Previous Issues Volume 2, Issue 2 - 2017
Bone Grafting In Posttraumatic Bone Deffects: Case Report and Review of Literature
Francisco Alonzo1*, Maria Bregni1, Mario Cahueque2, Andres Cobar3
1Orthopedic Surgeon, Guatemalan Social Security Institute, Guatemala, Guatemala.
2Orthopedic Surgeon, Centro Medico, Guatemala, Guatemala
3Orthopedic Surgeon, Mexican Social Security Institute, Guadalajara, Mexico.
Corresponding Author: Francisco Javier Alonzo, Orthopedic Surgeon, Guatemalan Social Security Institute, Guatemala, Guatemala,Mexico, E-Mail: [email protected]
Received Date: 14 Mar 2017 Accepted Date: 31 Mar 2017 Published Date: 03 Apr 2017
Copyright © 2017 Alonzo FJ
Citation: Alonzo FJ, Bregni M, Cahueque M and Cobar A. (2017). Bone Grafting in Posttraumatic Bone Deffects: Case Report and Review of Literature. Mathews J Case Rep 2(2): 028.
ABSTRACT
The management of segmental bone loss remains as an unsolved controversy. Bone defects after traumatic injuries are related to poor functional outcomes and prolonged periods or recuperation. In the management of large bone defects the selection of the adequate bone graft requires a deep acknowledgment of the advantages and disadvantages of each bone substitute available. Autologous bone grafts remain the gold standard to measure all of the available bone substitutes; the ideal bone substitute must provide properties such as osteoconduction, osteoinduction and the osteogenic property, it must also be readily available, provide mechanical support, manageable, biocompatible, provide a sufficient amount of substitute according to the situation and it has to be bioresorbable. We present the case of a patient with a large (>5 cm) diaphiseal defect of the tibia managed with a single, large bone block harvested from the iliac crest and its evolution after a one year follow up.
KEYWORDS
Bone Grafts; Posttraumatic Bone Defect; Bone Substitutes; Autografts; Allogeneic Bone Grafts; Ceramics; Bioactive Glass.
CASE SERIES
Bone formation can be defined as the process that results from the coordinated interaction between an appropriate substrate, growth factors and osteogenic cells. Despite the first two cited elements are already available in artificial preparations, the same affirmation cannot be applied to the source of osteogenic cells that can only be found in bone autografts [1, 2]. When assessing the selection between the different available options for bone grafting, autologous bone graft remains the gold standard to compare all others bone substitutes [3-5]. Autografts usually suffer from less resorption derived from their histocompatibility properties, the presence of viable cells and their osteoconductive and osteoinductive properties [6]. The autografts lack the risk of transmitting infectious diseases and are available at no cost [7]. The ideal graft must provide specific properties in the best combination to achieve bone growth, including: osteoconduction which refers to the ability of a graft to provide an adequate environment for bone formation and capillary ingrowth, osteoinduction: the capacity to recruit pluripotent mesenchymal stem cells that differentiate into osteoblasts and chondroblasts, osteogenic property which implies that the graft contains viable osteocytes or precursors. The ideal graft must also be readily available, manageable, provide mechanical support, biocompatible and bioreasorbable [7-10]. In some situations bone autografts are not available or do not provide a sufficient quantity to fill a large bone voids. Bone substitutes such as demineralized bone matrix, morselized and cancellous allografts ceramics and ceramics composites have osteoconductive properties and in some cases (morselized and cancellous allografts, osteochondral and cortical allografts) can provide mechanical support. They lack the osteogenic property of iliac crest autografts [7, 11, 12]. We present the case of a patient with a large (>5 cm) dia- phiseal defect of the tibia managed with large bone block harvested from the iliac crest and its evolution.
CASE REPORT
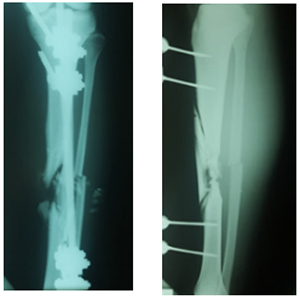
Figure 1: Initial X-rays, antero-posterior view.
Figure 2: Initial X-rays, lateral view.
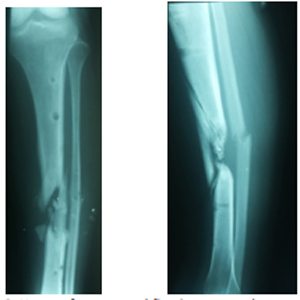
A 22-years old male, with no medical comorbidities, presented to the clinic after being managed for six months in other center for a tibial shaft fracture secondary to a firearm injury. He was initially managed with wound debridement and the application of an external fixator in the operating room. After a 6-month follow-up he was told he had a tibial non-union and needed a second surgery. The initial evaluation showed an afebrile patient with stable vital signs, without abnormalities in the thoracic and abdominal evaluation. He entered the clinic walking with the help of two crutches, and a monoplanar external fixator device in the left leg with four pins along de anterior tibial margin. None of the pins presented with local infection signs at the site of insertion. There was a marked muscular atrophy of the left leg. An anterior scar of about 2 cm wide in the mid-shaft tibial region with no infection signs was visible. Sensation, pulses and distal capillary refill showed no abnormalities. Radiographs showed a displaced multifragmentary tibial shaft fracture (AO 42C-3). The external fixator was removed one week prior to surgery.
Figure 3: X-rays after external fixation removal, anteroposterior view.
Figure 4: X-rays after external fixation removal, lateral view.
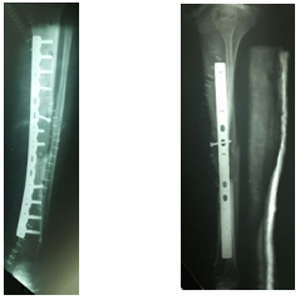
He was taken to the operating room where open reduction and internal fixation with a LCDCP 4.5 mm plate with the implantation of a tricortical iliac crest bone graft was performed.
Surgical technique In the Operating Room, with sterile technique, an anterior approach to the tibial shaft was used exposing the site of the fracture evidencing several large necrotic fragments of the tibial shaft without signs of infection and presence of abundant fibrotic tissue. The avascular fragments were removed until finding bleeding bone leaving a segmentary tibial shaft defect of 10 cm with an oblique proximal margin and a transverse distal margin. A second approach to the iliac crest was performed to obtain a tricortical iliac crest autograft. A segment of 11 cm long was obtained. The ends of the graft were molded to be fitted in the medullary cavity of the proximal and distal fragments. The fracture was fixed using a LCDCP 4.5 mm plate with 4 cortical screws in the proximal tibial fragment and 4 cortical screws in the distal fragment. The autograft was fixed to the plate with 2 cortical screws and an additional 3.5 mm cortical screw was placed to increase the stability in the oblique margin of the proximal shaft fragment. The wound was irrigated and closed.
Figure 5: Postoperative X-Rays, antero-posterior view.
Figure 6: Postoperative X-Rays, lateral view.
RESULTS AND DISCUSSION
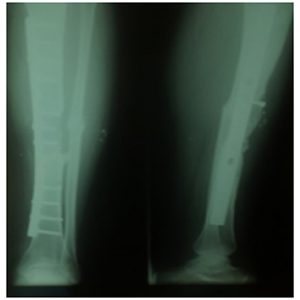
The patient reported early development of pain at the donor site requiring the prescription of analgesics that resolved after four weeks. After a 12 month follow up the patient shows a consolidation rate of 100% between the distal margin of the autograft and the tibial shaft and a rate of 85% between the proximal margin of the autograft and the tibial shaft. The patient tolerates full weight bearing and walks with one crutch.
Figure 7: X-rays after a 12 months follow up.
POST TRAUMATIC BONE DEFECTS
The surgical management of segmental bone loss remains as an unsolved controversy between orthopaedic surgeons. Treatment of extremities with multifragmentary fractures may be complicated by segmentary bone loss and damage to the soft tissue coverage. The challenge becomes even more complex when fibrosis and alteration of regional irrigation arises [13, 14]. Aggressive debridement of bone in multifragmentary open fractures reduces the risk of infection creating in exchange a posttraumatic segmental bone defect. Most authors recommend the removal of contaminated or avascular bone fragments and soft-tissue attachments. Performing an insufficient debridement will increase the risk of leaving contaminated tissues that can lead to chronic infection [14, 15]. The treatment of patients with bone loss is commonly associated to poor functional outcomes and prolonged periods of recuperation [16, 17]. When assessing the decision-making process, the knowledge about advantages and disadvantages for the different available surgical techniques and the different methods to manage soft tissues to create an environment that facilitates bone regeneration, play a crucial role in achieving the therapeutic goals [13, 18]. The treatment must be aimed to achieve a stable and functional extremity in the shortest, most tolerable way for the patient. Up to now, we have several available surgical techniques for handling segmental bone loss: Induced membrane technique, free vascularized autogenous bone graft, bone transport and distraction, demineralized bone matrix, non-structural and structural autologous bone graft between others [17, 20]. These options may be supplemented with the use of demineralized bone matrix or bone morphogenetic protein [16, 21, 22]. In order to achieve bone formation there are five requirements considered mandatory for uneventful consolidation: osteogenic cells, an osteoconductive scaffold, growth factors, a stable mechanical environment and vascularity [10, 23].
Table 1: Types of bone grafts.
| Properties | Presentations | Mechanical Support | Disadvantages | |
|---|---|---|---|---|
| Autologous bone grafts | Osteoinduction Oteoconduction -Osteogenic | -Cancellous, Cortical or corticocancellous grafts | -Depending of the type | -Donor site morbidity -Limited supply |
| Allogeneic bone Grafts | ||||
| Demineralized bone matrix | -Osteoconduction -Osteoinduction (arguable) | Commercial preparations including paste, mix, strip, inject and putty | -They can provide mechanical support depending of the shape of the defect. | -Risk of infectious diseases -slower integration |
| Osteochondral and cortical allografts | -Osteoconduction | -Whole bone or joint. | Provide mechanical support | -Higher Risk of infectious diseases -Slow integration |
| Ceramics and ceramics composites | ||||
| Calcium Phospate substitutes | -Osteoconduction | block, granular, powder or putty form | No mechanical support | Little tensile strength. |
| Calcium Sulfate | -Osteoconduction | Powder | No mechanical support | Slow integration |
| Bioactive Glasses | ||||
| Silica, calcium oxide, disodium oxide and pyrophosphate in different formulations | -Osteoconduction | Microspheres Fibers | No mechanical support | Variable rates of integration according to formulation. |
Autologous bone grafts The use of autologous bone grafts in the management of segmental bone loss is considered a useful tool for its osteoinductive and osteoconductive capacity becoming a biological stimulus for bone healing and regeneration, the grafts also provide with osteogenic cells to the site of implantation [9, 13, 24]. The iliac crest is the most common donor site for bone grafts, it can provide a large volume of autografts of different types (cancellous, cortico-cancellous, unicortical, bicortical or tricortical segments). Cancellous autografts can be harvested from this location preserving its trabecular architecture, this specific type of autograft does not provide mechanical stability. It is rapidly incorporated into the host site due to its osteogenic properties, the large area it provides for bone formation and the abundance of growth factors [25]. Cancellous bone grafts have the additional advantage of providing functional osteoblasts at the site of desired regeneration. The surviving osteocytes combined with graft porosity and local inflammatory response factors promote angiogenesis and recruitment of mesenchymal stem-cells that can differentiate into osteoblasts. New bone formation allows for the graft to completely turnover by one year. Autogenous cortical bone graft is a reliable option to achieve structural stability with or in the absence of a bone void, it provides an osteoconductive medium and immediate mechanical stability. Cortico-cancellous grafts offer the advantages of cancellous bone grafts (rapid integration, source of new osteoblasts, osteoinduction, osteoconduction) and the advantages of cortical autogenous bone grafts (mechanical stability) [8, 26]. Although in some publications the general recommendation is not to manage bone voids larger than 5 cm with autogenous grafts [4, 26]. The use of non vascularized autogenous bone grafting for larger segmental bone defects (up to 16 cm) has been described to provide satisfactory results in several case series [14, 25, 27-29] (Figure 1). In the case presented, we had a satisfactory functional outcome with the use of a single, iliac crest, block of 10 cm to manage the tibial defect. The bone segment appears to have recovered circulation on the 12 month follow up X-rays, despite its size. In patients that need more grafts than the quantity than can be supplied via autograft augmentation or patients with high risk of developing complications from autograft harvesting, other options should be considered for bone grafting such as allografts or synthetic bone fillers. The surgeon must make a decision based on the deep knowledgement of the properties and weakness of each allograft or bone substitute. The availability of the substitute and the personal experience with the materials should be considered as important factors.
Donor Site Morbidity of Bone Autografts of the Iliac Crest Complications associated to harvesting of bone grafts in the anterior iliac crest can be divided into mayor and minor. Minor complications are those that required no or minimal treatment and resulted in minimal disability whereas major complications are those that required repeated surgical intervention, readmission, and prolonged hospital stay or resulted in significant long term disability. Minor complications include persistent pain at donor site, superficial sensory nerve injury, superficial hematoma or seroma, superficial infection. Major complications are such as deep hematoma requiring surgical treatment, vascular injury, sacroiliac joint injury, ureteral injury, donor site fracture, incisional hernia, trendelenburg gait, deep infection [8, 20, 30]. The frequency of such complications are variable between studies. Arrington reported in retrospective review of 414 cases a frequency of 10% minor complications and 5.8% mayor complications [30]. Younger informed an overall rate of mayor complications of 8.6% (infection, prolonged wound drainage, large hematomas, reoperation, pain greater than 6 months, sensory loss, and unsightly scars) between 239 medical records reviewed [31]. Goulet studied 192 cases of atuogenous iliac crest bone harvesting reported 21.8 % (37 patients) with minor complications and 2.4 % (4 patients) with major complications [32]. Cockin reported a rate of minor complications of 6% and a rate of major complications of 3.4% in a series of 118 iliac crest bone grafts [33]. The incidences vary depending the inclusion criteria for minor and major complications. Related to the size of the harvested graft there has been studies that reported complications such as incisional hernia associated to the use of tricortical iliac crest bone grafts [8, 34, 35]. Some studies suggest that the larger the size of the graft, the higher the risk of major complications [34, 35]. The patient of our case reported early development of pain at the donor site. This complaint required the prescription of analgesics for approximately four weeks. We did not suffer any mayor complications despite the size of the harvested graft.
Allogeneic Bone Allogeneic bone is available in many preparations including: demineralized bone matrix, morselized and cancellous chips, corticocancellous and cortical grafts and whole bone segment [7, 36]. Demineralized bone matrix acts as an osteoconductive material and it has arguable osteoinductive properties [8, 9, 36]. Its osteoinductive property relies on the presence of proteins and growth factors, which can vary between donors and with a variable preparation process [8, 9, 37]. Osteochondral and cortical allografts are available as a whole bone or joint for limb salvage in large bone defects. They are osteoconductive, variably osteoinductive and carry a small risk of transmitting diseases, depending on their treatment and processing. The risk is increased when fresh allografts are used. In a clinicopathological study of retrieved human allografts, Enneking found that the union at cortical-cortical junctions occurs over a period of 12 months or more and the bone gap filled does not undergo stress-oriented remodeling, even after several years, thus failure occurred at this junction when the disruption was performed [38]. In other observational study on massive retrieved human allografts the findings of slow integration and osteconductive rather than osteoinductive property of allografts were also reported [39].
Ceramics and Ceramics Composites Synthetic bone grafts substitutes consist of hydroxyapatite, tricalcium phosphate, calcium sulfate or their combinations [10, 41]. When attached to vascularized bone, osteoid is produced into the surfaces of the ceramic without formation of an interface of soft tissue [23]. Although the unlimited supply, easy sterilization and storage can be cited as their strong points, their disadvantages include brittle handling properties, variable rates of resorption, poor performance in diaphyseal defects, and potentially adverse effects on normal bone remodeling [40, 41]. Calcium phosphate substitutes are osteoconductive synthetic bone fillers with no osteoinductive property unless osteoinductive substances (BMP, growth factors) are added resulting in a composite graft. They provide no structural support and have little tensile strength. Calcium phosphate ceramics include hydroxyapatite, coralline hydroxyapatite, tricalcium phosphate an biphasic calcium phosphate [8-10, 42]. Tricalcium phosphate is a frequently used resorbable ceramic. It can be obtained in different presentations: block, granular, powder or putty form. It provides limited biomechanical support due to lower tensile resistance [8-10, 42]. Hydroxyapatite is available in non-absorbable or absorbable solid forms as granules. Its bioresorption is related to its manufacturing process and it is mediated by macrophages, or giant cells [10, 42]. Degradation of tricalcium phosphate and calcium phosphate cement is done by osteoclasts in a period of time of about 1 year; hydroxyapatite degrades in a period of approximately 2-5 years. Tricalcium phosphate ceramic is removed as bone ingrowth is produced while hydroxyapatite is more permanent [23, 41, 42]. Coralline hydroxyapatite is based on certain coral species which produce a porous structure of calcium phosphate similar to human cancellous bone with osteoconductive properties as a bone substitute with high compressive resistance but brittle at low tensile strength [23, 42]. Calcium-collagen graft substitute is a composite of hydroyapatite, tricalcium phosphate and type I and III collagen mixed with autologous bone marrow to provide its osteogenetic property. It does not provide structural support and can be used to augment fracture healing in acute cases [7, 23, 42]. Calcium sulfate is a substitute available in a dry powder form, it is considered osteoconductive with no osteoinductive or osteogenetic property. It is completely dissolved in a period of 6-12 weeks. Its main use is to replace bone after tumor resections [10, 42]. The use of hybrid grafting (β- tricalcium phosphate and demineralized bone matrix) for larger defects has also been reported, with high levels of success [43]. The disadvantages of this method include the availability of the grafts and the high cost.
Bioactive Glasses Bioactive glasses are synthetic bone substitutes composed mainly of silica, calcium oxide, disodium oxide and pyrophosphate. They are available in the presentations of porous implants, microspheres and fibers. After implantation they bind to collagen, growth factors and fibrin to form a matrix that allows bone ingrowth by infiltration of osteogenic cells. The resorption rate of these materials depends on variations on their formulation. The matrix does not provide structural support despite its capacity to support some degree of compressive strength [7, 23, 44].
CONCLUSION
The management of posttraumatic segmental bone defects requires a thorough awareness of the advantages, disadvantages and singular properties of every option available for induce bone ingrowth or substitution, as well as the specific conditions and characteristics provided by the patient (age, comorbidities, weight, habits, etc) and the injury (contamination, infection, size of the defect, mechanism, etc.). Bone autografts remain the gold standard in bone grafting, they account for the principal properties to be sought on a bone substitute (osteoinduction, osteoconduction, osteogenetic property).
REFERENCES
- Hacking A and Vrahas M. (2012). Toward a new standard in autogenous bone graft?. J Bone Joint Surg Am. 94(23): e180.
- Sanders R. (2007). Bone Graft Substitutes: separating fact from fiction. J Bon Joint Surg Am. 89(3): 469.
- Sagi HC, Young ML, Gerstenfeld L, Einhorn TA, et al. (2012).Qualitative and Quantitative differences between bone graft obtained from the medullary canal (with a reamer/ irrigator/aspirator) and the iliac crest of the same patient. J Bone Joint Surg Am. 94 (23): 2128-2135.
- Mauffrey C, Thomas B and Smith W. (2015). Management of Segmental Bone Defects. J Am Acad Orthop Surg. 23(3): 143-153.
- Dimitriou R, Mataliotakis GI, Angoules AG, Kanakaris NK, et al. (2011). Complications following autologous bone graft harvesting from the iliac crest and using de RIA: a systematic review. Injury. 42(2): s3-s15.
- Kirkpatrick JS, Cornell CN,Hoang BH, Hsu W, et al. (2010). Bone void fillers. J Am Acad Orthop Surg. 18(9): 576-579.
- Finkermeier CG. (2002). Bone-Graftin and Bone-Graft Substitutes. J Bone Joint Surg Am. 84(3): 454-464.
- Meyeroff C and Arcdeacon M. (2011). Autogenous Bone Graft: Donor Sites and Techniques. J Bone Joint Surg Am. 93(23): 2227-2236.
- William G, Thomas A, Koval K, Mckee M, et al. (2007). Bone Grafts and Bone Grafts Substitutes in Orthopaedic Trauma Surgery. J Bone Joint Surg Am. 89(3): 649-658.
- Calori G, Mazza E, Colombo M and Ripamonti C. (2011). The use of bone-graft substitutes in large bone defects: any specific needs? Injury. 42: S56-S63.
- Kopylov P, Jonsson K, Thorngren K and Aspenberg P. (1996). Injectable Calcium Phosphate in the treatment of distal radial fractures. J Hand Surg Eur. 21B(6): 768-771.
- Yi- Loong C, Chong K and Merng- Koon W. (2010). Induced Membranes- A Staged Technique for segmental bone loss. J Bone Joint Surg Am. 92(1): 196-201.
- Fleming ME, Watson JT, Gaines RJ and O'Toole RV. (2012). Evolution of Orthopaedic Reconstructive Care. J Am Acad Orthop Surg. 20(1): s74-s79.
- Christian EP, Bosse MJ and Robb G. (1989). Reconstruction of large diaphyseal defects without free fibular transfer, in grade III-b Tibial fractures. J Bone Joint Surg Am. 71(7): 994-1004.
- Guerreschi F, Azzam W, Lovisetti L and Catagni M. (2010). Tetrafocal bone transport of the tibia with circular external fixation: a case report. J Bone Joint Surg Am. 92(1): 190-195.
- Andersen R, Schmidt A, Fitzgerald B, Tintle S, et al. (2015). Extremity War Injuries IX: Reducing Disability Within the Military. J Am Acad Orthop Surg. 23(8): e13-e26.
- Taylor BC, French BG, Fowler TT, Russell J, et al. (2012). Induced Membrane Tecnique for Reconstruction to Manage Bone Loss. J Am Acad Orthop Surg. 20(3): 142-150.
- Azzam W and Atef A. (2016). Our experience in the management of segmental bone loss defects caused by gunshot. Int Orthop. 40(2): 233-238.
- DeCoster T, Gehlert R, Mikola E and Pirela-Cruz M. (2004). Managament of Posttraumatic Segmental Bone Defects. J Am Acad Orthop Surg. 12(1): 28-38.
- Trigui M, Ayadi K, Ellouze Z, Gdoura F, et al. (2008). Treatment of bone loss in limbs by bone transport. Rev Chir Orthop Reparatrice Appar Mot. 94(7): 628-634.
- Cook SD, Baffes GC, Wolfee MW, Sampath TK, et al. (1994). The effect of recombinant human osteogenic protein-1 on healing of large segmental bone defects. J Bone Joint Surg Am. 76(6): 827-838.
- Iwata H, Sakano S and Bauer TW. (2002). Demineralized Bone Matrix and Native Bone Morphogenetic protein in Ortopaedic Surgery. Clin Orthop Relat Res. (395): 99-109.
- Giannoudis PV, Dinopoulos H and Tsiridis E. (2005). Bone substitutes: an update. INJURY. 36(suppl): S20-S27
- Lin D, Luo D, Lian K, Zhai W, et al. (2016). Reconstruction of Traumatic Bone Defect with In situ Implantation of Dropped Traumatic Segmental Bone Fragments. Orthopedics. 39(1): e14-e18
- Sen MK and Miclau T. (2007). Autologous iliac crest bone graft: Should it still be the gold standard for treating nonunions? Injury. 38(Suppl 1): S75-S80.
- Gutierrez M, Leyva F and Alvarez A. (2009). Delay in consolidation and pseudoarthrosis of the tibia. Rev Cub Med Mil.
- Gayito RC, Piruli G, Traore SY, Barbier O, et al. (2015). Treatment of large diaphyseal bone defect of the tibia bya the "fibula pro tibia" technique: application in developing countries. Acta Orthop Belg. 81(1): 17-22
- Lawal YZ, Garba ES, Ogirima MO, Dahiru IL, et al. (2011). Use of non-vascularized autologous fibula strut graft in the treatment of segmental bone loss. Ann Afr Med. 10(1): 25-28.
- Lin KC, Tarng YW, Hsu CJ and Renn JH. (2014). Free nonvascularized fibular strut bone graft for treatment of posttraumatic lower extremity large bone loss. Eur J Orthop Surg Traumatol. 24(4): 599-605.
- Arrington E, Smith W, Chambers H, Bucknell A, et al. (1996). Complications of iliac crest bone graft harvesting. Curr Orthop Prac. 329: 300-309.
- Younger EM and Chapman M. (1989). Morbidity at bone graft donor sites. J Orthop Trauma. 3(3): 192-5.
- Goulet JA, Senunas LE, DeSilva GL, Lou ML. (1997). Autogenous Iliac Crest Bone Graft Complications and Functional Assesment. Clin Orthop Relat Res. 339: 76-81.
- Cockin J. (1971). Autologous bone grafting-complications at the donor site. J Bone Joint Surg. 53B: 153.
- Banwart J, Asher M and Hassanein R. (1995). Iliac crest bone-graft harvest donor site morbidity a statistical evaluation. Spine. (20)9: 1055-1060.
- Caminiti MF, Sandor GK and Carmichael RP. (1999). Quantification of bone Harvested From the iliac crest using a Power-driven Trephine. J Oral Maxilofac Surg.
- Fillingham Y and Jacobs J. (2016). Bone grafts and their substitutes. Bone Joint J. 98-B(1 suppl A): 6-9.
- Schwartz Z, Hyzy S, Moore M, Hunter S, et al. (2011). Osteoinductivity of demineralized bone matrix is independent of donor biphosphonate use. J Bone Joint Surg Am. 93(24): 2278-2286.
- Enneking W and Campanacci D. (2001). Retrieved human allografts a clinicopathological study. J Bone Joint Surg Am. 83(7): 971-986.
- Enneking W and Mindell E. (1991). Observations on massive retrieved human allografts. J Bone Joint Surg Am. 73(8): 1123-1142.
- Bucholz RW. (2002). Nonallograft osteoconductive bone graft substitutes. Clin Orthop Real Res. (395): 44-52.
- Hak DJ. (2007). The use of osteoconductive bone grafts substitutes in orthopaedic trauma. J Am Acad Orthop Surg. 15(9): 525-536.
- Campana V, Milano G, Pagano E, Barba M, et al. (2014). Bone substitutes in orhtopaedic surgery: from basic science to clinical practice. J Mater Sci: Mater Med. (10):2445-2461.
- Mostafa A and Mahmoud A. (2014). Hybrid grafting of post-traumatic bone defects using β-tricalcium phosphate and demineralized bone matrix. Eur J Orthop Surg Traumatol. 24(5): 663-670.
- Auregan JC and Begue T. (2015). Bioactive glass for long bone infection: a systematic review. Injury. 8: S3-S7.