Previous Issues Volume 2, Issue 1 - 2017
Role of QRS Complex Fragmentation in Patients at High Risk of Cardiovascular Events
Osmar Antonio Centurión*, Nelson Javier Aquino-Martinez, Judith María Torales-Salinas, Luis Marcelo Miño, Orlando Robert Sequeira-Villar, Karina Elizabeth Scavenius-Aguilera
Department of Health Sciences’s Investigation, Sanatorio Metropolitano. Fernando de la Mora. Paraguay, Cardiology Department. Clinic Hospital. Asunción National University. San Lorenzo, Paraguay.
Corresponding Author: Osmar Antonio Centurión, Professor of Medicine, Asuncion National University, Department of Health Sciences’s Investigation, Sanatorio Metropolitano, Teniente Ettiene 215 c/ Ruta Mariscal Estigarribia, Fernando de la Mora, Paraguay, Tel: 595 21 585 540; E-Mail: [email protected]
Received Date: 28 Dec 2016 Accepted Date: 10 Jan 2017 Published Date: 10 Jan 2017
Copyright © 2016 Centurión OA
Citation: Centurión OA, Aquino-Martinez NJ, Torales-Salinas JM, Miño LM, et al. (2017). Role of QRS Complex Fragmentation in Patients at High Risk of Cardiovascular Events. Mathews J Cardiol. 2(1): 009.
ABSTRACT
The presence of fragmented QRS complexes (FQRS) on a routine 12-lead ECG is an interesting marker of depolarization abnormality. There is convincing data suggesting that the FQRS represents conduction delay from inhomogeneous activation of the ventricles due to myocardial scar. However, FQRS is not specific for coronary artery disease and is also encountered in other myocardial diseases such as cardiomyopathy and congenital heart disease. FQRS has also been described in other entities such as arrhythmogenic right ventricular cardiomyopathy and Brugada syndrome. The significance and the predictive value of FQRS complex as an ECG marker of cardiovascular events seem to be different in different entities. In patients with stable coronary artery disease and in patients with acute MI, FQRS seems to be a good predictor of cardiac events. In patients with non-ischemic cardiomyopathy, fragmentation of narrow QRS complex seems to correlate with the degree of fibrosis and dys-synchrony, and importantly may influence the response for cardiac resynchronization therapy. However, based on clinical studies with larger number of patients with current indications for cardiac resynchronization, FQRS does not influence and is not associated with poor response of resynchronization therapy. In patients with LV dysfunction, there is no clear evidence that the presence of FQRS could predict arrhythmic events. On the other hand, there was a statistical significant difference in mortality associated to the presence of fragmented QRS in patients with acute coronary syndrome and myocardial necrosis.
KEYWORDS
QRS Fragmentation; Cardiovascular Events; Electrocardiogram.
INTRODUCTION
Invasive and non-invasive tests for risk stratification of cardiovascular events were studied in the context of coronary artery disease, cardiomyopathy and heart failure [1-5]. One of the recent non-invasive tests, microwave T-wave alternans and signal-averaged electrocardiogram have high negative predictive values but have a low positive predictive value in patients with history of myocardial infarction (MI) or cardiomyopathy [3-5]. A low left ventricular ejection fraction in patients with structural heart disease is used routinely in clinical practice for risk stratification with a major limitation being the lack of desirable positive predictive value for the risk of cardiovascular events [4, 5]. The presence of fragmented QRS complexes (FQRS) on a routine 12-lead ECG is another marker of depolarization abnormality [6]. There is convincing data suggesting that the FQRS represents conduction delay from inhomogeneous activation of the ventricles due to myocardial scar. However, FQRS is not specific for coronary artery disease and is also encountered in other myocardial diseases such as cardiomyopathy and congenital heart disease. FQRS has also been described in other entities such as arrhythmogenic right ventricular cardiomyopathy and Brugada syndrome [7, 8]. In 1969, Flowers et al. named high-frequency components to the presence of fragmented QRS (FQRS) complexes [9]. They showed that these slurring and changes in the morphology of the QRS complex are more common among patients with prior myocardial infarction (MI) and among patients with either right or left ventricular (LV) enlargement. In experimental investigations, Friedman et al suggested that persistent changes in Purkinje fibers and myocardial fibrosis may cause slow and inhomogeneous myocardial activation in the canine heart with induced MI [10]. In the same animal experimental model, Gardner et al suggested that FQRS complex in infarcted canine heart is caused by slow and inhomogeneous activation associated to healed myocardial scar rather than changes in transmembrane resting or action potentials [11]. The presence of FQRS represents distortion of signal conduction and depolarization process within the ventricles which is related to myocardial scar/myocardial ischemia or myocardial fibrosis. Therefore, the FQRS but in particular late potentials, was investigated as a possible new tool to identify patients at the high risk of cardiovascular events.
Definition of Fragmented QRS
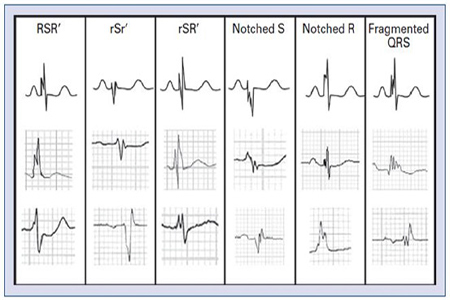
Das et al. proposed a definition for the FQRS and depicted the different types (Figure). They defined it as the presence of an additional R wave (R’) or notching in the nadir of the S wave, or the presence of > 1 R’ in 2 contiguous leads, corresponding to a major coronary artery territory on the resting 12-lead ECG with filter range 0.16–100 Hz, AC filter 60 Hz, paper speed 25 mm/s and 10 mm/mV. They also defined the fragmentation of wide QRS complex (WFQRS) in patients with bundle branch block and paced rhythms [12]. They defined WFQRS as various RSR patterns with or without a Q wave, with more than 2 R waves (R’) or more than 2 notches in the R wave, or more than 2 notches in the downstroke or upstroke of the S wave, in 2 contiguous leads corresponding to a major coronary artery territory. Later on, Das et al. defined fragmented premature ventricular complexes (PVC) in a similar fashion to fragmentation of bundle branch block and paced rhythms but also included PVC with 2 notches in the R wave which were more than 40 ms apart and present in 2 contiguous leads [13].
Figure 1:Examples of different types of QRS fragmentations are depicted in the Figure. Reproduced with permission from [14].
Fragmented QRS and Cardiovascular Events
Myocardial scars from a previous infarction may provide the anatomic substrate for lethal re-entrant ventricular arrhythmias in patients with ischemic heart disease and impaired left ventricular function. The initiation and perpetuation of a lethal re-entrant ventricular arrhythmia depend not only on a trigger event such as a premature ventricular complex, but also on the presence of a vulnerable myocardial substrate. The sensitivity and specificity of FQRS for detection of myocardial scar was investigated in 479 consecutive patients with and without prior history of coronary disease who underwent nuclear stress test [13]. The FQRS complexes were found to have higher sensitivity than Q waves for detecting regional myocardial scar as well as for detection of myocardial scar independently of the regional correlation. However, when specificity was compared, the FQRS was less specific than Q wave for myocardial scar (86% vs 99%). In a similar investigation, Das et al demonstrated that the presence of FQRS was associated with higher all-cause mortality in patients without FQRS and cardiac event rate [14]. In the multivariate Cox regression analysis, FQRS was an independent predictor of cardiac events (HR 1.62; p=0.0001). Pietrasik et al investigated the effect of FQRS on the risk of recurrent cardiac events in those patients with persistent or resolved Q waves at two months of their first Q-wave MI [15]. The risk of recurrent cardiac events was defined as presenting unstable angina, recurrent MI or cardiac death at follow-up. In this investigation, FQRS was associated with higher risk of recurrent events among those patients who had resolved Q wave. Therefore, it was suggested that FQRS may identify ischemic myocardium [15]. Kadi H et al found an association of FQRS within the context favourable to myocardial scar. In their study, FQRS seemed to correlate with chronic total coronary occlusion with poorly developed collateral coronary circulation in patients without prior MI [16]. However, contrary to those findings, two other studies failed to show significant association between FQRS and myocardial scar. First, Wang at al investigated the sensitivity and specificity of FQRS for the detection of myocardial scar by means of nuclear perfusion images [17]. They also assessed the presence of Q wave in 460 consecutive patients with known or suspected coronary artery disease. They showed that Q wave has better sensitivity than FQRS in detecting myocardial scar (32% vs 2%). Second, similar results were found by Carey et al [14, 18]. They studied 138 patients with severely depressed LV systolic function with a mean ejection fraction of 28% who had infarct volume assessed by positron emission tomography. They demonstrated that FQRS was not predictive for infarct size in both patients with narrow and wide QRS complexes [18]. Das et al demonstrated evidence relating fragmented wide QRS complexes to myocardial scar and poor prognosis in 879 patients with wide QRS [13]. These patients with wide QRS complexes equal to or more than 120 ms included those with bundle branch block, premature ventricular contractions, or paced QRS referred for nuclear stress testing or cardiac catheterization for evaluation of coronary artery disease. The presence of fragmented wide QRS complex was associated with high sensitivity and specificity for myocardial scar, as well as, a high positive predictive value and negative predictive value. The FQRS was associated with an increased risk of all cause-mortality after adjustment for age, ejection fraction, and history of diabetes mellitus [13]. The presence of FQRS and its association to mortality in patients with acute coronary syndrome was investigated in 896 patients [19]. The study patients were divided into two groups. One of the groups had 337 patients with MI (both STEMI and NSTEMI), and the other group comprised of 445 patients with unstable angina. There was a statistical significant difference in the presence of fragmented QRS between the two groups. FQRS was found in 224 patients with MI and only in 17 patients with unstable angina (51% vs 4%; p< 0.001). In addition, new Q waves developed in 122 (28%) patients with STEMI, in 76 (23%) patients with NSTEMI, and only in 2 (0.4%) patients with unstable angina [19]. In the multivariate Cox regression analysis, the presence of FQRS was associated with a 68% higher risk of all-cause mortality (HR 1.68; p=0.003) during a mean follow-up period of 34±16 months [19].
CONCLUSION
In conclusion, the significance and the predictive value of FQRS complex as an ECG marker of cardiovascular events seem to be different in different entities [20-26]. In patients with stable coronary artery disease and in patients with acute MI, FQRS seems to be a good predictor of cardiac events [14, 27]. In patients with non-ischemic cardiomyopathy, fragmentation of narrow QRS complex seems to correlate with the degree of fibrosis and dys-synchrony and importantly may influence the response for cardiac resynchronization therapy [28]. However, based on clinical studies with larger number of patients with current indications for cardiac resynchronization, FQRS does not influence and is not associated with poor response of resynchronization therapy [29-31]. In patients with LV dysfunction, there is no clear evidence that presence of FQRS could predict arrhythmic events [31-34]. On the other hand, there was a statistical significant difference in mortality associated to the presence of fragmented QRS in patients with acute coronary syndrome and myocardial necrosis.
REFERENCES
Myerburg RJ and Castellanos A. (2008). Cardiac arrest and sudden cardiac death. In: Braunwald’s Heart Disease: A Textbook of Cardiovascular Medicine (8/ed). Philadelphia, PA: WB Saunders; 933-974.
- Luqman N, Sung RJ, Wang CL and Kuo CT. (2007). Myocardial ischemia and ventricular fibrillation: pathophysiology and clinical implications. Int J Cardiol. 119: 283-290.
- Prystowsky EN. (2004). Primary prevention of sudden cardiac death: the time of your life. Circulation. 109(9): 1073-1075.
- Zipes DP, Camm AJ, Borggrefe M, et al. (2006). ACC/AHA/ ESC 2006 Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: a report of the American College of Cardiology/ American Heart Association Task Force and the European Society of Cardiology Committee for Practice Guidelines (writing committee to develop Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death): developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society. Circulation. 114: e385-e484.
- Huikuri HV, Castellanos A and Myerburg RJ. (2001). Sudden death due to cardiac arrhythmias. N Engl J Med. 345(20): 1473-1482.
- Das MK, Khan B, Jacob S, Kumar A, et al. (2006). Significance of a fragmented QRS complex versus a Q wave in patients with coronary artery disease. Circulation. 113(21), 2495-2501.
- Peters S, Trummel M and Koehler B. (2008). QRS fragmentation in standard ECG as a diagnostic marker of arrhythmogenic right ventricular dysplasia-cardiomyopathy. Heart Rhythm. 5(10): 1417-1421.
- Morita H, Kusano KF, Miura D, et al. (2008). Fragmented QRS as a marker of conduction abnormality and a predictor of prognosis of Brugada syndrome. Circulation. 118(17), 1697-1704.
- Flowers NC, Horan LG, Thomas JR, Tolleson WJ, et al. (1969). The anatomic basis for high-frequency components in the electrocardiogram. Circulation. 39(4): 531- 539.
- Friedman PL, Fenoglio JJ and Wit AL. (1975). Time course for reversal of electrophysiological and ultrastructural abnormalities in subendocardial Purkinje fibers surviving extensive myocardial infarction in dogs. Circ Res. 36(1): 127-144.
- Gardner PI, Ursell PC, Fenoglio JJ Jr and Wit AL. (1985). Electrophysiologic and anatomic basis for fractionated electrograms recorded from healed myocardial infarcts. Circulation. 72(3): 596-611.
- Das MK, Khan B, Jacob S, Kumar A, et al. (2006). Significance of a fragmented QRS complex versus a Q wave in patients with coronary artery disease. Circulation. 113: 2495-2501.
- Das MK, Suradi H, Maskoun W, Michael MA, et al. (2008). Fragmented wide QRS on a 12-lead ECG: A sign of myocardial scar and poor prognosis. Circ Arrhythm Electrophysiol. 1(4): 258-268.
- Das MK, Saha C, El Masry H, Peng J, et al. (2007). Fragmented QRS on a 12-lead ECG: a predictor of mortality and cardiac events in patients with coronary artery disease. Heart Rhythm. 4: 1385-1392.
- Goldenberg I, Zdzienicka J, Moss AJ, Zareba W, et al. (2007). Prognostic significance of fragmented QRS complex for predicting the risk of recurrent cardiac events in patients with Q-wave myocardial infarction. Am J Cardiol; 100(4): 583-586.
- Goldenberg I, Zdzienicka J, Moss AJ, Zareba W, et al. (2007). Prognostic significance of fragmented QRS complex for predicting the risk of recurrent cardiac events in patients with Q-wave myocardial infarction. Am J Cardiol; 100(4): 583-586
- Wang DD, Buerkel DM, Corbett JR, Gurm HS, et al. (2010). Fragmented QRS complex has poor sensitivity in detecting myocardial scar. Ann Noninvasive Electrocardiol. 15(4): 308-314.
- Carey MG, Luisi AJ Jr, Baldwa S, Al-Zaiti S, et al. (2010). The Selvester QRS Score is more accurate than Q waves and fragmented QRS complexes using the Mason-Likar configuration in estimating infarct volume in patients with ischemic cardiomyopathy. J Electrocardiol. 43: 318-325.
- Das MK, Michael MA, Suradi H, Peng J, et al. (2009). Usefulness of fragmented QRS on a 12-lead electrocardiogram in acute coronary syndrome for predicting mortality. Am J Cardiol. 104(12): 1631-1637.
- Schuller JL, Olson MD, Zipse MM, Schneider PM, et al. (2011). Electrocardiographic characteristics in patients with pulmonary sarcoidosis indicating cardiac involvement. J Cardiovasc Electrophysiol. 22(11): 1243-1248.
- Priori SG, Gasparini M, Napolitano C, Della Bella P, et al. (2012). Risk stratification in Brugada syndrome: results of the PRELUDE (Programmed ELectrical stimUlation preDictive valuE) registry. J Am Coll Cardiol; 59(1): 37-45.
- Haraoka K, Morita H, Saito Y, Toh N, et al. (2010). Fragmented QRS is associated with torsades de pointes in patients with acquired long QT syndrome. Heart Rhythm. 7(12): 1808-1814.
- Haraoka K, Morita H, Saito Y, Toh N, et al. (2010). Fragmented QRS is associated with torsades de pointes in patients with acquired long QT syndrome. Heart Rhythm. 7(12): 1808-1814.
- Wang J, Tang M, Mao KX, Chu JM, et al. (2012). Idiopathic ventricular fibrillation with fragmented QRS complex and J wave in resting electrocardiogram. J Geriatr Cardiol; 9(2): 143-147.
- Yamanari H, Nakayama K, Morita H, Miyazi K, et al. (2000). Effects of cardiac sympathetic innervation on regional wall motion abnormality in patients with long QT syndrome. Heart. 83(3): 295-300.
- Egidy Assenza G, Valente AM, Geva T, et al. (2013). QRS duration and QRS fractionation on surface electrocardiogram are markers of right ventricular dysfunction and atrialization in patients with Ebstein anomaly. Eur Heart J. 34(3): 191-200.
- Ari H, Cetinkaya S, Ari S, Koca V, et al. (2012). The prognostic significance of a fragmented QRS complex after primary percutaneous coronary intervention. Heart Vessels. 27(1), 20-28.
- Basaran Y, Tigen K, Karaahmet T, Isiklar I, et al. (2011). Fragmented QRS complexes are associated with cardiac fibrosis and significant intraventricular systolic dyssynchrony in nonischemic dilated cardiomyopathy patients with a narrow QRS interval. Echocardiography; 28: 62-68.
- Yu CM, Lin H, Zhang Q, Sanderson JE, et al. (2003). High prevalence of left ventricular systolic and diastolic asynchrony in patients with congestive heart failure and normal QRS duration. Heart. 89(1): 54-60.
- Rickard J, Zardkoohi O, Popovic Z, et al. (2011). QRS fragmentation is not associated with poor response to cardiac resynchronization therapy. Ann Noninvasive Electrocardiol. 16(2): 165-171.
- Brenyo A, Pietrasik G, Barsheshet A, Huang DT, et al. (2012). QRS fragmentation and the risk of sudden cardiac death in MADIT II. J Cardiovasc Electrophysiol. 23(12): 1343-1438.
- Tigen K, Karaahmet T, Gurel E, Cevik C, et al. (2009). The utility of fragmented QRS complexes to predict significant intraventricular dyssynchrony in nonischemic dilated cardiomyopathy patients with a narrow QRS interval. Can J Cardiol. 25(9): 517-522.
- Cheema A, Khalid A, Wimmer A, Bartone C, et al. (2010). Fragmented QRS and mortality risk in patients with left ventricular dysfunction. Circ Arrhythm Electrophysiol. 3(4): 339-344.
- Forleo GB, Rocca DGD, Papavasileiou LP, Panattoni G, et al. (2011). Predictive value of fragmented QRS in primary prevention implantable cardioverter defibrillator recipients with left ventricular dysfunction. J Cardiovasc Med. 12(11): 779-784.