Information Links
Related Conferences
Previous Issues Volume 11, Issue 1 - 2026
The Role of Relative Determination of the Right Lung Volume by the Diaphragm Index in the Diagnosis of Chronic Obstructive Pulmonary Disease
Michael D Levin*
Dorot. Medical Center for Rehabilitation and Geriatrics, Netanya, Israel
*Corresponding Author: Michael D Levin, MD, Ph.D, Dorot. Medical Center for Rehabilitation and Geriatrics, Department of Pediatric Radiology of the 1-st State Hospital, Minsk, Belarus, Dorot. Medical Center for Rehabilitation and Geriatrics, Amnon veTamar, Netanya, Israel, Tel: 972-538281393, Email: [email protected]
Received Date: February 27, 2026
Published Date: March 10, 2026
Citation: Levin MD. (2026). The Role of Relative Determination of the Right Lung Volume by the Diaphragm Index in the Diagnosis of Chronic Obstructive Pulmonary Disease. Mathews J Case Rep. 11(1):218.
Copyrights: Levin MD. © (2026).
ABSTRACT
Introduction: Chronic obstructive pulmonary disease (COPD) is defined as persistent airflow obstruction resulting from chronic bronchitis, emphysema, or both. Traditionally, spirometry had been considered the gold standard for diagnosing COPD. Modern imaging techniques show that determining lung volume makes it possible to more accurately differentiate between obstructive and restrictive causes of pulmonary insufficiency. However, all methods for determining lung volume record true lung volumes using standard values for comparison. The difficulty lies in the fact that these standards vary significantly depending on age, gender, and height. Secondly, complex engineering structures and/or mathematical programs are used to determine lung volume. Method: The relative volume of the right lung was determined on a standard plain radiograph. A horizontal isoline is drawn through the lower edge of the (D-10) thoracic vertebra, where the domes of the diaphragm are attached. The height of the right dome is defined as the vertical line in centimeters between the highest point of the dome and the isoline. It has sign (+) if it is above the isoline, or with a sign (-) if it is below. The depth of the sinus was defined as the vertical line in centimeters between the sinus and the intersection of the isoline with the lateral wall of the chest with signs + or - if it was above or below the isoline. The sum of the height of the diaphragm and the depth of the sinus (diaphragm index - DI) is an integral characteristic of the volume of the right lung. Results: This study included 66 patients aged 50–86 years. 46 of them were admitted to a medical center for rehabilitation and geriatrics with a diagnosis of COPD. In 21 of them (group 1), the lung volume was greater than normal (DI < -5), which confirmed the diagnosis of COPD. In 25 out of 46 patients (group 2), lung volume was not increased. The causes of respiratory failure in these patients included atelectasis, heart failure, and post-pulmonary embolism conditions. Twenty cases (Group 3) included patients with acute respiratory infections or post-aspiration. It was found that acute bronchitis also increases lung volume, but to a lesser extent than COPD. Conclusion: The diaphragm index (DI) is a simple, reliable, and inexpensive radiographic measure of lung volume. It enhances the diagnostic value of chest radiography by differentiating COPD from other causes of respiratory failure.
Keywords: Chronic Obstructive Pulmonary Disease, Spirometry, Computer Tomography, Chest Radiography, Diaphragm Index, Volume Lung.
INTRODUCTION
COPD is a condition characterized by chronic airflow obstruction resulting from chronic bronchitis, emphysema, or both. It is the third leading cause of death worldwide [1]. In COPD, pathological changes are observed in (a) the bronchi and bronchioles, (b) the alveolar tissue, and (c) the respiratory muscles. Inflammation of the peripheral airways contributes to the airflow limitation by increasing the thickness of the airway wall which, together with fibrosis and smooth muscle hypertrophy, causes airway narrowing. Bronchiole narrowing causes airflow limitation, leading to increased alveolar pressure and loss of lung elastic recoil due to parenchymal damage resulting from alveolar septal abnormalities. The increase in alveolar tissue volume, described as emphysema or hyperinflation, is accompanied by impaired gas exchange. The functional consequence of these abnormalities is expiratory flow limitation [1-4]. A sharp increase in lung volume disrupts the function of the respiratory muscles. Because large lungs limit the range of motion of the diaphragmatic domes, this reduces tidal volume. For example, surgeries that reduce lung volume improved lung function, improved exercise tolerance and 1-year all-cause mortality of 5%. [5,6].
Since the mid-20th century airflow obstruction was measured objectively. The gold standard measurements of forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) values, that can be compared manually with available predicted normal values. The definition of COPD based on the Global Initiative for Chronic Obstructive Lung Disease guidelines requires an FEV1 to FVC ratio of < 0.7. Because this ratio declines with age, it has been advocated to use the fifth percentile of the lower limit of normal of the FEV1 to FVC ratio to define true airways obstruction [7]. The spirometry criteria are frequently the topic of futile debates. Therefore, Vestbo believes that it is time to rethink diagnosis COPD [7]. For example, a study in 2015 reported that 54% of patients whose spirometry data did not correspond to COPD had a clinical picture or a CT scan showing emphysema, gas trapping, or airway disease, i.e., symptoms of COPD [8].
Aaron et al, showed that less than 60% of patients with a classical spirometry restrictive pattern had pulmonary restriction confirmed on lung volume measurements. For these patients, measurement of the total lung capacity is needed to confirm a true restrictive defect [9]. According to Cheng et al., total lung capacity in pulmonary function testing is very important for distinguishing between obstructive, restrictive, and mixed disorders. Therefore, they used special equations to determine mean total lung volumes based on the results of computer studies of individuals without lung pathology [10]. However, lung capacity depends on gender, height, and age, which increases the complexity of this method. Sharma and Sankari believe that body plethysmography, performed using a large plastic box, derives its accuracy from its ability to assess trapped air and detect small airway changes that are not always evident in other testing methods. The ability of body plethysmography to distinguish between obstructive and restrictive lung diseases help healthcare providers tailor treatment strategies more effectively [11]. A literature review shows that, unlike restrictive lung diseases, patients with COPD have larger lung volumes. Lung volume determination methods are significantly superior to other methods for the differential diagnosis of obstructive and restrictive lung diseases. However, lung volume determination methods require complex technical devices and sophisticated mathematical calculations. Furthermore, comparing the results obtained with normal values is difficult, as normal lung volumes vary among individuals of different ages, genders, heights, and age.
A full-inspiratory chest radiograph (CXR) is a standard part of the clinical evaluation of patients with COPD. Features suggestive of emphysema include hyperlucent lung fields, flattening of the diaphragm, vascular eruption, enlarged retrosternal airspaces, widened intercostal spaces, and a narrow and more vertical cardiac silhouette. The combination of these criteria has resulted in 90% sensitivity and 98% specificity of CXR for emphysema. CXR is generally considered insensitive for the diagnosis of the earliest stages of emphysema [1,12].
The aim of this study is to increase the diagnostic value of plain chest radiograph in the diagnosis of COPD. For this purpose, a method protected by a copyright certificate in 1988 was used [13]. The definition of normal values and the diagnostic accuracy of the method were presented in two articles [14,15].
Method for determining the relative volume of the right lung on a plain chest radiograph
It is known that the domes of the diaphragm are attached to the 10th thoracic vertebra. During inhalation, the domes of the diaphragm, like the wings of a bird, move down. At this moment, the volume of the lungs increases, as air is sucked into the lungs. During exhalation, the domes rise, squeezing air out of the lungs, because of which the volume of the lungs decreases. To determine the volume of the right lung we draw a horizontal line (isoline) through the lower edge of the 10th thoracic vertebra. On the right, along the midclavicular line, we determine the height of the dome in centimeters. It is a perpendicular, lowered (with the "+" sign) or erected (with the "-" sign) from the dome to the isoline. We also define the sinus depth as the distance from the intersection of the isoline with the right edge of the chest to the extreme point of the sinus. If the upper end of this line is above the isoline, it has a plus sign (+), if below, it has a minus sign (-). The integral characteristic of the lung volume, i.e. the diaphragm index (DI), is the sum of the dome height and the sinus depth in centimeters (Figure 1). In previous studies in patients without lung pathology, DI ranged from 0 to +5. [5,6].
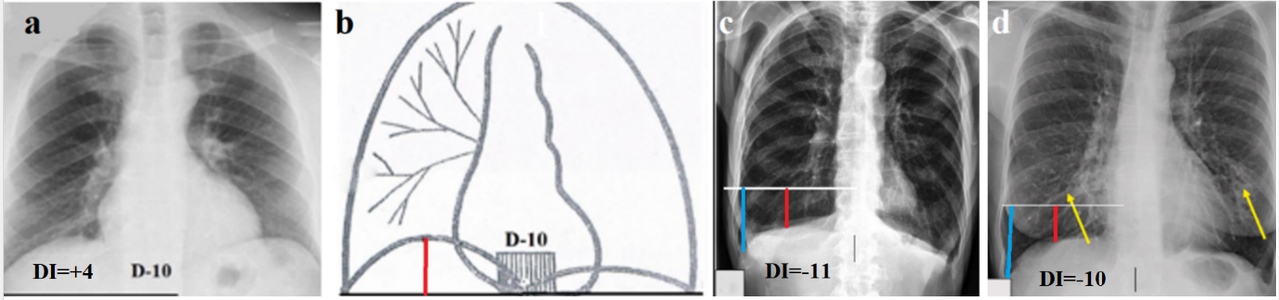
Figure 1. The principle of the proposed method. (a) Location of the diaphragm in a patient with a normal right lung volume. (b) Schematic for Figure 1a. The bottom of the sinus is located at the isoline level. The height of the diaphragm (red line) is located 4 cm about the isoline (DI = + 4). (c) Radiograph from Raoof et al. with the caption: - "A patient with emphysema showing increased lucency of the lungs with pruning of the peripheral vasculature, flattening of the diaphragms, widening of the intercostal spaces, kyphosis (barrel-shaped chest), and a narrowed cardiac silhouette" [1]. The use of the proposed method revealed a sharp increase in the volume of the right lung (DI = -11). The left lung is enlarged to the same extent. The exact values are obtained by calculating the projection magnification coefficient equal to the ratio of the true height of L-1 (2.2 cm) to the height of its image on the radiograph. The height of the dome is -3 cm, and the height of the sinus is -8 cm. DI = -11. (d) Second radiography from Raoof et al. with the caption: - "A Patient with COPD and bronchiectasis, as demonstrated by tubular lucencies (arrows) at the lung bases" [1]. The use of our method demonstrated a sharp increase in lung volume (DI=-10), despite the absence of many typical symptoms of COPD (the heart is not reduced in size, the shape of the chest and the course of the ribs are normal).
Patients: At the Center for Rehabilitation and Geriatrics, 66 patients aged 50 to 86 years with respiratory failure, bedridden were examined. Forty-six of them were diagnosed with COPD in the previous hospital. We divided these patients into two groups. Group 1 consisted of 21 patients, in whom a significant increase in the volume of the right lung was detected based on X-ray analysis and the calculation of the DI. Group 2 consisted of 25 patients in whom the DI was either within the normal range or significantly greater than +5. The 3rd group consisted of 20 patients in whom X-ray examination was performed against the background of acute conditions accompanied by inflammation of the bronchial mucosa (viral infections and aspirations) or with chronic bronchitis. They were selected to determine how acute bronchial and bronchiole constriction without damage to parenchymal tissue alters lung volume (DI).
RESULTS
1st group. In 21 patients, the ID was within -7 – (-23) (average -12.2) (Figure 2). The volume of the left lung was increased symmetrically to the right in 18 of them (Figure 2a, b, c). In 3 cases, the volume of the left lung was clearly smaller than the right (Figure 2d).
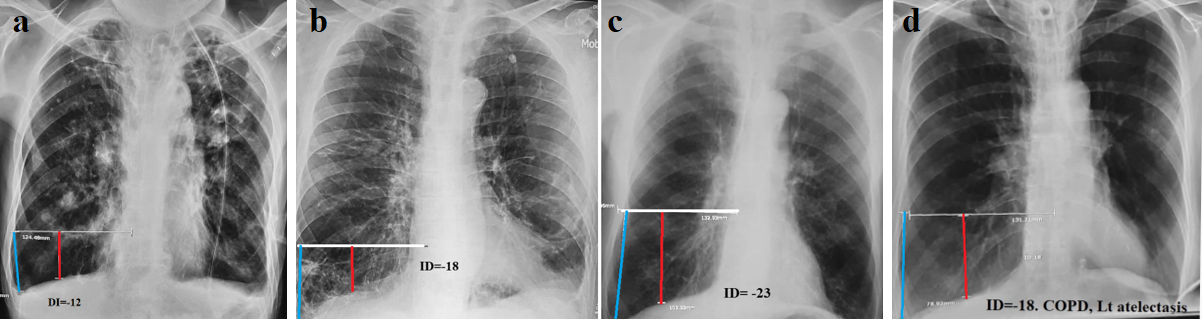
Figure 2 (a,b,c). Examples of COPD with different heart sizes and chest shapes. They are united by a sharp and uniform increase in lung volume, as indicated by DI values. (d). Severe form of COPD in a patient with a tracheostomy complicated by atelectasis at the base of the left lung. DI= -18.
All radiographs show radiological signs of chronic bronchitis (increased and deformed pulmonary pattern with signs of sclerosis, widening, and poor differentiation of the gates of the lungs). However, these signs are not specific. All radiographs in this group are characterized by a significant increase in lung volume, mathematically defined as DI -5 -23 (average -12.2).
In the 2nd group among 25 patients the DI ranged from -2 to (+18) (average 2.6) (Figure 3). In these patients, respiratory failure was due to either cardiac failure (8) or a decrease in lung volume due to atelectasis (12). Tumor-related atelectasis occurred in 3 cases. In addition, restrictive processes occurred in 4 cases, including 2 patients with COVID-19.
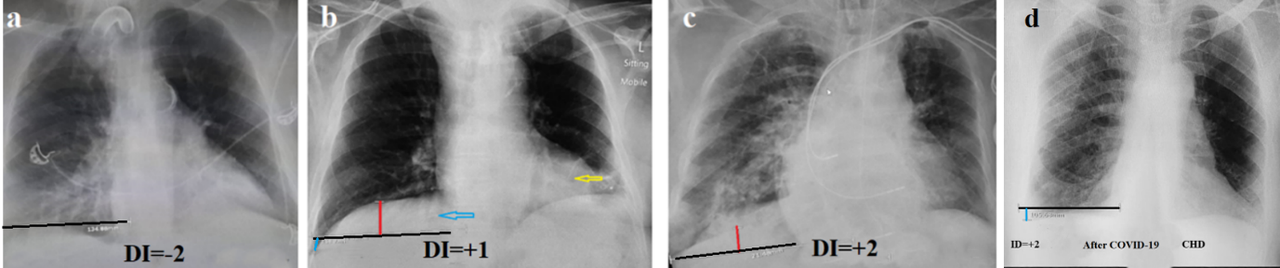
Figure 3. Radiographs of patients with respiratory failure in whom COPD was excluded by the proposed method. (a) Atelectasis of the lower left lobe and bronchiectasis in the right lower lobe. (b). The patient was admitted with a diagnosis of COPD exacerbation. The lung volume has not increased. DI= +1. The blue arrow shows bronchiectasis in the lower lobe on the right, and the yellow arrow shows atelectasis above the left diaphragm. (c). In a patient with chronic heart disease who suffered a pulmonary embolism. (d). CHD. The decreased volume and increased transparency of the left lung is due to atelectasis of the lower lobe in a patient after COVID-19.
In the 2nd group, the causes of respiratory failure were restrictive causes: atelectasis, bronchiectasis, conditions after pulmonary embolism, heart failure etc. We do not have data on the basis (research methods) by which COPD was diagnosed in the patients of the second group at the hospitals from which they arrived. An analysis of various diagnostic methods COPD will be provided in the "Discussion" section.
In 20 patients of the 3rd group, there were cases of acute bronchitis-bronchiolitis, which led to inflammation of the mucous membrane of the bronchi and bronchioles, creating an obstruction for breathing with a decrease in tidal volume. In some patients, the cause of inflammation was a viral infection, and in others, inflammation was caused by hydrochloric acid during aspiration (Figure 4). In acute obstructive bronchitis-bronchiolitis, a moderate increase in lung volume was observed. DI ranged from -3 to - 6 cm (mean -5.2 cm).
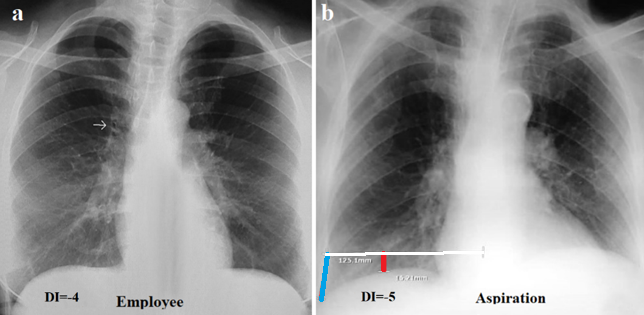
Figure 4. Radiographs of patients with acute obstructive pulmonary disease. (a) Bronchitis-bronchiolitis due to viral infection. The lung roots are enlarged and structureless. Thickened walls of the main bronchi are shown by the arrow on the right. Increased pulmonary markings in the lower fields. Deep sinuses are open. DI=-4. (b) Condition after clinically confirmed aspiration. Marked increase in pulmonary markings in the lower field on the right. The pulmonary roots are dilated and structureless. The volume of the right lung is increased. DI=-5.
The measurement in 3 groups results are summarized in Table 1.
Table 1. Diaphragm Index (DI) values three groups of patients
|
Groups |
Clinical characteristic |
N |
DI ranges |
Mean |
|
1-st |
COPD with increased lung volume |
21 |
-7 – (-23) |
12.2 |
|
2-nd |
Restrictive causes of respiratory failure |
25 |
-2 - (+18) |
2.6 |
|
3-rd |
Acute airway obstruction: bronchiolitis etc. |
20 |
- 3 –(- 6) |
-5.2 |
DISCUSSION
COPD begins as a mild form of the disease and develops slowly over decades as small airways narrow and disappear, causing lung function to decline. Therefore, it is important to promptly identify the disease in its early stages to prevent its progression [4]. Unfortunately, the etiology and pathogenesis of COPD are not fully understood. The role of smoking, which causes irritation and inflammation in the bronchial tree, is well known. However, many heavy smokers do not develop COPD despite chronic bronchitis. Therefore, other factors influence the development of the disease. For example, compared with nonsmokers, gamma delta T-lymphocytes were significantly increased in smokers with preserved lung function, however this response was blunted in patients with COPD [16]. Evidence from observational studies suggests that more immune cells are present in the lung tissue of COPD and asthma sufferers compared to healthy individuals, along with a significant immune response. Mendelian randomization (MR) studies found that a variety of immune cells and inflammatory factors are risk factors for the development of COPD and asthma [17]. By 2022, 283 articles devoted to the relationship between COPD and GERD had been published. However, the causal relationship between these diseases has not been established [18]. Recently, genetic studies by Wilson et al. identified several loci associated with GERD and COPD comorbidity that were previously associated with GERD or COPD separately, including HCG17, which plays a role in oxidative stress mechanisms, i.e., revealed a common genetic etiology of GERD in COPD [19]. Lin et al. Using a two-sample bidirectional Mendelian randomization (MR) study elucidated a notable causal relationship, with individuals genetically inclined towards GERD exhibiting a significantly elevated propensity to develop COPD. Conversely, reverse MR analyses did not identify a significant causal pathway between COPD or asthma and GERD onset [20]. Однако, известно, что non-esophageal GERD can exacerbate the pulmonary process and worsen the course of COPD [21, 22]. A review of the literature suggests that, in addition to smoking, another etiological factor for COPD is a common genetic etiology of oxidative stress mechanisms (GERD) and COPD. This connection is broader because GERD is one of the clinical forms of acid-dependent diseases [23]. Secondly, modern GERD diagnostics using pH monitoring underdiagnoses approximately 30% of patients with GERD [24]. Thus, patients with acid-related diseases, including GERD, are at risk for developing COPD, which requires not only a special examination, but also persistent treatment to prevent relapses of respiratory inflammation.
As shown by modern research methods, spirometry, which for nearly a century was considered the gold standard for the differential diagnosis of obstructive and restrictive causes of respiratory failure, has proven to be little ineffective. At the same time, lung volume determination methods are significantly superior to other methods for the differential diagnosis of obstructive and restrictive lung diseases [9-12,25,26]. All methods for determining lung volume record true lung volumes using standard values for comparison. The difficulty lies in the fact that these standards vary significantly depending on age, gender, and height. Furthermore, complex equipment and/or mathematical software are required to determine lung volume. Our proposed method determines the relative volume of the right lung based on the position of the right dome of the diaphragm relative to its attachment to the 10th thoracic vertebra on a standard chest radiograph taken during quiet breathing. In healthy individuals, the diaphragm volume index (DI) fluctuates between 0 and 5, regardless of age, gender, and height [13-15]. When there is restrictive causes respiratory failure DI increases above 5, depending on the degree of diaphragm elevation. If the volume of the right lung is increased, the right dome of the diaphragm descends, resulting in a decrease in DI below zero. The increase in lung volume in patients of the 3rd group according to the DI data, firstly, confirms that the inflammatory process in the bronchial system causes a violation of the air outlet from the lung, i.e., obstruction. Secondly, this indicates the high accuracy of this method. Since the proposed method determines lung volume increases even in acute bronchial inflammation, it seems likely that with its use, a chronic obstructive process can be recorded in the early stages of the disease. Based on this study, it can be assumed that an increase in lung volume with a DI (-10 or less) is a characteristic of COPD. However, a less significant increase in volume (-9 to -5) may be both in early stage of COPD and in chronic bronchitis. When a moderate increase in lung volume is combined with GERD and/or allergies [27] in patients with a chronic course of the disease, the possibility of an early stage of COPD can be assumed. The proposed DI method uses well-differentiated bone landmarks and simple mathematical analysis, which eliminates the possibility of discrepancies between researchers. There is no overlap between the DI in patients from groups 1 and 2, and this phenomenon does not depend on the height of adult patients, so there is no point in calculating the mean ± SD, exact p-values, or confidence intervals. We believe that in bedridden geriatric patients, the proposed method of radiograph evaluation is the best alternative for differential diagnosis of COPD and other diseases accompanied by respiratory failure, which will help to choose pathogenetically justified treatment.
A review of the literature reveals varying recommendations for the early diagnosis of COPD. For decades, spirometry has been regarded as the standard for diagnosing COPD; however, numerous studies have shown that COPD symptoms, pathology, and associated poor outcomes can occur despite normal spirometry [28]. Nevertheless, many publications are devoted to recommendations for the use of spirometry. Hundreds of articles publish recommendations from a physician community called "The Global Initiative for Chronic Obstructive Lung Disease (GOLD)." These recommendations represent the consensus of the participants. They do not compare different methods. Their reliability is compromised by the fact that all authors receive compensation from numerous sources for advertising equipment and medications [29].
Spirometry is not used or is poorly conducted at the primary care level for several identifiable reasons, including cost, inconvenience, and time. Questionnaires that include clinical symptoms of the disease are proposed as an alternative [30]. This study suggests that referring hospitals misdiagnosed COPD in 25 patients in Group 2 based on clinical symptoms alone, as developed questionnaires have not yet been widely used.
CONCLUSION
The diaphragm index (DI) is a simple, reliable, and inexpensive radiographic measure of lung volume. It enhances the diagnostic value of chest radiography by differentiating COPD from other causes of respiratory failure. We believe that in bedridden geriatric patients, the proposed method of radiograph evaluation is the best alternative for differential diagnosis of COPD and other diseases accompanied by respiratory failure, which will help to choose pathogenetically justified treatment.
ACKNOWLEDGEMENTS
None.
CONFLICT OF INTEREST
The author has no conflict of interest.
REFERENCES
- Raoof S, Shah M, Make B, Allaqaband H, Bowler R, Fernando S, et al. (2023). Lung Imaging in COPD Part 1: Clinical Usefulness. Chest. 164(1):69-84.
- Ritzmann F, Brand M, Bals R, Wegmann M, Beisswenger C. (2025). Role of Epigenetics in Chronic Lung Disease. Cells. 14(4):251.
- Hogg JC, Chu F, Utokaparch S, Woods R, Elliott WM, Buzatu L, et al. (2004). The nature of small-airway obstruction in chronic obstructive pulmonary disease. N Engl J Med. 350(26):2645-2653.
- Soriano JB, Polverino F, Cosio BG. (2018). What is early COPD and why is it important? Eur Respir J. 52(6):1801448.
- Congleton J. (2020). Lung volume in COPD: when less is more. Drug Ther Bull. 58(4):50.
- Hartman JE, Welling JBA, Klooster K, Carpaij OA, Augustijn SWS, Slebos DJ. (2022). Survival in COPD patients treated with bronchoscopic lung volume reduction. Respir Med. 196:106825.
- Vestbo J. (2014). COPD: definition and phenotypes. Clin Chest Med. 35(1):1-6.
- Regan EA, Lynch DA, Curran-Everett D, Curtis JL, Austin JH, Grenier PA, et al. (2015). Clinical and radiologic disease in smokers with normal spirometry. JAMA Intern Med. 175(9):1539-1549.
- Aaron SD, Dales RE, Cardinal P. (1999). How accurate is spirometry at predicting restrictive pulmonary impairment? Chest. 115(3):869-873.
- Cheng T, Li Y, Pang S, Wan H, Shi G, Cheng Q, et al. (2019). Normal lung attenuation distribution and lung volume on computed tomography in a Chinese population. Int J Chron Obstruct Pulmon Dis. 14:1657-1668.
- Sharma P, Sankari A. (2025). Body Plethysmography. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
- Miniati M, Monti S, Stolk J, Mirarchi G, Falaschi F, Rabinovich R, et al. (2008). Value of chest radiography in phenotyping chronic obstructive pulmonary disease. Eur Respir J. 31(3):509-515.
- Levin MD. (1988). Author's certificate for the invention "Method for diagnosing widespread pulmonary inflation." State Committee for Inventions and Discoveries. USSR. SU 1651861 A1.
- Levin MD. (1988). Method for determining lung volume and its role in the diagnosis of certain diseases in children. Zdravoochranenie Belorussii. 1988(11):51-55. (Russian).
- Levin MD, Averin VI, Degtyarev YG, Bolbas TM. (2022). Method for Determining the Volume of the Right Lung and its Role in the Diagnosis of Some Lung Diseases. Novosti Khirurgii. 30(5):447-452.
- Pons J, Sauleda J, Ferrer JM, Barceló B, Fuster A, Regueiro V, et al. (2005). Blunted gamma delta T-lymphocyte response in chronic obstructive pulmonary disease. Eur Respir J. 25(3):441-446.
- Guixing X, Yilin L, Huaying F, Fanrong L, Dehua L. (2025). Effect of the Antibody-mediated Immune Responses on COPD, Asthma, and Lung Function: A Mendelian Randomization Study. Arch Bronconeumol. 61(4):212-219.
- Zou M, Zhang W, Xu Y, Zhu Y. (2022). Relationship Between COPD and GERD: A Bibliometrics Analysis. Int J Chron Obstruct Pulmon Dis. 17:3045-3059.
- Wilson AC, Rocco A, Chiles J, Srinivasasainagendra V, Labaki W, Meyers D, et al. (2025). Novel risk loci encompassing genes influencing STAT3, GPCR, and oxidative stress signaling are associated with co-morbid GERD and COPD. PLoS Genet. 21(2):e1011531.
- Lin S, Lai D, He W, Zhan Q. (2025). Causal relationship between gastroesophageal reflux disease and chronic obstructive respiratory disease: A bidirectional Mendelian randomization study. Heliyon. 11(2):e42100.
- Ghisa M, Della Coletta M, Barbuscio I, Marabotto E, Barberio B, Frazzoni M, et al. (2019). Updates in the field of non-esophageal gastroesophageal reflux disorder. Expert Rev Gastroenterol Hepatol. 13(9):827-838.
- Levin MD. (2025). Aorto-Esophageal Sphincter and its role in the pathogenesis of the gastroesophageal reflux: cases report and analysis. Gastroenterol Hepatol Open Access. 16(1):1-8.
- Levin MD. (2025). Hydrochloric acid hypersecretion is the cause of pathology of the upper digestive tract. Literature analysis. Qeios. DOI: 10.32388/4QXH2Q.
- Levin MD. (2025). X-Ray Imaging of the Esophagus and Lower Esophageal Sphincter and Its Role in the Diagnosis of Gastroesophageal Reflux Disease. Mathews J Gastroenterol Hepatol. 10(2):31.
- Aalstad LT, Hardie JA, Espehaug B, Thorsen E, Bakke PS, Eagan TML, et al. (2018). Lung hyperinflation and functional exercise capacity in patients with COPD - a three-year longitudinal study. BMC Pulm Med. 18(1):187.
- Tanabe N, Sato S, Shimada T, Kaji S, Shiraishi Y, Terada S, et al. (2024). A reference equation for lung volume on computed tomography in Japanese middle-aged and elderly adults. Respir Investig. 62(1):121-127.
- Jin JM, Sun YC. (2017). Allergy and Chronic Obstructive Pulmonary Disease. Chin Med J (Engl). 130(17):2017-2020.
- Haynes JM, Kaminsky DA, Ruppel GL. (2023). The Role of Pulmonary Function Testing in the Diagnosis and Management of COPD. Respir Care. 68(7):889-913.
- Singh D, Stockley R, Anzueto A, Agusti A, Bourbeau J, Celli BR, et al. (2025). GOLD Science Committee recommendations for the use of pre- and post-bronchodilator spirometry for the diagnosis of COPD. Eur Respir J. 65(2):2401603.
- Frith PA. (2020). Detecting COPD using micro-spirometry and/or questionnaire. Respirology. 25(2):126-127.