Information Links
Related Conferences
Previous Issues Volume 6, Issue 1 - 2022
Fibrillation and Flutter of the Heart, Pathological Aspects of the Mechanism
Khabchabov RG*, Makhmudova ER
Candidate of Medical Sciences, Dagestan State Medical University, Russian Federation
*Corresponding Author: Khabchabov RG, Candidate of Medical Sciences,"Dagestan State Medical University" of the Ministry of Health of the Russian Federation, Makhachkala, Russian Federation. Tel: +79634136720 E-mail: [email protected]
Received Date: July 08, 2022
Published Date: Aug 20, 2022
Citation: Khabchabov RG, et al. (2022). Fibrillation and Flutter of the Heart, Pathological Aspects of the Mechanism. Mathews J Cardiol. 6(1):23.
Copyright: Khabchabov RG, et al. © (2022).
ABSTRACT
The article is scientific, theoretical and debatable, based on a deep analysis of the electrical activity of the heart and electrocardiogram data. Paroxysmal arrhythmia applies; paroxysmal tachycardia, flutter and fibrillation of the heart. The combination of these arrhythmias with structural changes in the heart should cause damage to the antioxidant barrier of source of generation. The article has a lot of pictures that should visually show you the cause and mechanism of the development of paroxysmal arrhythmias. But do not forget that the article and theoretical, written only for scientists.
Keywords: paroxysmal tachycardia, fibrillation, heart flutter.
INTRODUCTION
The title of the article with a question, what do we know about the cause and mechanism of development of paroxysmal tachycardia, flutter and fibrillation of the heart? Since ancient times, patients are treated by trial and error, because the mechanism of their development is unknown. What can the doctor say when the patient asks - why and how did paroxysmal tachycardia develop? And what can a student or professor say about this?
For example, in Russia, each student begins to study electrocardiography from the textbook Murashko V.V., Strutinsky A.V. "Electrocardiography" [4]. At the very beginning of the textbook there is a chapter on the “automatism of the heart,” it says in bold — the myocardium is devoid of automatism! But having reached the head of "heart fibrillation", the authors wrote — each group of myocardial muscles becomes a kind of ectopic source of generation! How to understand this, the authors contradict themselves? You can find many articles or textbooks written by professors, in which it is written that with paroxysmal tachycardia, the myocardium is the source of generation.
It has been proven by science that the myocardium conducts electricity, but it cannot generate, this is true!
Ignorance of the cause and mechanism of the development of paroxysmal arrhythmias led to confusion in the whole educational process. I can understand the chaos Murashko V.V. and Strutynsky AV, they were confused by research articles of 19th century. Here is one of them - Morphological substrate, which creates the risk of ventricular fibrillation after myocardial infarction, is a zone of myocardium bordering necrotic tissue, formed from islands of viable myocardial fibers and connective tissue interlaced [1].
We were very pleased with the article by Professor David Kass. In a study conducted by Professor David Kass [2], it was found that an enzyme called PDE-5 is responsible for disorders in the main cardiac pathways in patients with heart disease. In the same study, scientists found that the PDE-9 enzyme is responsible for disorders in other pathways of the heart. Earlier studies have shown that an excess of PDE-5 causes damage to the cardiac pathway, interfering with the cGMP signaling molecule and the PKG protein. In a further study, scientists found that an excess of PDE-9 causes heart failure, fibrillation and cardiac arrest, interfering with cGMP in the cardiac pathways. PDE-9 accelerates the breakdown of cGMP, which reduces the production of PKG protein. Scientists note that drugs that block PDE-9 activity are being tested for Alzheimer's disease and can be used to treat heart failure.
Our article states that there is an antioxidant barrier in the heart between the myocardium and pathways with ectopic generation nodes. Damage to this barrier, in combination with structural heart disease, is the cause of the development of paroxysmal arrhythmias.
The structural changes that determine the development of paroxysmal arrhythmias may be acute myocardial infarction, hypertrophy and dilatation of the ventricles, inflammation and swelling of myocardial tissue. These changes, according to many researchers, constitute an anatomical substrate with different mechanisms for the occurrence of paroxysmal arrhythmias. [6].
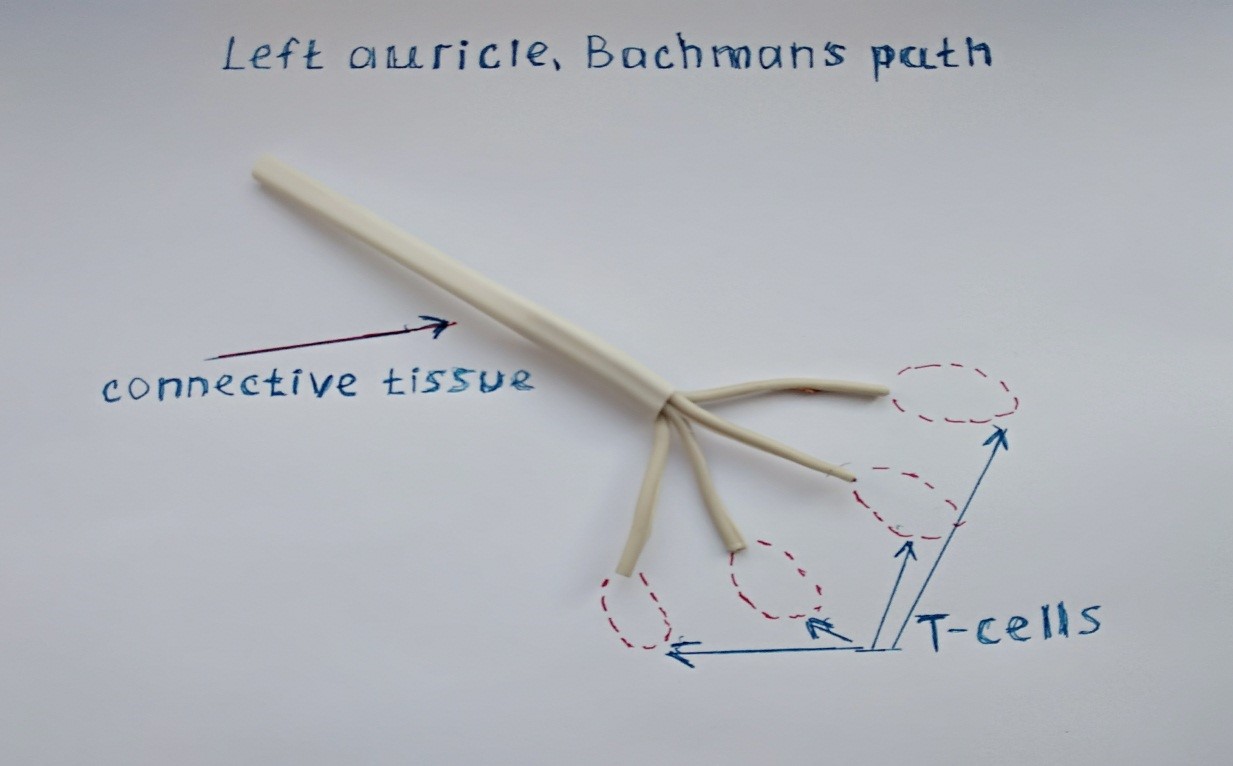
In the myocardium, the extracellular fluid contains a large amount of Na + and Cl- ions, especially during hypoxia and acidosis, the acidic environment prevails there, and the alkaline medium prevails in the pathways with ectopic nodes. To prevent the strong oxidation of ectopic nodes with the development of paroxysmal arrhythmias, there is an antioxidant barrier in the form of a connective tissue (fibrous) insulating sheath. This envelope starts from the sinus node and goes to Purkinje fibers, and Purkinje fibers envelop antioxidant T-cells (Fig. 1)
Figure 1: The antioxidant barrier of the pathways of connective tissue and T – cells.
And the structure of the pathways looks like this (Fig. 1-3), as an example, we have shown the left atrium with the Bachmann tract.
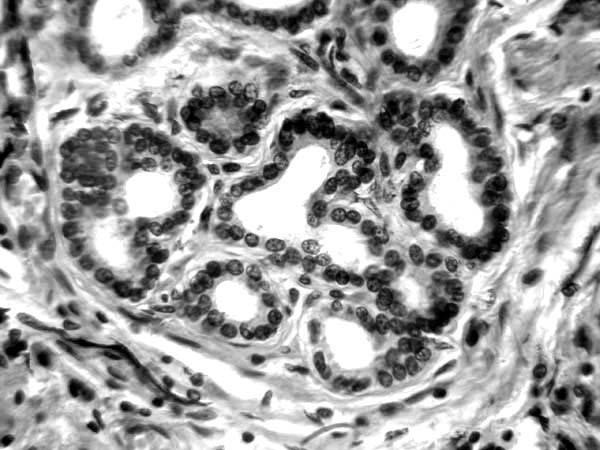
A microscopic examination of Purkinje fibers showed that many T-cells form a stable antioxidant wall (Fig. 2) [3].
Figure 2: Histology of T-cells Purkinje fibers.
Figure 3. 1: Ectopic generation nodes, 2, 5 - insulating connective tissue, 3 - Purkinje fibers (B cells), 4 - T – cells.
The famous researcher Purkinje described T - cells as conducting electricity between B - cells (Purkinje) and the myocardium. We believe that the main function of T - cells is the continuation of the protective barrier of Purkinje fibers against oxidation. It is the connective (fibrous) membrane and T-cells that create the antioxidant barrier of the cardiac pathways (Fig. 3). The destruction of this barrier is also possible with PDE 9-5 enzymes, which was described by Professor David Casse [2].
What happens after oxidation of the near, upper ectopic source of generation? Damage to the connective tissue occurs in the form of pores and cracks, then we will see only paroxysmal tachycardia on the ECG, most of the electricity will go to the Purkinje fibers and naturally trigger myocardium (Fig. 4).
Figure 4: The formation of pores or cracks in the fibrous sheath, the arrow indicates the oxidation path to the ectopic source of generation.
The damage to the oxidative barrier of the connective tissue can be significant (Fig. 5); most of the electrical impulse will be released to the myocardium through the damage window. A powerful wave of macro-reentri is formed in the myocardium; on the ECG we will see large F waves of atrial flutter.
Figure 5. 1: Large damage (window) in the fibrous membrane, ascending oxidation (arrow). 2 - ectopic focus generation. 3 - The release of a large wave of macro-reentri.
Fibrillation of the heart does not form one powerful release, as in trembling. Damage to the distal branches of the connective tissue or transitional T-cells will lead to the oxidation of 2-3 distal ectopic sites less powerful than proximal ones. This will form several micro-reentri waves that will collide with each other, these movements will be messy and chaotic (Fig. 6).
Figure 6. 1 - Damage to the fibrous membrane and T - cells. 2 - Branches and foci of ectopia (black dots). 3 - Wave micro-reentri. 4 - Focus of necrosis.
In some cases, the oxidation goes up and down along the conductive paths, then the oxidation of the proximal and distal nodes of generation occurs, this will lead to a combination of trembling and heart fibrillation.
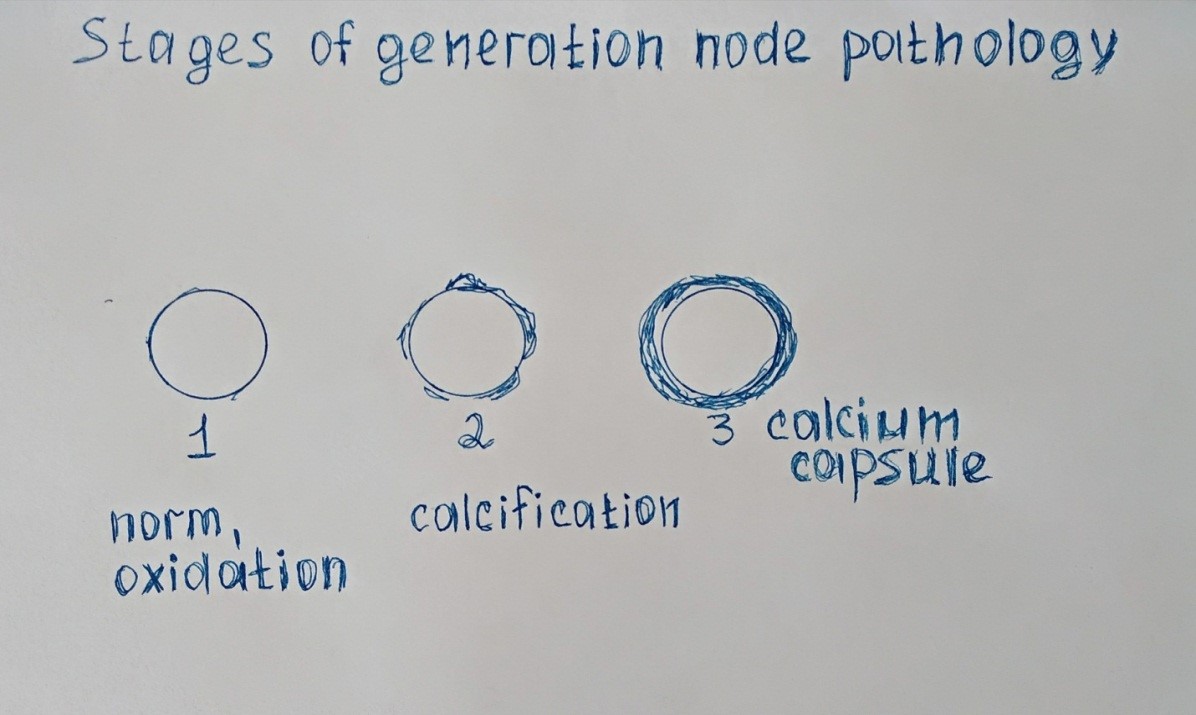
As you have noticed, at first the fibrillation has a high frequency of contractions (tachysystole), it takes 1-2 years and the frequency of contractions decreases (normysystole). What is the reason for this treatment? No, less treatment, more pathology of the node generation. At the beginning, the generation unit is irritated by oxidation, after 6-12 months, calcification forms on the surface, and then a calcium capsule (Fig. 7).
Figure 7: Stages of the pathology of the ectopic site of generation.
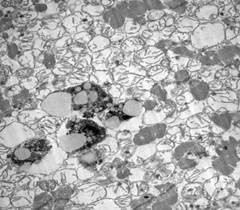
We were able to obtain the control autopsy material of the ectopia of the left atrium node of a man who died from a non-cardiac cause, as well as the material of a deceased patient with atrial fibrillation during life [3]. Electron microscopy results are shown for comparison (Fig. 8 and 9).
Figure 8: Electron microscopy of the normal state of the ectopic cells of the heart node. Mitochondria are visible with intact cristae inside the mitochondria of the cell [3].
Figure 9: Histology of the transitional type of cells of the cardiac conduction system in the area of ectopia of the left atrial node in a deceased patient with coronary heart disease and atrial fibrillation. Swelling of multiple mitochondria and destruction of mitochondrial cristae, calcification in mitochondria, which corresponds to the degeneration of cell ectopia.
DISCUSSION
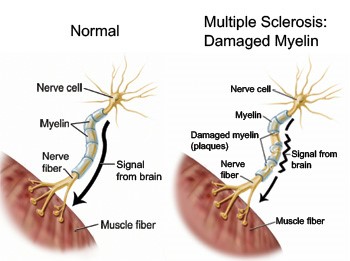
If you think that the mechanism of development of paroxysmal arrhythmias written by us is new in medicine, then you are mistaken! The described mechanism is similar to damage to the myelin sheath of axons in multiple sclerosis. In axons under the myelin sheath, an alkaline environment prevails, and acidic mycotoxins damage the myelin sheath, which covers and protects the nerves [5].
The myelin barrier helps to quickly carry out impulses from brain cells to various organs along axons, protecting axons from damage. Due to the fact that ion currents cannot pass through myelin, the entrance and exit of ions is carried out only in the area of interceptions. This leads to an increase in the speed of the nerve impulse (Fig. 10). Thus, the impulse is conducted in myelin fibers about 5-10 times faster than without the myelin layer [5].
Figure 10: Damage to the myelin layer.
CONCLUSION
We are confident that the treatment of paroxysmal arrhythmias in structural heart diseases should be comprehensive. To do this, you can use antiarrhythmic drugs, antioxidants and reparaty (restore the structure of the myocardium and pathways) [6,7].
REFERENCES
1. Kulakowski P. (2001). Ventricular signal averaged electrocardiography. Risk of Arrhythmia and Sudden Death. Ed / Kulakowski P // by Malik M. - London. Р: 167-179.
2. Lee DI, Zhu G, Sasaki T, Cho GS, Hamdani N, et al. (2015). Phosphodiesterase 9A controls nitric-oxide-independent c GMP and hypertrophic heart disease. Nature. 519: 472-47.
3. Mironova TF, Mironov VA, Kuvatova EV, Kuvatov VA. (2007). High-resolution analysis of heart rate variability in patients with angina during coronary artery bypass grafting. Modern problems of science and education. 6: 16-25.
3. Murashko VV, Strutynsky AV. (2000). Electrocardiography: a training manual. 4th edition. MEDpress. P: 312.
4. Miller DH, Barkhof F, Frank JA, Parker GJ, Thompson AJ. (2002). Measurement of atrophy in multiple sclerosis: pathological basis, methodological aspects and clinical relevance. Brain. 125(8): 1676-1695.
5. Savelieva IV. (1997). Stratification of patients with ventricular arrhythmias in groups of sudden death. IV Savelyev Cardiology. 8: 82-96.
6. Khabchabov RG, Makhmudova ER. (2016). Antiarrhythmic effect of antioxidants in patients with atrial fibrillation. J Atr Fibrillation. 8(6):1360.
7. Khabchabov RG, Makhmudova ER. (2018). Combined treatment of paroxysmal forms of atrial fibrillation and flutter. Herald of new medical technologies. Electronic edition. 5: 35-41.