Information Links
Related Conferences
Previous Issues Volume 5, Issue 1 - 2020
Delayed Appearance of Klein-Levin Syndrome Caused by Traumatic Brain Injury: A Case Report
Ali Javadpour1, Shekoofeh Yaghmaei2*, Nejla Mostafeenezhad1, Nima Derakhshan3, Peter Schofield4
1Department of psychiatry, Shiraz University of Medical Sciences, Shiraz, Iran
2Shiraz University of Medical Sciences, Shiraz, Iran
3Department of neurosurgery, Shiraz University of Medical Sciences, Shiraz, Iran
4The University of Newcastle, Australia *Corresponding author: Shekoofeh Yaghmaei, Medical Doctor, Shiraz University of Medical Sciences, Namazi Hospital, Namazi Square, Shiraz, Iran, Postal Code: 71937–11351, Tel/Fax: +98–713–6474259; E-mail: [email protected]
Received Date: July 28, 2020 Publication Date: October 20, 2020 Copyright: Yaghmaei S, et al. ©2020 Citation: Javadpour A. (2020). Delayed appearance of Klein-Levin Syndrome caused by traumatic brain injury: A case report. Mathews J Case Rep. (5)1:63.
ABSTRACT
Background: The combination of hypersomnia and any of: hyper-phagia, hyper-sexuality, or mood changes is known as Klein-Levin Syndrome. This syndrome is quite infrequent and mostly restricted to adolescent males.
Case Description: A young patient with KLS is presented in this report. He was presented with relapsing-remitting hypersomnia and irritability more than two years after incurring a traumatic brain injury. Magnetic resonance imaging (MRI) showed hyperintensity in the posterolateral part of the right temporal region, matched with the brain injuries caused by the old trauma. His extremely long sleep episodes forced medical team to provide him with some necessary basic cares such as nasogastric tube and urinary catheter. Five consecutive weeks of taking modafinil got his sleep pattern back to normal.
Conclusion: In addition to medical treatment which is inevitable for severe cases, family and social support is extremely vital for managing KLS patients.
KEYWORDS: Klein-Levin syndrome; Traumatic brain injury; Sleep disorder; Modafinil
ABBREVAIONS: KLS: Klein-Levin syndrome; TBI: Traumatic brain injury; GCS: Glasgow coma score; CT: Computed tomography; FAST: Sonography for trauma; MRS: Modified Rankin scale; MRI: Magnetic resonance imaging; SPECT: Single photon emission computed tomography; FDG-PET: Fluorodeoxyglucose- positron emission tomography.
BACKGROUND
Klein-Levin syndrome (KLS) is a rare sleep disorder mainly affecting adolescent males. The exact pathophysiology of this disease is still unknown. However, some studies have shown it is accompanied by hypo-perfusion in the hypothalamus, thalamus and caudate nucleus [1]. KLS is characterized by relapsing-remitting episodes of severe hypersomnia and at least one of the followings symptoms: hyper-phagia, hyper-sexuality, mood changes and cognitive impairment [2]. Some patients also experienced apathy, derealization, and irritability. Several triggering factors have been suspected. Some patients report a prior history of upper respiratory tract infection, acute bronchitis or bronchiolitis. Alcohol consumption, head trauma, and mental stress are also suggested as factors precipitating KLS [3]. Mild and moderate traumatic brain injuries (TBI) coupled with other factors, while in cases of severe TBI the prominent factor is severity of the injury [4]. Herein, we present a young man with KLS following a severe TBI and craniotomy.
CASE REPORT
On May 11th 2016 a 25–year-old male was involved in a car accident in Hormozgan province, in southern Iran and was brought to the nearest emergency trauma center. He was intubated few minutes after the crash by the emergency medical team and was transferred with a cervical collar to the emergency department in less than 20 minutes. According to the triage records, vital signs were stable with blood pressure of 135/85 mmHg, pulse rate of 108, respiratory rate of 18, and O2 Saturation of 98% (with 3 liters/minute oxygen through endotracheal tube). Upon arrival, he had Glasgow coma score (GCS) of 4/15, with bilateral midsized sluggish pupils and had multiple scalp lacerations which had been packed at the scene.
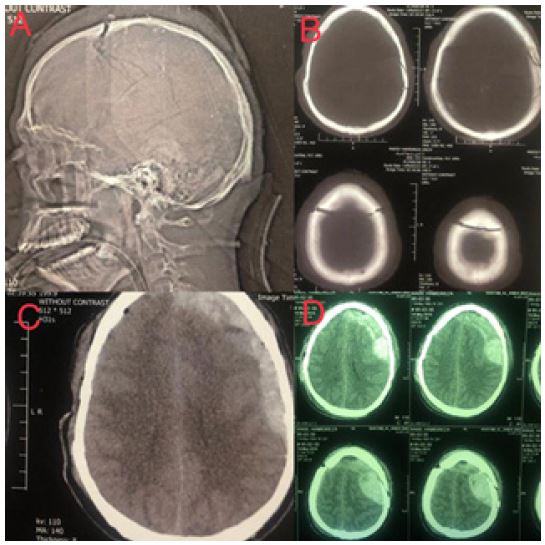
Figure 1: CT scan of brain in axial view.
Figure 2: MRI of brain with axial cut.
Brain computed tomography (CT) was performed which revealed a skull fracture with diastasis of the coronal suture bilaterally traversing the superior sagittal sinus, as well as bilateral epidural hematoma over cerebral convexities, with open ambient cisterns. Figure 1A, B and C. Focused assessment with Sonography for Trauma (FAST) and chest X-ray was unremarkable and after fluid resuscitation and blood sampling he was transferred to the intensive care unit. During the first five days, his GCS gradually improved to 7/15 but on the 5th day of admission his GCS suddenly dropped to 4/15 and he developed a fixed mydriasis of his left pupil. The brain CT scan revealed expansion of the epidural hematoma on the left side which required a prompt craniotomy to evacuate. Figure 1D Following surgery figure 2A, his GCS improved to 7/15 on the 7th day. On the 14th day of admission, he was weaned off from the mechanical ventilation and tracheostomy tube was inserted for him. He was transferred to the ward and, following a gradual improvement in his level of consciousness, he was discharged on the 24th day. Upon discharge, he had a Modified Rankin Scale (MRS) score of two without any neurologic deficits except for impairment of recent memory. During the next 31-months, his family reported normal social and personal functioning and he was able to carry out personal affairs as well as driving, going to a gym and socializing with family and his peers. However, he was not working.
In January 2019, after one week of anxiety, anorexia and restlessness, he experienced long and deep sleep episodes. The duration and depth of sleep episodes increased gradually up to the point that he was just forcefully awakened for eating (only 2 hours a day). No record of hyperphagia or hyper-sexuality was present in this period. He was admitted to his hometown hospital and a brain CT was performed. The CT was unremarkable apart from a hypodensity in the right temporal area and diffuse atrophy. Figure 2B there was no abnormal findings in his laboratory data, so he was discharged with a nasogastric tube and a urinary catheter. During the short awakening periods, he was speechless and did not establish eye contact.
Following a long episode of hypersomnia, he was transferred to our center for further evaluation. His neurological examination revealed: lack of orientation to time, place, and persons; flaccid motor response; normal deep tendon reflexes; no sensory pain response; and no pupil light response. His cerebrospinal fluid analysis was normal. Two 6-hour video electroencephalograms were compatible with normal sleep patterns. Brain MRI revealed the hyperintensity both in T2 and FLAIR sequences (Figure 2C and D) in the posterolateral part of the right temporal region, compatible with old ischemia.
His longest sleep episode lasted for 45 consecutive days and at this point, treatment with Modafinil was started at a dose of 100 mg/day for four days and 400 mg/day afterwards. After seven days of taking Modafinil, he woke up for a few minutes and, awakens time increased gradually. After five weeks, he had returned to a normal sleep pattern and normal daily function, although he continued to show evidence of impairment of recent memory. His sleepiness tended to recur when tapering of modafinil was attempted.
With the clinical diagnosis of post-traumatic KLS, he was discharged and prescribed Modafinil (200mg twice daily) to be taken indefinitely.
DISCUSSION
KLS is a combined neurological-psychiatric disorder. Depending upon the predominant symptoms, KLS cases may be referred either to neurologists or psychiatrists. This complex syndrome has diverse etiologies. Because KLS is a rare condition, it is important to have an index of suspicion for it in cases presenting with episodic hypersomnia after ruling out other differentials [5]. And, for such rare disorders, it is crucial to report cases whenever possible to help physicians keep them in mind.
Trauma is not considered as a precipitating event in the majority of KLS patients; however, there is a TBI-driven KLS form known as Posttraumatic KLS which counts for 9% of all KLS cases [6]. Therefore, in the appropriate clinical context a history of a head injury should be assiduously thought. The present case fulfils the criteria for KLS because of the periodic hypersomnia and irritability and, because he experienced a severe TBI before his first episode of hypersomnia, it seems probable that TBI was causally implicated in the emergence of symptoms.
Based on the literature, lesions of different parts of brain maybe associated with the development of KLS [7]. Frontotemporal and hypothalamus lesions have repeatedly been reported in KLS cases [8,9] reflecting the importance of hypothalamus in the regulation of sleep and eating. Frontotemporal hypoperfusion has been observed in some KLS cases investigated by a Single Photon Emission Computed Tomography (SPECT) study during their asymptomatic period or after remission [10]. By contrast, in a study of asymptomatic KLS patients using fluoro deoxy glucose- positron emission tomography (FDG-PET), Dauvilliers Y, et al. showed them to have frontotemporal cortex hypermetabolism compared to healthy control cases [11]. All abovementioned neuroimaging methods disclosed functional abnormalities in frontotemporal regions when patients were symptomatic [12]. Likewise, right temporal lesion was observed in the presented case.
Amnesia was a symptom of our patient and has been reported in other KLS cases in the literature. Temporal lobe dysfunction can cause amnesia, which may be the mechanism when it occurs as part of KLS. The short term memory dysfunction proceeded the onset of hypersomnic attacks in our case similar to what has been reported by Landtblom AM, et al. [13] in other KLS cases.
For most of the cases of TBI-associated KLS reported in the literature, the KLS symptoms, especially hypersomnia, are evident soon after the TBI and the delay between the trauma and onset of symptoms is uncommon [4]. In our case, about 32 months elapsed between TBI and the onset of the KLS symptoms. This unusual aspect of the history makes this case special, prompting us to submit this case report, as we wish to highlight this particular issue, i.e. that KLS may potentially arise as a consequence of trauma that has happened in distant past. Such a possibility should be considered in adopting therapeutic strategies when a patient is presented with signs like hypersomnia.
Among the recommended medications for KLS, none of them has been reported to be fully effective. The relative rarity of the syndrome presumably accounts for a lack of systematic studies to address this need. Unless the episodes are particularly severe, the best approach may be supportive treatment alone.
It is important to educate family members in how they may help the patient. The patient should have a safe and comfortable environment at home to rest. School and work activities should be postponed or adjusted until the symptoms resolve. The patient should not be allowed to wander unattended, drive a car, or operate heavy machinery. Symptoms of depression or anxiety should be monitored closely and treated if present.
A variety of different antidepressive and antiepileptic medications as well as mood stabilizers have been tried in patients with KLS. Lithium has been effective in several reports with a decrease in the frequency of relapses and duration of the episodes. However, the main drawback of Lithium is its associated side effects such as polydipsia, diarrhea, and subclinical hypothyroidism observed in more than half of 71 KLS patients receiving it [14]. This high prevalence of Lithium side-effects represents a major limitation in its use for KLS patients.
Arnulf I, et al. studied the results of three different treatments strategies in 108 KLS patients and obtained the following results [2]. Mood stabilizers were not effective but slight benefits were observed for some patients treated with carbamazepine, benzodiazepines, and other antiepileptics; Modafinil, methylphenidate, and amphetamine led to increased levels of alertness; after being treated with [6]. Moreover, Modafinil has significantly decreased the duration of symptomatic periods in KLS cases but it was not effective in reduction the frequency of relapses [15].
CONCLUSION
Sleep disorders following TBI are quite common. Traumatic brain injury should be in consideration as the risk factor for a rare sleep disorder entitled KLS. The main characteristics of KLS are hypersomnia coupled with cognitive or mood changes. Hyperphagia and hypersexuality are also presented in many cases.
Treating KLS has two main aspects,
- Psychological: In all cases it is necessary to provide the patient with a firm family and social support as well as a safe environment. Social activities should be postponed if needed.
- Psychiatric medication: In severe cases, such as the case presented, medical treatment is necessary to go along with family support. Modafinil is proved to be useful in treating severe KLS. Additionally, the adverse effects of Modafinil are easily tolerable. Receiving modafinil, in a few days, starts to take effect by suppressing the main problem of KLS patients i.e. decreases the duration of sleep periods.
DECLARATION OF INTEREST
All authors have none to declare.
ACKNOWLEDGEMENT
All authors have read and approved the manuscript and each author believes it represents honest work.
REFERENCES
- Usuda M, Kodaira M, Ogawa Y, Kanbayashi T, Yanagisawa S, et al. (2018). Fluctuating CSF hypocretin-1 levels in mild brain trauma-induced Kleine-Levin syndrome. Journal of the Neurological Sciences. 391:10-1.
- Arnulf I, Rico TJ, Mignot E. (2012). Diagnosis, disease course, and management of patients with Kleine-Levin syndrome. The Lancet Neurology. 11(10):918-28.
- Afolabi-Brown O, Mason TBA. (2018). Kleine-Levin Syndrome. Paediatric Respiratory Reviews. 25:9-13.
- Billiard M, Podesta C. (2013). Recurrent hypersomnia following traumatic brain injury. Sleep Medicine. 14(5):462-5.
- Mudgal S, Jiloha R, Kandpal M, Das A. (2014). Sleeping beauty: Kleine–Levin syndrome. Indian journal of psychiatry. 56(3):298.
- Shi Y-t, Tang B-s, Jiang H. (2013). Kleine–Levin syndrome with brain atrophy. Journal of Clinical Neuroscience. 20(7):1027-8.
- Papacostas S, Hadjivasilis V. (2000). The Kleine-Levin syndrome. Report of a case and review of the literature. European psychiatry. 15(4):231-5.
- Chiu HF, Li SW, Lee S. (1989). Kleine-Levin syndrome 15 years later. Australian and New Zealand journal of psychiatry. 23(3):425-7.
- Kostic V, Stefanova E, Svetel M, Kozic D. (1998). A variant of the Kleine-Levin syndrome following head trauma. Behavioural neurology. 11(2):105-8.
- Vigren P, Engström M, Landtblom A-M. (2014). SPECT in the Kleine–Levin syndrome, a possible diagnostic and prognostic aid? Frontiers in neurology. 5:178.
- Dauvilliers Y, Bayard S, Lopez R, Comte F, Zanca M, et al. (2014). Widespread hypermetabolism in symptomatic and asymptomatic episodes in Kleine-Levin syndrome. PLoS One. 9(4).
- Engström M, Latini F, Landtblom A-M. (2018). Neuroimaging in the Kleine-Levin syndrome. Current neurology and neuroscience reports. 18(9):58.
- Landtblom AM, Dige N, Schwerdt K, Säfström P, Granerus G. (2003). Short‐term memory dysfunction in Kleine–Levin syndrome. Acta Neurologica Scandinavica.108(5):363-7.
- Leu-Semenescu S, Le Corvec T, Groos E, Lavault S, Golmard J-L, et al. (2015). Lithium therapy in Kleine-Levin syndrome: an open-label, controlled study in 130 patients. Neurology. 85(19):1655-62.
- Huang Y-S, Lakkis C, Guilleminault C. (2010). Kleine-Levin syndrome: current status. The Medical clinics of North America. 94(3):557-62.
.JPG)