Previous Issues Volume 2, Issue 2 - 2017
The Basal, Peak and Maximum Heart Rate Determinants of the Cardiac Patients Who Underwent Dobutamine Stress Echocardiography
Rabindra Nath Das1 *, Youngjo Lee2 , Bansi Badan Mukhopadhya3
1Department of Statistics, University of Burdwan, Burdwan, West Bengal, India. 2Department of Statistics, Seoul National University, Seoul, Korea. 3Department of Community Medicine, Agartal Govt. Medical College and GB Hospital, Agartala, Tripura, India.
Corresponding Author: Das RN, Department of Statistics, University of Burdwan, Burdwan, West Bengal, India, Tel: +03422634975; E-Mail: [email protected]
Received Date: 11 May 2017 Accepted Date: 28 Jun 2017 Published Date: 31 Jun 2017
Copyright © 2017 Das RN
Citation: Das RN, Lee Y and Mukhopadhya BB. (2017). The Basal, Peak and Maximum Heart Rate Determinants of the Cardiac Patients Who Underwent Dobutamine Stress Echocardiography. Mathews J Cardiol. 2(2): 013.
ABSTRACT
Objectives: The present report aims to identify the determinants of the basal, peak and maximum heart rates of cardiac patients who underwent dobutamine stress echocardiography (DSE). Background: The determinants of basal, peak and maximum heart rates for the DSE data were not derived, considering the real situations that the responses were positive & heteroscedastic. Moreover, these determinants are little known in the literature. Methods: The considered data set (UCLA stress echocardiography data) in the current study consists of 31 factors/variables on 558 individuals, which is originally taken from a total of 1183 patients referred for DSE between March 1991 and March 1996 to the UCLA Adult Cardiac Imaging and Hemodynamics Laboratories. The responses basal, peak and maximum heart rates are heterogeneous, positive, and gamma distributed, therefore, joint generalized linear gamma model fitting is used. Results: The mean basal heart rate (bhr) increases as the percent maximum predicted heart rate (pctMphr) (P < 0.001), or maximum blood pressure (mbp) (P < 0.001) increases. The mean bhr decreases as the basal blood pressure (basebp) (P < 0.001), or the dobutamine dose at maximum double product (dobdose) (P = 0.074), or age (P = 0.003) increases. The mean peak heart rate (pkhr) increases as the maximum heart rate (maxhr) (P < 0.001), or dpmaxdo (P < 0.001) increases. The mean pkhr decreases as the systolic blood pressure (sbp) (P < 0.001) increases. The mean pkhr is higher for the DSE cardiac patients of male (P = 0.076) than female, or the patients with no history of diabetes mellitus (hxofDM) (P = 0.003). The mean maximum heart rate (maxhr) increases as the pkhr (P < 0.001), or sbp (P < 0.001), or pctMphr (P < 0.001), or dpmaxdo (P < 0.001), or ejection fraction on dobutamine (dobEF) (P = 0.062) increases. The mean maxhr decreases as the mbp (P < 0.001), or age (P < 0.001) increases. There are many determinants of the variances of bhr, pkhr and maxhr. Conclusions: Mean basal heart rate is positively associated with the maximum blood pressure (BP), or percent maximum predicted heart rate, or double product of basal BP & basal heart rate, while it is negatively associated with basal BP, or age. The mean peak heart rate is negatively associated with the systolic BP, or age, or sex, while it is positively associated with maximum heart rate, or percent maximum predicted heart rate. The mean maximum heart rate is negatively associated with the maximum BP, or age, while it is positively associated with systolic BP, or percent maximum predicted heart rate. Many of the present findings and their effects are almost new in the heart rates diagnosis literature.
KEYWORDS
Cardiac Parameters; Dobutamine Stress Echocardiography; Gamma Models; Heart Rates; Non-Constant Variance.
INTRODUCTION
It is too hard to determine the individual heart rate changes even by an experienced heart specialist. Practically, there are many determinants for a heart rate to slow down, or speed up, or vary inexplicably. The determinants are not well-known in the heart disease literature [1, 2]. The maximum, peak and basal heart rates are generally used values in clinical medicine and physiology [3-5]. Generally, the percentage of peak, or maximum, or a fixed percentage of heart rate is used to prescribe the intensity of exercise, or medicine in both the rehabilitation programs and the disease prevention [6, 7]. It is known that a delayed heart rate recovery during the first and second minute after acute exercise is an independent predictor of overall mortality [8, 9]. The peak, or maximum, or basal heart rate is widely used as a criterion for achieving peak exertion in the determination of maximal aerobic capacity [4, 10, 11]. Recently, some longitudinal and cross-sectional studies have pointed that aerobic endurance training accelerates heart rate recovery after exercise in healthy subjects [11, 12]. In many clinical settings, exercise testing is not used if subjects attain an arbitrary percentage of their age-predicted maximal heart rate [13, 14]. In earlier research articles [13, 14], mainly metaanalysis and regression analysis have been used to identify the determinants of heart rates, assuming the response variance is constant. Note that the three heart rates basal, peak and maximum are positive and heterogeneous. For a positive response variable, the variance may be non-constant, due to variance-to-mean relationship. Such data set should be analysed using joint Log-normal and gamma models [15-17]. The current data sets (basal, peak and maximum) are positive, their variances are non-constant, distributions are non-normal, and the previous statistical methodologies are inappropriate. In addition, best of our knowledge, there is little study of heart rates for the DSE data [18]. These issues have motivated us to locate the heart rate (basal, peak and maximum) determinants of the DSE data set [18]. The present report aims to examine the following hypotheses. What are the determinants of the basal, or peak, or maximum heart rate of the DSE data set? What are the associations of the determinants with the basal, or peak, or maximum heart rate? What are the effects of the determinants on the basal, or peak, or maximum heart rate? Answer of these hypotheses are little known in the cardiology literature. These issues are addressed in the current report. Statistical Data Analysis Methods Some continuous positive responses such as basal, peak, and maximum heart rates have non-normal error distributions. The class of generalized linear models is used for analysing such data. In practice, the gamma and the log-normal distributions are often used in modelling positive data [15, 16]. For some positive response variables, it is often observed that there exists a variance-to-mean relationship, consequently the response variance may be non-constant. For the analysis of such positive data yi ’s, Nelder and Lee [17] proposed the joint generalized linear models (JGLMs) when E( yi ) = µi and Var (Yi) = σi2 µi2 Where σi2’s are the dispersion parameters and V (*) is the variance function. The variance (in GLMs) consists of two components. One component is V(µi) which dependents on the mean changes, and the other is σi2 , independent of mean adjustment. Note that the variance function (in GLMs) characterizes the GLM family distribution. For example, the distribution is gamma if V(µ) = µ2, normal if V(µ)= 1, and Poisson if V(µ) = µ, etc. Joint generalized linear models for the mean and dispersion parameters (proposed by Nelder and Lee [17] are µi = g(µi) = xit β and εi = h(σi2) = wit γ Where g(•) and h(•) are GLM link functions (i.e., the relationship between the mean and the linear predictors, or the relationship between the variance and the linear predictors) for the mean and the dispersion respectively; and xit, are wit respectively, the row vectors (based on the levels of the variables) for the mean and the variance regression models. Maximum likelihood (ML) method is used for estimating the mean model parameters, and restricted ML (REML) estimators are used for the dispersion model [16]. Data Description and Descriptive Statistics Data: The current study has considered a secondary data set (UCLA stress echocardiography data) of 31 variables on 558 subjects from a total of 1183 patients referred to the UCLA Adult Cardiac Imaging and Hemodynamic Laboratories for DSE between March 1991 and March 1996. For every subject, 31 factors (Table 1) have been observed. In the current analyses, we have considered only 558 subjects with all non-missing information. This data set has been used by many authors [18-20]. A detailed description of the DSE data set, patient population, its collection method, and the DSE is given in [18]. We have not reproduced the data description herein as it would increase the length of the paper. It is well-known that the Dobutamine stress echocardiography is widely and successfully used to determine whether a patient with or without known coronary artery disease has ischemia [21-23].
Table 1: Operationalization of variables in the analysis.
| Variable name | Operationalization | Mean (S.D.)/Proportion |
| bhr (y1) | Basal heart rate (bpm) | 75.29 (15.42) |
| basebp (y2) | Basal blood pressure (mmHg) | 135.3 (20.77) |
| basedp (x1) | Basal double product (DP) bhr*basebp (bpm*mmHg) | 10181 (2579.75) |
| pkhr (x2) | Peak heart rate (bpm) | 120.6 (22.57) |
| sbp (x3) | Systolic blood pressure (mmHg) | 146.9 (36.53) |
| dp (x4) | DP pkhr*sbp (bpm*mmHg) | 17634 (5220.53) |
| dose (x5) | Dobutamine dose (DD) given | 33.75 (8.13) |
| maxhr (x6) | Maximum heart rate (bpm) | 119.4 (21.91) |
| pctMphr (x7) | Percent maximum predicted heart rate | 78.57 (15.12) |
| mbp (x8) | Maximum blood pressure (mmHg) | 156.0 (31.71) |
| dpmaxdo (x9) | DP on max DD (bpm¤mmHg) | 18550 (4901.43) |
| dobdose (x10) | DD at max double product (mg) | 30.24 (9.54) |
| age (x11) | Age (years) | 67.34 (12.05) |
| gender (F12) | Gender (male = 0, female = 1) | 0%=39.43, 1%=60.57 |
| baseEF (x13) | Baseline cardiac ejection fraction | 55.60 (10.32) |
| dobEF (x14) | Ejection fraction on dobutamine | 65.24 (11.76) |
| chestpain (F15) | Chest pain (yes (y) = 0, no (n) = 1) | 0%= 69.18, 1%= 30.82 |
| restwma (F16) | Resting wall motion abnormality on echocardiogram (ECDG) (y = 0, n = 1) | 0%= 53.94, 1%= 46.06 |
| posSE (F17) | Positive stress ECDG (y = 0, n = 1) | 0%= 75.63, 1%= 24.37 |
| newMI (F18) | new myocardial infraction (MI) (y = 0, n = 1) | 0%= 94.98, 1%= 5.02 |
| newPTCA (F19) | Recent angioplasty (y = 0, n = 1) | 0%= 95.16, 1%= 4.84 |
| newCABG (F20) | Recent bypass surgery (y = 0, n = 1) | 0%= 94.09, 1%= 5.91 |
| Death (F21) | death (y = 0, n = 1) | 0%= 95.70, 1%= 4.30 |
| hxofHT (F22) | History of hypertension (y = 0, n = 1) | 0%= 29.57, 1%= 70.43 |
| hxofDM (F23) | History of diabetes (y = 0, n = 1) | 0%= 63.08, 1%= 36.92 |
| hxofCig (F24) | History of smoking (non-smoker = 0, moderate = 1, heavy= 2) | 0%= 53.41, 1%= 24.73, 2%= 21.86 |
| hxofMI (F25) | History of MI (y = 0, n = 1) | 0%= 72.40, 1%= 27.60 |
| hxofPTCA (F26) | History of angioplasty (y = 0, n = 1) | 0%= 92.65, 1%= 7.35 |
| hxofCABG (F27) | History of coronary artery bypass surgery (y = 0, n = 1) | 0%= 84.23, 1%= 15.77 |
| any event (F28) | Death, newMI, newPTCA or newCABG (death = 0, no = 1) | 0%= 84.05, 1%= 15.95 |
| ecg (F29) | Baseline electrocardiogram diagnosis (normal = 0, equivocal = 1, MI=2) | 0%= 55.91, 1%= 31.54, 2%= 12.72 |
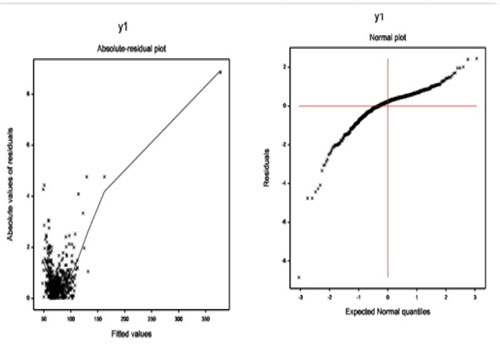
VARIABLES Dependent variables The current article aims to locate the determinants of basal, peak, and maximum heart rates. Therefore, three separate analyses have been derived. The first analysis considers basal heart rate as the dependent variable, and the remaining other variables in (Table 1) as the independent variables. Similarly, the second and the third analyses consider respectively, peak and maximum heart rate as the dependent variable, and the remaining others as the independent variables. Independent variables The DSE data set contains two sets of independent variables namely, qualitative and quantitative. Sixteen independent variables (Table 1) are qualitative and the remaining others are continuous variables. A description of each set of item and how they are operationalized for the current study is displayed in (Table 1). Descriptive Statistics The proportion of all the levels of the categorical variables, arithmetic means and standard deviations of all the continuous variables are displayed in Table 1. The average age of the study subject units is 67.34 years. The number of male (39.43%) study subjects are less than female (60.57%). The average basal, peak and maximum heart rates of the DSE patients are respectively, 75.29, 120.6, and 119.4. The percentages of the DSE patients with restwma, chest pain, newMI, posSE, newCABG and newPTCA are respectively, 53.94%, 69.18%, 94.98%, 75.63%, 94.09% and 95.16%. The percentages of non-smoker, moderate and heavy DSE patients are respectively, 53.41%, 24.73%, and 21.86%. The DES patient’s history of hypertension (hxofHT), diabetes (hxofDM), coronary artery bypass surgery (hxofCABG), angioplasty (hxofPTCA), and myocardial infraction (hxofMI) percentages are respectively, 29.57%, 63.08%, 84.23%, 92.65%, and 72.40%. The percentages of the ecg status of the DSE patients as normal, equivocal, and MI are respectively, 55.91%, 31.54%, and 12.72%. A descriptive idea of the factors/variables is given above. It is really interesting to identify what are the statistical significant determinants of the basal, peak, and maximum heart rates? In the following sections, the statistical significant determinants of the basal, peak and maximum heart rates are derived. Basal Heart Rate Analysis, Results & Discussion Basal heart rate analysis One can easily examine that the responses basal, peak and maximum heart rates of the considered DSE data are positive and heteroscedastic. Therefore, these responses should be examined using both the gamma and the log-normal models [15, 16, 24]. We have analysed these three responses data using both the gamma and the log-normal models. It is found that the gamma model analysis gives better results than the log-normal model for all the three cases. Thus, we have reported only the gamma model analysis results for these three cases. In the present section, we have derived the analysis of basal heart rate. In the present DSE data set, there are sixteen qualitative characters or factors. We have accepted the constraint for factors that the effects of the first levels are zero. Thus, we have taken that the first level of each factor as the reference level by estimating it’s as zero. Let ai for i = 1, 2, 3 be the main effect of A. Therefore, â2 = â2 -- â1 , as we have taken â1 = 0. For example, the effect of the difference between the second and the first levels (i.e., â2 -- â1) in the main effect A gives the estimate of the effect A2. The basal heart rate (a continuous non-constant variance response) has been analysed using the joint gamma models, based on the remaining other explanatory variables (Table 1), and the results are displayed in (Table 2). The selected models have the smallest Akaike information criterion (AIC) value in each class. Note that AIC selects a model which minimizes the squared error loss and predicted additive errors [25]. The final gamma fitted models (Table 2) have been selected based on minimum AIC value (2706.167). The selected models (Table 2) include some partially significant factors (dobutamine dose at max double product (dobdose), electrocardiogram diagnosis (ecg), history of angioplasty (hxofPTCA), which are known as confounders in epidemiology. It is well-known that all the selected effects are not necessarily significant [25, 26]. The diagnostic plots of joint generalized linear gamma fitted models (Table 2) have been examined in (Figure 1). The absolute residual values are plotted with respect to fitted values in Figure 1(a) for the gamma fitted models (Table 2). The residuals plot is almost flat with respect to running means except the right tail. The right tail is increasing due to the largest boundary value. The normal probability plot of the fitted gamma mean model (Table 2) is displayed in (Figure 1(b)) which does not show any systematic departure.
Table 2: Results for Mean and Dispersion Models of Basal Heart Rate.
| Model | covariate | estimate | standard error | tvalue | pvalue |
| Mean model | |||||
| Constant | 4.1999 | 0.0234 | 179.25 | < 0.001 | |
| basebp (y2) | -0.0067 | 0.0001 | -64.80 | <0.001 | |
| basedp (x1) | 0.0001 | 0.0001 | 102.97 | < 0.001 | |
| pctMphr (x7) | 0.0019 | 0.0004 | 4.39 | < 0.001 | |
| mbp (x8) | 0.0007 | 0.0002 | 3.35 | < 0.001 | |
| dpmaxdo (x9) | -0.0000 | 0.0001 | -3.67 | < 0.001 | |
| dobdose (X10) | -0.0003 | 0.0001 | -1.79 | 0.074 | |
| age (x11) | -0.0008 | 0.0002 | -3.03 | 0.003 | |
| posSE (Fx17 2) | -0.0070 | 0.0036 | -1.91 | 0.056 | |
| ecg (Fx29 2) | 0.0049 | 0.0033 | 1.45 | 0.148 | |
| ecg (Fx29 3) | 0.0078 | 0.0048 | 1.60 | 0.110 | |
| Dispersion model | |||||
| Constant | -3.0615 | 0.9509 | -3.220 | 0.001 | |
| basebp (y2) | -0.0070 | 0.0039 | -1.770 | 0.077 | |
| dose (x5) | 0.0268 | 0.0080 | 3.357 | <0.001 | |
| mbp (x8) | -0.0042 | 0.0022 | -1.925 | 0.055 | |
| age (x11) | -0.0132 | 0.0059 | -2.230 | 0.026 | |
| dobEF (x14) | -0.0358 | 0.0075 | -4.762 | <0.001 | |
| gender (Fx12 2) | 0.4859 | 0.1432 | 3.394 | <0.001 | |
| chestpain (Fx15 2) | 0.4779 | 0.1450 | 3.296 | <0.001 | |
| hxofPTCA(Fx26 2) | -0.4511 | 0.2520 | -1.790 | 0.074 |
Figure 1a and 1b: For the gamma fitted models of basal heart rate (Table 2), the (a) absolute residuals plot with respect to fitted values, and the (b) normal probability plot of the mean model.
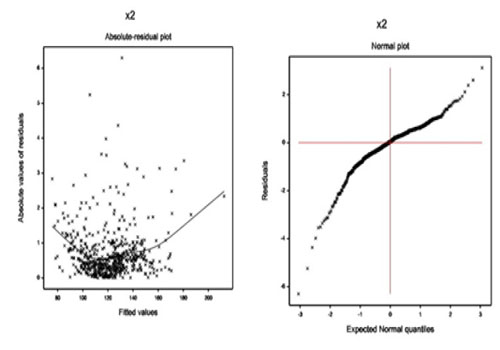
Results of Basal Heart Rate The summarized results of basal heart rate are given in (Table 2). It is observed (from Table 2) that mean basal heart rate (bhr) is separately negatively associated with the basal blood pressure (basebp) (P < 0.001), double product of maximum dobutamine dose (dpmaxdo) (P < 0.001), dobutamine dose at maximum double product (dobdose) (P = 0.074), age (P = 0.003), positive stress on echocardiography (posSE) (P = 0.056). Also the mean bhr is separately positively associated with the double product of basal blood pressure and basal heart rate (basedp) (P < 0.001), percent maximum predicted heart rate (pctMphr) (P < 0.001), maximum blood pressure (mbp) (P < 0.001), and ecg (P = 0.110). From the gamma fitted variance model (Table 2), the bhr variance is separately negatively associated with the basebp (P = 0.77), mbp (P = 0.055), age (P = 0.026), ejection fraction on dobutamine dose (dobEF) (P < 0.001), and hxofPTCA (P = 0.074). Also the bhr variance is separately positively associated with the dobutamine dose (dose) (P < 0.001), gender (P < 0.001), and chest pain (P < 0.001). Interpretations of Basal Heart Rate The mean model of basal heart rate (Table 2) interprets the followings: The mean basal heart rate (bhr) is inversely significantly associated with the basal blood pressure (basebp) (P < 0.001). This implies that the bhr increases or decreases according as the basebp decreases or increases. The mean bhr is directly significantly associated with the double product of basebp and bhr (basedp) (P < 0.001). Note that the basedp is a direct function of bhr. The mean bhr is directly significantly associated with the percent maximum predicted heart rate (pctMphr) (P < 0.001). The mean bhr is directly significantly associated with the maximum blood pressure (mbp) (P < 0.001). The mean bhr is inversely significantly associated with the double product of maximum dobutamine dose and mbp (dpmaxdo) (P < 0.001). The mean bhr is inversely partially associated with the dobutamine dose at maximum double product (dobdose) (P = 0.074), implying that the bhr increases or decreases according as the dobdose low or high. The mean bhr is inversely significantly associated with the age (P = 0.003). The mean bhr is inversely significantly associated with the positive stress on echocardiogram (yes = 0, no = 1) (posSE) (P = 0.056), implying that the bhr is higher or lower according as the DSE cardiac patients with having or not having posSE. The mean bhr is directly partially associated with the baseline electrocardiogram diagnosis (normal = 0, equivocal = 1, MI = 2) (ecg) (P = 0.110), implying that the bhr is higher of the DSE cardiac patients with ecg at the MI level. The variance model of basal heart rate (Table 2) interprets the followings: The variance of the basal heart rate (bhr) is inversely partially associated with the basebp (P = 0.077). Note that the basebp has the similar inverse association with both the mean and variance of bhr. The bhr variance is directly significantly associated with the given dobutamine dose (dose) (P < 0.001). The bhr variance is inversely significantly associated with the mbp (P = 0.055). Note that mbp has direct and inverse association with the mean and variance of bhr, respectively. The bhr variance is inversely significantly associated with the age (P = 0.026). The bhr variance is inversely significantly associated with the ejection fraction on dobutamine dose (dobEF) (P < 0.001). The bhr variance is directly significantly associated with the gender (male = 0, female = 1) (P < 0.001), implying that the bhr variance is lower for male than female. The bhr variance is directly significantly associated with the chest pain (yes = 0, no = 1) (P < 0.001), implying that the bhr variance is lower for the DSE cardiac patients having chest pain. The bhr variance is inversely partially associated with the history of angioplasty (yes = 0, no = 1) (hxofPTCA) (P = 0.074), implying that the bhr variance is lower for the DSE cardiac patients having no hxofPTCA. Peak Heart Rate Analysis, Results & Discussion Peak Heart Rate Analysis The peak heart rate (a continuous non-constant variance response) has been analysed using the joint gamma models, based on the remaining other explanatory variables (Table 1), and the results are displayed in Table 3. The final gamma fitted models (Table 3) have been selected based on minimum AIC value (3376.076). The final selected gamma models (Table 3) include some insignificant or partially significant factors (pctMphr, age, chest pain, bhr, basebp, basedp, dobEF, restwma) as confounders. The diagnostic plots of joint generalized linear gamma fitted models (Table 3) have been displayed in (Figure 2).
Table 3: Results for Mean and Dispersion Models of Peak Heart Rate.
| Model | covariate | estimate | standard error | tvalue | pvalue |
| Mean model | |||||
| Constant | 4.0546 | 0.0387 | 104.65 | < 0.001 | |
| sbp (x3) | -0.0009 | 0.0001 | -6.96 | <0.001 | |
| maxhr (x6) | 0.0063 | 0.0004 | 12.86 | < 0.001 | |
| pctMphr (x7) | 0.0002 | 0.0006 | 0.26 | 0.794 | |
| dpmaxdo (x9) | 0.0001 | 0.0001 | 6.39 | < 0.001 | |
| age (x11) | -0.0001 | 0.0004 | -0.24 | 0.810 | |
| gender (Fx12 2) | -0.0036 | 0.0021 | -1.73 | 0.084 | |
| chestpain (Fx15 2) | -0.0026 | 0.0022 | -1.16 | 0.246 | |
| restwma (Fx16 2) | 0.0048 | 0.0023 | 2.08 | 0.037 | |
| hxofDM (Fx23 2) | 0.0063 | 0.0021 | 2.93 | 0.003 | |
| Dispersion model | |||||
| Constant | 2.2361 | 2.5544 | 0.875 | 0.381 | |
| bhr (y1) | -0.0358 | 0.0343 | -1.042 | 0.269 | |
| basebp (y2) | -0.0225 | 0.0189 | -1.189 | 0.234 | |
| basedp (x1) | 0.0003 | 0.0002 | 1.148 | 0.251 | |
| sbp (x3) | -0.0438 | 0.0054 | -8.052 | <0.001 | |
| pctMphr (x7) | -0.0838 | 0.0068 | <0.001 | ||
| mbp (x8) | 0.0428 | 0.0064 | 6.679 | <0.001 | |
| dobdse (x10) | -0.0566 | 0.0084 | -6.765 | <0.001 | |
| age (x11) | 0.0257 | 0.0064 | 3.995 | <0.001 | |
| dobEF (x14) | 0.0093 | 0.0073 | 1.270 | 0.204 | |
| gender (Fx12 2) | 0.3865 | 0.1487 | 2.599 | 0.009 | |
| chestpain (Fx15 2) | 0.3100 | 0.1473 | 2.105 | 0.035 | |
| restwma (Fx16 2) | -0.0400 | 0.1621 | -0.247 | 0.805 | |
| hxofDM (Fx23 2) | -0.6411 | 0.1607 | -3.989 | <0.001 | |
| hxofCABG (Fx27 2) | -0.3316 | 0.1920 | -1.727 | 0.084 |
The absolute residual values are plotted with respect to fitted values in Figure 2(a) for the gamma fitted models (Table 3). The residuals plot is exactly fat with respect to running means, indicating that the variance is constant for the fitted model. The normal probability plot of the fitted gamma mean model (Table 3) is displayed in Figure 2(b) which does not show any lack of fit with respect to variables or outliers.
Figure 2: For the gamma fitted models of peak heart rate (Table 3), the (a) absolute residuals plot with respect to fitted values, and the (b) normal probability plot of the mean model.
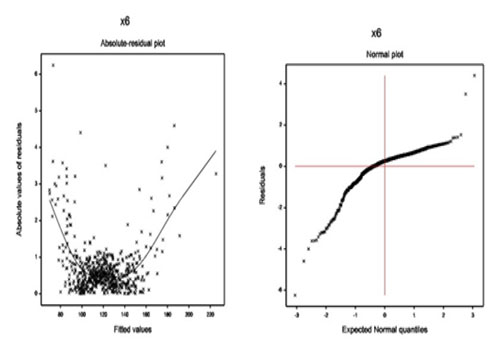
Results of Peak Heart Rate The summarized results of peak heart rate are displayed in Table 3. It is observed (from Table 3) that the mean peak heart rate (pkhr) is separately negatively associated with the systolic blood pressure (sbp) (P < 0.001) and gender (P = 0.084), Also the mean pkhr is separately positively associated with the maximum heart rate (maxhr) (P < 0.001), dpmaxdo (P < 0.001), resting wall motion abnormality on echocardiogram (restwma) (P = 0.037) and history of diabetes mellitus (hxofDM) (P = 0.003). From the gamma fitted variance model (Table 3), the pkhr variance is separately negatively associated with the sbp (P < 0.001), pctMphr (P < 0.001), dobdose (P < 0.001), hxofDM (P < 0.001), and history of coronary artery bypass surgery (hxofCABG) (P = 0.084). Also the pkhr variance is separately positively associated with the mbp (P < 0.001), age (P < 0.001), gender (P = 0.009) and chest pain (P = 0.035). Interpretations of Peak Heart Rate The mean model of peak heart rate (Table 3) interprets the followings: The mean peak heart rate (pkhr) is inversely significantly associated with the systolic blood pressure (sbp) (P < 0.001). The mean pkhr is directly significantly associated with the maximum heart rate (maxhr) (P < 0.001). This is due to the fact the maxhr is a function of pkhr. The mean pkhr is directly significantly associated with the double product of maximum dobutamine dose and mbp (dpmaxdo) (P < 0.001). The mean pkhr is inversely partially associated with the gender (male = 0, female = 1) (P = 0.084), implying that the pkhr is lower for DSE female cardiac patients than male. The mean pkhr is directly significantly associated with the resting wall motion abnormality on echocardiogram (yes = 0, no = 1) (restwma) (P = 0.037), implying that the pkhr is lower of the DSE cardiac patients with having restwma than the patients without restwma. The mean pkhr is directly significantly associated with the history of diabetes mellitus (yes = 0, no = 1) (hxofDM) (P = 0.003), implying that the pkhr is lower of the DSE cardiac patients with having hxofDM than the patients without hxofDM. The variance model of peak heart rate (Table 3) interprets the followings: The variance of the peak heart rate (pkhr) is inversely significantly associated with the sbp (P < 0.001). The pkhr variance is inversely significantly associated with the pctMphr (P < 0.001). The pkhr variance is directly significantly associated with the mbp (P < 0.001). The pkhr variance is inversely significantly associated with the dobdose (P < 0.001). The pkhr variance is directly significantly associated with the age (P < 0.001), implying that the pkhr variance increases of the DSE cardiac patients at the older ages. The pkhr variance is directly significantly associated with the gender (male = 0, female = 1) (P = 0.009), implying that the pkhr variance is lower for male DSE cardiac patients than female. The pkhr variance is directly significantly associated with the chest pain (yes = 0, no = 1) (P = 0.035), implying that the pkhr variance is lower for the DSE cardiac patients having chest pain than the patients with no chest pain. The pkhr variance is inversely significantly associated with the history of diabetes mellitus (yes = 0, no = 1) (hxofDM) (P < 0.001), implying that the pkhr variance is lower for the DSE cardiac patients having no hxofDM than the patients with hxofDM. The pkhr variance is inversely partially associated with the history of coronary artery bypass surgery (hxofCABG) (yes = 0, no = 1) (P = 0.084), implying that the pkhr variance is lower for the DSE cardiac patients having no hxofCABG than the patients with hxofCABG. Maximum Heart Rate Analysis, Results & Discussion Maximum heart rate analysis The maximum heart rate (a continuous non-constant variance response) has been analyzed using the joint gamma models, based on the remaining other explanatory variables (Table 1), and the results are displayed in (Table 4). The final gamma fitted models (Table 4) have been selected based on minimum AIC value (2748.75). The final selected gamma models (Table 4) include some insignificant or partially significant factors (dobEF, chest pain, newCABG, hxofDM, posSE, any event) as confounders. The diagnostic plots of joint generalized linear gamma fitted models (Table 4) have been displayed in (Figure 3). The absolute residual values are plotted with respect to fitted values in Figure 3(a) for the gamma fitted models (Table 4). The left and right tails of Figure 3(a) are respectively, decreasing and increasing due to the large values at the two boundaries. The middle part of the residuals plot is exactly fat with respect to running means, indicating that the variance is constant for the fitted model. The normal probability plot of the fitted gamma mean model (Table 4) is displayed in Figure 3(b) which does not show any systematic departure.
Table 4: Results for Mean and Dispersion Models of Maximum Heart Rate.
| Model | covariate | estimate | standard error | tvalue | pvalue |
| Mean model | |||||
| Constant | 4.1974 | 0.0251 | 166.70 | < 0.001 | |
| pkhr (x2) | 0.0024 | 0.0002 | 8.06 | <0.001 | |
| sbp (x3) | 0.0019 | 0.0002 | 7.47 | < 0.001 | |
| dp (x4) | -0.0001 | 0.0001 | -8.02 | <0.001 | |
| pctMphr (x7) | 0.0061 | 0.0004 | 12.68 | < 0.001 | |
| mbp (x8) | -0.0034 | 0.0002 | -12.34 | < 0.001 | |
| dpmaxdo (x9) | 0.0001 | 0.0001 | 13.05 | <0.001 | |
| age (x11) | -0.0031 | 0.0002 | -11.31 | <0.001 | |
| dobEF (x14) | 0.0002 | 0.0001 | 1.87 | 0.062 | |
| chestpain (Fx15 2) | 0.0030 | 0.0019 | 1.57 | 0.116 | |
| newCABG (Fx20 2) | 0.0044 | 0.0028 | 1.53 | 0.126 | |
| hxofDM (Fx23 2) | 0.0036 | 0.0019 | 1.89 | 0.059 | |
| hxofPTCA (Fx26 2) | 0.0078 | 0.0025 | 3.08 | <0.001 | |
| Dispersion model | |||||
| Constant | -3.7077 | 0.6781 | -5.468 | <0.001 | |
| dobdose (x10) | 0.0243 | 0.0066 | 3.698 | <0.001 | |
| age (x11) | -0.0113 | 0.0070 | -1.606 | 0.108 | |
| dobEF (x14) | -0.0543 | 0.0064 | -8.516 | <0.001 | |
| gender (Fx12 2) | 0.3988 | 0.1487 | 2.682 | 0.007 | |
| posSE (Fx17 2) | -0.3132 | 0.1741 | -1.799 | 0.072 | |
| newCABG (Fx20 2) | -1.2211 | 0.3863 | -3.161 | <0.001 | |
| death (Fx21 2) | -1.0345 | 0.4503 | -2.297 | 0.0219 | |
| hxofDM (Fx23 2) | -0.4870 | 0.1438 | -3.386 | <0.001 | |
| hxofCig (Fx24 2) | 0.3333 | 0.1630 | 2.045 | 0.041 | |
| hxofCig (Fx24 3) | 0.4661 | 0.1672 | 2.787 | 0.005 | |
| hxofPTCA (Fx26 2) | -1.1451 | 0.3058 | -3.745 | <0.001 | |
| any event (Fx28 2) | 0.4676 | 0.3044 | 1.536 | 0.125 | |
| ecg (Fx29 2) | -0.5130 | 0.1445 | -3.549 | <0.001 | |
| ecg (Fx29 3) | -0.1795 | 0.2307 | -0.778 | 0.436 |
Figure 3: For the gamma fitted models of maximum heart rate (Table 4), the (a) absolute residuals plot with respect to fitted values, and the (b) normal probability plot of the mean model.
Results of Maximum Heart Rate The summarized results of maximum heart rate are displayed in Table 4. It is observed (from Table 4) that the mean maximum heart rate (maxhr) is separately positively associated with the pkhr (P < 0.001), sbp (P < 0.001), pctMphr (P < 0.001), dpmaxdo (P < 0.001), dobEF ( P= 0.062), chest pain (P = 0.116), newCABG (P = 0.126), hxofDM (P = 0.059), and hxofPTCA (P = 0.002). The mean maxhr is separately negatively associated with the double product of pkhr and sbp (dp) (P < 0.001), mbp (P < 0.001), and age (P < 0.001). From the gamma fitted variance model (Table 4), the maxhr variance is separately negatively associated with the age (P = 0.108), dobEF (P < 0.001), posSE (P = 0.072), newCABG (P < 0.001), death (P < 0.021), hxofDM (P < 0.001), hxofPTCA (P < 0.001), ecg (P < 0.001), Also the maxhr variance is separately positively associated with the dobdose (P < 0.001), gender (P = 0.007), hxofCig (P = 0.041), any event (0.125). Interpretations of Maximum Heart Rate The mean model of maximum heart rate (Table 4) interprets the followings: The mean maximum heart rate (maxhr) is directly significantly associated with the pkhr (P < 0.001). This is due to the fact maxhr is a function of pkhr. The mean maxhr is directly significantly associated with the sbp (P < 0.001). The mean maxhr is inversely significantly associated with the double product of pkhr & sbp (dp) (P < 0.001). The mean maxhr is directly significantly associated with the pctMphr (P < 0.001). The mean maxhr is inversely significantly associated with the mbp (P < 0.001). The mean maxhr is directly significantly associated with the dpmaxdo (P < 0.001). The mean maxhr is inversely significantly associated with the age (P < 0.001), implying that the maxhr decreases or increases according as the older or younger ages. The mean maxhr is directly partially significantly associated with the dobEF (P = 0.062). The mean maxhr is directly partially significantly associated with the chest pain (yes = 0, no = 1) (chestpain) (P = 0.116), or the recent bypass surgery (yes = 0, no = 1) (newCABG) (P = 0.126), implying that the maxhr is lower of the DSE cardiac patients with having chestpain, or newCABG, and vice versa. The mean maxhr is directly significantly associated with the hxofDM (yes = 0, no = 1) (P = 0.059), or the history of angioplasty (yes = 0, no = 1) (hxofPTCA) (P = 0.002), indicating that the maxhr is higher of the DSE cardiac patients with no hxofDM, or hxofPTCA. The variance model of maximum heart rate (Table 4) interprets the followings: The variance of the maxhr is positively significantly associated with the dobdose (P < 0.001). The maxhr variance is inversely partially associated with the age (P = 0.108), indicating that the maxhr variance increases of the DSE cardiac patients at the younger ages. The maxhr variance is inversely significantly associated with the dobEF (P < 0.001). The maxhr variance is directly significantly associated with the gender (male = 0, female = 1) (P = 0.007), implying that the maxhr variance is higher for female DSE cardiac patients than male. The maxhr variance is inversely partially associated with the positive stress on echocardiogram (yes = 0, no = 1) (posSE) (P = 0.072), indicating that the maxhr variance is higher for the DSE cardiac patients having the posSE. The maxhr variance is inversely significantly associated with the newCABG (yes = 0, no = 1) (P < 0.001), indicating that the maxhr variance is higher for the DSE cardiac patients having the newCABG. The maxhr variance is inversely significantly associated with the death (yes = 0, no = 1) (death) (P = 0.021), implying that the maxhr variance is higher for the DSE cardiac patients who are close to death than the living patients. The maxhr variance is inversely significantly associated with the hxofDM (yes = 0, no = 1) (P < 0.001), implying that the maxhr variance is higher for the DSE cardiac patients having the hxofDM. The maxhr variance is directly significantly associated with the history of smoking (non-smoker = 0, medium = 1, high = 2) (hxofCig), implying that the maxhr variance is higher for the DSE cardiac patients with smoking status at medium level (P = 0.041) and at high level (P = 0.005) than the non-smokers. The maxhr variance is inversely significantly associated with the history of angioplasty (yes = 0, no = 1) (hxofPTCA) (P < 0.001), implying that the maxhr variance is higher for the DSE cardiac patients with hxofPTCA. The maxhr variance is directly partially associated with the factor any event (death = 0, no = 1) (P = 0.125), implying that maxhr variance is higher for the living DSE cardiac patients than the patients close to death. The maxhr variance is inversely significantly associated with the baseline electrocardiogram diagnosis (normal = 0, equivocal = 1, MI = 2) (ecg) at equivocal level (P < 0.001), implying that the maxhr variance is lower for the DSE cardiac patients with ecg at equivocal level than the others. CONCLUDING REMARKS The determinants of the basal, peak and maximum heart rates of the DSE cardiac patients are derived in (Tables 2 to 4). The effects of the determinants are described in the interpretation sections. From (Tables 2), it is observed that mean basal heart rate is negatively associated with the basal BP, or double product of maximum dobutamine dose, or dobutamine dose at maximum double product, or age, or positive stress on echocardiography, while it is positively associated with the double product of basal BP & basal heart rate, or percent maximum predicted heart rate, or maximum BP. Variance of basal heart rate is positively associated with the given dobutamine dose, or sex, or chest pain , while it is negatively associated with the basal BP, or maximum BP, or age, or ejection fraction at dobutamine dose, or history of angioplasty. (Table 3) shows that mean peak heart rate is positively associated with the maximum heart rate, or percent maximum predicted heart rate, or double product of maximum dobutamine dose, resting wall motion abnormality on echocardiogram, or history of diabetes mellitus, while it is negatively associated with the systolic BP, or age, or sex, or chest pain. The variance of peak heart rate is negatively associated with systolic BP, or percent maximum predicted heart rate, or dobutamine dose at maximum double product, or history of diabetes mellitus, or history of coronary artery bypass surgery, while it is positively associated with the maximum BP, or age, or sex, or chest pain. From Table 4, the mean maximum heart rate is positively associated with the peak heart rate, or systolic BP, or percent maximum predicted heart rate, or double product of maximum dobutamine dose, or ejection fraction at dobutamine dose, or history of diabetes mellitus, or history of angioplasty, while it is negatively associated with the double product of peak heart rate & systolic BP, or maximum BP, or age. The variance of maximum heart rate is positively associated with the dobutamine dose at maximum double product, or sex, or history of smoking, while it is negatively associated with the age, or ejection fraction at dobutamine dose, or positive stress on echocardiogram, or recent bypass surgery, or history of diabetes mellitus, or history of angioplasty, or status of echocardiography. It is observed from (Table 3 and 4) that the peak and maximum heart rates are positively associated with each other. The mean basal heart rate is negatively associated with the basal blood pressure, and it is positively associated with the maximum blood pressure (Table 2). The variance of basal heart rate is negatively associated with both the basal and maximum blood pressure (Table 2). The mean and the variance of peak heart rate are negatively associated with the systolic blood pressure, while the variance of peak heart rate is positively associated with the maximum blood pressure (Table 4). The mean maximum heart rate is positively associated with the systolic blood pressure, and it is negatively associated with the maximum blood pressure (Table 4). These results conclude that the heart rates (basal, peak and maximum) are highly associated with the basal, systolic and maximum blood pressures. Age is an important factor of heart rates. The mean and the variance of basal heart rate are significantly negatively associated with the age (Table 2), while the mean peak heart rate is insignificantly negatively associated with age, and the variance of peak heart rate is significantly positively associated with the age (Table 3). On the other hand, the mean and the variance of maximum heart rate are respectively, negatively significantly and partially associated with the age (Table 4). Smoking is also an important determinant of basal (Table 2) and maximum heart rate (Table 4). There are many more determinants of the heart rates (given in Tables 2-4). The present report attempts to derive the determinants of the heart rates of the DSE cardiac patients (Tables 2-4). It derives the complex relationships of the three heart rates with their determinants (Tables 2-4). It is true that a probabilistic model reveals the inherent truth that is covered in a complex system. There are two purposes in the present report. The first is to derive the heart rate determinants of the DSE data, and to compare the derived results with the previous research results. The second purpose is to compare the statistical assumptions used by previous researchers regarding the heart rate determinants with the present assumptions. Our concern is that the earlier research, using some inappropriate statistical assumptions, would draw important conclusions from erroneous assumptions. The given DSE data set is heterogeneous and positive, therefore, the joint generalized log-normal and gamma model fittings are more appropriate for this data set [24, 26]. We have computed both the JGLMs for this data set, and it is found that the gamma JGLM results are better than the log-normal fit (not shown), so only the gamma JGLM results are displayed. The current derived results, though not completely conclusive, are revealing: *The current findings have been derived based on the following statistical criteria. First, is that the final findings have been derived based on comparing both the gamma and the log-normal models. Second, is that the derived estimates have very small standard errors (Tables 2-4). Third, is the smallest AIC value within the model class. Fourth, is identifying the appropriate response distribution. Fifth, is the proper regression diagnostic using graphical analysis. *The present findings confirm some earlier research outputs, and some conflicts of earlier research outputs have been attempted to remove. *A fundamental conclusion has to do with the use of previous derived statistical models. While further research is called for, it is observed that the gamma joint GLMs (for basal, peak and maximum heart rates) are much more efficient than either classical regression analysis or joint log-normal, as they better fit the data. In short, research should have greater faith in the present results than those emanating from the previous models. The heart rate (basal, peak and maximum) determinants, their effects and associations are derived in the above sections. These results are almost new in the cardiology literature. It is too hard to determine the individual changes in the heart rate even by an expert cardiologist, as there are many factors are responsible for changing the heart rates in both the mean and variance models [Tables 2-4]. Cardiac patients and medical practitioners will be benefited from the present research outputs. The present findings are associated with the DSE data set, even though this report recommends the following for all individuals. Heart rates are related with the blood pressure, so care should be taken for both the heart rates and blood pressure together. Smoking should be stopped (Table 4). Heart rate should be considered along with the age, sex, patient conditions such as chest pain, positive stress, resting wall motion abnormality on echocardiogram, history of diabetes mellitus, history of angioplasty (Tables 2-4). Dobutamine dose and its interaction effect with other factors (Tables 2-4) should be considered on determining the heart rates.
CONFLICT OF INTEREST
The author confirms that this article content has no conflict of interest.
ACKNOWLEDGEMENT
The author is very much indebted to the referees who have provided valuable comments to improve the paper. The author thanks Dr. Alan Garfinkel, UCLA School of Medicine, Department of Physiology, Los Angeles, California, USA, who generously provided the data sets to freely distribute and use for non-commercial purposes. This research was supported by an NRF grant funded by Korea Government (MEST) (No. 2011- 0030810).
REFERENCES
- Jose AD and Collison D. (1970). The normal range and determinants of the intrinsic heart rate in man. Cardiovasc Res. 4(2): 160-167.
- Graettinger WF, Smith DH, Neutel JM, Myers J, et al. (1995). Relationship of left ventricular structure to maximal heart rate during exercise. Chest. 107(2): 341-345.
- American College of Sports Medicine. (2000). ACSM’s Guidelines for Exercise Testing and Prescription. 6th ed. Baltimore, MD: Lippincott Williams & Wilkins.
- Fletcher GF. (1997). How to implement physical activity in primary and secondary prevention: a statement for healthcare professionals from the Task Force on Risk Reduction, American Heart Association. Circulation. 96(1): 355-357.
- Boutcher SH and Stein P. (1995). Association between heart rate variability and training response in sedentary middle-aged men. Eur J Appl Physiol Occup Physiol. 70(1): 75-80.
- Lauer MS. (2009). Autonomic function and prognosis. Cleve Clin J Med. 76(Suppl 2): S18-S22.
- Cole CR, Foody JM, Blackstone EH and Lauer MS. (2000). Heart rate recovery after submaximal exercise testing as predictor of mortality in a cardiovascularly healthy cohort. Ann Intern Med. 132(7): 552-555.
- Danieli A, Lusa L, Potocnik N, Meglic B, et al. (2014). Resting heart rate variability and heart rate recovery after submaximal exercise. Clin Auton Res. 24: 53-61.
- Tanaka H, DeSouza CA, Jones PP, Stevenson ET, et al. (1997). Greater rate of decline in maximal aerobic capacity with age in physically active vs. sedentary healthy women. J Appl Physiol; 83(6): 1947-1953.
- Howley ET, Bassett DR and Welch HG. (1995). Criteria for maximal oxygen uptake: review and commentary. Med Sci Sports Exerc. 27(9): 1292-1301.
- Sugawara J, Murakami H, Maeda S, Kuno S, et al. (2001). Change in post-exercise vagal reactivation with exercise training and detraining in young men. Eur J Appl Physiol. 85(3-4): 259-263.
- Javorka M, Zila I, Balharek T and Javorka K. (2002). Heart rate recovery after exercise: relations to heart rate variability and complexity. Braz J Med Biol Res. 35(8): 991- 1000.
- Gibbons RJ, Balady GJ, Beasley JW, Bricker JT, et al. (1997). ACC/AHA guidelines for exercise testing: a report of the American College of Cardiology/ American Heart Association Task Force on Practice Guidelines (Committee on Exercise Testing). J Am Coll Cardiol. 30(1): 260-315.
- Tanaka H, Monahan KD and Seals DR. (2001). Age-predicted maximal heart rate revisited. J. Ame. College of Cardiology. 37(1): 153-156.
- Firth D. (1988). Multiplicative errors: log-normal or gamma? J. R. Statist. Soc. B. 50(2): 266-268.
- Das RN and Lee Y. (2009). Log normal versus gamma models for analyzing data from quality-improvement experiments. Qual. Engineering. 21(1):79-87.
- Nelder JA and Lee Y. (1991). Generalized linear models for the analysis of Taguchi-type experiments. Applied Stochastic Models and Data Analysis. 7: 107-120.
- Krivokapich J, Child JS, Walter DO and Garfinkel A. (1999). Prognostic value of dobutamine stress echocardiography in predicting cardiac events in patients with known or suspected coronary artery disease. J Am Coll Cardiol. 33(3): 708-716.
- Das RN. (2016). Hypertension risk factors who underwent dobutamine stress echocardiography. Interv Cardiol. 8(1): 595-605.
- Das RN. (2017). The mean heart rate associations of the DSE data. Interv Cardiol J. 3(1): 1-3.
- Marcovitz PA and Armstrong WF. (1992). Accuracy of dobutamine stress echocardiography in detecting coronary artery disease. Am J Cardiol. 69(16): 1269-1273.
- Davila-Roman VG, Waggoner AD, Sicard GA, Geltman EM, et al. (1993). Dobutamine stress echocardiography predicts surgical outcome in patients with an aortic aneurysm and peripheral vascular disease. J Am Coll Cardiol. 21(4): 957-963.
- Secknus MA and Marwick TH. (1997). Evolution of dobutamine echocardiography protocols and indications: safety and side effects in 3,011 studies over 5 years. J Am Coll Cardiol. 29(6): 1234-1240.
- Lee Y, Nelder JA and Pawitan Y. (2006). Generalized Linear Models with Random Effects (Unified Analysis via H-likelihood). Chapman & Hall, London.
- Hastie T, Tibshirani R and Friedman J. (2001). The Elements of Statistical Learning. Springer-Verlag, NewYork.
- Das RN. (2014). Robust Response Surfaces, Regression, and Positive Data Analyses. Chapman & Hall, London.