Previous Issues Volume 4, Issue 3 - 2019
Prevalence and Factors Associated with Low Bone Mass and Osteoporosis in Syrian Postmenopausal Women
Nermeen Hijazi1, Zaynab Alourfi1,2*
1Department of Internal Medicine, Endocrinology, Damascus University, Syria
2Faculty of Medicine, Syrian Private University, Syria
Received Date: Sep 17, 2019
Published Date: Oct 04, 2019
*Corresponding author: Zaynab Alourfi, Department of Internal Medicine, Endocrinology, Damascus University, Damascus, Syria.
Copyright © 2019 Alourfi Z.
Citation: Alourfi Z. (2019). Prevalence and Factors Associated with Low Bone Mass and Osteoporosis in Syrian Postmenopausal Women. Mathews J Case Rep 4(3): 58.
ABSTRACT
Aim: Osteoporosis is a silent asymptomatic disease until it is complicated with a fracture. The aim of this cross-sectional study was to assess the prevalence of low bone mass and osteoporosis in postmenopausal women and determine their relationship with age, menopause duration, body mass index and other potential risk factors.
Methods: 813 postmenopausal women were included, aged between 40-96 yrs. Body weight, height, body mass index (BMI), body mineral density (BMD), were measured. Risk factors for low BMD derived from questionnaire were documented.
Results: Using world health organization criteria 24% had osteoporosis and 45.2% had low bone mass. Incidence of osteoporosis and low bone mass increased significantly with age and menopause duration. Femoral neck BMD decreased by 0.0055 g/cm2 and total lumbar BMD decreased by 0.0035 g/cm2 per year. However, increased BMI was associated with increasing in BMD in both femoral neck BMD and total lumbar BMD by 0.0056 g/cm2 and 0.0057 g/cm2 per 1 kg/m respectively.
Conclusions: The prevalence of low bone mass and osteoporosis in Syrian postmenopausal women was in alignment with results of most other studies. BMD in lumbar vertebrae and femoral neck is negatively correlated with age, duration of menopause, smoking, physical inactivity and the use of proton pump inhibitors. However, it is positively correlated with BMI.
Keywords: Bone mineral density; Low bone mass; Osteoporosis; Body mass index; Postmenopausal women.
Abbreviations: BMD: Body Mineral Density; BMI: Body Mass Index; DXA: Dual-energy X-ray Absorptiometry; PPI: Proton Pump Inhibitors.
INTRODUCTION
Osteoporosis is a systemic skeletal disease characterized by deterioration of bone micro-architecture leading to increased bone fragility and fractures risk. It is silent, asymptomatic until it is complicated with a fracture [1]. Diagnosis of low bone mass and osteoporosis increases with age. In women, due to estrogen deficiency, bone loss accelerates around the time of menopause [1]. About 200 million women are diagnosed world wild with osteoporosis yearly [2] and about 19 billion dollars was the cost of managing fractures in the United States of America in 2005 [3]. This heavy economic burden illustrates the early diagnosis and treatment of osteoporosis [4]. Hence the importance of early detection by conducting periodic surveys and modifying the adjustable risk factors associated with bone [3] low mineral density is the single best predictor of fracture risk in asymptomatic postmenopausal women [5,6], Dual-energy x-ray absorptiometry (DXA) of the hip and spine is the “gold standard” for measurement of BMD [7].
Despite the reports of the incidence of 4000 hip fractures yearly in Syria, information about the prevalence of osteoporosis and low bone mass are limited [8]. In this study, prevalence of osteoporosis in postmenopausal Syrian women was determined and compared with reports of other studies in the Middle East and the west. BMI, smoking, physical inactivity and the use of proton pump inhibitors (PPI) and loop diuretics were studied as risk factors.
METHODS
A cross-sectional study was conducted between November 2018 and March 2019 at Al-Mouwasat University Hospital, Damascus, Syria. Participants were apparently healthy postmenopausal women aged ≥40 yrs. Subjects who were previously diagnosed with osteoporosis or took medication proved to increase bone mineral density (such as Bisphosphonate, Hormone Replacement Therapy, Selective Estrogen Receptor Modulator, Strontium Ranelate, Calcitonin, PTH Analog) were excluded.
Information were collected by a questionnaire included potential risk factors for osteoporosis include: age, menopause duration, weight, height, work, exercise, cigarette smoke, maternal history of osteoporosis, and the use of thyroid hormone, proton pump inhibitors (PPI), and loop diuretics.
Weight (with light clothes) and barefoot height were measured using the Seca Scale Model 713 device (Boian Surgical, Padstow, Australia). Body mass index was calculated by the equation (BMI=weight (kg)/height (m2)) and used to categorize the participants according to the criteria by World Health Organization (WHO) as follows: BMI < 25 kg/m2 for normal weight, 25≤ BMI < 30 kg/m2 for overweight, and BMI ≥30 kg/m2 for obesity.
Total body measurement of BMD was made using a dual-energy X-ray absorptiometry (Medilink, MEDIX DR VER v4.0.3) at total lumbar spine (L1-L4) and left hip. Lumbar vertebrae were excluded from BMD measurement if degenerative diseases, fusions, or fractures were recorded at the study visit or noted in the image. The hip was not scanned when a positive history of fracture or surgery was present. The scanner was used by the same well trained nurse and calibrated daily against the standard calibration block supplied by the manufacturer to control possible baseline drift. The diagnosis of osteoporosis was done according to WHO criteria, using T-score values shown in Table 1.
Table 1: T-Score values and diagnosis by the WHO.
|
Diagnosis |
T score |
|
Osteoporosis |
T ≤ -2.5 |
|
Low bone mass |
-1≤ T < -2.5 |
|
Normal |
-1 > T |
Data analysis was performed using the SPSS software version 23.0 (IBM, Armonk, New York, USA), p values <0.05 were considered statistically significant. Mean, standard deviation and relative risk were calculated. The Chi square test was used for nominal data and Spearman’ Rho for correlation.
RESULTS
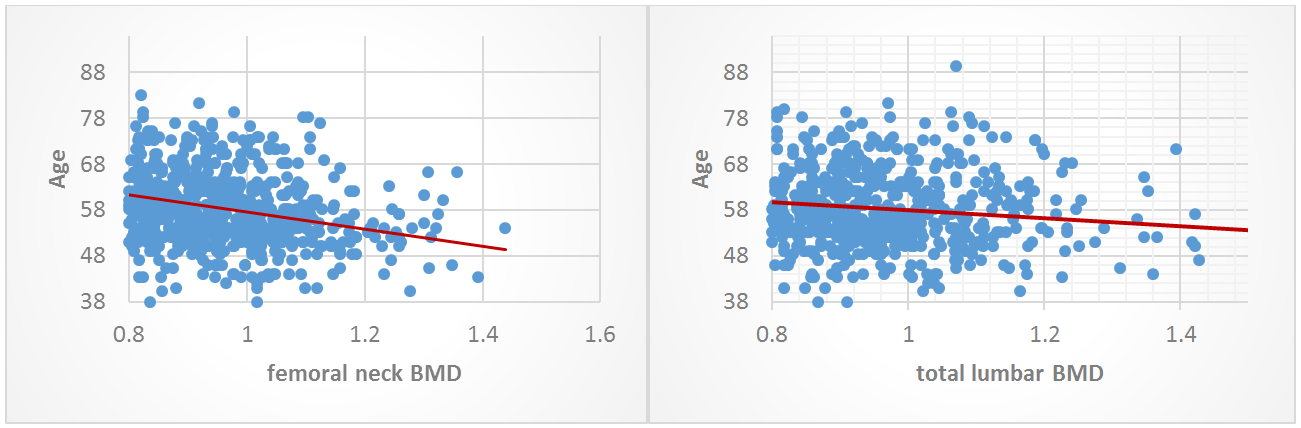
813 postmenopausal women were included, their mean age was 58.92 ± 8.6 yr, and their mean menopause duration was 10.1 ± 8.3 yr, with an average height of 152.16 ± 6.38 cm; weight 77.09 ± 14.51 kg; BMI 33.35 ± 6.13 kg/m2; total lumbar BMD 0.905 ± 0.162 g/cm2; and femoral neck BMD 0.928 ± 0.148 g/cm2. Osteoporosis was found in 195 women (24%) and low bone mass was found in 368 women (45.2%) while normal bone density was found in 250 women (30.8%). The incidence of osteoporosis and low bone mass increased significantly with age and menopause duration (p: 0.0001) (Table 2). By comparing age with total lumbar BMD and femoral neck BMD a statistically significant negative correlation was found (r: -0.32 p: 0.0001), (r: -0.165 p: 0.0001) respectively, with a decreased of 0.0055 g/cm2 of femoral neck BMD and 0.0035 g/cm2 of total lumbar BMD decreased per year (calculated by Slopeline equation) (Figures 1 and 2).
Figure 1: Correlation between age and femoral neck BMD. Figure 2: Correlation between age and total lumbar BMD.
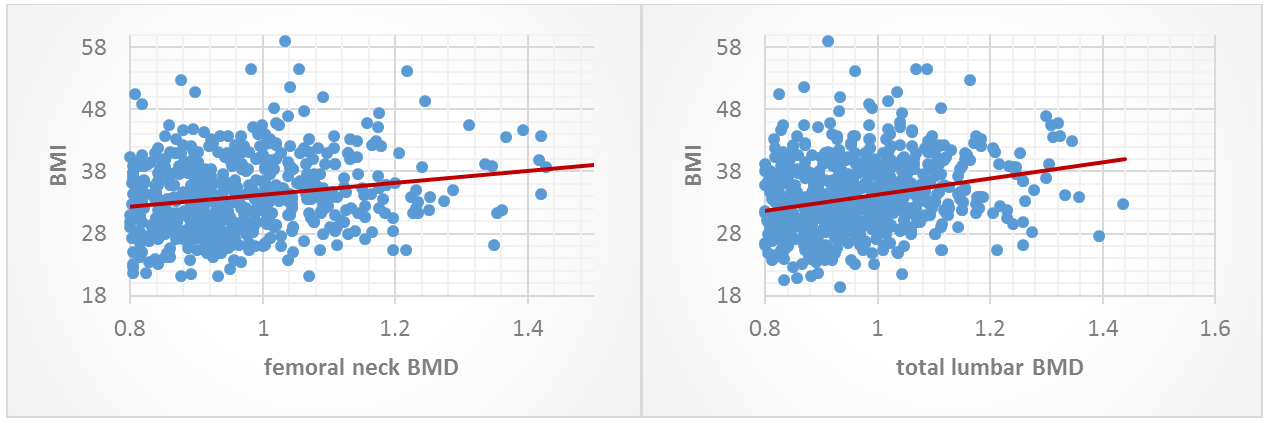
Obesity, over weight and normal weight prevalence in our study were 69.6%, 23.1% and 7.3% respectively. The normal weight group had the highest osteoporosis incidence with a significant difference (p: 0.0001) (Figures 3 and 4). Spearman’s rho showed a statistically significant positive correlation between BMI and total lumbar BMD and femoral neck BMD (r: 0.318 p: 0.0001) and (r: 0.252 p: 0.0001) respectively, with an increased ratio of 0.0056 g/cm2 of femoral neck BMD and 0.0057 g/cm2 of total lumbar BMD decreased per 1 kg/m2 (calculated by slope line equation). Smoking was significantly associated with increased prevalence of osteoporosis (p: 0.004), and while significant lower percentage of osteoporotic patients were among women who had regularly exercised (p: 0.046), higher percentage (not significantly important) of osteoporotic patients were among non-worker women (p: 0.367) (Table 2).
Figure 3: Correlation between BMI and femoral neck BMD. Figure 4: Correlation between BMI and total lumbar BMD.
Maternal history of osteoporosis and the use of loop diuretic or PPI were associated with increased prevalence of both osteoporosis and low bone mass but only the use of PPI had a significantly important association with the diagnosis (p: 0.016) (Table 2). On the other hand, the prevalence of osteoporosis and low bone mass were almost equal among women who took or didn’t take thyroid hormone therapy (p: 1) (Table 3).
Table 2: Characteristic of women screened for low bone mass and osteoporosis using DXA BMI: body mass index. PPI: proton pump inhibitors.
|
Normal |
Low bone mass |
Osteoporosis |
P-value |
||
|
Age group (Years) |
40-50 |
52(47.7%) |
44(40.4%) |
13(11.9%) |
0.0001† |
|
50-60 |
129(32.2%) |
179(44.6%) |
93(23.2%) |
||
|
60-70 |
50(23.3%) |
108(50.2%) |
57(26.5%) |
||
|
>70 |
19(21.6%) |
37(42%) |
32(36.4%) |
||
|
Menopause duration (Years) |
<10 |
167(38.7%) |
186(43%) |
79(18.3%) |
0.0001† |
|
10-20 |
68(22.5%) |
152(50.3%) |
82(27.2%) |
||
|
>20 |
15(19%) |
30(38%) |
34(43%) |
||
|
BMI Kg/m2 |
<25 |
1(1.7%) |
25(42.4%) |
33(55.9%) |
0.0001† |
|
25-30 |
46(24.5%) |
91(48.4%) |
51(27.1%) |
||
|
>30 |
203(35.9%) |
252(27.1%) |
111(19.6%) |
||
|
Smoke cigarette |
Yes |
43(23.6%) |
80(44%) |
59(32.4%) |
0.004† |
|
No |
207(32.8%) |
288(45%) |
136(21.6%) |
||
|
Exercise |
Yes |
103(35.8%) |
126(43.7%) |
59(20.5%) |
0.046† |
|
No |
147(28%) |
242(46.1%) |
136(26.9%) |
||
|
Work |
Yes |
79(34%) |
103(44.4%) |
50(21.6%) |
0.367 |
|
No |
171(29.4%) |
265(45.6%) |
145(25%) |
||
|
Maternal history of osteoporosis |
Yes |
44(27.5%) |
74(46.3%) |
42(26.2%) |
0.567 |
|
No |
206(31.6%) |
294(45%) |
153(23.4%) |
||
|
Use of thyroid hormone |
Yes |
40(30.5%) |
59(45%) |
32(24.4%) |
1 |
|
No |
210(30.8%) |
309(45.3%) |
163(23.9%) |
||
|
Use of PPI |
Yes |
46(30.5%) |
56(37.1%) |
49(32.5%) |
0.016† |
|
No |
204(30.8%) |
312(47.1%) |
146(22.1%) |
||
|
Use of loop diuretic |
Yes |
7(20.6%) |
16(47.1%) |
11(32.3%) |
0.324 |
|
No |
243(31.2%) |
352(45.2%) |
184(23.6%) |
||
|
† Statistically significant |
|||||
Table 3: Shows the relative risk of osteoporosis for the potential risk factors.
|
Risk factor |
RR* |
|
Smoke cigarette |
1.5 |
|
Not regularly exercise |
1.3 |
|
Non-worker |
1.16 |
|
Maternal history of osteoporosis |
1.12 |
|
Use of thyroid hormone |
1.02 |
|
Use of PPI |
1.47 |
|
Use of loop diuretic |
1.37 |
|
RR: relative risk |
|
DISCUSSION
Osteoporosis is currently considered one of the common metabolic diseases, resulting from the imbalance between bone resorption and formation. This reduces bone strength, micro-architecture disruption and increases risk of fractures [9]. This is to the best of our knowledge, the first study in Syria [8]. Among the 813 postmenopausal women (45.2%) had low bone mass and, nearly the fourth (24%) had osteoporosis. These results are aligned with those of many other studies from the Middle East and the west [10-17]. The incidence of osteoporosis in our study was higher than that reported in Kuwait [10] (19.3%), Qatar [11] (17%), Puerto Rican [12] (11.5%) and the United States of America [13] (7.2%) and lower than that reported in Jordan [14] (30%), Italy [15] (36.6%) and turkey [16] (30%). However, the prevalence of low bone mass among those studies and our study were approximately the same (Table 4).
Table 4: Comparison of this study with other similar studies.
|
Country & Authors |
N |
Osteoporosis prevalence |
Low bone mass prevalence |
|
Syria, 2019 |
813 |
24% |
45.2% |
|
Kuwait-Gupta, et al. [10] |
2296 |
19.3% |
45.6% |
|
Saudi Arabia-Al-Quaiz, et al. [17] |
362 |
39% |
|
|
Jordan-Shilbayeh, et al. [14] |
400 |
30% |
43% |
|
Qatar-Hammoudeh, et al. [15] |
147 |
17% |
43% |
|
Italy-Cipriani, et al. [15] |
3247 |
36.6% |
|
|
Puerto Rican-Noel, et al. [12] |
953 |
11.5% |
43% |
|
Turkey-Demir, et al. [16] |
1522 |
30% |
40% |
|
USA-Siris, et al. [13] |
2000160 |
7.2% |
39.6% |
Several factors should be taken into account in the evaluation of these findings, mostly related to genetic, environmental and lifestyle determinants. Given to the complex pathogenesis of the disease [18], difference in the genetic susceptibility to osteoporosis and fracture across countries is advocated as one of the main mechanisms [19-21]. The results of our study showed a significant negative correlation between age and BMD, with about 0.5% decreased of the total bone mass. This study focused on the entire post menopause phase, However, other prospective studies showed that rates of bone loss from the spine and hip were 1% to 2% of its total mass during the early post menopause phase and about 35% to 55% slower loss during the late menopause phase [22,23]. A significantly positive correlation between BMI and BMD was found in our study, with an increased ratio of 0.0056 g/cm2 of femoral neck BMD and 0.0057 g/cm2 of total lumbar BMD decreased per 1 kg/m2, which was consistent with many previous studies [24,25].
The mechanisms whereby adipose tissue exerts positive effects on BMD status are not entirely clear. The putative mechanism relevance of adipose tissue for skeletal integrity probably resides in the role of several adipokines in bone remodeling through effect on both formation and resorption [26,27]. On the other hand, the prevalence of osteoporosis was much lower in women who regularly exercised, reflecting the importance of exercise in prevention of osteoporosis [28]. Smoking was significantly associated with increased prevalence of osteoporosis in this study, which may be explained by recent date that indicate Tobacco influences bone mass indirectly through alteration of bodyweight, parathyroid hormone-vitamin D axis, adrenal hormones, sex hormones, and increased oxidative stress on bony tissues [29].
The risk ratio of having osteoporosis was 1.37 higher among women who took loop diuretic than the women who did not, the fact that Loop diuretics increase urinary calcium excretion by inhibiting the Na-K-2Cl co-transporter in the Loop of Henle of the kidney [30] may explain why chronic use of loop diuretics might result in higher rates of bone loss. The effect of PPIs on calcium absorption/metabolism has received much attention recently because of the important relationship between PPIs use and bone loss showed by several studies [31-33]. This was in consistence with our study which shows that the women who took PPIs had higher prevalence of diagnosing osteoporosis (RR: 1.47).
Limited animal and human studies suggest that gastric acid secretion can facilitate calcium absorption and ionized calcium release from insoluble calcium salts, and by that acid suppressants such as PPIs can decrease bone density. Also Limited experimental evidence indicates that PPIs may also potentially affect bone resorption by inhibiting the osteoplastic proton transport system; this may ameliorate the negative effect of PPIs increasing osteoporosis by decreasing calcium absorption [33].
CONCLUSION
The prevalence of low bone mass and osteoporosis in Syrian postmenopausal women was in alignment with results of most other studies. BMD in lumbar vertebrae and femoral neck is negatively correlated with age, duration of menopause, smoking, physical inactivity and the use of proton pump inhibitors. However, it is positively correlated with BMI.
REFERENCES
- Poole KES, Compston JE (2006) Clinical review: Osteoporosis and its management. 333(7581): 1251-1256.
- Gourlay M, Brown SA (2004) Clinical considerations in premenopausal osteoporosis. Arch Intern Med 164(6): 603-614.
- Cosman F, de Beur SJ, LeBoff MS, Lewiecki EM, Lindsay BT, S Randall, et al. (2014) Clinician’s guide to prevention and treatment of osteoporosis. Osteoporos Int 25(10): 2359-2381.
- Wang M, Bolland M, Grey A (2016) Management recommendations for osteoporosis in clinical guidelines. Clin Endocrinol (Oxf) 84(5): 687-692.
- Miller CJ, Bonnick SL, Rosen CJ (1996) Consensus of an international panel on the clinical utility of bone mass measurements in the detection of low bone mass in the adult population. Calcif Tissue Int 58(4): 207-214.
- Lips P (1997) Epidemiology and predictors of fractures associated with osteoporosis. Am J Med 103(2A): 3S-8S; discussion 8S-11S.
- Marcus R, Feldman D, Dempster DW, Luckey M, Cauley JA (2013) Osteoporosis (4th Ed.). Academic Press, Cambridge, MA, USA.
- International psoriasis Foundation (2011) The Middle East & Africa regional audit, epidemiology, cost & burden of osteoporosis in 2011.
- Lane NE (2006) Epidemiology, etiology, and diagnosis of osteoporosis. Am J Obstet Gynecol 194(2 Suppl): S3-11.
- Gupta R, Al-saeed O, Azizieh F, Albusairi A, Gupta P, Mohammed A (2012) Evaluation of bone mineral density in postmenopausal women in Kuwait. J Clin Densitom 15(2): 211-216.
- Hammoudeh M, Al-khayarin M, Zirie M, Bener A (2005) Bone density measured by dual energy X-ray absorptiometry in Qatari women. Maturitas 52(3-4): 319-327.
- Noel SE, Mangano KM, Griffith JL, Nicole C, Dawson-hughes B, Tucker KL (2017) Prevalence of osteoporosis and low bone mass among Puerto Rican older adults. J Bone Miner Res 33(3): 396-403.
- Siris ES, Miller PD, Barrett-Connor E, Faulkner KG, Wehren LE, Abbott TA, et al. (2015) Identification and fracture outcomes of undiagnosed low bone mineral density in postmenopausal women results from the national osteoporosis risk assessment. JAMA 286(22): 2815-2822.
- Shilbayeh S (2003) Prevalence of osteoporosis and its reproductive risk factors among Jordanian women. Osteoporos Int 14(11): 929-940.
- Cipriani C, Pepe J, Bertoldo F, Bianchi G, Cantatore FP, Corrado A (2018) The epidemiology of osteoporosis in Italian postmenopausal women according to the National Bone Health Alliance (NBHA) diagnostic criteria: a multicenter cohort study. J Endocrinol Invest 41(4): 431-438.
- Demir B, Haberal A, Geyik P, Baskan B, Ozturkoglu E, Karacay O, et al. (2008) Identification of the risk factors for osteoporosis among postmenopausal women. Maturitas 60(3-4): 253-256.
- Alquaiz AM, Kazi A, Tayel S, Ahamed Shaikh S, Al-Sharifet A, Othman S, et al. (2014) Prevalence and factors associated with low bone mineral density in Saudi women: A community based survey. BMC Musculoskelet Disord 15: 5.
- Pepe J, Cipriani C, Cantatore FP, Fabbri A, Pola E, Vinicola V (2017) The effect of parathyroid hormone (1-84) treatment on serum bone morphogenetic protein 4 and vascular endothelial growth factor in postmenopausal women with established osteoporosis. J Endocrinol Invest 40(6): 663-667.
- Urano T, Inoue S (2014) Genetics of osteoporosis. Biochem Biophys Res Commun 452(2): 287-293.
- Cauley JA, Chalhoub D, Kassem AM, Fuleihan GE (2014) Geographic and ethnic disparities in osteoporotic fractures. Nat Rev Endocrinol 10(6): 338-351.
- Alonso N, Ralston SH (2014) Unveiling the mysteries of the genetics of osteoporosis. J Endocrinol Invest 37(10): 925-934.
- Kanis JA, Odén A, Mccloskey EV, Johansson H, Wahl DA, Cooper C, et al. (2012) A systematic review of hip fracture incidence and probability of fracture worldwide. Osteoporos Int 23(9): 2239-2256.
- Finkelstein JS, Brockwell SE, Mehta V, Greendale GA, Sowers MR, Ettinger B, et al. (2008) Bone mineral density changes during the menopause transition in a multiethnic cohort of women. J Clin Endocrinol Metab 93(3): 861-868.
- Bjarnson NH, Christiansen C (2000) The influence of thinness and smoking on bone loss. J Clin Endocrinol Metab, 85(2): 590-596.
- Salamat MR, Salamat AH, Abedi I, Janghorbani M (2013) Relationship between weight, body mass index, and bone mineral density in men referred for dual-energy X-ray absorptiometry scan in Isfahan, Iran. 2013: 205963.
- Gomez-ambrosi J, Rodríguez A, Catalán V, Frühbeck G (2015) The bone-adipose axis in obesity and weight loss. Obes Surg 6(6): 273-286.
- Migliaccio S, Francomano D, Bruzziches R, Greco EA, Fornari R, Donini LM, et al. (2013) Trunk fat negatively influences skeletal and testicular functions in obese men: Clinical implications for the aging male. Int J Endocrinol 2013: 182753.
- Benedetti MG, Furlini G, Zati A, Mauro GL (2018) The effectiveness of physical exercise on bone density in osteoporotic patients. BioMed Research International 2018: 1-10.
- Al-bashaireh AM, Haddad LG, Weaver M, Chengguo X, Kelly DL, Yoon S (2018) The effect of tobacco smoking on bone mass: An overview of pathophysiologic mechanisms. J Osteoporos 2018: 1206235.
- Wiener CM, Brown CD, Hemnes AR (Eds) Harrison’s Principles of Internal Medicine (18th Ed.). McGraw Hill Professional, NY, USA.
- Nesgaard B, Birger P, Abrahamsen B (2016) Proton pump inhibitors and osteoporosis. Curr Opin Rheumatol 28(4): 420-425.
- Vestergaard P, Rejnmark L, Mosekilde L (2006) Proton pump inhibitors, histamine H2 receptor antagonists, and other antacid medications and the risk of fracture. Calcif Tissue Int 79(2): 76-83.
- Sheen E, Triadafilopoulos G (2011) Adverse effects of long-term proton pump inhibitor therapy. Dig Dis Sci 56(4): 931-950.