Previous Issues Volume 2, Issue 2 - 2017
Cutaneous Vasculitis as A First Sign of Isolated Peptoniphilus Spp in Urinary Tract Infection: A Case Report
Eduardo Esteban-Zubero1*, Rubén Castejón-Moreno1, Moisés Alejandro Alatorre-Jiménez2, Lourdes del Carmen Rizo-De La Torre3, Daniel Barberá-Pérez4 , Subramanian Panchu Ravindra Rajan Subala5 , Alejandro Marín-Medina3 , Sara Anabel Alonso Barragán6 , Carlos Arturo López-García7 .
1Medical Doctor in Emergency Department of Hospital San Pedro. Calle Piqueras, 98, 26006, Logroño, Spain.
2Department of Neurosciences, Western biomedical research center, Mexican Institute of Social Security (Guadalajara, Mexico)
3Department of Genetics, Western biomedical research center, Mexican Institute of Social Security (Guadalajara, Mexico).
4Primary Care Resident of Delicias Sur Health Center. Manuel Dronda, 1, 50009 Zaragoza, Spain.
5Molecular Entomology Laboratory, Department of Biotechnology, School of Biosciences, Periyar University, Salem-636011, Tamil Nadu, India.
6Department of Genetics, University of Guadalajara. Av. Juárez No. 976, Centro, 44100 Guadalajara, JAL, México.
7Mission Regional Medical Center. 900 S Bryan Rd, Mission Tx 78572, USA.
Corresponding Author: Esteban-Zubero E, Emergency Department of Hospital San Pedro. Calle Piqueras 98. ZIP Code: 26006. Logroño, Spain, Tel : +34654123994; E-Mail: [email protected]
Received Date: 13 Jul 2017 Accepted Date: 19 Oct 2016 Published Date: 21 Oct 2016
Copyright © 2017 Esteban-Zubero E
Citation: Esteban-Zubero E, Castejón-Moreno R, AlatorreJiménez MA, Barberá-Pérez D et al. (2017). Cutaneous Vasculitis as A First Sign of Isolated Peptoniphilus Spp in Urinary Tract Infection: A Case Report. Mathews J Case Rep 2(2): 031
ABSTRACT
Cutaneous vasculitis (CV) is a disease that may occur as a primary pro¬cess or be secondary to a wide and het¬erogeneous group of disorders, including drug reactions, infections, connective tissue diseases, autoimmune disorders and malignancies. There are several different cutaneous manifestations, including palpable purpura, urticarial papules and plaques, nodules, ulcers, and livedo reticularis. Skin biopsy is the gold standard for the diagnosis of CV. However, the presence of a leukocytoclastic vasculitis is not sufficient to establish the specific underlying aetiology. Herein, we summarize a case report of a 15-year-old woman affected with non-pruriginous skin lesions, predominantly in the lower limbs (up to thighs), without fever or signs of systemic or localized infection. The hysthopatological study of skin lesions showed orthokeratotic epidermis with underlying dermis with vascular changes with slight proliferation of capillaries, small vessels lesions, endothelial cell swelling, deposits of eosinophilic fibrin bands, and fibrinoid degradation. Neutrophil polymorphonuclear predominant inflammatory infiltrate around vessels with endothelial damage and nuclear dust (leukocytoclastia). In addition, an urine cultura showed a Peptoniphilus spp infection. With the diagnosis of of CV caused by a Peptoniphilus spp isolated in an urinary tract infection, the patient was treated with corticosteroids and amoxicilin. After 1 week, all the skins lesions were resolved.
KEYWORDS
Vasculitis; Cutaneous Vasculitis; Urinary Tract Infection.
INTRODUCTION
Cutaneous vasculitis (CV) is a disease that may occur as a primary pro¬cess or be secondary to a wide and het¬erogeneous group of disorders, including drug reactions, infections, connective tissue diseases, autoimmune disorders and malig nancies [1-7]. Clinically, it is mainly characterised by palpable purpura, involving usually the lower extremities [1,4] with histopathologic findings that have in common vascular inflammation and blood vessel damage [1-8]. Skin biopsy is the gold standard for the diagnosis of CV. However, the presence of a leukocytoclastic vasculitis is not sufficient to establish the specific underlying aetiology [8]. Current pharmacological treatment of primary CV is mainly based on corti¬costeroids, while, immunosuppressive agents can be added in the most severe cases. However, when CV is secondary to an underlying bacterial infection, this treatment may lead to a deleterious effects in patients [6,7].Herein, we describe a case of CV secondary to a urinary tract infection produced by Peptoniphilus spp with a short review of the literature. For our knowledge, this is the first case reported of CV caused by a Peptoniphilus spp isolated in an urinary tract infection.
CASE REPORT
A previously healthy 15-year-old woman with no prior significant medical history was admitted in our Emergency Department affected with non-pruriginous skin lesions, predominantly in the lower limbs (up to thighs), without fever or signs of systemic or localized infection. No apparent possibility of insect bite. Requesting any abnormalities in the previous weeks, the patient referred a respiratory tract infection treated symptomatically 1 week ago.
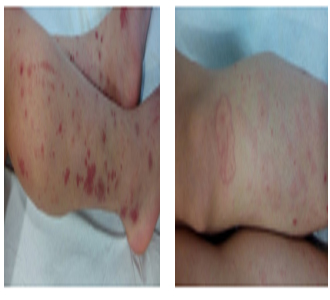
Attending to the physical examination, cardiopulmonary as well as abdominal explorations were anodyne. Any disturbs were observed at oropharyngeal level. The patient presented edemas in distal region of lower extremities without fovea. At skin level in lower extremities, macular and purpuric lesions of different sizes were observed. These lesions were not erasable to the digital pressure. The smallest lesions were petechiae like, while the largest lesions were confluent up to 4-5 cm in their largest diameter (Figure 1 and 2). On left forearm, the patient showed 2-3 petechiae lesions on extensor face.On right hand, 2 millimetric petechiae on the dorsal side were also observed.
Figure 1: Lower extremities showing edemas in distal region without fovea. At skin level, macular and purpuric lesions of different sizes not erasable to the digital pressure. The smallest lesions were petechiae like, while the largest lesions were confluent up to 4-5 cm in their largest diameter
Figure 2: Lower extremities showing edemas in distal region without fovea. At skin level, macular and purpuric lesions of different sizes not erasable to the digital pressure. The smallest lesions were petechiae like, while the largest lesions were confluent up to 4-5 cm in their largest diameter.
During her admission, a blood test was performed revealing a discrete increase of C-reactive protein (16.0 mg/L, normal range between 0.0 - 10.0 mg/L). Attending to the coagulation blood results, prothrombin time (62.0%, normal range between 80.0 - 120.0%), prothrombin ratio (1.44, normal range between 0.8 - 1.2), and fibrinogen levels (513.0 mg/dl, normal range between 184.0 - 480.0 mg/dl) were altered. A chest x-ray study was also realized without disturbs. A rapid strep test was also performed to complete the study. The result was negative to Streptococcus infection. An urine cultura was realized showing a Peptoniphilus spp infection sensitive to ampicilin, amoxicilin and moxifloxacin.
With the diagnosis of CV, the patient was accepted in the Department of Internal Medicine for study. During her admission, a blood test measuring rheumatoid factor, anti-streptolysin O (ASLO), immunoglobuline G, M and A, C3, C4 and CH 50 complement levels was performed. The results did not show any disturbs. Autoimmune disorders were also studied measuring ANA, pANCA, cANCA, AntiMPO, and AntiPR3 values. All of them were negative.
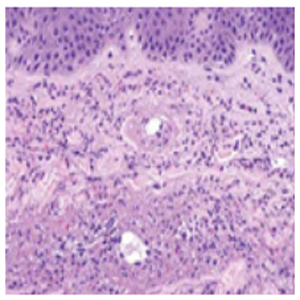
The histopathological study of skin lesions showed orthokeratotic epidermis with underlying dermis with vascular changes with slight proliferation of capillaries, small vessels lesions, endothelial cell swelling, deposits of eosinophilic fibrin bands, and fibrinoid degradation. Neutrophil polymorphonuclear predominant inflammatory infiltrate around vessels with endothelial damage and nuclear dust (leukocytoclastia) were also observed (Figure 3). In the direct immunofluorescence study, fibrinogen deposition was observed in the wall of small vessels with absence of IgM, IgG, IgA, C3, C4, albumin, Kappa and Lambda deposits.
Figure 3: Histopathologic features of leukocytoclastic vasculitis, a form of small-vessel vasculitis. Neutrophils, neutrophilicnuclear debris, and amorphous protein deposits are present within the expanded wall of a cutaneous venule.
During her admission, the patient progressed favorably with a decreasing administration of corticosteroids (60 mg during 3 days, 45 mg during 3 days, 30 mg during 3 days, 15 mg during 3 days, 5 mg during 3 days and retire), amoxicilin (1 gr every 8 hours during days) and symptomatic tratment (analgesics as well as antihistamines). After three days, all the drugs were administered orally and the patient was discharged of the Department of Internal Medicine. After 1 week, all the skins lesions were resolved.
DISCUSSION
Peptoniphilus spp are Gram-positive anaerobic cocci (GPAC) that were formerly classified in the genus Peptostreptococcus. There are more than 15 species of this cocci, and are frequently observed in the human vagina and gut as commensals. In the literarure, Peptoniphilus spp. have most commonly been associated with bone and joint infections, diabetic skin and soft tissue infections, and surgical site infections [9-13]. However, this specie has been also isolated recently in the amniotic fluid and cord blood from pregnancies complicated by preterm birth and early-onset neonatal sepsis. This study isolated Peptoniphilus harei in 1 of 36 singleton pregnancies using cultura independent methods [14].
Cutaneous vasculitis (CV) is a disease that may occur as a primary pro¬cess or be secondary to a wide and het¬erogeneous group of disorders, including drug reactions, infections, connective tissue diseases, autoimmune disorders and malignancies [1-7]. While, in most cases, an underlying cause is not found and the dis¬ease is generally self-limited, CV can be the associated with severe bacte¬rial infection in the 3.5% of the cases [6].
There are several different cutaneous manifestations, including palpable purpura, urticarial papules and plaques, nodules, ulcers, and livedo reticularis. The presentation of these lesions depend on the size of the involved vessels and the extensión of the vascular bed that is affected. Skin biopsy is the gold standard for the diagnosis of CV, and is charactericed by the morphological evidence of both angiocentric infiltration of inflammatory cells and damage of the vessel wall. In small vessels, is especially showed endotelial oedema, intra and perivascular inflammatory infiltrate, vascular wall necrosis, fibrin deposition and extravasation of red blood cells, like our case reported [15-16].
Several studies have been studied a relationship between the histopathologic lesions of CV and the severity of the underlying disease. However, the results are not conclusive. A study disclosed that patients with deep infiltrates had more commonly cutaneous ulceration and pulmonary involvement than those patients with CV who had superficial inflammatory infiltrates [7,17]. In addition, a case series observed that systemic disease may be related with disturbs at reticular dermis level as well as subcutaneous tissue [18]. However, other studies have been not observed a relation between the severity of the disease and the histopathological disorders showed [19].
The knowledge of of an underlying si specially relevant due to the current pharmacological treatment of primary CV is mainly based on corti¬costeroids and immunosuppressive agents (in the most severe cases). However, when CV is secondary to an underlying bac-terial infection, this treatment may lead to a deleterious effects in patients. When CV is secondary to an infectious disease, the treatment of it is essential to resolve both clinical affectations: skin and systemic disease [6,7]. In our case report, amoxicilin as well as corti¬costeroids were necessary for treatment.
In conclussion, CV is a disease that may occur as a primary pro-cess or be secondary to an infectious disease, including a several systemic infection. Suspicion of an underlying disease is relevant because the histopathological findings are not correlated with the severity of the condition. In addition, the treatment varies according to the diagnosis. Prospective studies are recquired to confirm the correlated theories of histopahtological findings and the prevalence of an underlying disease. For our knowledge, this is the first case report of a CV caused by a Peptoniphilus spp isolated in an urinary tract infection.
REFERENCES
- González-Gay MA, García-Porrúa C and Pujol RM. (2005). Clinical approach to cutaneous vasculitis. Curr Opin Rheumatol. 17: 56-61.
- Loricera J, Calvo-Río V, Ortiz-Sanjuán F, González-López MA, et al. (2013). The spectrum of paraneoplastic cutaneous vasculitis in a defined population: incidence and clinical features. Medicine (Baltimore). 92(6): 331-343.
- Loricera J, Calvo-Río V, Mata C, Ortiz-Sanjuán F, et al. (2014). Urticarial vasculitis in northern Spain: clinical study of 21 cases. Medicine (Baltimore). 93(1): 53-60.
- Stagnaro C, Cioffi E, Talarico R and ella Rossa A. (2015). Systemic vasculitides: a critical digest of the most recent literature. Clin Exp Rheumatol. 33: S145-S154.
- Loricera J, Blanco R, Ortiz-Sanjuán F, Hernández JL, et al. (2015). Single-organ cutaneous small-vessel vasculitis according to the 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides: a study of 60 patients from a series of 766 cutaneous vasculitis cases. Rheumatology (Oxford). 54(1): 77-82.
- Loricera J, Blanco R, Hernández JL, Calvo-Río V, et al. (2015). Cutaneous vasculitis associated with severe bacterial infections. A study of 27 patients from a series of 766 cutaneous vasculitis. Clin Exp Rheumatol. 33: S36- S43.
- . Loricera J, González-Vela C, Blanco R, Hernández JL, et al. (2016). Histopathologic differences between cutaneous vasculitis associated with severe bacterial infection and cutaneous vasculitis secondary to other causes: study of 52 patients. Clin Exp Rheumatol. 34: S93-S97.
- Carlson JA. (2010). The histological assessment of cutaneous vasculitis. Histopathology. 56: 3-23
- Dowd SE, Wolcott RD, Sun Y, McKeehan T, et al. (2008). Polymicrobial nature of chronic diabetic foot ulcer biofilm infections determined using bacterial tag encoded FLX amplicon pyrosequencing (bTEFAP). PLoS One. 3: e3326.
- Wolcott RD, Gontcharova V, Sun Y and Dowd SE. (2009). Evaluation of the bacterial diversity among and within individual venous leg ulcers using bacterial tag-encoded FLX and titanium amplicon pyrosequencing and metagenomic approaches. BMC Microbiol. 9: 226.
- Smith DM, Snow DE, Rees E, Zischkau AM, et al. (2010). Evaluation of the bacterial diversity of pressure ulcers using bTEFAP pyrosequencing. BMC Med Genomics. 3: 41.
- . Walter G, Vernier M, Pinelli PO, Million M, et al. (2014). Bone and joint infections due to anaerobic bacteria: an analysis of 61 cases and review of the literature. Eur J Clin Microbiol Infect Dis. 33(8): 1355-1364.
- Brown K, Church D, Lynch T and Gregson D. (2014). Bloodstream infections due to Peptoniphilus spp.: report of 15 cases. Clin Microbiol Infect. 20(11): 857-860.
- Wang X, Buhimschi CS, Temoin S, Bhandari V, et al. (2013). Comparative microbial analysis of paired amniotic fluid and cord blood from pregnancies complicated by preterm birth and early-onset neonatal sepsis. PLoS One. 8: e56131.
- Carlson JA, Eng BT and Chen KR. (2005). Cutaneous vasculitis update: diagnostic criteria, classification, epiodemiology, etiology, pathogenesis, evaluation and prognosis. Am J Dermatopathol. 27: 504-528.
- Chen KR and Carlson JA. (2008). Clinical approach to cutaneous vasculitis. Am J Clin Dermatol. 9: 71-92.
- Hodge SJ, Callen JP and Ekenstam E. (1987). Cutaneous leukocytoclastic vasculitis: correlation of histopathological changes with clinical severity and course. J Cutan Pathol. 14: 279-284.
- Sánchez NP, Hale HM and Su WP. (1985). Clinical and histopathologic spectrum of necrotizing vasculitis: report of findings in 101 cases. Arch Dermatol. 121: 220-224.
- Cribier B, Couilliet D, Meyer P and Grosshans E. (1999). The severity of histopathological changes of leukocytoclastic vasculitis is not predictive of extracutaneous involvement. Am J Dermatopathol. 21: 532-536.