Information Links
Related Conferences
Previous Issues Volume 5, Issue 2 - 2020
Endoscopic Keyhole and Microsurgery Approach to Cerebellopontine angle Tumors: Surgical outcomes
Orestes López Piloto*, Tania Margarita Cruz Hernández, Gabriela Tania Liñán Barreto, Claudia Díaz Villalvilla Flores, Orestes López Ayala, Ernesto Torres Garcia
Neurology and Neurosurgery Institute, Havana, Cuba
*Corresponding Author: Orestes López Piloto, Neurology and Neurosurgery Institute, Havana, Cuba, Email: [email protected]
Received Date: November 09, 2020 Publication Date: December 27, 2020 Copyright: Piloto OL, et al. (2020). Citation: Piloto OL, et al. (2020). Endoscopic Keyhole and Microsurgery Approach to Cerebellopontine angle Tumors: Surgical outcomes. Mathews J Case Rep. 5(2):65.
ABSTRACT
Objective: To evaluate the results of endoscopic approach versus the conventional approach in the surgical treatment of pontocerebellar angle tumors.
Methods: An observational, comparative, ambispective and cross-sectional study was carried out in 105 patients with tumors of the pontocerebellar angle, approached at the Neurology and Neurosurgery Institute (NNI) in the period between January 2015 and December 2019. They used clinical, surgical, pathological, auditory and imaging variables.
Results: 28.6% of the cases and 30% of the controls were between 50 and 59 years old. 60% of the cases and 57.1% of the controls were women. The tumor was on the right side in 68.6% of the cases and 67.1% of the controls. Hearing loss was found in 80% and 84.3% of cases and controls, respectively. Schwannoma was found in 82.9% of cases and 85.7% of controls. Postoperative facial function was classified as grade I in 60% and 48.6% of cases and controls, respectively, while auditory was classified as class I in 74.3% and 61.4% for both groups, respectively. The frequency of complications was 40% in cases and 51.4% in controls.
Conclusions: The age group of 50-59 years prevails in the study, female sex, hearing loss and headache as a clinical picture and schwannoma as a histological variant. The retrosigmoid endoscopic keyhole achieves a greater degree of tumoral resection, postoperative facial and auditory function compared to the conventional approach. Facial paralysis is identified as the most frequent complication.
KEYWORDS: Keyhole; Cerebellopontine angle; Endoscopic; Microsurgery
INTRODUCTION
The first successful excision of a tumor in cerebellopontine angle region was a vestibular schwannoma performed by Dr. Charles Ballance in 1894 [1]. Harvey Cushing described the cerebellopontine angle tumor syndrome in 1917 [2] and Walter Dandy in 1934, favored the total removal of the vestibular schwannoma, being the first neurosurgeon to achieve total resection of a vestibular schwannoma with a relatively low operative mortality [3-5].
In 1960 William House introduced the surgical microscope by describing the translabyrinthine approach to treat cerebellopontine angle tumors, emphasizing the early identification and importance of preserving the facial nerve [6]. Vestibular schwannomas represent 78% of tumors in this region and most originate from the vestibular branch of the eighth cranial nerve [7,8].
The development of endoscopic techniques, has contributed to a significant improvement in surgical results. The endoscopic technique has shown significant advantages in the treatment of various lesions using different approaches [9-11].
The Retrosigmoid Keyhole for cerebellopontine angle surgery has been well studied and applied in tumor resection, vascular decompression and aneurysm surgery [12-14]. The advantage of this technique is given in the context of minimally invasive craniotomies, increased visualization of hidden anatomical areas and inspection around all surgical angles [15,16].
METHODS
An observational, comparative, ambispective and cross-sectional study was conducted in 105 patients with cerebellopontine angle tumors, who underwent surgery at the Neurology and Neurosurgery Institute (NNI) in the period between January 2015 and December 2019.
Inclusion criteria
1. Patients over 18 years of age.
2. Patients of either sex.
3. Any type of tumor of the cerebellopontine angle.
4. Patients who express their written willingness to enter the study by signing the informed consent.
Exclusion criteria
1. Patients with non-tumor conditions of the cerebellopontine angle.
2. At the time of inclusion they present any chronic disease or in a decompensation phase (Example; heart disease, diabetes, arterial hypertension).
The sources of information were the clinical history of each patient operated on for a cerebellopontine angle tumor included in the study. The information collection models were included in a data collection sheet in which all the general information related to the study was filed. A 95% confidence interval was estimated for a significance level of p < 0.05.
To assess the degree of tumor resection, the Kanzaki et al [17] classification was used. This classifies tumor resection into four types, taking into account the subjective intraoperative opinion of the neurosurgeon, which in this study was modified by measuring the volume by performing MRI in the first 72 hours of surgery.
Total resection: when no tumor remains were left. The tumor was completely removed.
Almost total resection: when a volume of less than 2% of tumor remains was left.
Subtotal resection: when a volume between 2 and 5% of tumor remains was left.
Partial resection: when a volume greater than 5% of tumor remains was left.
Facial function was assessed before surgery, one week after the intervention, and 3 months later, using the House-Brackmann [11] classification, which establishes the categories or degrees of nerve dysfunction.
Auditory function was evaluated before surgery, one week after the intervention and three months later, the classification by logo audiometry [18,19] was used, which establishes four categories or classes of dysfunction. The study was carried out in accordance with the provisions of the Declaration of Helsinki, modification of Fortaleza, Brazil, on research in humans. This study was examined and approved by the bioethics commission of the Institute of Neurology and Neurosurgery [20].
RESULTS Similar behavior was observed with a predominance of the age group between 50 and 59 years old with 10 (28.6%) years in the group of endoscopic approaches and 21 (30%) in the group of microsurgical approaches. The mean age in the first group was 52.04 ± 9.17 with a minimum of 20 and a maximum of 67 years and for the second it was 52.89 ± 9.11 with a minimum of 19 and a maximum of 69 years, not statistically significant results.
Both in the endoscopic approaches group and in the microsurgical approaches group, a predominance of women was observed with 60% (n = 21) and 57.1% (n = 40), respectively, results without statistical significance.
The laterality of the tumor, was mostly right in the group with endoscopic approaches with 24 (68.6%) and microsurgical approaches group with 47 (67.1%), results not statistically significant.
Regarding the clinical manifestations, the most frequent was hearing loss with 80% (n = 28) in the endoscopic approaches group and 84.3% (n = 59) in microsurgical approaches group, followed by headache with 42.6% (n = 15) among those approached by endoscopy and 44.2% (n = 31) among those approached with a microscope and tinnitus with 34.3% (n = 12) for the first group and 31.4% (n = 22) for the second, results without statistical significance.
The main tumor diagnosis in both groups was schwannoma, with 29 (82.9%) for the group with endoscopic approaches and 60 (85.7%) for the group with microsurgical approaches, results without statistical significance.
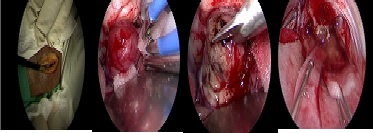
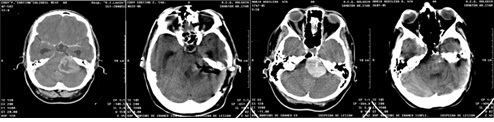
In the patients in the group approached by endoscopy, resection was total in 23 (21.8%) patients, almost total in 7 (6.6%), subtotal in 4 (3.8%) and partial in 1 (0.9%) (Figure 1). In the control group it was total in 31 (29.5%), almost total in 15 (14.7%), subtotal in 11 (10.4%) and partial in 13 (12.3), (Figure 2) results without statistical significance p 0.072. The patients studied underwent a postoperative CT scan in the first 24 hours after surgery, as well as an MRI of the skull in the first 72 hours after surgery, where the pre and postoperative volume was compared to define the degree of resection.
Figure 1: Intraoperative picture and procedure with Gross Total Resection using Key Hole Retrosigmoid endoscopy approach.
Figure 2: Pre and Postoperative CT scan image and result of Microsurgical approach with Gross Total Resection and big bone defect.
|
|
Grade Tumor Resection |
||||
|
APPROACH |
Gross Total |
Almost total |
Subtotal |
Partial |
TOTAL |
|
Endoscopic Keyhole |
No % 23 21.8 |
No % 7 6.6 |
No % 4 3.8 |
No % 1 0.9 |
No % 35 33.4 |
|
Microsurgical |
31 29.5 |
15 14.7 |
11 10.4 |
13 12.3 |
70 66.6 |
|
Total |
54 51.3 |
22 21.3 |
15 14.2 |
14 13.2 |
105 100 |
Table 1: Degree of tumor resection in both groups. p < 0.05. Test Fischer: p 0.072.
According to the degree of postoperative facial function in those approached by endoscopy, 60% (n = 21) were classified as grade I, 5.7% (n = 2) as grade II, and 17.1% (n = 6) as grade III , 11.4% 8n = 4) as grade IV and 2.9% (n = 1) as grade V and VI, respectively, on the other hand, in the group approached with a microscope, 48.6% (n = 34) were classified as grade I , 15.7% (n = 11) as grade II, 12.9% (n = 9) as grade III, 10% (n = 7) as grade IV, 8.6% (n = 6) as grade V and 4.3% (n = 3) as grade VI, results that presented significance when statistically analyzed.
Regarding the degree of postoperative auditory function in those approached by microscope, 74.3% (n = 26) were classified as class I, 14.3% (n = 5) as class II, 11.4% (n = 4) as class III and 5.7% (n = 2) as class IV, while, in those approached by microscope, 61.4% (n = 43) were classified as class I, 17.1% (n = 12) as class II, the 12.9% (n = 9) as class III and 8.6% (n = 6) as class IV, statistically significant results.
Complications occurred in 14 (40%) of the group with endoscopic approaches and in 36 (51.4%) of the microsurgical approaches, results with statistical significance. In both groups, a predominance of facial paralysis was observed, which occurred in all patients in the group approached by endoscopy, as well as in all patients in the group approached by microscope. The rest of the complications also behaved similarly in the two groups.
DISCUSSION
Cerebellar angle tumors are more frequent between the fifth and sixth decades of life 21. The mean age of the patients with cerebellar angle tumors studied by He et al [12] was 55.8 years, with a range between 36 and 78 years and a predominance of those aged 50 or over with 69.4% (n = 34). The patients with cerebellopontine angle tumors included in the series by Larrañaga Sigwald [13] had a mean age of 51 years; with ages that were between 32 and 74 years.
The greatest affectation of the female sex in this series is a fact that has been previously reported in the literature. Shirvani et al [18] found that of the 29 patients with cerebellopontine angle tumors, 75.8% (n = 22) were female and 24.2% (n = 7) male. In the study by Attia et al [19] of the patients with cerebellopontine angle tumors, 63% (n = 19) were women and 37% (n = 11) were men.
In our research and in accordance with the literature reviewed, hearing loss, headache, facial paresis or paralysis, vomiting and alterations in balance, are the clinical manifestations found most frequently in patients diagnosed with tumors of the angle cerebellum [22-24].
The pathological anatomy of cerebellopontine angle tumors according to Díaz Anadón et al [25] reported that in 163/170 (95.9%) patients the diagnosis was vestibular schwannoma, in 5/170 (2.9%) meningioma and in 2/170 (1.2 %) remaining cases, it was a choristoma and an arachnoid cyst, respectively.
The goal of surgical treatment is to perform complete tumor resection, although there is some terminological controversy as to what it means to perform a complete resection; it can mean an extracapsular excision or even an apparently complete excision on intraoperative visualization, but with tumor remains in the surgical bed not macroscopically visible [22,25].
In this investigation, the degree of total tumor resection achieved in patients operated on endoscopically was higher than those operated on conventionally. Shirvani et al [18] the percentage of resection of cerebellopontine angle tumors was total in 79.3% and subtotal in 17.2%. Wackym et al [26] reported more than 96% success in the endoscopic total resection of cerebellopontine angle tumors. In a series of 178 vestibular schwannomas operated endoscopically, Gerganov et al [27] found that total tumor resection was not achieved in only 1 (0.6%) patient. For their part Kankane et al [28] report that of the 224 cases with a diagnosis of cerebellopontine angle tumor treated by endoscopy, total tumor resection was achieved in 79.3%. Hasegawa et al [29] report a total tumor resection percentage of 86.5% after endoscopic surgery for cerebellopontine angle tumor.
Although these results are superior to those found in our study, they coincide in demonstrating the benefits of the endoscopic approach to achieve total tumor resection in these cases, due to the better visualization of the areas that are blind for microscopy and around the corners. In addition, this approach reduces the need for retraction of the cerebellum and the release of CSF to address the cerebellopontine angle and perform total tumor removal [30,31].
Due to its proximity to the VIII cranial nerve, the facial nerve is susceptible to being damaged by different mechanisms. Some patients with normal facial function present with subclinical preoperative dysfunction, which can be demonstrated on electromyography. During surgery, the facial can be injured immediately by physical mechanisms (manipulation, stretching or section). It can also suffer thermal or vascular damage. Delayed alterations in facial function may be due to entrapment of the nerve in the meatal foramen, edema, or vasospasm [32-34].
The functional results found by He et al [12] report that 5 (10.2%) patients presented a House-Brackmann grade II, another 4 (8.2%) a grade 3 and 3 (6.1%) a grade IV-V. The remaining 75.5% (n = 37) were classified as grade I. Similarly, the functional results reported by Marchioni et al [35] showed that after endoscopic resection of cerebellopontine angle tumors, 3/20 (15%) patients presented House-Brackmann grade II transient facial palsy that recovered in the follow-up period, while the remaining 17 (85%) showed normal facial function.
Also at discharge, patients with vestibular schwannoma who received endoscopic resection by del Río Arroyo et al [36] presented grade I facial nerve function in 28 (35.4%) patients, grade II in 17 (21.5%), grade III; IV and V in 7 (8.9%), respectively and grade VI in 13 (16.5%). Similarly, Samii et al [37] published 98.5% anatomical preservation and 81% functional preservation grade I-III according to House-Brackmann, in 200 patients with cerebellopontine angle schwannoma, operated by keyhole endoscopic retrosigmoid approach.
On the other hand, in the work published by Kabil and Shahinian [38] it was observed that in a total of 112 patients with a diagnosis of vestibular schwannoma who underwent endoscopic tumor resection, anatomical preservation was achieved with grade I functionality III of the facial nerve in 100%. Likewise, according to the report by Yang et al33 in the 19 (100%) patients operated on for a cerebellopontine angle tumor endoscopically, the anatomical integrity of the facial nerve was maintained and according to the functionality one week after surgery, 3 (15.8%) were classified as grade I, another 10 (52.6%) as grade II and the remaining 6 (31.6%) as grade III, results that in both cases were higher than those achieved in this thesis.
Hearing preservation is an important aspect in the quality of life of patients with cerebellopontine angle tumors, and regardless of tumor size, its preservation should be attempted in all patients with useful hearing before surgery [39]. As observed in this series, the endoscopic approach to the cerebellopontine angle allows a better visualization of the structures included within it, which has repercussions, as in this investigation, in a better preservation of the VIII nerve and a better degree of function postoperative hearing than in the conventional approach.
In the group of patients operated on for cerebellopontine angle tumor, in the series by Agarwal et al, [40] maintenance of hearing was achieved in 75.3% of the patients after endoscopic resection. In the study by Kankane et al [28], in patients with cerebellopontine angle tumor who received endoscopic tumor resection, preservation of the vestibulocochlear nerve was achieved in 83% of cases, while hearing loss was found in 35.3%.
Surgery is not without potential risks and complications, even vital ones, especially the injury to the facial nerve. Although, both the injury to this nerve and that of the different cranial nerves and other vascular structures is determined more in relation to the tumor size than to the type of approach, the patients operated with Endoscopic Keyhole presented fewer complications than those operated via conventional.
The complications reported by Khan et al [23] after endoscopic resection of cerebellar angle tumors were the deterioration of facial nerve function in 7.6% of patients, CSF fistula and hydrocephalus in 5.4%, each of them and intracranial hematoma in 3.6%.
After endoscopic resection of cerebellopontine angle tumors, Huang et al [24] found 186 (28.3%) cases with balance alterations as complications, (9.3%) with tinnitus, 313 (47.6%) with hearing loss and 250 (44.2%) with permanent facial paralysis. Other complications reported were meningitis in 50 (7.6%) patients, lower cranial nerve deficit in 49 (7.5%), CSF fistula in 18 (2.7%), and intracranial hematoma in 8 (1.2%). In the series by Nonaka et al [41] the main complication presented by patients undergoing endoscopic surgery for cerebellar angle schwannoma was facial paralysis in 14%.
CONCLUSIONS Histologically the predominant tumor was schwannoma in both groups. Using the keyholeretrosigmoid endoscopic approach, a gross total tumor resection was achieved in most of cases (21.8%), compared with 29.5% in the Microsurgery group, as well as a degree of postoperative facial function with a House-Brackmann I in 60% of the cases and CLASS I hearing function of 74.3% compared to those approached by microsurgery with results of 48.6% and 61.4% respectively. The most frequent complication observed was facial paralysis in 40% of the cases approached by endoscopy and 51.4% in microsurgery approached.
DECLARATION OF CONFLICTING INTERESTS
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
FUNDING
The author(s) received no financial support for the research, authorship, and/or publication of this article.
REFERENCES
- Brackmann DE, Arriaga MA. (2018). Extra-axial neoplasms of the posterior fossa. In: Cummings CW, Gates GA, editors. Otolaryngology. Head & neck surgery. Missouri: Mosby. 3294-313.
- Cushing H. (1963). Tumors of the nervus acusticus and the syndrome of the cerebellopontile angle (reprint of the 1917 edition). New York: Hafner Publishing Company.
- Dandy W. (1934). Removal of cerebellopontile (acoustic) tumors through a unilateral approach. Arch Surg. 29:337–344.
- Bebin J. (2018). Pathophysiology of acoustic tumors. In: House WF, Luetje CM, editors. Acoustic tumors, vol. 1. Baltimore: University Park Press. 45-83.
- Canalis R, Lambert P. (2015). The ear: comprehensive otology. Lippincott Williams & Wilkins, Philadelphia 2015.
- Al-Mefty O, Pravdenkova S, Gragnaniello C. (2010). A technical note on endonasal combined microscopic endoscopic with free head navigation technique of removal of pituitary adenomas. Neurosurg Rev. 33:243-9.
- Komotar RJ, Starke RM, Raper DM, Anand VK, Schwartz TH. (2011). The endoscope-assisted ventral approach compared with open microscope-assisted surgery for clival chordomas. World Neurosurg. 76:318-27.
- Hori T, Maruyama T, Chernov M. (2015). Clinical experience with endoscope-controlled removal of intrameatal vestibular schwannomas. In: Kanno T, Kato T (eds): Minimally Invasive Neurosurgery and Neurotraumatology. Tokyo: Springer. 1760-802.
- Betka J, Chovanec M, Zverina E, Profant O, Lukes P, et al. (2016). Minimally invasive endoscopic and endoscopy-assisted microsurgery of vestibular schwannoma. In: Iancu C (ed): Advances in Endoscopic Surgery. Rijeka, Croatia: InTech. 191-216.
- Little AS, Almefty KK, Spetzler RF. (2014). Endoscopic surgery of the posterior fossa: strengths and limitations. World Neurosurg. 82:322-24.
- House JW, Brackmann DE. (1985). Facial nerve grading system. Otolaryngol Head Neck Surg. 93:146-7.
- He X, Liu W, Wang Y. (2017). Surgical management and outcome experience of 53 cerebellopontine angle meningiomas. Cureus. 9(8):e1538.
- Larrañaga Sigwald L. (2018). Control postoperatorio y seguimiento del neurinoma acústico intervenido por vía suboccipital retrosigmoidea. Trabajo de fin de grado de Medicina. Universidad de San Sebastián. País Vasco, España.
- Pikis S, Margolin E. (2016). Malignant transformation of a residual cerebellopontine angle epidermoid cyst. J Clin Neurosci. 33:59–62
- Master AN, Roberts DS, Wilkinson EP, Slattery WH, Lekovic GP. (2018). Endoscope-assisted middle fossa craniotomy for resection of inferior vestibular nerve schwannoma extending lateral to transverse crest. Neurosurg Focus. 44(3):E7.
- Moosa S, Ding D. (2015). Role of stereotactic radiosurgery in the management of cerebellopontine angle tumors. Austin J Radiat Oncol & Cancer. 1(1): 1004.
- Kanzaki J, Tos M, Sanna M, Moffat DA, Monsell EM, et al. (2003). New and modified reporting systems from the consensus meeting on systems for reporting results in vestibular schwannoma. Otology & neurotology: official publication of the American Otological Society, American Neurotology Society [and] European Academy of Otology and Neurotology. 24(4):642-8; discussion 8-9.
- Shirvani M, Shahmohammadi M, Kabir NM, Arjmand Y. (2017). Surgical management of patients with cerebellopontine angle meningiomas in a 15 year period. Sci J Neurol Neurosurg. 3(2):48-51.
- Attia M, Alaghoy I, El Hawary M, Abo-Shosha M. (2018). Endoscopic assisted microscopic skull base surgery. Open Journal of Modern Neurosurgery. 8:187-200.
- OMC. Debate sobre Declaración de Helsinki. Última modificación de la Declaración de Helsinki, ¿qué beneficios aporta? Rev OMC. 2009 [citado 16 Jun 2014]; (9):29-34.
- Chen BS, Roberts DS, Lekovic GP. (2016). Endoscopic-assisted middle fossa craniotomy for resection of vestibular schwannoma. J Neurol Surg Rep. 77:e001–e007.
- Jawad MSM. (2017). Cerebellopontine angle tumors; tumor size and surgical outcome. Open Access J Neurol Neurosurg. 3(4).
- Khan B, Ikramalam AM, Khanzada K. (2018). Surgical complications following removal of cerebello-pontine angle (CP) lesions. Pak J Neurol Surg. 22(4):183-6.
- Huang X, Xu J, Xu X, Chen M, Ji K, et al. (2017). Functional outcome and complications after the microsurgical removal of giant vestibular schwannomas via the retrosigmoid approach: a retrospective review of 16-year experience in a single hospital. BMC Neurology. 17:18.
- Díaz Anadon A, Lassaletta L, Roda JM, Gavilán Bouzas J. (2009). Fistulas de LCR tras la cirugía de tumores del ángulo pontocerebeloso y su relación con el índice de masa corporal. Acta Otorrinolaringol Esp. 60(5):318-24.
- Wackym AP, King WA, Poe Dennis S, Meyer GA, Ojemann RG, et al. (1999). Adjunctive use of endoscopy during acoustic neuroma surgery. Laryngoscope. 109(8):1193-201
- Gerganov VM, Giordano M, Herold C, Samii A, Samii M. (2010). An electrophysiological study on the safety of the endoscope-assisted microsurgical removal of vestibular schwannomas. EJSO. 36:422-7
- Kankane VK, Warade AC, Misra BK. (2019). Nonvestibular schwannoma tumors in the cerebellopontine angle: A single surgeon experience. Asian Journal of Neurosurgery. 14(1):154-61.
- Hasegawa M, Nouri M, Nagahisa S. (2016). Cerebellopontine angle epidermoid cysts: clinical presentations and surgical outcome. Neurosurg Rev. 39(02):259-66,discussion 266-7.
- Matthies C. (2018). Functional microsurgery of vestibular schwannomas in; Ramina R, Aguiar PHP, Tatagiba M. Samii’s essentials in Neurosurgery. Springer-Verlag Heidelberg. 189-203.
- Martuza RL. Suboccipital Retrosigmoid Surgical Approach for Vestibular Schwannoma (Acoustic neuroma). In; Hinojosa AQ. Schmidek & Sweet operative neurosurgical techniques: indications, methods, and results. Elsevier Saunders, 2017.
- Cruz García O. (2006). Abordajes quirúrgicos combinados a la base craneal posterior estudio de 44 lesiones en 42 pacientes. Tesis para optar al grado científico de Doctor en Ciencias Médicas. Instituto Superior de Ciencias Médicas de la Habana. La Habana, Cuba. 2006.
- Yang SM, Yu LM, Z YH, Yu LM, JI Fei, et al. (2009). Endoscope-assisted cerebellopontine angle surgery. J Otology. 4(1):44-9.
- Yamakami I, Ito S, Higuchi Y. (2014). Retrosigmoid removal of small acoustic neuroma: curative tumor removal with preservation of function. J Neurosurg. 4:1-10.
- Marchioni D, Alicandri-Ciufelli M, Rubini A. (2015). Endoscopic transcanal corridors to the lateral skull base: initial experiences. Laryngoscope. 125(suppl 5):S1-S13.
- Del Río Arroyo L. (2009). Factores predictivos de la función facial tras la cirugía del schwannoma vestibular. Tesis doctoral. Universidad Autónoma de Madrid. Madrid, España.
- Samii M. (2018). Vestibular schwannomas. In: Springer-Verlag, editor. Surgery of cerebellopontine lesions. Berling Heidelberg. 147-304.
- Kabil MS, Shahinian HK. (2006). A series of 112 fully endoscopic resections of vestibular schwannomas. Minim Invas Neurosurg. 49:362-8.
- Moffat DA, Parker RA, Hardy DG, Macfarlane R. (2014). Factors affecting final facial nerve outcome following vestibular schwannoma surgery. J Laryngol Otol. 128(5):406-15.
- Agarwal V, Babu R, Grier J, Adogwa O, Back A, et al. (2013). Cerebellopontine angle meningiomas: postoperative outcomes in a modern cohort. Neurosurg Focus. 35(6):E10
- Nonaka Y, Fukushima T, Watanabe K. (2013). Contemporary surgical management of vestibular schwannomas: analysis of complications and lessons learned over the past decade. Neurosurgery. 72:103–15. discussion 115.