Information Links
Related Conferences
Previous Issues Volume 8, Issue 8 - 2023
Absence of Viral DNA Suggests Sri Lanka as a Non-Endemic Country for Kaposi’s Sarcoma Associated Virus (KSHV): A Case Series Study
Manosha Lakmali Perera1,*, Irosha Rukmali Perera2, Sharika Gunathilake3
1School of Dentistry and Oral Health, Griffith University, Queensland, Australia
2Preventive Oral Health Unit, The National Dental Hospital (Teaching), Colombo, Sri Lanka
3Oral & Maxillofacial Unit D, National Dental Hospital (Teaching) Sri Lanka, Ward Place, Colombo 7, Sri Lanka
*Corresponding Author: Manosha Lakmali Perera, School of Dentistry and Oral Health, Griffith University, Queensland, Australia (Alumni), Tel +94 71 3271945, ORCID: 0000-0001-7275-2736; Email: [email protected].
Received Date: August 16, 2023
Published Date: September 20, 2023
Citation: Perera ML, et al. (2023). Absence of Viral DNA Suggests Sri Lanka as a Non-Endemic Country for Kaposi’s Sarcoma Associated Virus (KSHV): A Case Series Study. Mathews J Case Rep. 8(8):123.
Copyrights: Perera ML, et al. © (2023).
ABSTRACT
Amidst innovations in early detection strategies and treatment options, oral cancer constitutes the Head and neck cancers (HNSCCs) echelons as the number one among Sri Lankan males and, also continues as a global public health menace. Oral squamous cell carcinoma (OSCC) is the commonest oral malignancy in most countries, with no exception in Sri Lanka. Persistent immunosuppression in oral cancer patients triggers the activation of the lytic cycle of latent viruses demonstrating B cell tropism. Of them, Human Herpes virus 8 (HHV 8) better known as Kaposi’s sarcoma-associated herpes virus (KSHV) relates to malignancies, especially among human immunodeficiency virus (HIV) patients. In the light of these findings, this case series study aimed to detect the HHV-8 status in oral squamous cell carcinoma tissues of Sri Lankan male patients. Ethical clearance was obtained from Sri Lanka and Australia. Written informed consent was obtained from the participants. Incisional biopsies of cases were collected, transported, stored, and dispatched as frozen tissues at -800 C. The DNA was extracted from frozen specimens using Gentra Puregene Tissue kit (Qiagen, Germany), solid tissue protocol according to the manufacturer’s instructions. The real-time PCR was performed with the specific primers to detect HHV- 8 in these OSCC cases. However, the HHV-8 DNA was not found d in 22 OSCC cases. Prevalence and associated risk factors of KSHV differ within populations and geographic locations across the world establishing the reservoir of this virus in high-risk groups of endemic countries. Sri Lanka seems a non-endemic country for this oncogenic virus. Small sample size is a limitation of this study. Powered studies based on Acquired Immune Deficiency Syndrome (AIDS) patients will confirm Sri Lanka as a non- endemic country for one of the two gamma herpes viruses.
Keywords: Oral Cancer, Human Herpes Virus 8, Kaposi’s Sarcoma-Associated Herpes Virus, Risk Factors, Non-Endemic.
INTRODUCTION
(HNSCCs) persist as a public health menace and are categorized as the 8th commonest cancer type according to a 2020 global estimation [1]. Oral squamous cell carcinoma is the most common subgroup of head and neck squamous cell carcinoma which includes malignant neoplasms, beginning from the lining mucosae of the lips and the mouth (oral cavity) including the anterior two-thirds of the tongue as defined by the WHO International Classification of Disease [2]. Sri Lanka contributes to the global oral cancer burden with Papua New Guinea, Bangladesh, Hungary, having the highest incidence rates [3]. This non-communicable disease becomes a health economic disaster when considering the cost of treatment modalities for instance: surgery, radiation and chemotherapy as well as palliative care for oral cancer survivors, despite the poor 5 year survival rate. This easily preventable malignancy links with geographic and population-specific risk factors which introduce aetiological agents or known carcinogens mainly polycyclic-aromatic hydrocarbons (PAH) and nicotine-derived nitrosamine ketone (NNK), ranks 1st among Sri Lankan males, exclusively due to life course exposure to smokeless tobacco and areca nuts as a habit of betel quid chewing [3-5]. The oncogenic Human Herpes virus 8 (HHV 8) also famous as Kaposi sarcoma (KS)-associated with the herpes virus KSHV discovered by Chang and colleagues in 1994 and well characterized since then [6,7]. As the name implies this virus is the causative agent of Kaposi’s sarcoma (KS) the commonest HIV-associated malignancy in the world [1,7]. Molecular pathological evidence unveiled the mechanisms of oncogenesis of HHV-8. Via molecular mimicry, viral encoded proteins are found to be activated by several cellular signaling cascades whilst evading immune surveillance [1,8]. Immunosuppressive conditions in oral cancer patients may provide an ideal opportunity to activate oncogenic viruses from their latent stage [3]. Viral infections due to Epstein Bar Virus (EBV one of the oncogenic γ-herpes viruses among OSCC cases) reported previously in local [3,9], regional [10,11] and global [12] oral cancer patients. Nevertheless, there are no published studies on the status of HHV-8/KSHV, the other γ-herpes virus in Sri Lankan OSCC patients and the prevalence of this virus in Sri Lankan HIV patients who are infected with HIV in the endemic countries of KSHV.
Against this backdrop, the present case series study aims to assess the HHV-8 status in a group of Sri Lankan male, oral squamous cell carcinoma (OSCC) cases confirmed by histopathologically diagnosis.
MATERIALS AND METHODS
Ethical Approval
Ethical approval for present case series study was taken from the Faculty Research Committee, Faculty of Dental Sciences, University of Peradeniya, Sri Lanka (FRC/ FDS/UOP/E/2014/32) and Griffith University Human Research Ethics Committee, Australia (DOH/18/14/ HREC).
Study design, sample size calculation, setting, and subjects
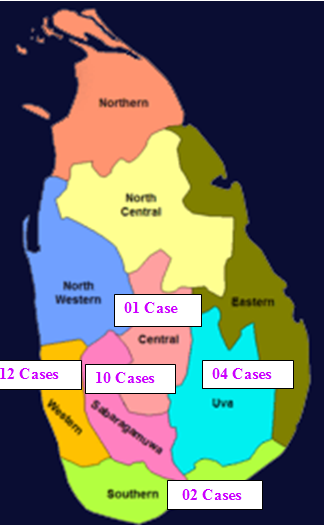
Present case series study was centered on a representive subsample of multicenter hospital based large case control study conducted in nine Oral and Maxillo-Facial (OMF) Units located in six provinces (Figure 1&2) of Sri Lanka as described previously [3,5]. The scientific sample size calculation for the unmatched case-control study was based on Kelsey et al., 1996 as described previously [3]. Data and sample collection performed as described previously [3,5]. Written informed consent was obtained from each participant [3,5]. A case series of 29 clinically suspected (Figure 2) but histologically confirmed OSCC, tissue samples were selected to proceed with as follows.
Tissue sampling and DNA extraction
Deep tissue pieces (~3 mm3) from incisional OSCC biopsies were taken. Subsequently, DNA extraction from frozen samples (=800C) was performed according to a solid tissue protocol as described previously [3,5].
Real Time PCR for HHV 8
β-globin PCR with the primers PCO3 and PCO4 was performed to ensure the quality of DNA and to confirm the absence of PCR inhibiting agents [3]. Amplification of KSHV Target DNA was done as described previously [3]. The real time PCR assay was set up to amplify 106 bp of HHV-8, from published primer sequences as described previously [3]. Then, the real time PCR was performed on a Quant Studio 6- real -time machine with an initial step of hold stage of polymerase activation step at 950 C for 5 minutes, followed by 45 cycles of amplification (5 seconds denaturation at 950 C for 5 minutes; 30 seconds annealing (TM)/extension at 550 C) and melt curve stage of 3 steps (950 C for 10 minutes, 500 C for 10 minutes and 950 C for 15 minutes) [3]. Overall run duration was 72 minutes and 24 seconds. Amplicon detection was determined via rt PCR screen and melt curve analysis [3].
RESULTS
Distribution of Samples by the Province in Sri Lanka
Figure 1. Number Cases obtained from each province out of six provinces in Sri Lanka.
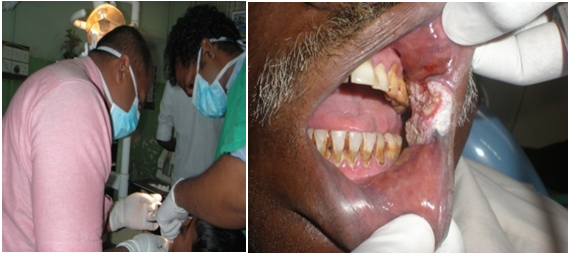
Clinical examination of Oral Squamous Cell Carcinoma (OSCC) patient at an OMF Unit
Figure 2. Oral examination by Dental Surgeons of a buccal mucosa-affected OSCC patient.
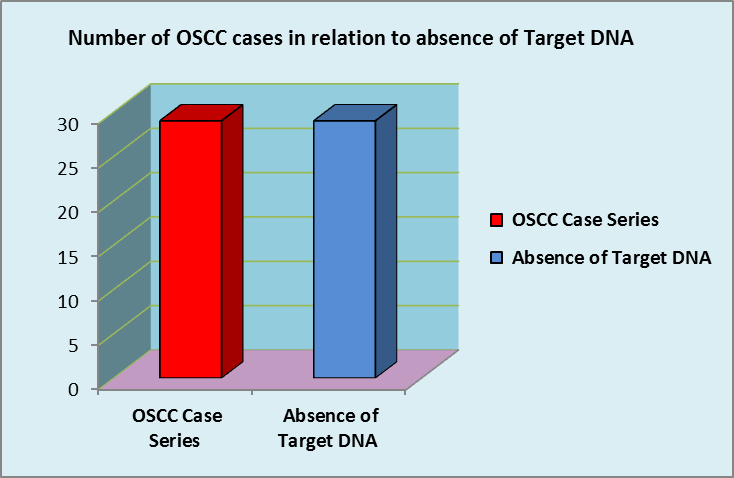
KSHV status in 29 OSCC case series
Figure 3. Absence of KSHV DNA in Case Series.
According to Figure 3 this case series comprising 29 OSCC tissues did not contain KSHV DNA. This finding provides rationale to speculate KSHV was not a matter to concern in OSCC tissues in a group of Sri Lankan male patients.
DISCUSSION
In the present study KSHV/ HHV-8 DNA was not detected at least in one sample out of 29 of case series. Comparable findings were reported in a study conducted by Yang and colleges [12] as this virus were not detected in any of 25 archival Formalin-Fixed Paraffin-Embedded (FFPE) samples. In another molecular epidemiology study, HHV-8 was detected in one of 21archival FFPE samples of head and neck squamous cell carcinomas in a HIV positive patient [13]. However, it has been revealed that the KSHV infection is not ubiquitous with seroprevalence. Risk factors and transmission routes exhibit considerable variations across populations in different geographic region which may justify the negative finding of present study [14]. KSHV transmits via saliva during childhood in endemic areas. Whereas, KSHV transmission could be associated with homosexual contact in non-endemic countries. Small sample size is a limitation of the present study [14]. Anti- apoptotic properties of KSHV infected primary cells was observed previously [15,16]. The presence of latent virus in KSHV-associated cancers provides a potential target for therapy.
CONCLUSION
Sri Lanka seems to be a non-endemic country for KSHV as the target DNA was not found in any sample.
RECOMMENDATIONS
Further case control studies with large sample sizes, representing younger oral cancer patients and HIV patients are recommended to confirm the conclusion of the present case series study.
AUTHOR CONTRIBUTIONS
M. Perera, contributed to conception, design, data acquisition and interpretation, scientifically improved the manuscript; I. Perera, contributed to design, data acquisition, interpretation and statistical analysis.
FUNDING AGENT
This study was funded by Griffith University Australia under the account No s2864780.
ACKNOWLEDGMENTS
First and foremost, I would like to sincerely thank to Dr. D. J. Speicher for establishing the real time PCR technique to detect KSHV at School of Dentistry and Oral Health, Gold Coast Campus, Griffith University, QLD 411. I deeply regret not making him as coauthor in this case review when considering the contribution of him to achieve success in laboratory performance in this regard. Then I extend my gratitude to my Principal Supervisor, Prof. Newell W. Johnson for allowing me to work under him. Big thanks to Prof. G.C. Ulett for his overall supervision in the DNA extraction phase of my Ph.D. under the guidance of Dr. D. Ipe in his lab.
CONFLICT OF INTEREST
The authors declare no potential conflicts of interest with respect to the authorship and/or publication of this article.
REFERENCES
- Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. (2021). Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J Clin. 71(3):209-249.
- Gupta B, Lalloo R, Johnson NW. (2015). Life course models for upper aero-digestive tract cancer. Int Dent J. 65(3):111-119.
- Perera ML. (2017). The Microbiome Profile of Oral Squamous Cell Carcinoma Tissues in A Group Of Sri Lankan Male Patients. Griffith University, Queensland, Australia. Available at: https://research-repository.griffith.edu.au/bitstream/handle/10072/370682/Perera%2cManosha_Final%20Thesis_redacted.pdf?sequence=1&isAllowed=y
- Perera M, Al-hebshi NN, Speicher DJ, Perera I, Johnson NW. (2016). Emerging role of bacteria in oral carcinogenesis: a review with special reference to perio-pathogenic bacteria. J Oral Microbiol. 8:32762.
- Perera M, Al-hebshi NN, Perera I, Ipe D, Ulett GC, Speicher DJ, et al. (2018). Inflammatory bacteriome and oral squamous cell carcinoma. J Dent Res. 97(6):725-732.
- Chang Y, Cesarman E, Pessin MS, Lee F. (1994). Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi's sarcoma. Science. 266(5192):1865-1869.
- Speicher DJ, Johnson NW. (2012). Detection of human herpesvirus 8 by quantitative polymerase chain reaction: development and standardisation of methods. BMC Infect Dis. 12:210.
- Jalouli J, Jalouli MM, Sapkota D, Ibrahim SO, Larsson P-A, Sand L. (2012). Human papilloma virus, herpes simplex virus and epstein barr virus in oral squamous cell carcinoma from eight different countries. Anticancer Res. 32(2):571-580.
- D' Costa J, Saranath D, Sanghvi V, Mehta AR. (1998). Epstein-Barr virus in tobacco-induced oral cancers and oral lesions in patients from India. J Oral Pathol Med. 27(2):78-82.
- Acharya S, Ekalaksananan T, Vatanasapt P, Loyha K, Phusingha P, Promthet S, et al. (2015). Association of Epstein-Barr virus infection with oral squamous cell carcinoma in a case–control study. J Oral Pathol Med. 44(4):252-257.
- Kis A, Fehér E, Gáll T, Tar I, Boda R, Tóth ED, et al. (2009). Epstein–Barr virus prevalence in oral squamous cell cancer and in potentially malignant oral disorders in an eastern Hungarian population. Eur J Oral Sci. 117(5):536-540.
- Yang Y-Y, Koh L-W, Tsai J-H, Tsai C-H, Wong EF-C, Lin S-J, et al. (2004). Involvement of viral and chemical factors with oral cancer in Taiwan. Jpn J Clin Oncol. 34(4):176-183.
- McLemore MS, Haigentz M, Smith RV, Nuovo GJ, Alos L, Cardesa A, et al. (2010). Head and neck squamous cell carcinomas in HIV-positive patients: a preliminary investigation of viral associations. Head Neck Pathol. 4(2):97-105.
- Zhang X, Xu Y, Li Y, Yuan H, Liu Z, Zhang T. (2022). Prevalence and correlates of Kaposi's sarcoma-associated herpesvirus and herpes simplex virus type 2 infections among adults: evidence from the NHANES III data. Virol J. 19(1):5.
- Aalam F, Nabiee R, Castano JR, Totonchy J. (2020). Analysis of KSHV B lymphocyte lineage tropism in human tonsil reveals efficient infection of CD138+ plasma cells. PLoS Pathog. 16(10):e1008968.
- Lu J, Jha HC, Verma SC, Sun Z, Banerjee S, Dzeng R, Robertson ES. (2014). Kaposi's sarcoma-associated herpesvirus-encoded LANA contributes to viral latent replication by activating phosphorylation of survivin. J Virol. 88(8):4204-4217.