Information Links
Related Conferences
Previous Issues Volume 6, Issue 1 - 2021
A Rare Case of Peliosis Hepatis in Infancy
Siromahov M1, N Rasheva1, M Georgieva1, V Kaleva2, Hr Hristosova2, P Drenakova3, I Krasnaliev4, B Balev5
1Department of pediatrics, Second pediatric clinic, UMHAT St. Marina, Varna, Bulgaria
2Department of pediatrics, Pediatric clinic of oncohaematology, UMHAT St. Marina, Varna, Bulgaria
3 Department of General surgery, Pediatric surgical clinic, UMHAT St. Marina, Varna, Bulgaria
4 Department of General and clinical pathology, UMHAT St. Marina, Varna, Bulgaria
5Department of Radiology, UMHAT St. Marina, Varna, Bulgaria
*Corresponding author: Mihail Siromahov, Department of pediatrics, Second pediatric clinic, UMHAT St. Marina, Varna, Bulgaria, E-mail: [email protected]
Received Date: July 9, 2020
Publication Date: January 28, 2021
Copyright: Siromahov M, et al. © 2021. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Siromahov M, et al. (2021). A Rare Case of Peliosis Hepatis in Infancy. Mathews J Case Rep. (6)1:67.
ABSTRACT
Peliosis hepatis is a rare condition characterized by numerous vascular formations in the liver measuring from a few millimeters to several centimeters in diameter. Such formations may also be found in the spleen, lymph nodes, lungs, kidneys, adrenals, bone marrow and other parts of the gastrointestinal tract. The condition is typically asymptomatic and diagnosed accidentally. Rarely, patients present with severe symptoms such as jaundice, hepatomegaly, liver failure and haemoperitoneum.
Clinical case: We present the case of a 5 months old infant who presented to her family physician with gastroesophageal reflux. An abdominal ultrasound was performed which showed numerous oval lesions in the liver parenchyma. The patient’s physical examination was remarkable for a palpable solid mass localized above the umbilicus. Further imaging and laboratory investigations revealed that the patient was suffering from neuroblastoma with concurrent peliosis hepatis.
Conclusion: Peliosis hepatis is typically a benign condition, which may be caused by infectious diseases, malignancy, prolonged intake of medicines or toxins. Treating the underlying disease often leads to resolution of the vascular lesions. This necessitates the formation of a wide differential diagnosis and through investigation.
KEYWORDS: Peliosis hepatis; Infancy; Neuroblastoma
Abbreviations
PH: Peliosis hepatis; AST: Aspartate Aminotransferase; ALT: Alanine Aminotransferase; GGT: Gamma-Glutamyl Transferase; AFP: Alpha Fetoprotein; LDH: Lactate Dehydrogenase; TORCH: Toxoplasmosis, other, Rubella, Cytomegalovirus, Herpes simplex; HIV: Human-Immunodeficiency Virus; CT: Computed Tomography; MRI: Magnetic Resonance Imaging; MIBG scan: Iodine-123 meta-iodobenzylguanidine scan.
INTRODUCTION
Peliosis hepatis (PH) is a rare condition, characterized by multiple cyst-like vascular formations in the liverparenchyma ranging from several millimeters to a few centimeters in diameter. The same vascular formations could be found in other organs such as the spleen, lungs, kidneys, adrenals, bone marrow or in other parts of the gastrointestinal tract [1]. The condition is typically asymptomatic and an accidental finding. In rare cases it may present with jaundice, hepatomegaly, hematoperitoneum etc.
CLINICAL PRESENTATION
Patient history
A five month old female infant (I.H.) presented to her family physician with regular regurgitations after feeding for the past month. The patient was consulted with a pediatric gastroenterologist who performed an abdominal ultrasound which revealed multiple oval lesions in the liver.
I.H. is a five months old infant from a first complicated pregnancy (the mother had acute renal colic during the 5th month of gestation). She was born from an elective caesarean section four days post term; birth weight was 4200g, height – 52cm, head circumference – 36cm. The patient’s early neonatal period was uneventful except for postnatal jaundice which was successfully managed with oral glucose solutions.
Family history
Family history of the patient included a third degree relative with breast cancer. The family had also had an unvaccinated house cat up to the 8th month of gestation.
Patient work up
On admittance at the pediatric ward, I.H. was in fairly well appearing general state, reaching adequate developmental milestones for her age. Her body weight was 6,830kg, height was 66cm, body temperature was 36.6°C. The patient’s physical examination was unremarkable except for a palpable solid mass localized under the umbilicus measuring around 0.5-1cm in diameter, which mobile, did not infiltrate the overlying skin and showed no erythema.
|
Leukocytes |
12,75.109/L |
|
Hemoglobin |
121 g/L |
|
Platelets |
384.109/L |
|
AST |
84 U/L |
|
ALT |
58 U/L |
|
GGT |
85 U/L |
|
AFP |
612 ng/L |
|
LDH |
773 U/L |
|
Fibrinogen |
1,49 g/L |
|
Prothrombin activity |
55% |
Table 1: Shows the initial laboratory findings.
The patient underwent abdominal ultrasound at admittance, which revealed multiple well-perfused oval lesions in both liver lobes with hypoechogenic halo and hyperechogenic center ranging between 7-8mm in diameter (Figure 1).
Figure 1: Abdominal ultrasound showing hypoechogenic halo and hyperechogenic center.
Because of the wide differential diagnosis, the patient underwent additional laboratory investigations (Table 2).
|
TORCH serologi |
Negative |
|
HIV serology |
Negative |
|
Bartonellahanselae serology |
Negative |
|
Autoimmune hepatitis autoantibodies |
Negative |
|
Hemocultures |
Negative |
|
Myelogram |
Benign cell morphology |
|
Flow cytometry |
Unremarkable |
Table 2: Additional laboratory investigations.
An abdominal CT-scan was performed which revealed multiple hepatic lesions up to 15mm in diameter. The lesions appeared hypodense in the arterial phase, hyperdense in late venous phase. The investigation also revealed multiple borderline-sized retroperitoneal, gastrohepatic, and periportal lymph nodes. The aforementioned findings were suggestive of hepatic peliosis; also included in the differential was multiple hepatic hemangiomas (Figure 2).
Figure 2: CT-scan showing hypodense hepatic lesions in the arterial phase which are hyperdense in late venous phase.
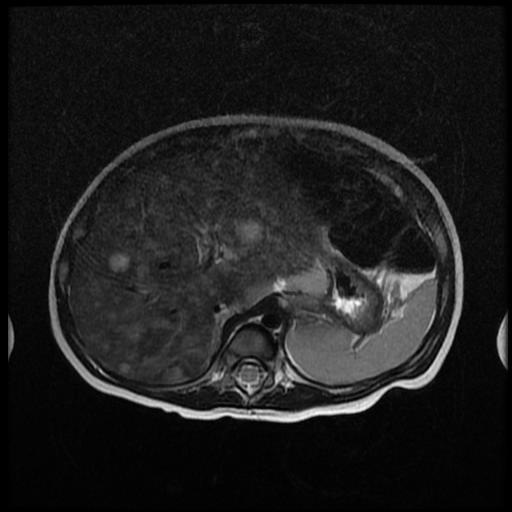
The patient underwent an abdominal MRI, which revealed multiple oval lesions in both hepatic lobes, which were hypointense on T1 and hyperintense on T2, and DW1 weighted imaging. There was an oval formation with well-demarcated hypointense capsule in front of the upper pole of the left kidney, with dimensions of 26/20/35 mm. The formation had heterogeneous structure and was hypointense in T1 and hyperintense in T2 weighted imaging. On phase contrast, the formation heterogeneously increased its signal intensity. MRI imaging also revealed multiple enlarged lymph nodes in multiple sites – around vena portae, superior mesenteric artery and abdominal aorta (Figure 3).
Figure 3: Multiple different in size oval lesions in both hepatic lobes which were hyperintense on T2 weighted imaging.
The periumbilical mass was excised under general anesthesia. Histology and immunohistochemistry was consistent with metastatic neuroblastoma (high expression of Sinaptophysin, CD56, Chromogranin A).
The patient was then scheduled for neuroblastoma specific laboratory investigations, which revealed neuron-specific enolase of 105mcg/l, deletion of the long arm of 11th and 3rd chromosome,which are associated with unfavorable prognosis. MIBG-scan was performed which showed intensive acquisition at the region of the left adrenal gland, the paraaortal region and along the large abdominal blood vessels; there were also zones of higher activity at the right infraclavicular and paracardial regions; bone-marrow metastasis was suspected; the liver showed unhomogenic acquisition.
The work up of the presented case revealed that the patient had metastatic abdominal neuroblastoma with concurrent PH.
Even though liver histology could have made a definite diagnosis of PH, it would not have had an added benefit to the treatment of the patient or her overall prognosis.
DISCUSSION
PH is a rare and often benign condition characterized by multiple blood-filled cavities in the liver parenchyma. The condition is classified histologically into two types: parenchymal and phlebectatic. In the parenchymal type, the cystic, blood-filled spaces are not lined by endothelial cells whereas in the phlebectatic type is lined with endothelium representing a dilated central vein within the hepatic lobule [2]. The two histologic types of PH may be observed in the same patients which leads some investigators to believe that they represent different stages of the same pathologic process [3].
The etiology of PH remains uncertain yet factors associated with it may be divided into: factors related to drug exposure such as steroids [4,5], immunosuppressants [6,7] as well as several toxins including alcohol [8]. Factors related to infectious diseases such as tuberculosis, HIV [9] and Bartonellahensalae [10], factors related to autoimmunity [11] and factors related to oncologic processes [12,13] are also associated with PH.
PH is typically asymptomatic and may come to the attention of the clinician during routine imaging investigations. However, several cases have been described in the literature, in which the PH presents with spontaneous lesion rupture, hematoperitoneum, jaundice, hepatomegaly, acute liver failure or portal hypertension.
Imaging studies are supportive of the diagnosis. Abdominal ultrasound may reveal pseudocystic hepatic lesions which may appear either hypoechoic in normal parenchyma, or hyperechoic in the presence of steatosis [3]. CT-scan may reveal multiple oval lesions ranging from a few mm to several centimeters in diameter, with variable density. CT-angiography usually shows multiple hypervascularized lesions during the late arterial phase, with increased enhancement during the parenchymal and venous phase [2,14]. Due to the limitations of the CT-scan detection capabilities, MRI studies are considered the golden standard in PH imaging. MRI-angiography is the preferred method revealing multiple oval contrast accumulations in the late arterial phase, which persist into the venous phase [2,5,15].
Definitive diagnosis of PH is made through liver histology. Because of the high bleeding risk which may be associated with percutaneous liver biopsy, excision biopsy is preferred [14,15].
When PH is asymptomatic, treatment is aimed at the etiologic cause if it can be identified. Cessation of the offending drug [4] or treatment of the underlying condition [10,12] led to total regression of the hepatic lesions in several cases. In symptomatic patients, presenting with acute signs of intraperitoneal bleeding, treatment is directed towards controlling the bleeding and liver sparing surgery [15,16].
A thorough investigation of the literature revealed that no other case of PH had been associated with neuroblastoma in the pediatric age. Even though liver histology could have made a definitive diagnosis of PH it would not have had an added benefit to the treatment of the patient or her overall prognosis.
CONCLUSION
PH is a rare condition, characterized by multiple vascular formations in the liver, often associated with malignancy, autoimmunity, drug exposure or toxins. The presented case is most likely the first described case of PH associated with neuroblastoma in children. Because timely treatment of hepatic peliosis can lead to complete remission, forming a broad differential diagnosis in atypical liver formations is crucial.
CONFLICT OF INTEREST
The authors have no conflict of interest to declare.
BIBLIOGRAPHY
- Guandalini S, Dhawan A, Branski D. 2016. Textbook of Pediatric Gastroenterology, Hepatology and Nutrition. Springer International Publishing. 91.
- Elsayes KM, Shaaban AM, Rothan SM, Javadi S, Madrazo BL, et al. (2017). A comprehensive approach to hepatic vascular disease. Radiographics. 37(3):813–836.
- Bracero LA, Gambon TB, Evans R, Beneck D. (1995). Ultrasonographic findings in a case of congenital peliosis hepatitis. J Ultrasound Med. 14(6):483–486.
- Solimini R, Rotolo MC, Mastrobattista L, Mortali C, Minutillo A, et al. (2017). Hepatotoxicity associated with illicit use of anabolic androgenic steroids in doping. Eur Rev Med Pharmacol Sci. 21(1):7–16.
- Battal B, Kocaoglu M, Atay AA, Bulakbasi N. (2009). Multifocal peliosis hepatis: MR and diffusion-weighted MR-imaging findings of an atypical case. Ups J Med Sci. 115(2):1–4.
- C.-Y. Yu. (2014). Peliosis hepatis complicated by portal hypertension following renal transplantation. World J Gastroenterol. 20(9):2420.
- Calabrese E, Hanauer SB. (2011). Assessment of non-cirrhotic portal hypertension associated with thiopurine therapy in inflammatory bowel disease. J Crohns Colitis. 5(1):48–53.
- Elsing C, Placke J, Herrmann T. (2007). Alcohol binging causes peliosis hepatis during azathioprine therapy in Crohn’s disease. World J Gastroenterol. 13(34):4646–8.
- Sanz-Canalejas L, Gómez-Mampaso E, Cantón-Moreno R, Varona-Crespo C, Fortún J, et al. (2014). Peliosis hepatis due to disseminated tuberculosis in a patient with AIDS. Infection. 42(1):185–189.
- Angelakis E, Raoult D. (2014). Pathogenicity and treatment of Bartonella infections. Int J Antimicrob Agents. 44(1):16–25.
- Cordeiro RA, Hoff LS, Garcia MVF, Leão Filho HM, Borba EF. (2018). Peliosis hepatis and systemic lupus erythematosus: A rare condition identified by magnetic resonance imaging. Rev Assoc Med Bras. 64(1):19–21.
- Kleger A, Bommer M, Kunze M, Klaus J, Leithaeuser F, et al. (2009). First Reported Case of Disease: Peliosis Hepatis as Cardinal Symptom of Hodgkin’s Lymphoma. Oncologist. 14(11):1088–1094.
- Yadav R, Mallick S, Mittal D, Madhusudhan KS, Jana M, et al. (2015). Giant hepatocellular adenoma with peliosis hepatis in a child: A diagnostic dilemma. Trop Gastroenterol. 36(3):200–202.
- D. Crocetti. (2015). Peliosis hepatis: Personal experience and literature review. World J Gastroenterol. 21(46):1318.
- Wang SY, Ruggles S, Vade A, Newman BM, Borge MA. (2001). Hepatic rupture caused by peliosis hepatis. J Pediatr Surg. 36(9):1456–1459.
- Mascagni P, Melandro F, Larghi Laureiro Z, Mennini G, Rossi M. (2018). Spontaneous hepatic rupture in a bodybuilder: A case report and review of the literature. Rev Esp Enfermedades Dig. 110(4):254–256.
.jpg)
.jpg)