Previous Issues Volume 1, Issue 2 - 2016
Pathological Changes in Pulmonary Vasculature after Atrial Fibrillation Ablation
Jessica Huston1, Craig Selzman2, Lyska Emerson3, Leif Jensen4, Nathan Hatton5, Frederick Han6, John J Ryan6
1Department of Medicine, Veterans Affairs Medical Center, Salt Lake City, UT, United States.
2Division of Cardiothoracic Surgery, Department of Surgery, University of Utah, Salt Lake City, UT, United States.
3Department of Pathology, University of Utah School of Medicine, Salt Lake City, UT, United States.
4Department of Radiology, University of Utah School of Medicine, Salt Lake City, UT, United States.
5Division of Pulmonary Medicine, Department of Medicine, University of Utah, Salt Lake City, UT, United States.
6Division of Cardiovascular Medicine, Department of Medicine, University of Utah, Salt Lake City, UT, United States.
Corresponding Author: John J Ryan, Assistant Professor, Division of Cardiovascular Medicine, University of Utah Health Science Center, 30 North 1900 East, Room 4A100, Salt Lake City, UT 84132, United States, Tel: 801-585-2341; E-Mail: john.ryan@hsc.
Received Date: 22 Aug 2016 Accepted Date: 29 Aug 2016 Published Date: 31 Aug 2016
Copyright © 2016 Ryan JJ
Citation: Ryan JJ, Huston J, Selzman C, Emerson L, et al. (2016). Pathological Changes in Pulmonary Vasculature After Atrial Fibrillation Ablation. Mathews J Cardiol. 1(2): 005.
ABSTRACT
A well-known complication of radiofrequency ablation is pulmonary vein stenosis. We present a complex case of a patient with pulmonary vein occlusion, which developed within a few months after radiofrequency ablation for atrial fibrillation. The pathologic changes seen on lung biopsy, which developed in the months after the ablation are similar to those seen in pulmonary arterial hypertension. A 67-year-old Caucasian male with a history of rheumatoid arthritis and radiofrequency ablation for atrial fibrillation presented with hypoxic respiratory failure and hemoptysis. In the preceding months he had been treated for recurrent pneumonia and high dose corticosteroids for a histopathologic diagnosis of pulmonary capillaritis. A contrast-enhanced computed tomography of the chest revealed total occlusion of the left inferior, left superior, and right superior pulmonary veins with patent flow only through the right inferior pulmonary vein from the right lower lobe. Surgical specimens revealed vascular remodeling similar to Group 1 pulmonary arterial hypertension. This case is important because of the impressive radiographic imaging and histopathological changes seen in a patient with pulmonary veins stenosis and radiofrequency ablation
KEYWORDS
Pulmonary Vein Stenosis; Pulmonary Hypertension; Pulmonary Vascular Disease; Hypoxia.
INTRODUCTION
We present the case of a patient with known rheumatoid arthritis (RA) who underwent radiofrequency ablation (RFA) for atrial fibrillation (AF). Less than a year after the ablation the patient developed significant pulmonary vein occlusion involving three out of four pulmonary venous outflow tracts. The surgical lung specimens demonstrated marked vascular remodelling and other pathological changes consistent with an occlusive pulmonary vasculopathy.
CASE PRESENTATION
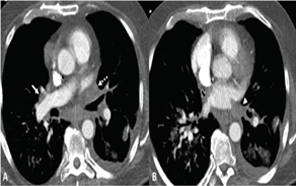
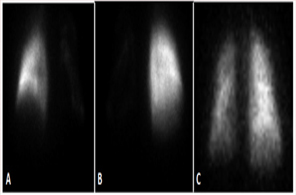
This case is a 67 year-old Caucasian male who presented with a chief complaint of “coughing up blood.” He had three-month history of hemoptysis and acute-on-chronic hypoxic respiratory failure. His past medical history consisted of RA and AF, having undergone RFA nine months prior to the onset of symptoms. His RA had been treated with methotrexate, sulfasalazine and hydroxychloroquine. In the three months prior to his admission, he had been treated for recurrent pneumonia. Admission physical exam revealed hypoxia with left sided basilar crackles and respiratory accessory muscle use. Contrastenhanced computed tomography of the chest revealed total occlusion of the left inferior, left superior, and right superior pulmonary veins with patent flow only through the right inferior pulmonary vein from the right lower lobe (Figure 1). Ventilation-perfusion scan revealed absence of perfusion in all lobes except the right lower lobe (Figure 2).
Figure 1:Contrast-enhanced CT at level of superior (A) and inferior (B) pulmonary veins (PV). The left superior PV (dashed arrow) is occluded, the right superior PV (white arrow) is severely narrowed. The left inferior PV (white arrow head) is occluded with clot extending into the left atrium. The right inferior PV (black arrow) is narrowed at the ostium.
Figure 2:Ventilation-perfusion scan: Tc-99 MAA perfusion scan. A. anterior, B. posterior C. Xe-133 ventilation scan.
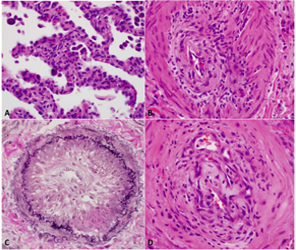
Due to increasing respiratory distress and clinical decompensation, the patient underwent surgical resection of infarcted lung tissue and pulmonary vein repair and was placed on extracorporeal membrane oxygenation. The lung resection specimens revealed a background diffuse interstitial mononuclear cell infiltrate and striking pulmonary vascular changes characterized by marked concentric intimal fibrosis and medial hypertrophy of small and medium size arteries. Occasional true vasculitis and complex re-canalization within vessels was present (Figure 3). Eccentric intimal fibrosis suggestive of possible thrombotic arteriopathy was also noted. During the post-operative course, the patient experienced fatal cerebrovascular event and expired.
Figure 3: A. Dense predominantly lymphocytic interstitial infiltrate of background lung. Hematoxylin and eosin stain, 40X magnification. B. Lymphocytic vasculitis of small artery with marked intimal fibrosis and medial hypertrophy. Hematoxylin and eosin stain, 40X magnification. C. Near-occlusive concentric intimal fibrosis of small pulmonary artery. Elastic-van Gieson stain, 40X magnification. D. Complex re-canalization of a small vessel with scattered inflammatory cells. Hematoxylin and eosin stain, 40X magnification.
Currently the mainstay of treatment of pulmonary vein stenosis consists of angioplasty and stenting to relieve proximal venous obstruction. Robbins and colleagues described two adult cases of pulmonary vein stenosis after AF ablation treated with angioplasty without stenting with relief of obstruction and symptoms in both patients [1]. These interventions relieve proximal obstruction but do not address the peripheral vascular remodelling that occurs due to pulmonary venous hypertension and occlusion. The pathologic changes found in this case report are noteworthy because they imply the potential for pulmonary vein stenosis to progress into a pathological phenotype consistent with Group 1 PAH, in this case in the setting of concomitant RA. Further pathological investigation is required to determine if the pulmonary vascular remodeling seen in this case report, in particular the concentric obliterative arteriopathy and vascular inflammation, are a feature of end-stage iatrogenic pulmonary vein stenosis from RFA.
CONCLUSION
Prior case reports of pulmonary vein stenosis have described pathologic specimens with intimal and medial hyperplasia indicating vascular remodelling secondary to elevated pulmonary pressures [2, 3]. In this case the patient had RA, and the striking diffuse lymphocytic cellular pneumonitis and capillaritis seen suggested RA involvement of the lung, yet the severity of the vascular changes seen is not commonly encountered in RA. This case is challenging because of the interplay between vascular arterial changes and changes induced in the pulmonary veins from RFA. The combination of complications of AF ablation on the hemodynamics within the pulmonary vascular bed superimposed on a pre-existing pulmonary disease and the interplay of these factors on the pulmonary vasculature has not been previously described.
AUTHORS CONTRIBUTIONS
JH drafted the manuscript and figures. CS aided in the conception and drafting of the manuscript. LE provided pathologic figures and descriptions as well as participated in drafting of the manuscript. LJ provided radiographic figures and descriptions. FH participated in drafting of the manuscript. NH helped to draft the manuscript. JJR conceived of the project and drafted the manuscript. All authors read and approved the final manuscript.
ACKNOWLEDGEMENT
The authors have no further acknowledgements.
REFERENCES
- Robbins IM, Colvin EV, Doyle TP, Kemp WE, et al. (1998). Pulmonary vein stenosis after catheter ablation of atrial fibrillation. Circulation. 98, 1769-1775.
- Yang HM, Lai CK, Patel J, Moore J, et al. (2007). Irreversible intrapulmonary vascular changes after pulmonary vein stenosis complicating catheter ablation for atrial fibrillation. Cardiovascular pathology, the official journal of the Society for Cardiovascular Pathology. 16, 51-55.
- Ernst S, Ouyang F, Goya M, Lober F, et al. (2003). Total pulmonary vein occlusion as a consequence of catheter ablation for atrial fibrillation mimicking primary lung disease. Journal of cardiovascular electrophysiology. 14, 366-370.