Previous Issues Volume 1, Issue 2 - 2016
Atrial Fibrillation: A Theoretical Assumption and a New Perspective
Vladimir Tilman1
1The Sheba Medical Center, Rehabilitation Hospital, Ramat Gan, Israel.
Corresponding Author: Vladimir Tilman, Tne Sheba Medical Center, Rehabilitation Hospital, Ramat Gan, Israel, Tel: 972-0547345525; E-Mail: [email protected]
Received Date: 13 Oct 2016 Accepted Date: 31 Oct 2016 Published Date: 04 Nov 2016
Copyright © 2016 Tilman V
Citation: Tilman V. (2016). Atrial Fabrillation: A Theoretical Assumption and a New Perspective. Mathews J Cardiol. 1(2): 008.
ABSTRACT
The most common cardiac arrhythmia in clinical practice is atrial fibrillation (AF). The cause for the development of AF has been investigated by many studies, however, the question remains unanswered. A recently published hypothesis suggested that atrial fibrillation is a physiological mechanism, of protective value, based on the discontinuation of atrial mechanical systole. This lowers the pressure within the system of the pulmonary veins and alveolar capillaries, in pathological situations, and as a result reduces the probability for development of pulmonary congestion and edema. This hypothesis explains the connection between left ventricle (LV) dysfunction and AF, helps with designing algorithms of relationships between AF and LV dysfunction, and with various diseases and conditions, suggests a new approach to the location of the source of AF and shows a protective function of the atriums in the development of LV dysfunction. There is a correlation between this hypothesis and the known facts and phenomenons associated with AF.
INTRODUCTION
It is known that the most common arrhythmia in clinical practice is atrial fibrillation (AF) and is very often associated with LV dysfunction / heart failure (HF). It is called the epidemic of the century. An arrhythmia that is particulary common in the elderly, AF is growing in prevalence with the ageing of the population. In addition to age, there are many types of cardiac and medical conditions that are also closely linked to AF. These include hypertention, coronary artery disease, heart failure, valvular heart diseases, hyperthyroidism and so on. Despite the fact that there have been many studies carried out on the subject of “AF-a cause or consequence?”, the question remains unanswered [1].
THE FIRST QUESTION – WHY DOES AF DEVELOPE?
In the years 2015-2016 the new hypothesis was proposed, which assumes that AF is a protective physiological mechanism in the cases of LV dysfunction/heart failure and it answers many questions related to the AF [2]. The reasoning of the hypothesis is as follows. The end diastolic pressure (EDP) is the algebraic sum of the left ventricle diastolic pressure (LVdp) and left atrial systolic pressure (atrial kick pressure, AKp). EDP=LVdp+AKP. Normal range of EDP is 8-12 mm Hg, AKp is 2-6 mm Hg. At the end of the diastole when the mitral valve(MV) is still open, EDP immediately transmitted and equalized (according to hydrodynamic law) throughout the communicating system of the LV, LA, pulmonary veins(PV), and alveolar capillaries(AC), as there are no obstacles to the spread of the EDP wave. EDP is thus equal to pulmonary wedge pressure (PWP) with a normal range of 8-12 mm Hg. In the conditions of LV dysfunction (HF varying degrees), EDP increases due to failure of the LV (i.e. LV diastolic pressure component increases). EDP ↑ = LVsp ↑ + AKp. This increased EDP at the end of diastole with opened MV transmits to the entire communicating system of the LA-PV-AC and with the increased PWP, but with the threat of exit of fluid through the alveolar-capillary membrane. When PWP increases to 20 mm Hg, pulmonary edema is usually present. The assumption of the existence of a physiological protective mechanism is logical, as it would prevent prolonged pressure in the system described. But to reduce the LV diastolic pressure (LVdp) is impossible because it is caused by the pathological condition of the LV myocardium and is not subject to rapid change. However, there is a possibility of reducing the total EDP (and thus reducing pressure in the entire system!) by stopping the mechanical systole of left atrium, which will exclude the LA pressure component from the total EDP. EDP = LVdp + AKp. This is accomplished by AF which halts the mechanical systole of left atrium.The right atrium is also involved in the AF. Termination of RA systole leads to decrease EDP of right ventricle (RV) and to decrease of systemic venous inflow to the heart, which results in the reduction of preload. The reduction of preload is one of the most urgent treatments of pulmonary edema. Thus, due to the termination of a mechanical systole in both the LA and RA, AF leads to a synergy in the hydrodynamic results. Many facts confirm that the cessation of mechanical systole of both atriums by the development of AF in the cases of LV dysfunction/ HF creates more optimal hydrodynamic conditions in the heart when the pressure in the communicating system of LA-PV-AC increases, which results in the appearance of the heart as “needing” AF:
- Clinical trials (AFFIRM, RACE, STAF, AF-CHF etc.) have failed to determine the clinical advantage of sinus rhythm over optimal rate-control. There was no benefit provided by rhythmcontrol and actually an inclination towards harm in the conditions of LV dysfunction/HF compared with rate-control strategy [3 - 5].
- In many cases, after successful treatment of AF (by medications, cardioversion, or ablation) AF spontaneously returns. This susceptibility of AF reccurence is consistent with the above proposed hypothesis that in conditions of LV dysfunction there is a “need” within the heart for AF, which provides more optimal hydrodynamic benefit [6, 7].
- It is not uncommon in clinical practice cases to find a recurrence of pulmonary edema after successful return of AF to sinus rhythm. This phenomenon is explained by the fact that in the conditions of LV dysfunction, there is a preference in the heart for hydrodynamics with AF [8].
- Cases where pulmonary edema developed after implantation of pacemaker for AF can be explained in the same way (so-called pacemaker syndrome). In these cases, not only is there desynchronization of atriums and ventricles, but also an increase of pressure in the system of LA-PV-AC as a result of imposing atrial regular rhythm instead of the former AF [9].
Thus, the assuption that AF is a physiological protective mechanism in the conditions of LV dysfunction/HF explains well all the facts and phenomena associated with the AF and allows us to build the algorithms of their relationships in various diseases [10]. It is possible that AF is a mechanism, programmed genetically for the purpose of essential protection from increasing pressure in the LA-PV-AC system due to pathological conditions. Aberrant triggering of this mechanism at the genetic level, perhaps as result of mutation, may explain Lone AF, which is the development of AF with no apparent organic pathology. As with other well-known protective and compensatory mechanisms, AF is not without drowbacks, the most notable of which is the tendency towards thrombosis in the LA. Rapid rate of AF (a function of the AV node) can have the potential to aggravate HF.
THE SECOND IMPORTANT QUESTION – WHERE IS THE SOURCE OF AF?
There are several theories as to the development of AF, but no one answeres the question where is its source. There still has not been found a localisation of an anatomical or histological substrate as a source of AF. The positive outcomes as a result of termination of AF by ablation of the pulmonary vein orifices are a solid argument that the patients with AF have a focal (e.g. pacemacer or triggered dischages) mechanism in the pulmonary viens region, and it is probable that ablation only “cuts” the advancement of AF. This is consistent with the proposed hypothesis because the predicted source of AF is in a strategically important localization in the LA-PV-AC system, where increase of pressure may result in complications (pulmonary cogestion or edema). This increase of pressure in the entire system can activate the mechanism of AF localised in the pulmonary veins. Therefore, the development of AF serves the purpose of reducing the pressure in the system. There is also a possibility that the source of AF has functions of baroreceptors that can “turn on” AF in response to a high risk of increased pressure in the LA-PV-AC system and “turn off” when the danger dissipates. In clinical practice, it is known that AF occurs in acute pulmonary edema, with spontaneous termination after successful treament of the pulmonary edema without using antiarrhythmic medicines. Thus, the proposed hypothesis can suggest a sequence of events which results in the development of AF. Each individual has a protective physiological mechanism in the pulmonary veins, most likely genetically programmed. When the pressure in the LA-PV-AC system increases to a specific critical value, the AF mechanism is set in motion. AF terminates mechanical systole of the LA excluding the component of systolic left atrial pressure from the total pressure in the LA-PV-AC system, and of the RA with effect of lowering preload, and as a result decreases the threat of the development of pulmonary congestion or edema.
THE PROTECTIVE FUNCTION OF THE ATRIUMS
In the ligh of the proposed hypothesis reveals the special role of the left atrium, as the protective organ in the conditions of the LV dysfunction. From hydrodynamics, it is known that reducing pressure in a system is possible, either directly by decreasing the pressure, or due to an increase of the volume of the system. It is applicable also to the hydrodynamics system of the heart. Three protective mechanisms reducing pressure in the LA-PV-AC system are known.
- Direct decreasing pressure in the LA-PV-AC system - the terminating of mechanical systole of the LA at the AF while excluding the component of systolic LA pressure from total EDP (the proposed hypothesis). (Table 1) Thus, AF is the the first aid in the conditions of the LV dysfunction!
Table 1
EDP – total end dyastolic pressure at the end of LV dyastole, DPlv – dyastolic pressure of left ventricle at the end of dyastole, AKp – systolic left atrial pressure (atrial kick).SINUS RHYTHM ATRIAL FIBRILLATION EDP = DPlv + AKp EDP = DPlv - In the case of prolonged pressure overload in the LA-PV-AC system - increasing the volume of the system due to significant dilatation of left atrium, reduces pressure in the system, protecting the patient from the death. In mitral stenosis, there is significant enlargement of the LA, sometimes reaching a very big size, as a result of prolonged increased pressure in the LA-PV-AC system [11]. While this prolonged influence of an elevated pressure in the LA-PV-AC system obviously causes expansion of LA, due to it’s thin walls, this increase in capacity volume of the system would also bring to reduction of pressure, and may operate as a protective mechanism (damper effect) that prevents death. The increase in capacity volume of the system would lead to reduction of the pressure similar to dampers used in technical hydrodynamic systems, and may server as a compensatory mechanism that saves the patient’s life. There is a direct correlation between the size of the LA with AF, calling attention to their synergy of action.
- Humoral protective mechanism – pressure decreases in the LA-PV-AC system due to reduction of volume of liquid – diuretic effect of atrial-natriuretic peptid (ANUP), allocation of which increases in the cases of elevated pressure within the system [12].
- As mentioned above the right atrium is also involved in the AF and has very importent protective function in cases of LV dysfunction. Termination of RA mechanical systole leads to decrease of systemic venous inflow to the heart, resulting in a decreased preload.
AN EXAMPLE OF THE ALGORITHM OF RELATONSHIP OF AF AND HYPERTENTION (on the basis of the proposed hypothesis)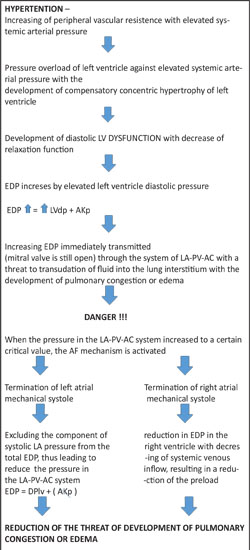
Figure 1: The Algorithm of relationship between AF and hypertention. Similary, we can build algorithms of the development of AF in various diseases.
CONCLUSION
The above proposed hypothesis suggests that AF is a physiological mechanism that serves a protective role and is most likely genetically programmed. The primary purpose of AF is to reduce pressure in the LA-PV-AC system, through elimination of the mechanical systole of the LA and of the RA, with the effect of reducing preload that creates conditions that are more favorable for the heart in the LV dysfunction /HF and thus decreases the threat of pulmonary congestion or edema. This hydrodynamic approach explains the direct correlation of AF with all diseases as well as LV dysfunction and age, and with all facts and phenomena associated with AF.
REFERENCES
- Ringborg A, Nieuwlaat R, Lindgren P, Jonsson B, et al. (2008). Costs of atrial fibrillation in five European countries: results from the Euro Heart Survey on atrial fibrillation. Europace. 10(4), 403-411.
- Tilman V. (2016). Atrial fibrillation: In the light of new hypotheses. Med Hypotheses. 86(3), 10-13.
- Wyse DG, Waldo AL, DiMarco JP, Domanski MJ, et al. (2002). For the Atrial Fibrillation Follow-up investigation of Rhythm Management (AFFIRM) investigators. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 347(23), 1825-1833.
- Camm AJ, Kirchhof P, Lip GY, Schotten U, et al. (2010). Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Eur Heart J. 31(19), 2369-2429.
- Chung MK, Shemanski L, Sherman DG, Greene HL, et al. (2005). Functional status in rate- versus rhythm-control strategies for atrial fibrillation: results of the Atrial Fibrillation Follow- Up Investigation of Rhythm Management (AFFIRM) Functional Status Substudy. J Am Coll Cardiol. 46(10), 1891-1899.
- Vikman S, Mkikallio TH, Yli-Myry S, Nurmi M, et al. (2003). Heart rate variability and recurrence of atrial fibrillation after electrical cardioversion. Ann Med. 35(1), 36-42.
- Alcaraz R and Rieta JJ. (2009). Time and frequency recurrence analysis of persistent atrial fibrillation after electrical cardioversion. Physiol Meas. 30, 479-489.
- Gowda RM, Misra D, Khan IA and Schweitzer P. (2003). Acute pulmonary edema after successful cardioversion of atrial fibrillation. Am JTher. 10(1), 73-74.
- Balley SM, Wilkoff BL. (2006). Complications of pacemakers and defibrillations in the elderly. Am J Geriatr Cardiol. 15(2), 102-107.
- Tilman V. (2016). Algorithm of Relationships between Left Ventricle Dysfunction and Atrial Fibrillation. J cardiovasc Dis Diagn. 4, 247.
- Tung R and DeSanctis R. (2004). Giant left atrium. NEJM. 351(14), 1437.
- Yoshihiko Saito. (2010). Roles of atrial natriuretic peptide and its therapeutic use. J of Cardiol. 56(3), 262-270.