6 | 60.4±24.8 | * |
| Carbohydrate intake(g/day) | 198.6±89.4 | 271.5±91.3 | ** |
| blood glucose(mg/dl) | 91.7±16.3 | 78.9±13.1 | ** |
| insulin((μIU/ml) | 6.19±3.79 | 6.87±4.19 |
Protein, lipid, and carbohydrate uptakes were calculated from self-administered questionnaires. Tukey's test was used for statistical analysis *; p < 0.05,**; p < 0.01
Ethanol uptakes were not shown here. (Table 1) indicates the values of various variables of participants. We compared these variables with those reported by the Japanese Ministry of Welfare. It is evident that participants of the present experiments fell within the average ranges of height and weight. The uptake rates of energy and protein were similar between young and aged men; however, the young men consumed more lipids and carbohydrates whereas the aged men consumed more sugar. Although not shown here, aged men consumed more alcoholic drinks than did young men. For the reasons given above, aged men consumed more energy than young men. Blood glucose levels were higher in aged men. In the present research p value of less than 0.05 was considered statistically significant.
Table 2: Amino acids levels in plasma before and after the administration of glucose or sucrose with water as a control in aged men.
| Amino Acids | Aged(n=44) | |||
|---|---|---|---|---|
| 0 min. | 120 min. | |||
| (n=44) | control(n=13) | glucose(n=15) | sucrose(n=16) | |
| Histidine | 81.7±8.7+ | 86.2±9.0 | 74.2±6.9*## | 73.1±8.5**## |
| Lysine | 190.6±25.8 | 189.9±22.9 | 164.8±15.6**# | 164.8±20.7**# |
| Methionine | 27.6±4.6 | 25.5±3.0 | 17.6±4.2**## | 19.7±3.8**## |
| Phenylalanine | 63.0±8.5++ | 60.7±8.9 | 48.0±9.7**## | 47.6±7.6**## |
| Treonine | 131.8±28.4 | 126.6±25.7 | 97.7±23.2**# | 109.8±23.8* |
| Tryptophan | 59.1±8.9++ | 56.9±8.1 | 56.0±7.2 | 51.5±7.6* |
| Isoleucine | 66.6±12.5 | 65.5±13.2 | 37.9±8.2**## | 42.5±4.9**## |
| Leucine | 130.4±17.0 | 126.8±18.8 | 78.5±12.2**## | 89.4±11.0**## |
| Valine | 230.3±30.4 | 231.5±35.3 | 167.7±20.8**## | 183.6±19.4**## |
| Tyrosine | 58.1±9.3++ | 49.8±9.4* | 38.6±5.3**# | 41.1±6.0** |
| Alanine | 352.1±68.1+ | 327.9±52.2 | 309.6±48.3 | 370.2±51.6 |
| a-Aminobutyric acid | 20.7±5.4 | 18.9±6.6 | 18.4±3.1 | 16.7±4.6 |
| Arginine | 77.7±15.8+ | 64.5±11.9* | 51.1±10.1** | 51.6±11.3** |
| Asparagine | 44.7±6.4 | 37.5±10.5* | 30.9±8.3** | 34.4±7.6** |
| Aspartic acid | 3.29±1.31 | 5.48±2.16* | 6.03±2.92** | 4.35±2.43 |
| Citrulline | 22.2±3.6++ | 21.6±3.1 | 16.8±2.7**## | 15.5±3.7**## |
| Cystine | 14.1±4.0++ | 10.7±7.4 | 9.24±7.0 | 9.88±5.54 |
| Glutamic acid | 38.0±12.1++ | 130.1±119.2 | 156.3±124.7** | 113.0±97.4* |
| Glutamine | 542.6±64.7 | 399.9±182.1* | 308.9±165.7** | 386.9±137.3** |
| Glycine | 207.8±25.2 | 194.4±27.4 | 184.6±13.4* | 190.9±30.1 |
| Monoethanolamine | 8.39±1.16 | 7.90±1.59 | 7.11±0.87 | 7.38±0.93 |
| Ornithine | 59.2±13.5+ | 65.2±15.1 | 55.2±9.7 | 59.7±10.5 |
| Proline | 167.2±46.0 | 144.0±27.7 | 136.5±44.4 | 157.8±51.8 |
| Serine | 122.2±17.3++ | 112.7±8.7 | 95.1±11.7**# | 108.0±16.3* |
| Taurine | 50.2±9.5++ | 59.0±23.2 | 68.3±27.8* | 53.5±11.5 |
| Total AAs | 2727.8±209.8+ | 2508.0±197.7** | 2203.1±145.6**## | 2359.3±196.2** |
| EAAs | 964.2±95.0 | 883.3±64.8# | 731.9±74.9**## | 760.4±70.5**## |
| NEAAs | 1763.7±141.3++ | 1624.6±142.8* | 1471.2±104.0** | 1598.9±149.9** |
| EAAs/NEAAs | 0.548±0.048 | 0.546±0.033 | 0.499±0.054* | 0.477±0.044**## |
| BCAAs | 419.9±50.9 | 380.6±33.2 | 296.9±46.0**## | 304.3±36.5**## |
| BCAAs/AAAs | 3.69±0.39 | 3.87±0.45 | 3.76±0.52 | 3.65±0.42 |
| BCAAs/Total AAs | 0.154±0.013++ | 0.153±0.013 | 0.135±0.018**# | 0.129±0.016**## |
Blood samples were collected at 0 min and 120 min after the administration of solutions containing 50 g of glucose or sucrose in young men. The concentration is expressed in (μM. *; p < 0.05; **; p < 0/01 for 0 min vs. 120 min. Different marks such as a vs. b or c are significant; *; p < 0.05, **, p < 0.01, ?control group vs. ?glucose or ?sucrose administered group. a vs. ac or ab; not significant, b vs. ac; significant. Tukey's test was used for the calculation of significance among the 3 groups (water, glucose, and sucrose), and the t-test was used for the calculation of significance between 0 min and 120 min. AAs: amino acids, EAAs: essential amino acids, NEAAs: non-essential amino acids, BCAAs: branched chain amino acids, AAAs: aromatic amino acids.
Amino acids levels in plasma of 36 young men at 0 min and 120 min after the administration of glucose (12 men) or sucrose (13 men) were measured with 11 men of a control group taking water only. Similarly, a decrease in the EAAs and BCAAs levels was observed compared with the control group (11 men) after the administration of glucose or sucrose to young men. EAAs such as histidine, lysine, methionine, phenylalanine, threonine, tryptophan, leucine, isoleucine, and valine and BCAAs such as leucine, isoleucine, and valine decreased significantly after the administration of glucose or sucrose in young groups too. NEAA levels decreased after the administration of glucose in aged and young men.
Table 4: Changes in plasma levels of 3 branched-chain amino acids, total amino acids, total essential amino acids and total nonessential amino acids.
| Amino Acids | ?120-0min. | |||||
|---|---|---|---|---|---|---|
| Aged | Young | |||||
| Control (n=13) | Glucose (n=15) | Sucrose (n=16) | Control (n=11) | Glucose (n=12) | Sucrose (n=13) | |
| Isoleucine | -7.57± 6.4a | -23.6±6.6b | -23.8±4.8b | -9.0±8.8a | -27.2±14.7b | -28.3±8.5b |
| Leucine | -11.9 ± 12.5a | -45.3 ±9.0b | -40.6 ±11.0b | -13.9 ±13.1a | -47.2 ±28.1b | -45.7 ±14.4b |
| Valine | -12.9 ±13.9a | -52.3±21.4b | -45.0±12.9b | -21.2 ±16.1a | -59.2 ±46.9b | -50.6±16.2ab |
| Total AAs | -151.9 ±111.0a | -493.3 ±132.9b | -316.4 ±128.8c | -255.3 ±89.7a | -648.4 ±566.6b | -393.8 ±147.1ab |
| NEAAs | -103.0 ± 96.1a | -275.1± 87.5b | -130.0 ±108.5ac | -169.4 ±67.3a | -376.4 ±362.6b | -174.8±98.5a |
| EAAs | -48.9 41.0a | -218.2 54.2b | -186.3 42.8b | -85.9 45.1a | 272.1 207.3b | -219.0 57.6b |
| EAAS/NEAAs | 0.0±0.0a | -0.0 ±0.0b | -0.1±0.0b | 0.0±0.0a | -0.1±0.2b | 0.1 ±0.0ab |
| BCAAs | -32.2±26.6a | -121.2±31.7b | -109.4±24.4b | -44.1 ±35.3a | -133.6±87.9b | -124.5 ±35.6b |
| Amino Acids | 0.01±0.20 | -0.1 ±0.2 | 0.05±0.18 | 0.09±0.18 | -0.21±0.94 | 0.00±0.31 |
| BCAAs/Total AAs | -0.00±0.01a | -0.02 ±0.01b | -0.03±0.01b | -0.00±0.01a | -0.03±0.04b | -0.03±0.04b |
The concentration is expressed in μM Blood samples were collected at 0 min and 120 min after the administration of solutions containing 50 g of glucose or sucrose in aged men. The concentration is expressed in μM. Decrease in branched chain amino acids levels from 0 min were shown Different marks such as a vs. b or c are significant. P < 0.05; ? control group vs. ? glucose or ?sucrose administered group. a vs. ac; not significant, b vs. ac; significant. Tukey's test was used for the calculation of significance among 3 groups (water, glucose, and sucrose). AAs: amino acids, EAAs: essential amino acids, NEAAs: non-essential amino acids, BCAAs: branched chain amino acids.
When the extent of the decrease was compared between the control group and the glucoseor the sucrose-administered groups, the levels of EAAs decreased at 120 min after the administration of glucose or sucrose in both aged and young men. The levels of NEAAs significantly decreased only after glucose administration in young men. This decrease was not significant in aged men. BAAs, such as leucine, isoleucine, and valine, decreased significantly after the administration of glucose and sucrose in both aged and young men.
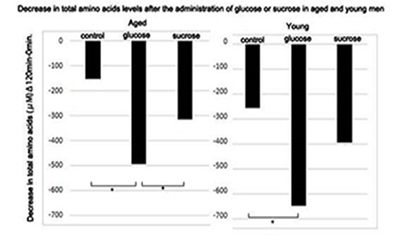
Figure 1: Decrease in total amino acids levels after the administration of glucose or sucrose in aged and young men.
Decrease in plasma levels of total amino acids at 120 min. after the administration of solutions containing 50 g. of glucose or sucrose in young and aged men were calculated. Statistical difference was calculated between control and glucose or sucrose administered groups by the Tukey's test. *; p < 0.05 Control; n = 13, glucose; n = 15, sucrose; n = 16 for aged men. Control; n = 11, glucose; n = 12, sucrose; n = 13 for young men. Changes in the levels of total amino acids at 120 min in aged and young men after the administration of glucose or sucrose compared to those levels in the control group. Glucose uptake resulted in a significant decrease in the amounts of total amino acids in young and aged people. Sucrose administration resulted in a significant decrease in the total amino acids only in aged men.
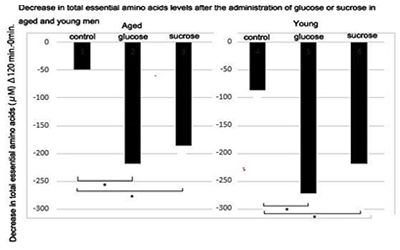
Figure 2: Decrease in total essential amino acids after the administration of glucose or sucrose in aged and young men.
Decrease in plasma levels of total essential amino acids at 120 min after the administration of solutions containing 50 g of glucose or sucrose in young and aged men were calculated. Statistical difference was calculated between the control and glucose or sucrose administered groups by the Tukey's test. *; p < 0.05 (Figure 2) shows changes in the decrease of total EAA levels at 120 min compared with the control group after glucose or sucrose administration in aged and young men. Both glucose and sucrose administration significantly decreased total EAAs in young and aged men at 120 min after the administration compared with the control group.
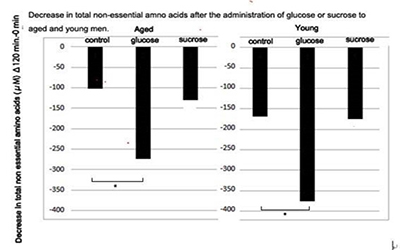
Figure 3: Decrease in total non-essential amino acids after the administration of glucose or sucrose to aged and young men.
Decrease in plasma levels of total non-essential amino acids at 120 min after the administration of solutions containing 50 g of glucose or sucrose in young and aged men were calculated. Statistical difference was calculated between control and glucose or sucrose administered groups by the Tukey's test. *; p < 0.05 (Figure 3) shows decrease in total NEAAs levels at 120 min compared with the control group after glucose or sucrose administration in aged men. Glucose uptake resulted in a significant decrease in the amounts of total NEAAs levels in aged men. There was practically no further decrease in the total NEAAs after the administration of sucrose compared with the control group in either aged or young men.
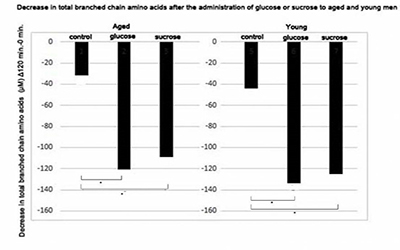
Figure 4: Plasma levels of total non-essential amino acids after the administration of glucose or sucrose to aged and young men.
Decrease in plasma levels of total branched amino acids at 120 min after the administration of solutions containing 50 g of glucose or sucrose in young and aged men were calculated. Statistical difference was calculated between control and glucose or sucrose administered groups by the Tukey's test. *; p < 0.05 (Figure 4) shows a decrease in the plasma levels of total BCAAs at 120 min after the administration of glucose or sucrose to aged and young men. Both glucose and sucrose administration significantly decreased total BCAA (leucine, isoleucine, and valine) levels in young and aged men at 120 min after the administration compared with the control group.
DISCUSSION
As mentioned earlier, the Wurtman's group has shown that the increased uptake of carbohydrates resulted in the transfer of tryptophan to the brain by the transporter by reducing the concentration of competing amino acids, which are transported to muscles [8, 9]. Their results suggested that the main determinant of brain tryptophan and serotonin concentrations is not only plasma tryptophan but also the ratio of this amino acid to other plasma neutral amino acids (that is, tyrosine, phenylalanine, leucine, isoleucine, and valine) that compete for uptake into the brain. These results imply that carbohydrate intake results in the reduction of plasma amino acid levels. To compare their results with ours, tryptophan was considered to be transported to the brain after the administration of glucose or sucrose, which explains the decrease of tryptophan levels after the administration of glucose or sucrose in humans (Table 2, 3). The results shown in Tables 2 and 3 were similar to those of Wurtman's group. Of interest was that valine, leucine isoleucine, and phenylalanine decreased together with tryptophan, which appears to confirm the proposal by Wurtman's group that the amino acids competing with tryptophan for transport to the brain are transported to some tissues, possibly muscles. In regards to the relationship between the levels of plasma insulin and amino acids, Chevalier et al. [14] indicated that during hyperinsulinemic states, many amino acids, notably the BAAs, declined markedly, suggesting that the increased insulin levels reduced the levels of some amino acids. It has been shown that the levels of alanine, phenylalanine, valine, leucine, isoleucine, and tyrosine increase, whereas the levels of histidine and glutamine decrease during hyperglycemia, indicating that the changes in amino acid levels occur in parallel with the changes in fasting plasma glucose levels [5]. Insulin has been shown to increase transport of amino acids to skeletal muscles [15]. These results suggest that insulin assists the transfer of some amino acids (mostly EAAs and BCAAs) from the blood to tissues, possibly muscle, thus decreasing the plasma levels of some amino acids [14, 15]. Some research reports regarding the relationship between insulin sensitivity and plasma amino acid deprivation exist [16, 17]. Although the plasma levels of total NEAAs decreased after the administration of glucose in aged men, the decrease was also observed in the control group. Therefore, compared with that in the control group, the extent of the decrease in plasma levels of total NEAAs was significant at 120 min after glucose administration in young and aged men (Figure 3). We measured the levels of glucose or insulin in plasma over time and observed an increase in the plasma insulin levels after the administration of glucose or sucrose in human adults [18]. In particular, the insulin levels increased more significantly in young men than in aged men after the administration of sucrose [18]. Since insulin was suggested to be one of the factors to decrease the plasma levels of amino acids, sucrose administration may not be as effective as glucose administration in decreasing plasma amino acids levels in general [8-10, 14, 15]. However, sucrose administration most effectively decreased plasma levels of EAAs and BCAAs. Our results confirmed that the levels of EAAs, such as phenylalanine, leucine, isoleucine, valine, and tryptophan, decreased after the administration of glucose in humans. We also found that both EAA and BCAA levels after the administration of sucrose significantly decreased in both young and aged men. Probably changes in the plasma levels of EAAs and BCAAs are sensitive to an increase in the insulin levels because sucrose administration resulted in lower levels of insulin than glucose administration [17]. These results suggest that there were significant decreases in the plasma levels of total amino acids, EAAs, and NEAAs after the administration of glucose in both aged and young men. Sucrose administration resulted in a significant decrease in the plasma levels of total EAAs and BCAAs. A decrease in the plasma levels of total NEAAs was not significant in both aged and young men after the administration of sucrose. It is speculated that the transfer of EAAs and BCAAs from the blood to the tissues, such as muscles, is sensitive to a small increase in the insulin levels. In the present experiment, participants did not move. The exercise may change the results. The reason why we compared the effects of glucose administration with sucrose administration is that we wanted to show the potency of sucrose in changing plasma amino acids levels. Probably, sucrose uptake together with essential amino acids may be effective in increasing muscular movement.
ACKNOWLEDGEMENTS
We are grateful to Professor William S. Harris, Department of Medicine, Univ. of South Dakota School of Medicine for reading this manuscript and giving valuable suggestions. Experiments were designed and performed by all of the authors. AT wrote a manuscript. Statistical analyses were done by TT. All authors read the manuscript and approved the final version. All the authors had responsibilities for the final content. Ogawa,M.: No conflicts of interest, Takao,T.: No conflicts of interest, Ishii Y: No conflicts of interest, Shimizu F: no conflicts of interest, Takada T: No conflicts of interest.
FINANCIAL SUPPORT
This study was supported by grants by Ito Memorial Foundation, grant in aide of Showa Womens University and NPO "International Projects on Food andHealth." Word count: 3100
REFERENCES
- Noto H, Goto A, Tsujimoto T and Noda M. (2013). Low-carbohydrate diets and all-cause mortality: a systematic review and meta-analysis of observational studies. PLoS One. 8(1), e55030.
- Statements on Atkins' death. (2004). USA Today February 2nd, 2004.
- Felig P. (1975). Amino acid metabolism in man. Annu Rev Biochem. 44, 933-955
- Jefferson LS, Li JB and Rannels SR. (1977). Regulation by insulin of amino acid release and protein turnover in the perfused rat hemicorpus. J Biol Chem. 252(4),1476-1483.
- Biolo G, Williams BD, Fleming RY and Wolfe RR. (1999). Insulin action on muscle protein kinetics and amino acid transport during recovery after resistance exercise. Diabetes. 48(5), 949-957.
- Robinson MM, Soop M, Sohn TS, Morse DM, Schimke JM, et al. (2014). High insulin combined with essential amino acids stimulates skeletal muscle mitochondrial protein synthesis while decreasing insulin sensitivity in healthy humans. Clin Endocrinol Metab. Dec. 99(12), 2574-2583.
- Stancakova A, Civelek M, Saleem NK, Soininen P, et al. (2012). Hyperglycemia and a common variant of GCKR are associated with the levels of eight amino acids in 9,369 Finnishmen. Diabetes. 61(7), 1895-1902.
- Fernstrom JD and Wurtman RJ. (1971). Brain serotonin content: increase following ingestion of carbohydrate diet. Science. 174(4013), 1023-1025.
- Fernstrom JD and Wurtman RJ. (1972). Brain serotonin content: physiological regulation by plasma neutral amino acids. Science. 178(4059), 414-416
- Lipsett D, Madras BK, Wurtman RJ and Munro HN. (1973). Serum tryptophan level after carbohydrate ingestion: selective decline in non-albumin-bound tryptophan coincident with reduction in serum free fatty acids. Life Sci II. 12(2), 57-64.
- Pan RM, Mauron C, Glaeser B and Wurtman RJ. (1982). Effect of various oral glucose doses on plasma neutral amino acid levels. Metabolism. 31(9), 937-943.
- Shimbo K, Kubo S, Harada Y, Oonuki T, et al. (2010). Automated precolumn derivatization system for analyzing physiological amino acids by liquid chromatography/mass spectrometry. Biomed Chromatogr. 24(7). 683-691.
- Yoshida H, Kondo K, Yamamoto H, Kageyama N, et al. (2015). Validation of an analytical method for human plasma free amino acids by high-performance liquid chromatography ionization mass spectrometry using automated precolumn derivatization. J Chromatogr B Analyt Technol Biomed Life Sci. 998-999, 88-96.
- Chevalier S, Gougeon R, Kreisman SH, Cassis C, et al. (2004). The hyperinsulinemic amino acid clamp increases whole-body protein synthesis in young subjects. Metabolism. 53(3), 388-396.
- Bonadonna RC, Saccomani MP, Cobelli C and DeFronzo RA. (1993). Effect of insulin on system A amino acid transport in human skeletal muscle. J Clin Invest. 91(2), 514-521.
- Xiao F, Yu J, Guo Y, Deng J, et al. (2014). Effects of individual branched-chain amino acids deprivation on insulin sensitivity and glucose metabolism in mice. Metabolism. 63(6), 841-850.
- Spegel P, Lindqvist A, Sandberg M and Wierup N. (2014). Glucose-dependent insulinotropic polypeptide lowers branched chain amino acids in hyperglycemic rats. Regul Pept. 189, 11-16
- Takao T, Ogawa M, Ishii Y, Shimizu F, et al. (2016). Different glycemic responses to sucrose and glucose in old and young male adults. J Nutr Food Sci. 6, 460-465