Information Links
Related Conferences
Previous Issues Volume 11, Issue 1 - 2026
Cytokine-induced Neurogenesis for the Treatment of IVF-induced ASD
Takuji Shirasawa1,2,*, Luis Carlos Aguilar Cobos3
1Ochanomizu Health and Longevity Clinic, Tokyo 101-0062, Japan
2Shirasawa Anti-Aging Medical Institute, Tokyo 101-0062, Japan
3Livant Neurorecovery Center, Guadalajara, Mexico
*Corresponding Author: Takuji Shirasawa, Ochanomizu Health and Longevity Clinic, Tokyo 101-0062, Japan & Shirasawa Anti-Aging Medical Institute, Tokyo 101-0062, Japan, Email: [email protected]
Received Date: March 11, 2026
Published Date: March 25, 2026
Citation: Shirasawa T, et al. (2026). Cytokine-induced Neurogenesis for the Treatment of IVF-induced ASD. Mathews J Case Rep. 11(1):219.
Copyrights: Shirasawa T, et al. © (2026).
ABSTRACT
In this case report, three cases of autism spectrum disorder (ASD) with a potential etiology of in vitro fertilization were presented. To the best of our knowledge, this is the first report suggesting that epigenetic gene modifications due to in vitro fertilization play an important role in the pathology of ASD. Furthermore, we demonstrated that cytokine-induced neurogenesis can improve ASD symptoms in all three cases presented in this paper. This innovative therapeutic approach for treating ASD has the potential to be extrapolated to other forms of ASD with different etiologies. An imbalance between GABAergic inhibitory neurons and glutamatergic excitatory neurons plays an important role in the etiology of ASD. This imbalance has been linked to the emergence of ASD symptoms, and the alleviation of these symptoms has been associated with cytokine-induced neurogenesis. Written informed consent was obtained from the patients.
Keywords: IVF, Epigenesis, Cytokine, Neurogenesis, ASD.
INTRODUCTION
Autism spectrum disorder (ASD) is a complex neurodevelopmental disorder that affects an individual’s capacity for social interaction and is associated with specific patterns of abnormal behavior and development [1]. A multitude of biological factors have been observed to be associated with the onset of the disorder [2]. Recent studies have indicated a correlation between ASD and aberrant neural mechanisms, such as issues with synaptic plasticity and communication, as well as genetic mutations [3]. Previous studies have indicated that, in addition to genetic variations, individuals diagnosed with ASD exhibit structural and functional differences in various regions of the brain, including the amygdala, prefrontal cortex, and cerebellum [4]. Comorbidities demonstrate additional diagnostic and therapeutic challenges, thus necessitating a holistic evaluation and treatment plan [5].
At present, all available treatments for ASD fall into three categories: behavioral, pharmacological, and new and innovative approaches. Despite the plethora of approaches that have been attempted, the most prevalent type of treatment involves some form of applied behavior analysis (ABA), which is predicated on the principles of behaviorism [6,7]. Different pharmacological treatments, such as the use of selective serotonin reuptake inhibitors and atypical antipsychotics, are available for the treatment of the core symptoms of ASD [1]. Recent advancements in this field have led to the development of novel interventions, including research endeavors focused on the utilization of neuromodulation and digital therapeutics as complementary modalities to existing therapeutic modalities [8,9]. A plethora of studies have demonstrated the necessity of customizing intervention strategies to suit the unique characteristics of individuals with ASD [1]. This necessity arises from the observation that individuals with ASD exhibit a considerable degree of heterogeneity in their symptoms and profiles [2]. Research endeavors aimed at acquiring further knowledge concerning the genetic and epigenetic factors underlying this disorder are imperative. This enhanced understanding will facilitate the development of novel and precise medications for this condition.
In this case report, we described three cases of ASD with a potential etiology of in vitro fertilization. To the best of our knowledge, this is the first report suggesting that epigenetic gene modifications due to in vitro fertilization play an important role in the pathology of ASD. Furthermore, we demonstrated that cytokine-induced neurogenesis can improve ASD symptoms in all three cases presented in this paper. This innovative therapeutic approach for treating ASD has the potential to be extrapolated to other forms of ASD with different etiologies. In this discussion, the central theme involves the significance of an imbalance between GABAergic inhibitory neurons and glutamatergic excitatory neurons in the pathogenesis of ASD. This imbalance is considered to be a key factor in the development of ASD symptoms, with the resolution of these symptoms being linked to cytokine-induced neurogenesis.
METHODS
The cytokine and exosome cocktail formulation used in this study was designed and developed by Luis Carlos Aguilar Cobos at the Livant Neurorecovery Center, Mexico, as previously described [10-16]. In this case study, 8 cytokine and exosome formulations were used. Our group has focused specific attention on the cytokines that can facilitate the neurogenesis of GABAergic neurons and glutamatergic neurons. Another cytokine (such as that involved in OST and Muss) includes progranulin, which facilitates neurogenesis in the frontal and temporal cortex. Exosomes derived from young adult porcine brains with a predominance of miR-124 are also included in Neurogen EP. A cytokine cocktail containing HPLC-purified GDNF, omega 3 and exosomes extracted from Salmon was also used for Renotrof (SAL)23 EM. A cytokine cocktail was sublingually administered 3 times per day.
CASE DESCRIPTION
Case 1
On March 8, 2019, a 3-year-old male patient was referred to the Ochanomizu Health & Longevity Clinic and presented with the primary complaint of communication difficulties in his nursery school and speech impediments, including limited vocabulary. The patient was born through in vitro fertilization with C-section due to prematurity. The patient exhibited developmental delays in both speech and communication. The patient was subsequently diagnosed with autism spectrum disorder (ASD) at a local hospital. A review of the patient’s family medical history revealed a history of frontotemporal dementia (FTD) in the patient’s paternal grandmother. The neurological examination revealed no abnormalities in either motor or sensory functions.
A subsequent electroencephalography (EEG) examination performed on March 8, 2019, revealed the presence of abnormal slow waves at the frontal, temporal, and parietal leads at rest (data not shown). P300 electroencephalogram (EEG) data were analyzed by using Neuroscan Software (https://compumedicsneuroscan.com/). The results of the study indicated that after utilization of the target stimulus in the form of a high-pitched sound, hyperexcitable and asymmetric P300 responses were detected at the frontopolar, frontal, temporal, parietal, and occipital leads (Figure 1A, red lines). As illustrated in Figure 1B, the magnified recordings from the left frontopolar lead demonstrated a P300 EEG response with a peak voltage of 400 µV2 at 250 msec and a peak voltage of 600 µV2 at 950 msec (Figure 1B, left panel, red lines). The right panel of Figure 1B illustrates analogous hyperexcitable P300 EEG responses in the right frontopolar lead (Figure 1B, right panel, red lines). The magnification of the recordings from the parietal leads also revealed biphasic or triphasic hyperexcitable P300 EEG responses in the right and left parietal lobes (Figure 1C). Visual evoked potential (VEP) analysis revealed advanced dissociation between the right and left frontal leads (F7 and F8; Figure 1D, left panel). These findings suggest the presence of disorganized neuronal circuits within the cerebral cortex of the frontal lobe. Additionally, coherence analysis of the P300 response revealed low fluctuations in the left frontal, central, and left central leads (Figure 1E, left panel). These findings indicate the underdeveloped state of the neuronal network in the left frontal lobe.
The induction of cortical neurogenesis was achieved through the therapeutic application of cytokines, including Gabatrof (EPI), Gabatrof (ADPK), Epatrof (H-23 AZ D5), and OST treatments, which occurred from March 8, 2019, to August 24, 2020, as outlined in the Materials section. The communication problem experienced a substantial improvement with the augmentation of the vocabulary.
A subsequent re-evaluation of the patient’s electroencephalogram (EEG) on August 24, 2020, revealed a substantial decrease in slow wave activity (data not shown). The hyperexcitable P300 EEG response evoked by a target stimulus of high-pitched sound was significantly normalized in all of the leads (Figures 1A, 1B, 1C; black lines). These findings indicate that inhibitory GABAergic interneurons may have regenerated to suppress the hyperexcitability of the neuronal network of the cerebral cortex. The results of the flash visual evoked potential (VEP) analysis demonstrated a substantial improvement in the dissociated evoked potentials between the right and left frontolateral leads on August 24, 2020, after treatment (Figure 1D, right panel). These data imply that cortical neuronal networks were regenerated with functionally relevant synaptic functions. Coherence analysis of P300 signals recorded on August 24, 2020, revealed improvements in neural circuit connectivity at the left frontal and central leads after treatment (Figure 1E, right panel indicated by red arrows). As of the present date (February 2026), the patient is 10 years of age and is undergoing a maintenance cytokine protocol resulting in normal social communication skills.
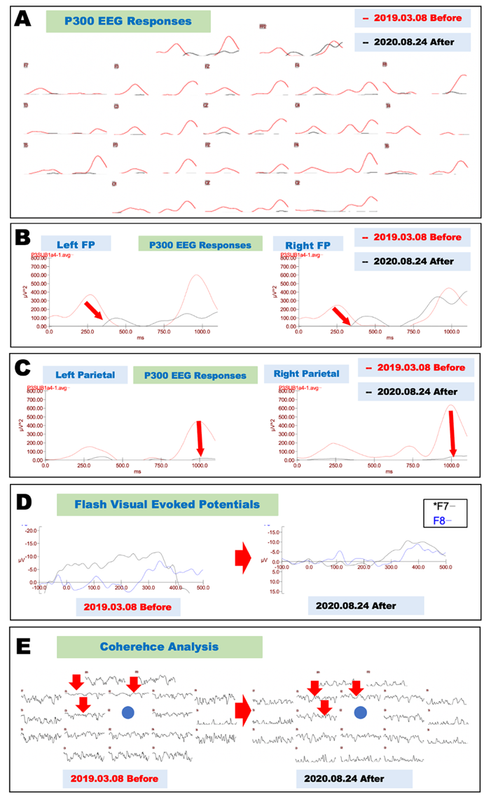
Figure 1. Neurophysiological evaluation before and after cytokine treatment (Case 1).
A. Electrophysiological evaluation of P300 EEG responses before and after cytokine-induced neurogenesis. P300 EEG responses recorded on March 8, 2019, before treatment are shown as red lines, and those recorded on August 24, 2020, after treatment are shown as black lines.
B. Magnified recordings of the frontopolar leads shown in Fig. 1A reveal that the hyperexcited premature P300 components recorded on March 8, 2019 (red lines), were significantly improved at both frontopolar (FP) leads recorded on August 24, 2020 (black lines).
C. Magnified recordings of the parietal leads shown in Fig. 1A reveal that the hyperexcited P300 components recorded on March 8, 2019 (red lines), were significantly improved at both parietal leads recorded on August 24, 2020 (black lines).
D. Electrophysiological records of flash visual evoked potentials before and after cytokine treatment. Asymmetrical potentials were recorded on March 8, 2019, between F7 (left frontolateral lead, black line) and F8 (right frontolateral lead, blue line); these potentials were significantly improved on August 24, 2020 (right panel).
E. Coherence analysis of P300 before and after cytokine-induced neurogenesis. Impaired neural network connections recorded on March 8, 2019 (left panel), in the frontal electrodes (F3, FZ, and C3, as shown by red arrows) were significantly improved on August 24, 2020 (right panel).
Case 2
An 8-year-old male patient was referred to the Ochanomizu Health & Longevity Clinic due to his difficulty in communicating with others and in controlling his emotions. The patient was born through in vitro fertilization with a normal delivery. The patient does not cooperate at home or at school. The patient was then diagnosed with ASD at a local hospital. A review of the patient’s family medical history revealed no neurological diseases. The neurological examination yielded no findings that would suggest any issues with motor or sensory functions.
On December 26, 2022, an electroencephalogram (EEG) showed abnormal slow wave activities in the frontal and temporal leads at rest (data not shown). The P300 EEG data recorded on December 26, 2022, revealed asymmetric low-voltage P300 EEG responses in the frontopolar, frontal, temporal, parietal, and occipital regions after stimulation with high-pitched sounds (Figure 2A, red lines). As shown in Figure 2B, P300 responses in the left frontopolar lead demonstrated a peak voltage of 10 µV2 at 300 msec and a peak voltage of 20 µV2 at 900 msec (Figure 2B, left panel, red lines). The right panel shows similar asymmetric biphasic P300 responses in the right frontopolar lead (Figure 2B, right panel, red lines). Electrophysiological recordings during the visual spatial memory test on December 26, 2022, revealed hyperexcited responses in both the frontopolar and frontolateral leads (Figure 2C, red lines) compared with control responses (blue lines). Coherence analysis of the P300 response revealed asymmetric fluctuations in the frontopolar leads (Figure 2D). These findings suggest that the neural network in the frontal lobe is underdeveloped.
The induction of neurogenesis was achieved through the therapeutic application of cytokines, including OST EM, OST, Neurogen EP, CAL, Gabatrof (ADPK), Muss EM, and Renotrof (SAL) 23 EM applications. This cytokine treatment was administered from December 26, 2022, to June 24, 2023, as outlined in the Materials section. After cytokine treatment, the ability of the patient to communicate and work together with others significantly improved both at home and at school.
A subsequent re-evaluation of the patient’s electroencephalogram (EEG) on June 24, 2023, revealed a substantial decrease in slow wave activity (data not shown). The asymmetric P300 EEG responses evoked by a target stimulus of high-pitched sound were significantly improved in all of the leads (Figure 2A, black lines). High-voltage symmetric P300 responses with a peak voltage of 90 µV2 at 750 msec in both frontopolar leads are shown in Figure 2B. These findings suggest that excitatory glutamatergic pyramidal neurons were regenerated. These neurons were able to functionally respond to the high-pitched sound stimulus. Moreover, the results of the visual spatial memory test revealed that hyperexcited responses in both the frontopolar and frontolateral regions (Figure 2C, red lines) were improved and were observed to be closer to the responses of the controls (blue lines) on June 24, 2023, after treatment (Figure 2C, black lines and blue lines). These findings indicate that the inhibitory GABAergic networks had undergone regeneration, thereby resulting in substantial suppression of the hyperexcitable responses that had been observed prior to treatment during the spatial memory test. A subsequent coherence analysis of the P300 electroencephalogram (EEG) responses recorded on June 24, 2023, revealed enhanced neuronal connections between the frontopolar leads and the central parietal lead (Figure 2D, indicated by red arrows). As of the present date (February 2026), the patient is 10 years of age and is undergoing a maintenance of the cytokine protocol, with normal social communication skills and stable emotional control being reported. Although the patient had previously demonstrated a dislike of sporting activities, he had recently joined the baseball club and is currently enthusiastically enjoying baseball.
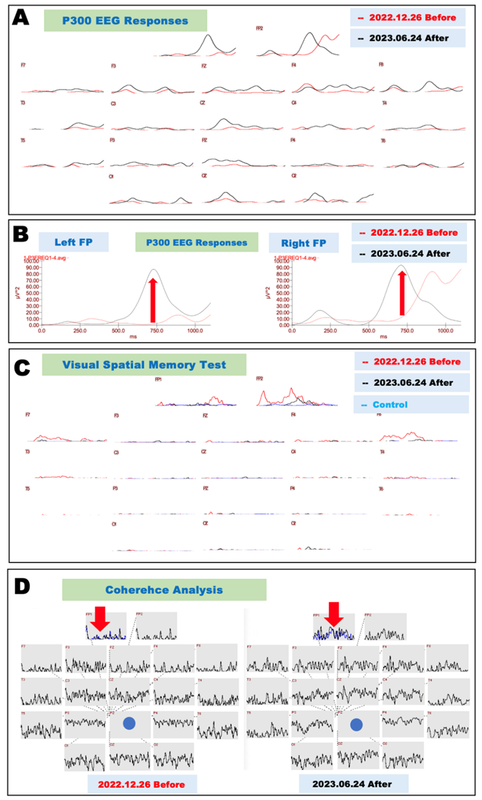
Figure 2. Neurophysiological evaluation before and after cytokine treatment (Case 2).
A. Electrophysiological evaluation of P300 EEG responses before and after cytokine-induced neurogenesis. P300 EEG responses recorded on December 26, 2022, before treatment are shown as red lines, and those recorded on June 24, 2023, after treatment are shown as black lines.
B. Magnified recordings of the frontopolar leads in Fig. 2A show that the underdeveloped P300 components recorded on December 26, 2022 (red lines), were significantly improved at both frontopolar (FP) leads recorded on June 24, 2023 (black lines).
C. Electrophysiological records of visual space memory tests before and after cytokine treatment. Hyperexcitable potentials were recorded in both frontopolar and frontolateral leads on December 26, 2022; these potentials were significantly improved on June 24, 2023 (right panel). Control potentials are drawn as blue lines.
D. Coherence analysis of P300 before and after cytokine-induced neurogenesis. Impaired neural network connections recorded on December 26, 2022 (left panel), in frontopolar electrodes (FP1, as shown by red arrows) were significantly improved on June 24, 2023 (right panel). The FP1 recording (black line) was overlaid with the FP2 lead (blue line) (as shown by red arrows).
Case 3
An 11-year-old boy visited the Ochanomizu Health & Longevity Clinic on December 26, 2022, with chief complaints of communication problems, fatigue, disinterest, and sleep disturbances. The patient was born by in vitro fertilization with a normal delivery. At times, the patient was reported as lacking cooperation both at home and at school. The patient is a cousin of the patient described in Case 2. The neurological examination revealed no abnormalities in motor or sensory functions.
Electroencephalography (EEG) performed on December 26, 2022, revealed abnormal slow waves in the frontal and temporal leads at rest (data not shown). P300 EEG data analyzed via Neuroscan software (https://compumedicsneuroscan.com/) revealed asymmetric P300 EEG responses in the frontopolar, frontal, and temporal leads after a high-pitched target stimulus (Figure 3A, red lines). Magnification of the recordings from the left frontal lead in Figure 3A shows triphasic P300 responses with a peak voltage of 20 µV2 at 500 msec in the left frontopolar lead (Figure 3B, left panel, red lines). The right panel shows similar asymmetric triphasic P300 EEG responses in the right frontal lead (Figure 3B, right panel, red lines). Magnification of the recordings from the temporal leads in Figure 3C shows asymmetric P300 EEG responses with a single peak in the left temporal lead (Figure 3C, left panel, red lines) and triphasic peaks in the right temporal lead (Figure 3C, right panel, red lines). As demonstrated in Figure 3D, an analysis of the patient’s emotional response indicated a relative absence of emotional response regarding the evaluated emotions, including joy, sadness, happiness, neutral feelings, and anxiety, as measured from the left frontopolar lead (Figure 3D, left panel).
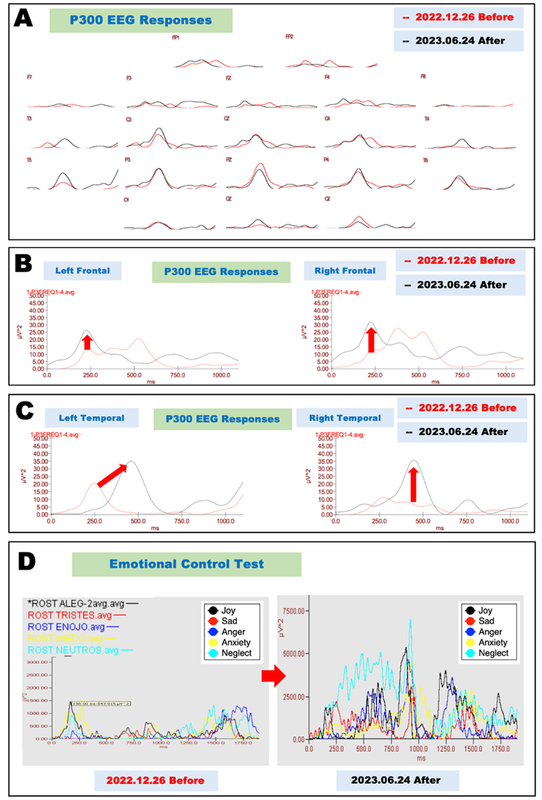
Figure 3. Neurophysiological evaluation before and after cytokine treatment (Case 3).
A. Electrophysiological evaluation of P300 EEG responses before and after cytokine-induced neurogenesis. P300 EEG responses recorded on December 25, 2022, before treatment are shown as red lines, and those recorded on June 24, 2023, after treatment are shown as black lines.
B. Magnified recordings of the frontal leads shown in Fig. 3A reveal that the irregular P300 components recorded on December 25, 2022 (red lines), were significantly improved at both frontopolar (FP) leads recorded on June 24, 2023 (black lines).
C. Magnified recordings of the temporal leads shown in Fig. 1A reveal that the asymmetrical P300 components recorded on December 25, 2022 (red lines), were significantly improved at the left temporal leads recorded on June 24, 2023 (black lines).
D. Electrophysiological evaluation of emotional control tests performed before and after cytokine-induced neurogenesis. The left frontopolar lead recorded on December 25, 2022 (left panel), showed low voltages for all emotions, such as joy (black), sadness (red), anger (blue), anxiety (yellow), and neglect (light blue). After treatment, all emotions were significantly upregulated (right panel).
Magnetic resonance imaging (MRI) data obtained on December 26, 2022, revealed an increase in the volume of the frontal and temporal lobes, as depicted in the 3D MR image from the left lateral view. Furthermore, a reduction in the lateral ventricle volume was evident, as demonstrated by the 2D MR images in the right panel (Figure 4A). This result suggests the possibility of an increased volume of white matter, as described in a previous publication [17]. However, in silico endoscopic images of the hippocampus revealed atrophy of the head part of the left hippocampus (Figure 4B, yellow arrow) and atrophy of the neck of the right hippocampus (Figure 4C, yellow arrows). These findings suggest that the limbic system may also be involved in the pathology of ASD, as described in a previous publication [18].
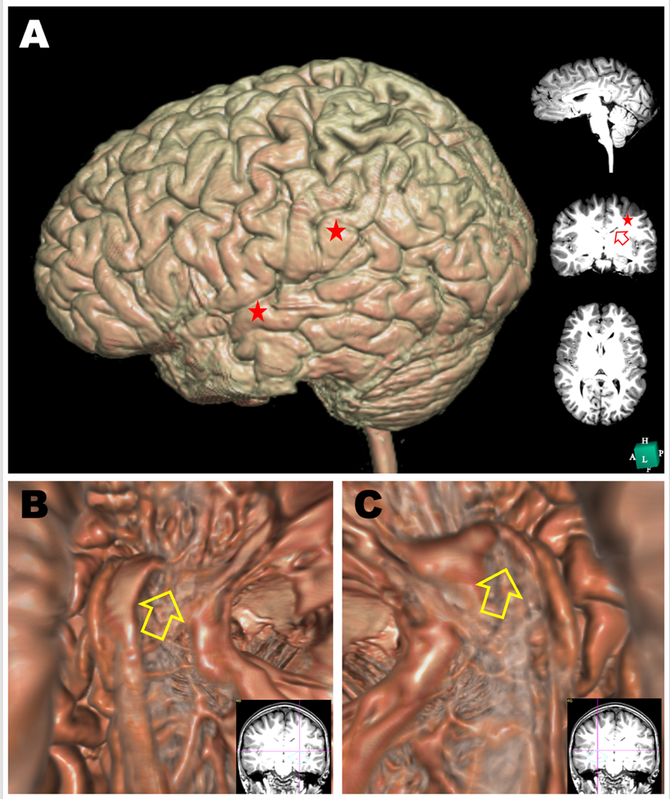
Figure 4. Morphological evaluation before cytokine treatment in Case 3.
A. An MRI scan was performed on December 25, 2022, before cytokine treatment. 3D structure of the brain reconstructed in silico (using Expert INTAGER software) from MRI T1-weighted images with 1-mm sagittal slices. The right panel shows conventional 2D T1-weighted images of sagittal (upper), coronal (middle), and horizontal (lower) sections.
B&C. Endoscopic in silico views of the left hippocampus (B) and right hippocampus (C). The head part of the left hippocampus and upper neck part of the right hippocampus were degenerated, as indicated by the yellow arrows.
To induce neurogenesis, we therapeutically applied the cytokines OST EM, OST, and Neurogen EP from December 26, 2022, to June 24, 2023, as described in the Materials section. The patient's ability to communicate with others and to control their emotions improved both at home and at school.
A subsequent re-evaluation of the patient’s EEG on June 24, 2023, revealed a substantial decrease in slow wave activity (data not shown). The asymmetrical P300 EEG responses evoked by a target stimulus of high-pitched sound were significantly improved in all of the leads (Figure 3A, black lines). Symmetric P300 EEG responses in the frontal leads are shown in Figure 3B. These responses exhibited a peak voltage of 30–35 µV2 at 250 msec at both frontal leads. These findings suggest that excitatory glutamatergic pyramidal neurons were functionally regenerated. Moreover, these neurons were able to respond in a physiologically relevant manner to the high-pitched sound stimulus. As shown in Figure 3C, the previously observed asymmetric P300 EEG responses became significantly improved to symmetrical P300 EEG responses after treatment on June 24, 2023. This change demonstrated a peak voltage of 35 µV2 at 450 msec at both temporal leads (see black lines). The emotional responses for joy, sadness, happiness, neutral feelings, and anxiety are shown in Figure 3D. All of these responses increased in a balanced manner (Figure 3D, right panel).
DISCUSSION
In the present case report, we demonstrated for the first time that cytokine-induced neurogenesis successfully regenerated the disorganized cortical neural networks of three patients with IVF-induced ASD and improved symptoms such as speech and social communication problems. The findings from the neurophysiological assessments in all three cases presented in this paper suggest an imbalance between inhibitory and excitatory neuronal networks after a high-pitched sound stimulus. This observation suggests the potential influence of the maturation of inhibitory GABAergic neurons and excitatory glutamatergic neurons on the manifestation of symptoms associated with ASD.
We have previously published studies focusing on the exploration of therapeutic applications of cytokine-induced neurogenesis in Alzheimer's disease [8,9,14], vascular dementia [10], and bipolar disorder [12]. In this study, we demonstrated that the same therapeutic strategy could be applied and was effective for the treatment of ASD. The induction of differentiation in somatic neuronal precursor cells residing in the cerebral cortex was facilitated by the use of cytokines and exosomes, as previously described [14]. The neurophysiological examination revealed that all three cases exhibited an imbalance between inhibitory and excitatory neuronal networks in the cerebral cortex. This finding suggests that the maturation of inhibitory GABAergic neurons, as well as excitatory glutamatergic neurons, may be the primary neuropathology underlying ASD, as suggested by large-scale exosome sequencing studies focusing on ASD [19].
We employed a combination of pharmaceutical agents, including Gabatrof (EPI), Gabatrof (ADPK), and Epatrof (H-23 AZ D5), to facilitate the regeneration of inhibitory GABAergic neurons, excitatory glutamatergic neurons, and cortical neurons, respectively. Additionally, we utilized OST, OST EM, Muss EM, and Neurogen EP to promote the regeneration of cortical neurons in general. In the second case, Renotrof (SAL) 23 EM was administered to facilitate the regeneration of dopaminergic neurons, thereby modulating emotional control.
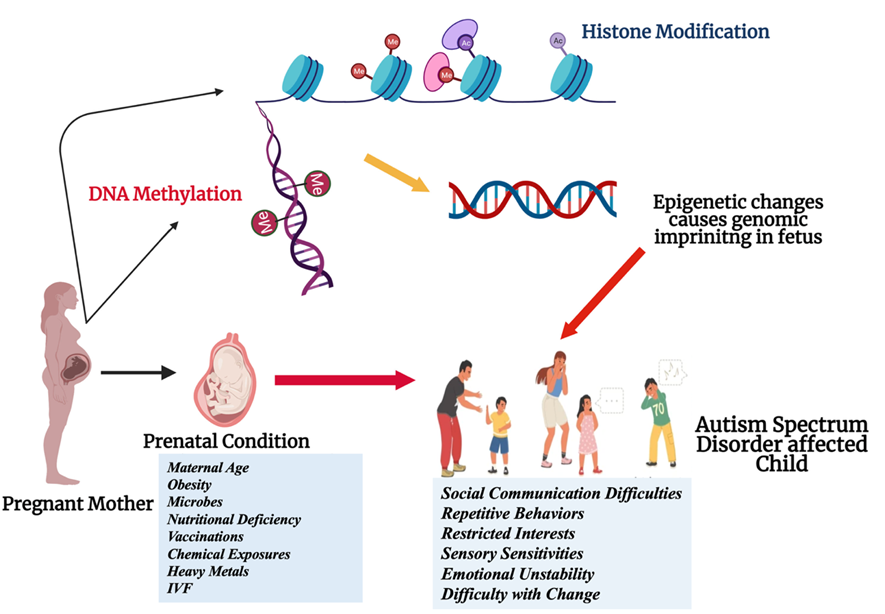
Figure 5. Prenatal conditions influencing the epigenetic control of genes responsible for neurodevelopmental disorders such as ASD.
As shown in Figure 5, the underlying mechanisms explaining the diverse manifestations of autism have begun to be elucidated based on the notion of epigenetics, which demonstrates how external factors can modulate the expression of specific genes. In individuals who are at risk for autism, their distinct epigenetic profiles are shaped by interactions between genes and life conditions. These profiles have been observed to exert a greater influence on outcomes than previously anticipated [20]. A variety of factors have been demonstrated to influence molecular pathways involved in brain development, including maternal stress levels, chemical constituents in the ambient environment or the subject's body, and dietary choices [21]. In the study “Genes and Environments in the Development of Different Forms of ASD”, Bhandari, Paliwal, and Kuhad (2020) examined the interplay between genetic factors and environmental influences in the manifestation of various forms of ASD [22]. Exposure to heavy metals during critical phases of development has the potential to disrupt epigenetic control, thereby influencing the growth and function of synapses and, consequently, cognitive processes [23]. In 2014, Hall and Kelley made an observation that has since received significant attention in the field [24]. Specifically, they noted the importance of incorporating epigenetic shifts into the exploration of genetics in autism research. These shifts could reveal previously unobserved risks, thereby offering novel insights into the etiology of autism [24]. Notably, Robert F. Kennedy Jr. (the United States Secretary of Health and Human Services) has prohibited the use of mercury in vaccines. Prenatal exposure to microbes or suboptimal nutrition has been demonstrated to disrupt gene regulation, thereby increasing the likelihood of developing autism [25]. The implementation of local changes also has the effect of obscuring straightforward genetic explanations for neurodevelopmental disorders [26].
In this study, all three patients were born through in vitro fertilization (IVF). A review of the extant research conducted by Lo and colleagues in 2022 indicated that offspring conceived through IVF may experience divergent neurodevelopmental challenges compared to those offspring derived from intracytoplasmic sperm injection (ICSI) procedures [27]. Contrary to expectations, studies have suggested that these shifts in risk are not uniform across different fertility treatments. A 2023 study revealed a unique correlation between infertility and an elevated risk of autism, thus suggesting the potential influence of underlying parental factors on the outcome [28]. As illustrated in Figure 6, a multitude of factors can influence epigenetic control during IVF procedures. Hyperovulation with hormones, in vitro culture, intracytoplasmic sperm injection, and the cryopreservation of oocytes in liquid nitrogen can result in alterations to epigenetic gene modifications (Figure 6). These factors may serve as etiological agents associated with the development of ASD.
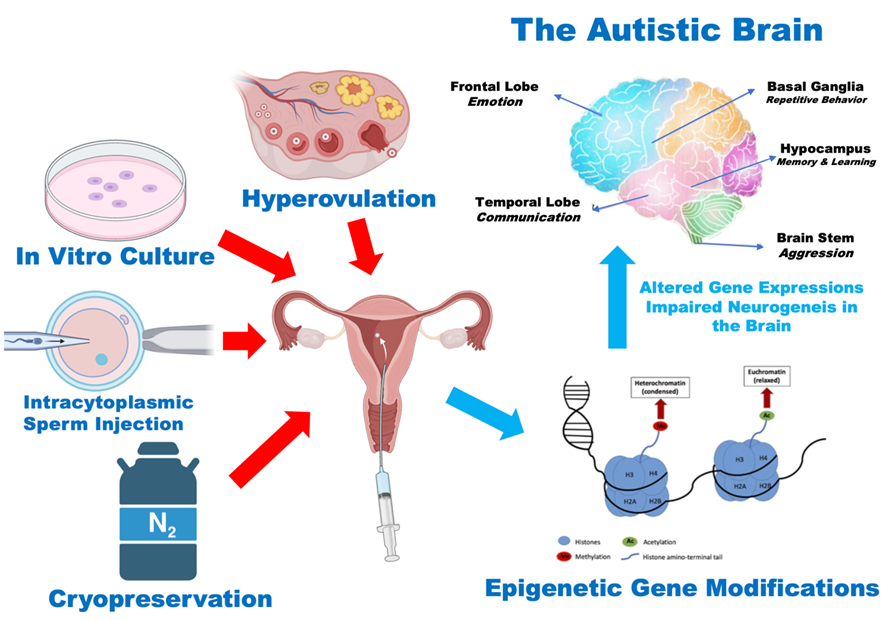
Figure 6. Factors involved in the in vitro fertilization procedure that can induce epigenetic gene modifications and alter gene expression, thereby causing impaired neurogenesis in ASD.
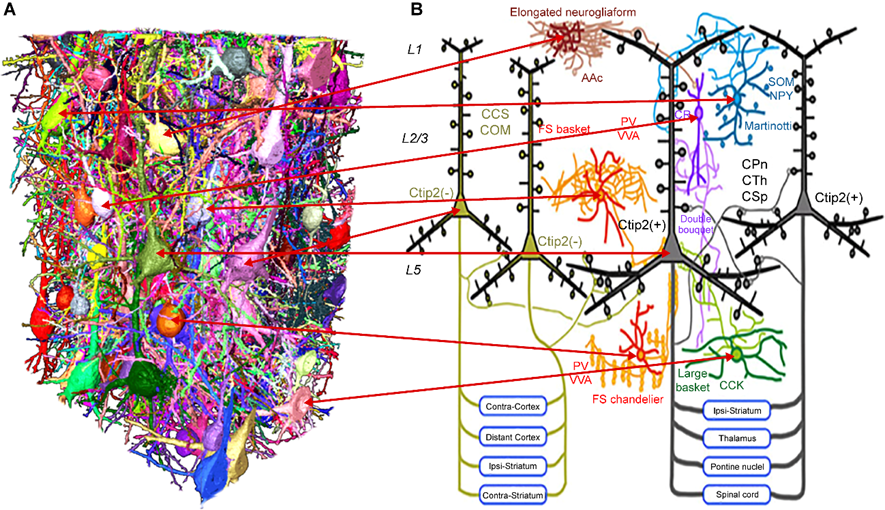
Figure 7.
A. 3D structure of neuronal networks in the cerebral cortex. Glutamatergic pyramidal neurons are densely surrounded by various types of interneurons in the human cerebral cortex. The figure is modified from MPNeuro (MAX PLNCK NEUROSCIENCE) (https://maxplanckneuroscience.org/silence-for-thought/).
B. The fundamental subtypes and interconnections of GABAergic cells and layer 5 pyramidal cells in the frontal cortex. Molecules expressed in GABAergic cells: Ac, alpha-actinin-2; CCK, cholecystokinin; CR, calretinin; NPY, neuropeptide Y; PV, parvalbumin; SOM, somatostatin; and VVA, binding with Vicia villosa. For the pyramidal cell groups, the following abbreviations were utilized for the considered cell types: CCS, crossed-corticostriatal cell; COM, commissural cell; CPn, corticopontine cell; CTh, corticothalamic cell; and CSp, corticospinal cell. The figure has been modified from the Division of Cerebral Circuitry (www.nips.ac.jp).
As illustrated in Figure 7, excitatory glutamatergic pyramidal neurons are surrounded by at least six types of GABAergic interneurons in the cerebral cortex, which include Chandelier cells, large basket cells, FS basket cells, double bouquet cells, Martinotti cells, and elongated neurogliaform cells (Figure 7). The inhibitory regulatory GABAergic network is a highly intricate system that receives a multitude of signals from dopaminergic neurons, cholinergic neurons, and serotonergic neurons [23,29,30]. A higher ratio of GABA to glutamate was observed in patients who demonstrated a more favorable response to bumetanide, which is a pharmaceutical agent known to influence GABA levels [31]. This finding suggests the potential for developing treatments that could modulate the balance between these two neurotransmitters. As demonstrated in this study, the patient in Case 1 exhibited hyperexcitability of glutamatergic neurons after the auditory stimulus, whereas patients in Cases 2 and 3 demonstrated a dysregulated balance between inhibitory GABAergic and excitatory glutamatergic neurons. The present study proposes the working hypothesis that cytokine-induced neurogenesis recapitulates natural brain development. This process is predicted to reconstruct complex inhibitory interneurons that regulate the physiologically relevant signals of pyramidal neurons in the cerebral cortex.
ASD is a multifactorial condition arising from a complex interplay among genetic, environmental, and neurobiological factors. Multiple genetic studies have repeatedly substantiated the multiplex etiology of ASD, thus underscoring the significance of genes implicated in immune and inflammatory responses [2]. In addition, recent studies have indicated that environmental factors (including nutritional deficiency, chemical exposure, and heavy metal exposure) can influence the manifestation of autistic behaviors in susceptible individuals (see Figure 5) [32]. The prognosis of ASD is contingent upon the etiology; however, the three cases presented in this paper demonstrate a favorable prognosis. In the initial two cases, the patients are undergoing maintenance cytokine therapy, which is a treatment that necessitates only a low dose of cytokines for maintenance. However, in the third case, the patient has been free of receiving both cytokines and medications. Given the established etiology of these cases (involving epigenetic gene modifications), the potential for a more favorable outcome in cases of cytokine-induced neurogenesis compared to those caused by gene mutations is a promising avenue for further research. We believe that the induction of neurogenesis by cytokines has the potential to ameliorate a range of neurological diseases, including neurodegenerative and neurodevelopmental disorders. This novel technology is poised to transform the field of regenerative medicine in the context of neurological treatment.
CONCLUSION
In this case report, three cases of ASD with a potential etiology of in vitro fertilization were discussed. To the best of our knowledge, this is the first report suggesting that epigenetic gene modifications due to in vitro fertilization play an important role in the pathology of ASD. Furthermore, we demonstrated that cytokine-induced neurogenesis can improve ASD symptoms in all three cases presented in this paper. This innovative therapeutic approach for treating ASD has the potential to be extrapolated to other types of ASD with different etiologies. An imbalance between GABAergic inhibitory neurons and glutamatergic excitatory neurons plays a pivotal role in the emergence of ASD symptoms, with the alleviation of ASD symptoms being associated with cytokine-induced neurogenesis.
ACKNOWLEDGEMENTS
The authors would like to thank Ms. Sayuri Sato and Ms. Fernanda Diaz for the preparation of this manuscript.
ETHICAL APPROVAL OF STUDIES AND INFORMED CONSENT
Written informed consent was obtained from the patients.
CONFLICT OF INTEREST
The authors have no conflicts of interest to disclose.
REFERENCES
- Hirota T, King BH. (2023). Autism Spectrum Disorder: A Review. JAMA. 329(2):157-168.
- Ayoub G. (2025). Autism Spectrum Disorder as a Multifactorial Disorder: The Interplay of Genetic Factors and Inflammation. Int J Mol Sci. 26(13):6483.
- Kuo HY, Liu FC. (2022). Pathophysiological Studies of Monoaminergic Neurotransmission Systems in Valproic Acid-Induced Model of Autism Spectrum Disorder. Biomedicines. 10(3):560.
- Frye RE, Rossignol DA. (2016). Identification and Treatment of Pathophysiological Comorbidities of Autism Spectrum Disorder to Achieve Optimal Outcomes. Clin Med Insights Pediatr. 10:43-56.
- Raza MH, Bhutta ME, Siddique MH. (2024). Shifting focus: the need for early intervention and safer alternatives in autism spectrum disorder treatment. Trends Psychiatry Psychother. 46:e20240953.
- Roane HS, Fisher WW, Carr JE. (2016). Applied Behavior Analysis as Treatment for Autism Spectrum Disorder. J Pediatr. 175:27-32.
- Eissa N, Al-Houqani M, Sadeq A, Ojha SK, Sasse A, Sadek B. (2018). Current Enlightenment About Etiology and Pharmacological Treatment of Autism Spectrum Disorder. Front Neurosci. 12:304.
- Wu X, Huangfu J, Zhang W, Hu Y, Yang L, Wei J, et al. (2025). Efficacy and Safety of Neuromodulation Interventions for Autism Spectrum Disorders with Comorbidities: A Systematic Review. Neuropsychiatr Dis Treat. 21:2759-2782.
- Zhuang H, Liang Z, Ma G, Qureshi A, Ran X, Feng C, et al. (2024). Autism spectrum disorder: pathogenesis, biomarker, and intervention therapy. MedComm (2020). 5(3):e497.
- Shirasawa T, Cobos LCA. (2022). Cytokine-induced Neurogenesis for Alzheimer’s Disease and Frontotemporal Dementia. Personalized Medicine Universe. 11(1):27-32.
- Shirasawa T, et al. (2023). Cytokine-induced Neurogenesis Can Reverse Cognitive Decline in Alzheimer’s Disease. Mathews J Case Rep. 8(3):97.
- Shirasawa T, Cobos LCA. (2023). Cytokine-Induced Neurogenesis and Angiogenesis Reversed Cognitive Decline in a Vascular Dementia Patient with Hashimoto’s Thyroiditis. ES J Case Rep. 4(1):1036.
- Shirasawa T, et al. (2023). Cytokine-Induced Neurogenesis in Charcot-Marie-Tooth neuropathy with Connexin 32 Gene Mutation. Mathews J Case Rep. 8(7):118.
- Shirasawa T, et al. (2024). Cytokine-Induced Neurogenesis for Bipolar Disorder: A Case Study. Mathews J Case Rep. 9(4):163.
- Shirasawa T, Hatayama K, Masuyama H, Cobos LCA. (2023). Cytokine-Induced Neurogenesis and Gut Microbiota in Alzheimer’s Disease Biomed J Sci & Tech Res. 51(5):43080-43088.
- Shirasawa T, et al. (2024). Cytokine-induced Neurogenesis for the Prevention and Treatment of Alzheimer’s Disease. Mathews J Case Rep. 9(9):187.
- Currey L, Harvey T, Pelenyi A, Piper M, Thor S. (2025). Mechanisms of brain overgrowth in autism spectrum disorder with macrocephaly. Front Neurosci. 19:1586550.
- Zheng X, Wang X, Song R, Tian J, Yang L. (2024). Executive function, limbic circuit dynamics and repetitive and restricted behaviors in children with autism spectrum disorder. Front Neurosci. 18:1508077.
- Satterstrom FK, Kosmicki JA, Wang J, Breen MS, De Rubeis S, An JY, et al. (2020). Large-Scale Exome Sequencing Study Implicates Both Developmental and Functional Changes in the Neurobiology of Autism. Cell. 180(3):568-584.e23.
- Tordjman S, Somogyi E, Coulon N, Kermarrec S, Cohen D, Bronsard G, et al. (2014). Gene × Environment interactions in autism spectrum disorders: role of epigenetic mechanisms. Front Psychiatry. 5:53.
- Kubota T, Mochizuki K. (2016). Epigenetic Effect of Environmental Factors on Autism Spectrum Disorders. Int J Environ Res Public Health. 13(5):504.
- Bhandari R, Paliwal JK, Kuhad A. (2020). Neuropsychopathology of Autism Spectrum Disorder: Complex Interplay of Genetic, Epigenetic, and Environmental Factors. Adv Neurobiol. 24:97-141.
- Masini E, Loi E, Vega-Benedetti AF, Carta M, Doneddu G, Fadda R, et al. (2020). An Overview of the Main Genetic, Epigenetic and Environmental Factors Involved in Autism Spectrum Disorder Focusing on Synaptic Activity. Int J Mol Sci. 21(21):8290.
- Hall L, Kelley E. (2014). The contribution of epigenetics to understanding genetic factors in autism. Autism. 18(8):872-881.
- Hall MB, Willis DE, Rodriguez EL, Schwarz JM. (2023). Maternal immune activation as an epidemiological risk factor for neurodevelopmental disorders: Considerations of timing, severity, individual differences, and sex in human and rodent studies. Front Neurosci. 17:1135559.
- Sealey LA, Hughes BW, Sriskanda AN, Guest JR, Gibson AD, Johnson-Williams L, et al. (2016). Environmental factors in the development of autism spectrum disorders. Environ Int. 88:288-298.
- Lo H, Weng SF, Tsai EM. (2022). Neurodevelopmental Disorders in Offspring Conceived via In Vitro Fertilization vs Intracytoplasmic Sperm Injection. JAMA Netw Open. 5(12):e2248141.
- Velez MP, Dayan N, Shellenberger J, Pudwell J, Kapoor D, Vigod SN, et al. (2023). Infertility and Risk of Autism Spectrum Disorder in Children. JAMA Netw Open. 6(11):e2343954.
- Tremblay R, Lee S, Rudy B. (2016). GABAergic Interneurons in the Neocortex: From Cellular Properties to Circuits. Neuron. 91(2):260-292.
- Clain J, Couret D, Planesse C, Krejbich-Trotot P, Meilhac O, Lefebvre d'Hellencourt C, et al. (2022). Distribution of Adiponectin Receptors in the Brain of Adult Mouse: Effect of a Single Dose of the Adiponectin Receptor Agonist, AdipoRON, on Ischemic Stroke. Brain Sci. 12(5):680.
- Zhang L, Huang CC, Dai Y, Luo Q, Ji Y, Wang K, et al. (2020). Symptom improvement in children with autism spectrum disorder following bumetanide administration is associated with decreased GABA/glutamate ratios. Transl Psychiatry. 10(1):9.
- Caporale N, Leemans M, Birgersson L, Germain PL, Cheroni C, Borbély G, et al. (2022). From cohorts to molecules: Adverse impacts of endocrine disrupting mixtures. Science. 375(6582):eabe8244.