Information Links
Related Conferences
Previous Issues Volume 8, Issue 6 - 2023
More Insidious Development and Fulminant Malignant Hyperthermia: A Case Report
Luiz Eduardo Imbelloni1, Bruna Mukarzel2, Jorge Campello3, Thiago da Cruz4, Antônio Abílio de Santa Rosa5, Sylvio Valença de Lemos Neto6, Ana Cristina Pinho7, Anna Lúcia Calaça Rivoli8
1Senior Nacional Cancer Institute (INCA) Researcher, Rio de Janeiro, RJ, Brazil. Co-responsible for CET-SBA Hospital Clínicas Municipal de São Bernardo do Campo, SP, Brazil
2Resident in the 3rd year of Anesthesiology at INCA, Rio de Janeiro, RJ, Brazil
3Anesthesiologist at the National Cancer Institute (INCA), Brazil
4Anesthesiologist at the National Cancer Institute (INCA), Rio de Janeiro, RJ, Brazil
5Geneticist at the National Cancer Institute (INCA), Rio de Janeiro, RJ, Brazil. CEP-INCA Coordinator
6Anesthesiologist at the National Cancer Institute (INCA), Responsible for the CET-SBA of the National Cancer Institute, Rio de Janeiro, RJ, Brazil
7Anesthesiologist at the National Cancer Institute (INCA), Co-Responsible for the CET-SBA of the National Cancer Institute, Rio de Janeiro, RJ, Brazil
8Anesthesiologist at the National Cancer Institute (INCA), Responsible Coreme Residence INCA, Brazil
Corresponding Author: Dr. Luiz Eduardo Imbelloni, Senior Nacional Cancer Institute (INCA) Researcher, Rio de Janeiro, RJ, Brazil. Co-responsible for CET-SBA Hospital Clínicas Municipal de São Bernardo do Campo, Av. Epitácio Pessoa, 2356/203 – Lagoa, 22411-072- Rio de Janeiro, RJ – Brazil; Tel:+ 55.11.99429-3637; Email: [email protected]; [email protected]
Received Date: April 15, 2023
Publication Date: May 03, 2023
Citation: Imbelloni LE, et al. (2023). More Insidious Development and Fulminant MalignantHyperthermia: A Case Report. Mathews J Case Rep. 8(6):109.
Copyright: Imbelloni LE, et al. © (2023)
ABSTRACT
Background: Malignant hyperthermia (MH) is a pharmacogenetic condition, secondary to exposure to inhalational anesthetics and succinylcholine and related to the exacerbated release of calcium in the myoplasm, leading to hypermetabolism and rhabdomyolysis. The classical clinical presentations are of rapid onset and evolution. This case describes an MH atypical presentation. Case Report: Male, 41 years old, 85 kg, brown, admitted for a hepatic tri-segmentectomy to treat a Klatskin's tumor. Surgery was performed under balanced general anesthesia with sevoflurane and epidural block. The patient presented with hyperthermia, profuse sweating, need for a progressive increase in minute ventilation to control etCO2 values, metabolic lactic acidosis, and hyperkalemia, progressing to circulatory shock. The acute condition was controlled with dantrolene and supportive measures, but there was progression to death after one week. Conclusion: This case showed a rare presentation of MH. Despite having a slow and insidious onset, that progressed with greater severity than reported in other cases, culminating in the patient's death on the 7th postoperative day.
Keywords: Inhalational Anesthetics, Malignant Hyperthermia, Dantrolene, Epidural Anesthesia, Anesthetic Complications, Death
INTRODUCTION
Malignant hyperthermia (MH) is a pharmacogenetic disorder that manifests as a hypermetabolic response to virtually all potent inhaled agents, the depolarizing muscle relaxant, and, in humans, stressors such as vigorous exercise and heat. This situation, common in marathon runners and military, is also known as Exertional Heat Stroke. The key clinical features include unexplained elevation of expired carbon dioxide, despite increased minute ventilation, muscle rigidity, rhabdomyolysis, hyperthermia, tachycardia, acidosis, and hyperkalemia, with sudden onset [1]. The syndrome is likely to be fatal if untreated. The incidence of fulminant MH ranges from 1:62,000 to 1:84,000 in general anesthesia cases if succinylcholine and inhalation agents are used [2]. Usage of new modern anesthetic techniques resulted in the clinical presentation of malignant hyperthermia being more frequently indolent and/or insidious rather than truly fulminant, as was already known in the anesthetic community, with reports of four cases [3]. We present one recently referred case to illustrate this point, presenting with late-onset clinical picture, 4 hours after sevoflurane use, with profuse sweating, slowly progressive increase in CO2 (42-50 mmHg), increase in temperature, and need for successive doses of rocuronium.
CASE REPORT
The case report was registered on Plataforma Brasil (CAEE: 64036722.5.0000.5274) and approved by Institutional Review Board (Number: 5.752.387) and written informed consent was obtained. Male, 41 years old, 85 kg, brown, admitted to hospital National Cancer Institute (INCA) for a hepatic tri-segmentectomy to treat a Klatskin's tumor. With no other comorbidities or known allergies, the patient had previously been submitted to anesthesia, however, he never to any halogenated agent or succinylcholine. He had external biliary drainage performed in the previous month, uneventfully. Preoperative laboratory without relevant changes.
The proposed anesthetic technique was balanced general anesthesia with sevoflurane and epidural block. Continuous epidural blockade was performed under venous sedation with midazolam 5 mg, fentanyl 150 µg, ketamine 15 mg, and propofol 50 mg., The puncture was performed between T9-T10 and 10 ml ropivacaine 0.375% was injected without intercurrences. After blockade installation, anesthetic induction was performed with fentanyl 250 µg, lidocaine 100 mg, propofol 100 mg, and rocuronium 100 mg. Orotracheal intubation was performed under direct laryngoscopy with tube 8 and the initial ventilator parameters were: volume-controlled ventilation, tidal volume 550 ml, PEEP 8, RR 16, FiO2 30%. Throughout the surgery, other doses of local anesthetic were administered: lidocaine 2% 10 ml and ropivacaine 0,5% 10 ml.
Sevoflurane was started at a low dose (CAM 0.5), and the dose was increased approximately 1 hour after induction (CAM 0.8). Dexmedetomidine was used in an infusion pump as an adjuvant (0,2 µg/kg/h to 0.4 µg/kg/h). The patient began to show an increase in etCO2, maintaining a rate between 42 - 50 mEq/L at the expense of a high minute volume (about 10 to 12 L/min), and the need to administer multiple doses of rocuronium for adequate surgical relaxation. About 4 hours after sevoflurane administration, the patient developed an increase in temperature and profuse sweating, which remained high despite the removal of the thermal blanket and exposure of a significant body area (open abdominal surgery).
Initially, fever due to bacteremia was considered, secondary to manipulation of the biliary tract, and piperacillin-tazobactam (4.5 grams) and 2 grams of dipyrone were administered. Without improvement in temperature, blood gas analysis collected before performing hepatic ischemia already showed that the patient began to evolve with metabolic acidosis associated with significant hyperlactatemia.
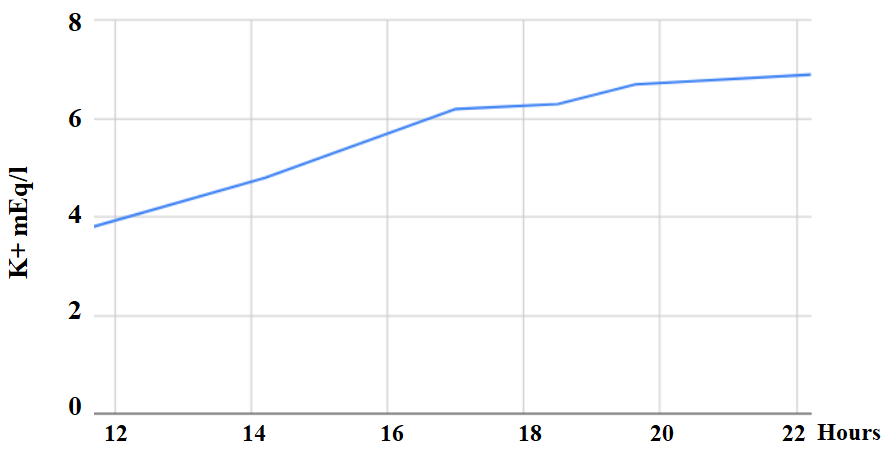
The patient showed a progressive increase in potassium in the blood gases collected since the beginning of the procedure (Figure 1). Laboratory results in 8 hours after the start of exposure showed potassium at 8 mEq/L and phosphorus at 6.8 mEq/L. Measures were taken to control hyperkalemia (glycoinsulin - two steps of regular insulin 10 IU + 100 ml of hypertonic glucose and 10% calcium gluconate 20 ml).
Table 1: Evaluation of blood gas analysis in time, before and after Dantrolene.
|
ARTERIAL BLOOD GASES |
11:40 h |
17:00 h |
19:38 h |
22:13 h |
|
Dantrolene |
Before |
Before |
30 min After |
3 hours After |
|
pH |
7.32 |
7.24 |
7.11 |
7.08 |
|
pCO2 (mmHg) |
45.9 |
44 |
40 |
46 |
|
pO2 (mmHg) |
88 |
168 |
444 |
396 |
|
HCO3 (mmol/L) |
23.6 |
29 |
34 |
13.9 |
|
Basic excess |
- 2.4 |
- 8.7 |
- 16.6 |
- 16.1 |
|
Oxygen saturation (%) |
95 |
98 |
99 |
99 |
|
Lactate (mmol/L) |
0.6 |
4.3 |
8.2 |
9.1 |
|
Volume-minute (ml/kg) |
7 |
14 |
12 |
7 |
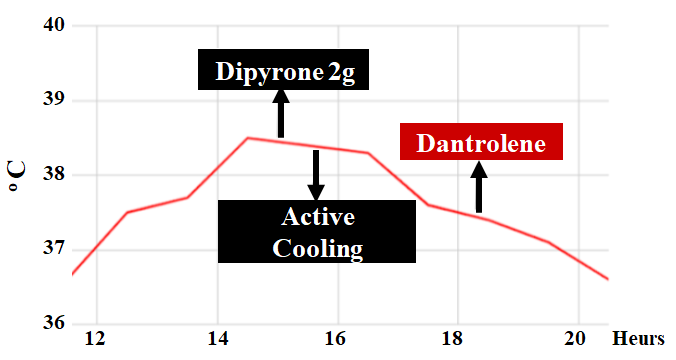
The patient evolved with worsening of metabolic acidosis and hyperlactatemia, associated with circulatory shock with the need to start high-dose amine (norepinephrine 0.5 to 0.7 µg/kg/min) and sodium bicarbonate 8.4% 40 ml was administered. At this moment, the diagnosis of malignant hyperthermia was considered, conversion to total intravenous anesthesia was performed, the flow of fresh gases increased, the ventilatory circuit and soda lime were changed, and active cooling of the patient was started using frozen saline bags administered dantrolene 2.5 mg/kg (total dose 212.5 mg).
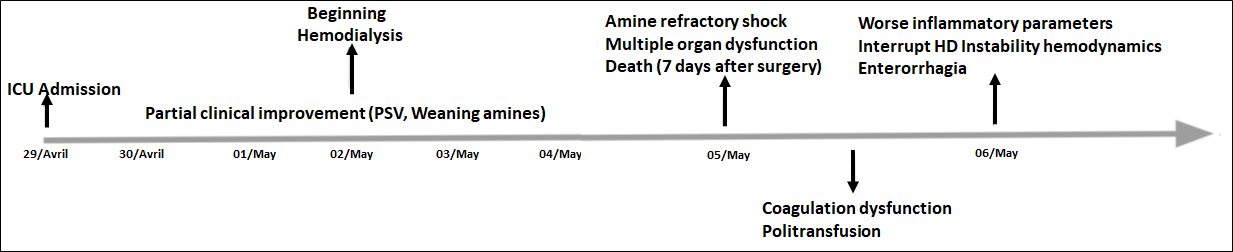
The patient evolved with improvement in hyperthermia, enabling a steady reduction of minute volume, reaching values of 8 ml/kg without a significant increase in etCO2 (Figure 2). However, he maintained hemodynamic shock and metabolic and electrolyte changes despite the measures employed. He was referred to the ICU care, in serious general condition, maintaining a picture of mixed acidosis, with evolution to liver failure and acute renal failure requiring dialysis. He evolved to amine-refractory shock and multiple organ dysfunctions, followed by death 7 days after the procedure (Figure 3). The family members were referred to genetic counseling.
Figure 2: Evaluation of temperature over time and ducts.
Figure 3: Chronological line of clinical evolution during the period of stay in the ICU.
DISCUSSION
In a prospective analysis of all telephone calls to the Brazilian hotline service for MH, from January to December 2009, 22 calls were received, showing that the number of calls per year is still low and the characteristics of the crises were similar to those described in other countries [4]. In Brazil, there is no current data on MH incidence. It is estimated that in 2009 about 1:50,000 episodes occurred for patients undergoing general anesthesia how much one would expect 77 cases reported in this period [5]. The present case is about a probable malignant hyperthermia crisis of insidious development and with some attenuated clinical signs, we believe that this form of appearance made the diagnosis difficult. The patient was young, with good functional capacity, and without reports of having previously undergone general anesthesia. He did not have any other comorbidity besides the Klatskin tumor and laboratory alterations related to it (elevation of canalicular enzymes). Despite the slow and insidious onset and fast implementation of treatment by a medical team, the patient progressed to death.
Malignant hyperthermia is a pharmacogenetic disorder that manifests as a hypermetabolic response to exposure to triggering agents: halogenated inhalational anesthetics (halothane, sevoflurane, isoflurane, desflurane) and succinylcholine, a depolarizing neuromuscular blocker [1].
The clinical features of MH reflect disturbances of calcium homeostasis by defects in genes and proteins involved in excitation-contraction coupling generating the dihydropyridine-ryanodine receptor complex, which acts as the main calcium release channel in muscles [6]. Malfunction of this complex causes constant activity of the channel function. This consequent overload of calcium in the sarcolemma induces maintained muscle contraction leading to sudden and potentially lethal rhabdomyolysis.
MH may occur at any time during anesthesia as well as in the early postoperative period [7]. Initially, the increase in temperature was considered bacteremia, secondary to manipulation of the biliary tract. The use of antibiotics and antipyretics did not improve, and blood gas analysis was performed showing metabolic acidosis associated with hyperlactatemia. The initially developed etCO2 elevation was difficult to correct, only successfully adjusting the minute volume to 10-12 L/min, quite high values, was it possible to correct the etCO2 elevation, with the need to change the soda lime twice during the surgery. Despite the classic description of MH with sudden and uncontrolled increases in etCO2, in this case, the onset was insidious and slow, making early diagnosis difficult. Small increases of etCO2 (2-3 mm Hg) were treated with small increases in minute volume [8], unsuccessful conduct in the case.
Early postoperative fever in the absence of signs of hypermetabolism is unlikely to be MH [7]. If it occurs, postoperative MH with signs and symptoms that begin more than one hour after the interruption of anesthesia, it is probably such a rare presentation of this disease that it should justify the investigation of alternative diagnoses [7]. This case showed a rare presentation of MH.
A large spectrum of manifestations from subclinical to fulminant has always been reported, and elevated temperature may be an early MH sign [3]. Between 1992 and 2011, 373 Canadian individuals with adverse anesthetic reactions were referred to the MH Unit [9]. One hundred twenty-nine proband survivors of adverse anesthetic reactions were selected, whose MH susceptible status was confirmed by caffeine halothane contracture testing [10]. And the authors agree with previous studies that early diagnosis and rapid dantrolene treatment reduce MH-associated complications.
Guidelines published in 2021 on MH, suggest that there are no specific clinical features of malignant hyperthermia, and the condition may prove fatal unless it is recognized in its early stages and treatment is promptly and aggressively implemented [11].
CONCLUSION
It is important for anesthesiologists to be aware of this change in the clinical presentation profile of MH, provided by modern anesthetic techniques such as total intravenous, free opioid, and free halogenated inhalational anesthetics. It is essential to always have a high index of suspicion, especially in the case of atypical clinical conditions such as the one described, and to consider that the diagnostic criteria used for the classic presentation of MH crises may not be applicable to all atypical cases. This case showed a rare presentation of MH. That, despite having a slow and insidious onset, progressed with greater severity than reported in other cases, culminating in patient's death on the 7th postoperative day.
REFERENCES
- Rosenberg H, Pollock N, Schiemann A, Bulger T, Stowell K. (2015). Malignant hyperthermia: A review. Orphanet Journal of Rare Diseases. 10(93): 1-19.
- Halliday NJ. (2003). Malignant hyperthermia. J Craniofac Surg. 14(5): 800-802.
- Heytens L, Forget P, Scholtès JL, Veychemans F. (2015). The changing face of malignant hyperthermia: less fulminant, more insidious. Anaesth Intensive Care. 43(4): 506-511.
- da Silva HC, Almeida Cdos S, Brandão JC, Nogueira e Silva CA, de Lorenzo ME, Ferreira CB, et al. (2013). Malignant Hyperthermia in Brazil: Analysis of Hotline Activity in 2009. Rev Bras Anestesiol. 63(1): 13-26.
- Ministry of Health. SUS Hospital Information System (SIH/SUS).
- Melzer W, Dietze B. (2001). Malignant hyperthermia and excitation-contraction coupling. Acta Physiol Scand. 171: 367-378.
- Litman RS, Flood CD, Kaplan RF, Kim YL, Tobin JR. (2008). Postoperative malignant hyperthermia: an analysis of cases from the North American Malignant Hyperthermia Registry. Anesthesiology. 109(5): 825-829.
- Karan SM, Crowl F, Muldoon SM. (1994). Malignant hyperthermia masked by capnographic monitoring. Anesth Analg. 78: 590-592.
- Larach MG, Gronert GA, Allen GC, Brandom BW, Lehman EB. (2010). Clinical presentation, treatment and complications of malignant hyperthermia in North America from 1987 to 2006. Anesth Analg. 110: 498-507.
- Riazi S, Larach MG, Hu C, Wijeysundera D, Massey C, Kraeva N. (2014). Malignant hyperthermia in Canada: Characteristics of index anaesthetics in 129 malignant hyperthermia susceptible probands. Anesth Analg. 118: 381-387.
- Hopkins PM, Girard T, Dalay S, Jenkins B, Thacker A, Patteril M, et al. (2021). Malignant hyperthermia 2020. Guideline from the Association of Anaesthetists. Anaesthesia. 76: 655-664.