Previous Issues Volume 2, Issue 1 - 2018
The Cytopathologist in the Hospital - Based Fnac Clinic: US Image Guidance is Our New Tool to an Even Better Fnac Practice
Richard William Doughty1 , Vivian Orszagh1 , Berit Gravdehaug2 , Torill Sauer1 *
1Akershus University Hospital, Department of Pathology. 2Akershus University Hospital, Department of Breast- and Endocrine Surgery. Corresponding Author: Torill Sauer, Department of Pathology, Akershus University Hospital, N-1478 Lørenskog, Norway, Tel: +47 67964599 E-Mail: [email protected]
Received Date:14 May 2018 Accepted Date: 29 May 2018 Published Date: 30 May 2018
Copyright © 2018 Sauer T
Citation: Sauer T, Doughty RW, Orszagh V and Gravdehaug B. (2018). The Cytopathologist in the Hospital - Based Fnac Clinic: US Image Guidance is Our New Tool to an Even Better Fnac Practice. Mathews J Cytol Histol 2(1): 007.
ABSTRACT
Cytopathologists performing fine-needle aspirations (FNAC) on palpable lesions has a long history. Palpation guided FNAC still has its place, but an increasing number of lesions are being investigated by ultrasound and ultrasound guided FNAC. Naturally this applies to all non-palpable lesions, however the use of ultrasound in the investigation of non-palpable lesions may provide more representative samples and generally improved diagnostic quality. In a hospital based setting, ultrasound imaging and often also FNAC or CNB (core needle biopsy) have traditionally been the domain of radiologists. Today, ultrasound is increasingly being used in a number of specialities for specific investigations, and may now be regarded as a general investigative tool. Currently few cytopathologists (outside the US) have adopted this practice in a hospital- based setting. In this review we describe the model used at a large university hospital in Norway.
KEYWORDS
Ultrasound Imaging; US-Guided FNAC; ROSE; Thyroid Cytology; Head and Neck Cytology
INTRODUCTION
Cytopathologists performing their own fine needle aspiration (FNAC) started in Sweden in the late 1950s and early 1960s, with Sixten Franzen, Joseph Zajicek and Torsten Löwhagen [1] as the early pioneers. This was the beginning of an era where cytopathologists performed FNAC of palpable lesions, in both hospital- and community-based practice. In Europe, hospital based practice was the most common and it was established and performed in collaboration with the specialists from the relevant clinical disciplines and radiologists. In Norway the use of FNAC as a diagnostic tool began in the mid 1980s and has continually evolved and adapted with technological developments advancements, including image-guided FNAC. Ultrasound (US)- and Computer Tomography (CT)-guided FNAC of non-palpable and/or deep - seated lesions have typically been the responsibility of interventional radiologists, with or without collaboration with cytopathologists or cytotechnologists. Today, a major part of FNAC is performed under US-guidance (including EBUS (endoscopic bronchial US) and EUS (endoscopic US)), partly because lesions are not palpable, but also to ensure optimal sampling from representative areas of the lesions. Even in palpable lesions, adequacy has been shown to be better with US-guided FNAC [2-6]. Collaboration with cytopathologists or cytotechnologists is essential to handle the material correctly, both technically and with ROSE (rapid onsite evaluation) [7,8].
During the last decades the use of ultrasound devices in medical diagnostics has expanded enormously. Today, the use of ultrasound in clinical investigations is routine, and often a mandatory part of the work up and follow-up in several specialties. It is no longer seen as a modality only used by radiologist, but has become a general medical tool and is, so to speak, medicines new stethoscope. Cytopathologists have a long history of collaborating with the radiologists, and during the last 10-15 years an increasing number of cytopathologists have started performing ultrasound-guided fine needle sampling with (FNAC) or without (FNC) aspiration [4, 9-13]. The numbers of palpable FNAC have been declining and core needle biopsy (CNB) has replaced FNAC in a number of anatomic locations and indications in large parts of the world. However, FNC is still the primary method in investigation of thyroid nodules, and is also still commonly used in the investigation of lesions in the salivary glands and superficial lymph nodes.
Cytopathologists practicing FN(A)C are a unique hybrid of clinician and laboratory - based physicians. This integrated diagnostic approach ensures optimal representative material (aspiration and primary preparation including smearing and fixation), effective triage of samples for further investigations as well as a final diagnosis that integrates first hand knowledge of clinical history and radiology. As with learning effective technique for the sampling of palpable lesions, learning ultrasound –guided FNAC requires good in-house training to acquire the necessary skills as well as one-to-one instruction from an experienced aspirator [14-16]. Cytopathologists performing US-guided FNC report adequacy, sensitivity and specificity as good as or better than clinicians or radiologist [4,9,10,17].
Ultrasound imaging is used as an aid to visualize the lesion(s) the clinician would like to sample. When the lesion is nonpalpable US guidance is necessary, however it is also recommended, and is advantageous to use ultrasound in palpable lesions. The lesions might be complex, as is often the case in thyroid nodules, situated deeper or somewhat eccentric in comparison to the palpable findings and there may be vessels (Case 1) or other anatomic structures close to our nodule that are optimal not to puncture. Furthermore, the US image might give an indication that the lesion is not what the pathologist or the referring clinician might have thought (Cases 2-4).
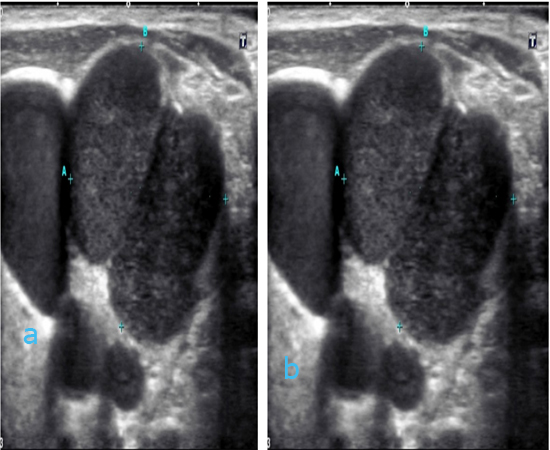
Case descriptions Case 1 (figures 1a-b): 65 yrs old female with known ovarian cancer with metastases. She presented with palpable lymph nodes in the left side of the neck (level 3). Ultrasound revealed multilobulated, well-defined nodules, each with a homogenous, hypoechoic internal echostructure and focal, strong internal vascularity. These lesions represented two enlarged, pathological lymph nodes (1a) in close proximity to the left carotid artery (1b), both measuring 2.2 cm x 1.9 cm. Finding the carotid body among a neck mass, can be anticipated even without Doppler, due to its biphasic pulsatile nature. The same applies to the jugular vein, which, in contrast, has triphasic flow. FNAC showed metastatic adenocarcinoma consistent with origin from the ovaries.
Figure 1a: ultrasound image showing polylobulated, well-defined nodules, each with a homogenous, hypoechoic internal echostructure. They represented two enlarged, pathological lymph nodes and the left carotid artery as shown in the Doppler image 1b. Figure 1b: ultrasound image as above showing that one of the “nodules” is the carotid artery and that the two pathological lymph nodes show focal, strong internal vascularity.
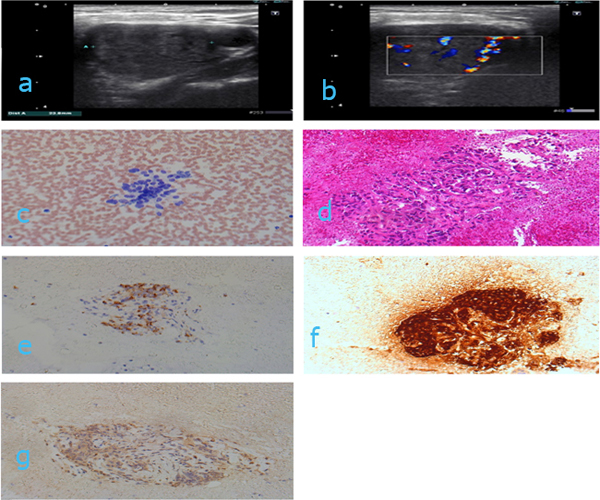
Case 2 (figures 2a-g): 87 yrs old female with palpable, firm tumor in the left side of the neck (level 3). The tumor had been aspirated three times elsewhere, but only blood had been obtained. No ultrasound imaging had been previously performed. Ultrasound examination revealed a triangular welldefined, hypoechoic tumor 2.38 cm in diameter. The carotid artery was situated lateral of the lesion (2a). US Doppler from other angles showed that the tumor actually surrounded the carotid artery (2b). Knowledge of close proximity of the carotid artery to the tumor was crucial to the precise placement and angle of the aspiration needle. Cell block (2d) and air dried, MGG (May Grünwald Giemsa) stained smears were bloody with groups of cohesive tumor cells (2c). Immunostaining on cell block material showed tumor cells patchy positive for chromogranin (2e), strongly positive for synaptophysin (2f), indeterminate for S-100 (2g) and negative for pancytokeratin (image not shown). Both morphology and immunophenotype were consistent with a carotid body tumor. The tumor was not resected.
Figure 2a: Ultrasound image of a triangular, well defined and hypoechoic tumor in the left side of the neck. Laterally there is a vessel, most probably the carotid artery. Figure 2: ultrasound image as 2a showing the vessel going through the tumor. Figure 2c: Direct air dried smear stained with MGG. Magnification x 200. Figure 2d: Cell block stained with HE. Magnification x 100. Figure 2e: Cell block immunostained with chromogranin. Magnification x 100. Figure 2f: Cell block immunostained with synaptophysin. Magnification x 100. Figure 2g: Cell block immunostained with S-100. Magnification x 100.
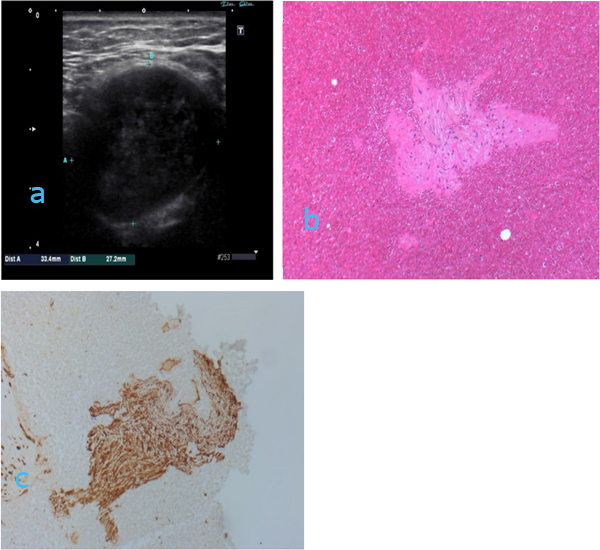
Case 3 (figures 3 a-c): 66 yrs old woman with a suspicious tumor/lymph node in the left side of the neck (level 3). Several previous FNAC had been performed elsewhere using palpation - guided aspiration without ultrasound guidance. These samples had been unsatisfactory due to inadequate material. Ultrasound showed a well-defined hypoechoic tumor (3a) without the typical characteristics of a lymph node (fatty hilus, disk/oval shape). The size was 3.34 cm (width) x 2.72 cm (depth). US color Doppler showed the tumor had poor vascularization. The aspirates (not shown) and the cell block (3b) had scant spindle cell aggregates. Immunostaining revealed these cell groups were positive for S-100 (3c) and negative for desmin (image nor shown). The findings were consistent with a benign schwannoma.
Figure 3a: ultrasound image showing well defined hypoechoic (somewhat heterogeneous echo) tumor in the left side of the neck. Ultrasound characteristics of a lymph node (fatty hilum, disk shape) are not seen. Figure 3b: Cell block stained with HE. Magnification x 100. Figure 3c: Cell block immunostained with S-100. Magnification x 100
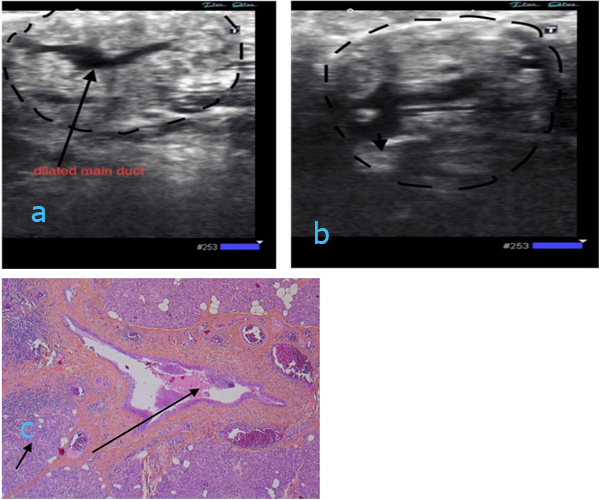
Case 4 (figure 4 a-c): 48 yrs old woman presenting with a tender nodule thought to be an enlarged lymph node in the left submandibular region (level 1). The nodule had appeared after a common cold. The patient had night sweats and the clinician considered the clinical picture was “strongly suspicious for malignant lymphoma”. On ultrasound the nodule did not have the typical characteristics of an enlarged lymph node, but instead it was noted that the submandibular gland was enlarged and the main duct was dilated (4a) with possible occlusion due to a stone (4b, arrow). Due to lack of a focal lesion, FNAC was not indicated. The gland was surgically resected a number of weeks later and sent for histological examination. Histologically there was a chronic inflammation and dilated main duct (4c) with a stone occluding the main duct (not shown).
Figure 4a: ultrasound image showing submandibular gland with dilated central duct (long arrow).
Figure 4b: ultrasound image of the same and with possible occlusion by a stone (arrow) Figure 4c: Histological section from removed submandibular gland (short arrow) with dilated duct (long arrow) stained with HE. Magnification x 50.
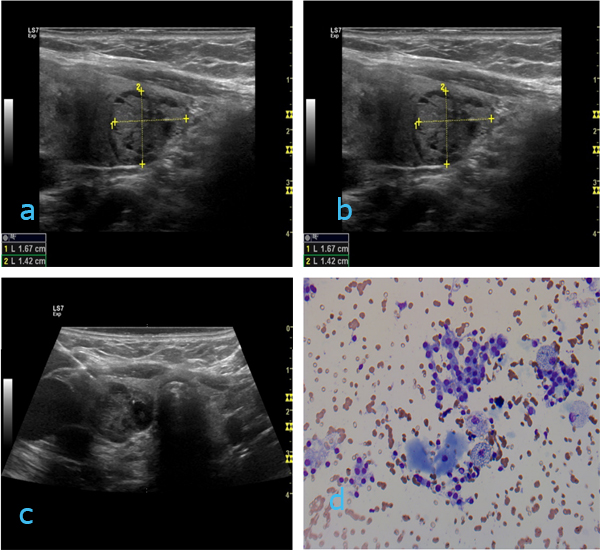
Case 5 (figures 5a-d):72 yrs old woman with two nodules in the right thyroid lobe. The dominant nodule was solid, isoechoic and centrally located (figure 6). The second was situated at the caudal pole. This nodule was lobulated with heterogeneous echogenicity with multiple cystic areas (figure 5a). The lesion measured 1.67x1.42x1.35 cm. US color Doppler showed predominantly peripheral vessels (figure 5b). FNC was performed on both nodules and is visualized in figure 5c. The cytological diagnosis for both lesions was benign follicular nodule (Bethesda II) (figure 5d).
Figure 5a: Ultrasound image of the right thyroid lobe with a lobulated nodule with heterogeneous echo and cystic parts in the caudal part of the lobe. Size 1.67x1.42x1.35 cm. Figure 5b: Ultrasound Doppler image showing peripheral vessels. Figure 5c: Ultrasound image demonstrating needle in place. Figure 5d: Direct smear (MGG) from benign nodule (Bethesda II). Magnification x 200.
In cases 2 -4 the referring clinician strongly suspected a malignant lesion. The ultrasound imaging was an important contributor in making the correct diagnosis, securing a representative material and prompting additional sampling to obtain material for immunostaining (cases 2 and 3). This intergrated approach allowed the pathologist to deliver high-quality definite diagnoses.
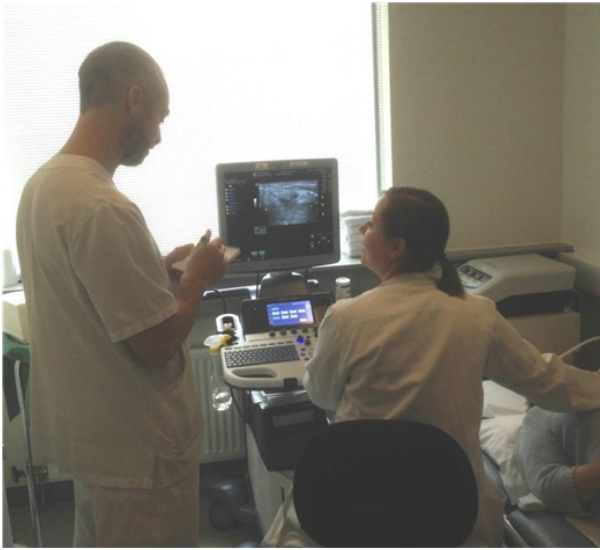
Figure 6: US imaging with two cytopathologists at work in the out patient clinic.
DISCUSSION
As cytopathologists we would describe and document the US findings (location, size in three dimensions, echogenicity, vascularization, microcalcifications), but we would not make a radiological diagnosis. Instead the cytopathologist uses this information to aid the cytological diagnosis. Examples of image descriptions are given in tables 1 and 2. An introductory US course to get familiar with normal anatomy and some common pathological findings is very useful [18-21]. With a basic skill set acquired, the next step would be attending one of the dedicated courses on US-guided FNC. For pathologists in North America the course given by the College of American Pathologists (CAP): Ultrasound-Guided Fine-Needle Aspiration Advanced Practical Pathology Program (https://learn.cap. org/Activity/3371965/Detail.aspx) is a very good option.
Table 1:Example description of thyroid ultrasound findings.
- The left lobe measures 2,4 x 1,8 x 3,3 cm
- Isthmus thickness 0, 2 cm
- The right lobe measures 2,8 x 2,5 x 3,8cm and harbors a centrally located solid nodule, well-defined and hypoechoic without halo, as well as scattered microcalcifications and peripheral vessels. The nodule measures 2,2 x 1,8 x 2,5 cm.
- Ultrasound-guided FNC done
- ROSE: adequate material
Table 2: Example description of lymph node ultrasound findings
- Palpable, enlarged lymph node situated laterally and behind the left m. sternocleidomastoideus (Level 3)
- Hypoechoic, round and without fatty hilus, measures 2,5 x 3 cm. No necrosis
- FNC done
- ROSE: adequate material
- Additional material obtained for immunocytochemistry
TS established FN(A)C clinic on palpable lesions in a university hospital in the late 1980s. Ultrasound guidance was added to this clinic in 2011 and run for about two years. That experience was used to establish the US FN(A)C clinic at Akershus University Hospital, Norway (Ahus).
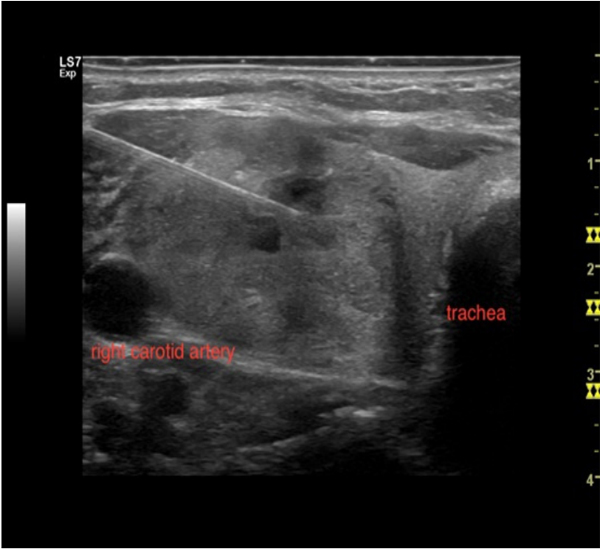
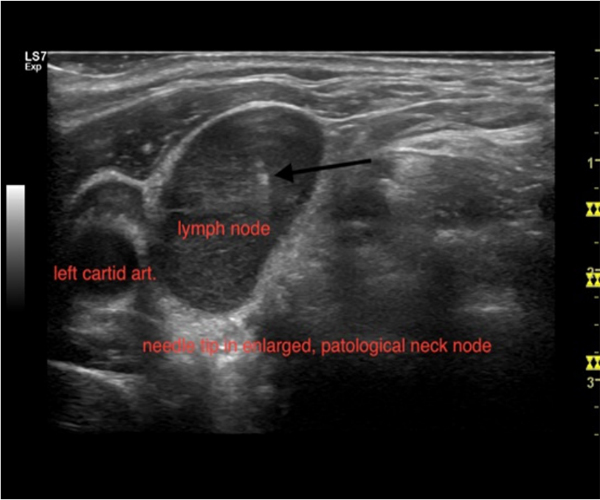
In-house training at Ahus takes place as part of the general pathology residency. Training in FN(A)C, with and without ultrasound guidance is offered and takes place during the last 6 months of the cytopathology training. Phantom training is not used. The residents always work alongside a cytopathologist with experience in US-guided FNAC (figure 6). The residents start by performing US-FNAC on large lesions that are relatively easy to sample. As they gain experience and confidence they gradually expand the size and type of lesions they sample until they can perform US-FNAC on the whole range of lesions typically seen in the clinic. During this period there is always an experienced cytopathologist as backup. In most cases at the Ahus clinic, the longitudinal (figure 7) approach is used, but depending on the location of a lesion, a transversal may also be applied (figure 8).
Figure 7: ultrasound image of longitudinal needle insertion in a solid thyroid nodule. The whole length of the needle is visible. clinic.
Figure 8: u ultrasound image of transversal needle insertion in an enlarged and pathological neck node. Only the needle tip is visible (arrow).
The US machine is usually situated on the left side of the bench and near the patient’s head. Where to sit during aspiration depends on the location of our target, but in most cases on the same side as the US machine and quite near. Personal preferences may differ, though.
The final report typically includes both US imaging and cytological findings. National guidelines and/or recommendations for the work up of thyroid nodules and other lesions are in place in many countries [22-28]. In the setting of pathologist directed US-guided FN(A)C, we can provide the patient and the referring doctor with a high quality of information for that particular patient with optimal integration of US imaging findings and cytology.
In hospital based practices, there might be conflicts with other specialties such as radiology, endocrinology, and surgery as to who should perform US-guided FNC and a working relationship with one of these disciplines should be sought.
At Ahus (a university hospital near Oslo, Norway) our main collaborator is the breast-and endocrine department. The pathologist FN(A)C clinic is located at the breast-and endocrine outpatient clinic, where the cytopathologists work alongside or physically together with the endocrine surgeons. Thyroid referrals are seen here on 4 out of five working days per week. Through this collaboration with the breast- and endocrine surgeon three different working systems have developed. The current Ahus model:
1. Pathologist –only with referrals from outside the hospital as well as in house. Referrals represent lesions in superficial locations and consist of 50 % thyroid lesions whereas the rest are lymph nodes, salivary glands and soft tissue. Breast lesions are handled in the breast radiology and are not referred to the cytopathologist. The cytopathologist does the ultrasound to visualize the lesion and the surrounding tissue. Image still photos for documentation are digitally stored. The images can be printed out, but this is not routinely done and printed images are not a part of the final report. ROSE is done in most cases (figure 9).
Direct air dried smears are prepared and eventually also liquid based fixation specimens (SurePath), depending on the amount of material and the need for adjunct ICC (immunocytochemistry) or other special investigations. In cases with no ultrasound findings in superficial lesions, the referring clinician is notified, but the patient is not referred from us to specialist (radiological) ultrasound. If a previous CT scan indicates a deeper, not superficial lesion, the patient is referred directly to radiology and not to the cytopathologist. Ultrasound of axillary lymph nodes (ALN) can sometimes be located deep in the axillae and not within the range of our US probes. Such patients are referred to the radiology department for further work up.
Figure 9: Transportable ROSE table
2. Endocrine surgeon-only with new thyroid referrals and follow up visits including repeat US examination and FNC as well as parathyroid diagnostics including ultrasound and US-guided FNC of thyroid nodules. The cytopathologist is not present at the clinic and no ROSE is performed.
3. Endocrine surgeon and pathologist together with new thyroid referrals and follow up visits. The endocrine surgeon and the cytopathologist alternate in performing ultrasound and US-guided FNC. ROSE is performed when there is doubt about the representativity of the material, but not in all cases. This collaboration is unique and allows immediate interdisciplinary discussion about findings and interpretation of these as well as further work-up and handling of the patients. Such discussions may be regarding selection for surgery, whether adjunct genetic molecular analysis should be performed, and/or the need for further follow up visits or not.
In-house clinicians quite often have a desire to look in the microscope while the patient is still with us and discuss our preliminary findings. There are regular informal meetings between the responsible endocrine surgeon and one of the cytopathologists (TS) to discuss findings and further handling of patients with thyroid lesions Bethesda III-VI. Currently there are no consensus conferences or tumor board meetings.
The aim of investigating thyroid nodules is to diagnose neoplastic lesions, in order to select those patients that require surgery, to avoid too may non-neoplastic nodules in the operated patient group and to safely diagnose and identify the non-neoplastic nodules that can be left in situ. Tables 3 and 4 show some preliminary results and demonstrate the effect of ROSE and the participating cytopathologist. Inadequate samples are reduced to 2.2 % and 0.1 %, respectively. Sensitivity, specificity, NPV and PPV are based on samples with histological confirmation. Bethesda IV comprised the category “neoplastic lesions”, whereas Bethesda V and VI were included in the malignant category. Few cases of Bethesda III had histological confirmation and they are not included in the calculations. Bethesda III cases are operated if they have a BRAF mutation, indicating that the lesion might be a papillary carcinoma or due to size or clinical symptoms, but otherwise just have clinical follow up. The number of patients with histological confirmation is limited, but shows a better selection of cases for operation of cytologically neoplastic (Bethesda IV), suspicious (Bethesda V) or malignant (Bethesda VI) nodules. The number of unnecessary surgeries of non-neoplastic nodules is reduced. The sensitivity for malignancy is increased and this results in improved selection for surgery.
Table 3: Relationship with paitent’s pathoclinic features and CD47 staining intensity
| Bethesda categories | Surgeon alone | Cytopathologist w/surgeon | Cytopathologist alone | Numbers and percentages of cases |
|---|---|---|---|---|
| Unsatisfactory (Bethesda I) | 112 (12.2 %) | 14 (2.2 %) | 4 (0.1 %) | 130 (5.2 %) |
| Benign (Bethesda II) | 729 | 504 | 867 | 2100 (83.4 %) |
| AUS/FLUS (Bethesda III) | 13 | 15 | 6 | 34 (1.4 %) |
| Follicular neoplasm (Bethesda IV) | 50 | 76 | 61 | 187 (7.4 %) |
| Suspicious for malignancy (Bethesda V) | 3 | 6 | 4 | 13 (0.5 %) |
| Malignant (Bethesda VI) | 8 | 14 | 32 | 54 (2.1%) |
| Total number of cases with precentages | 915 (36.3 %) | 629 (25 %) | 974 (38.7 %) | 2518 |
Table 4: Some comparative results thyroid FNC (unpublished data based on 184 cases with histological confirmation, Ahus) (neoplastic lesions= BIV, malignant lesions = BV and BVI).
| Endocrine Surgeon N= 79 | Surgeon + Cytopathologist N= 29 | Cytopathologist N=76 | |
|---|---|---|---|
| Sensitivity neoplastic lesions | 61.2 % | 91.6 % | 97.8 % |
| Specificity neoplastic lesions | 82.8 % | 53.9 % | 93.1 % |
| PPV neoplastic lesions | 65.5 % | 64.7 % | 95.8 % |
| NPV neoplastic lesions | 80 % | 87.5 % | 96.4 % |
| Sensitivity malignant lesions | 33.3 % | 60 % | 83.3 % |
| Specificity malignant lesions | 96.2 % | 96.1 % | 95.4 % |
| PPV malignant lesions | 60 % | 66.2 % | 69.9 % |
| NPV malignant lesions | 93.2 % | 95.2 % | 96.9 % |
Numerous cytopathologists are already skilled aspirators with very good FNAC results. Ultrasound imaging has become a general medical tool used in a number of specialities. In cytopathology ultrasound examination is used as a tool for visualization and allows guided sampling of the lesions. By adding ultrasound examination and sampling to their skill-set cytopathologists performing FN(A)C in a hospital-based setting have the opportunity to secure high quality diagnoses, integrate and interact with other clinicians and improve patient care .
CONFLICT OF INTEREST
All authors state that they have no conflicts of interest.
REFERENCES
- Linsk JA. (1985). Aspiration cytology in sweden: The Karolinska group. Diagn Cytopathol. 1(4): 332-335.
- Danese D, Sciacchitano S, Farsetti A, Andreoloi M, et al. (1998). Diagnostic Accuracy of Conventional Versus Sonography-Guided Fine-Needle Aspiration Biopsy of Thyroid Nodules. Thyroid : official journal of the American Thyroid Association. 8(1): 15-21.
- Krishnappa P, Ramakrishnappa S and Kulkarni MH. (2013). Comparison of Free Hand versus Ultrasound-Guided Fine Needle Aspiration of Thyroid with Histopathological Correlation. JEnvironPatholToxicolOncol. 32(2):149-155.
- Wu M. (2011). A comparative study of 200 head and neck FNAs performed by a cytopathologist with versus without ultrasound guidance: evidence for improved diagnostic value with ultrasound guidance. Diagn Cytopathol. 39(10): 743-751.
- Mangla G, Aora VK and Singh N. (2015). Clinical audit of ultrasound guided fine needle aspiration in a general cytopathology service. J Cytol. 32(1): 6-11.
- Mehrotra P, Viswanathan H, Johnson SJ, Wadehra V, et al. (2006). Ultrasound guidance improves the adequacy of our preoperative thyroid cytology but not its accuracy. Cytopathology. 17(3): 137- 144.
- Witt BL and Schmidt RL. (2013). Rapid Onsite Evaluation Improves the Adequacy of Fine-Needle Aspiration for Thyroid Lesions: A Systematic Review and Meta-Analysis. Thyroid : official journal of the American Thyroid Association. 23(4): 428-435.
- Sauer T, Freng A and Djupesland P. (1992). Immediate Interpretation of FNA Smears From the Head and Neck Region. Diagn Cytopathol. 8(2):116-118.
- Bellevicini C, Vigliar E, Malapelle U, Pisapia P, et al. (2016). Cytopathologists can reliably perform ultrasound-guided thyroid fine needle aspiration: a 1-year audit on 3715 consecutive cases. Cytopathology. 27(2) :115 -121.
- Pagni F and Di Bella C. (2013). The Interventional Pathologist. Arch Pathol Lab Med. 137(1):11-12.
- Abele J. (2008). The Case for Pathologist Ultrasoundguided Fine-Needle Aspiration Biopsy. Cancer Cytopathol. 114(6): 463-468.
- Lieu D. (2008). Cytopathologist-performed ultrasoundguided fine-needle aspiration and core-needle biopsy: a prospective study of 500 consecutive cases. Diagn Cytopathol. 36(5): 317-324.
- Paksoy N and Ozbek B. (2018). Cytopathologist‑performed and ultrasound‑guided fine needle aspiration cytology enhances diagnostic accuracy and avoids pitfalls: An overview of 20 years of personal experience with a selection of didactic cases. CytoJournal. 15(9): 31-39.
- Britt-Marie Ljung. Papanicolaous society of cytopathology.
- Abele JS, Miller TR, King EB and Lowhagen T. (1985). Smearing techniques for the concentration of particles from fine needle aspiration biopsy. Diagn Cytopathol. 1(1): 59-65.
- Abele JS. The science and art of optimal fine needle biopsy and smear making. 2nd edition ed. LA O, editor. San Diego: Plural Publishing; 2017.
- Graciano AJ, Fischer CA, Chone CT, Bublitz GS, et al. (2017). Efficacy of ultrasound-guided fine-needle aspiration performed by surgeons newly trained in thyroid ultrasound. Head Neck. 39: 439-442.
- Baskin J, Duick DS, Levine RA. Thyroid Ultrasound and Ultrasound-Guided FNA 3rd Edition. New York: Springer; 2013.
- Orloff LA. (2017). Head and Neck Ultrasonography: Essential and Extended Applications, 2nd Edition. San Diego: Plura Publishing.
- Milas M, Mira, Mandel SJ, Langer JE, et al. (2017). Advanced Thyroid and Parathyroid Ultrasound. New York: Springer.
- Sofferman RA and Ahuja A. (2012). Ultrasound of the Thyroid and Parathyroid Glands. New York: Springer.
- Anil G, Hegde A and Chong FH. (2011). Thyroid nodules: risk stratification for malignancy with ultrasound and guided biopsy. Cancer Imaging. 11: 209-223.
- Cooper DS, Doherty GM, Haugen BR, Kloos RT, et al. (2009). American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid CancerRevised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid : official journal of the American Thyroid Association.19(11): 1167-1214.
- Frates MC, Benson CB, Charboneau JW, Cibas ES, et al. (2005). Management of thyroid nodules detected at US: Society of Radiologists in Ultrasound consensus conference statement. Radiology. 237(3): 794-800.
- Abele JS. (2012). Putting aspiration back into thyroid fineneedle biopsy-the re-emerging role of vacuum assistance. Cancer Cytopathol. 120(6): 366-372.
- Ljung BM, Langer J, Mazzaferri EL, Oertel YC, et al. (2008). Training, credentialing and re-credentialing for the performance of a thyroid FNA: a synopsis of the National Cancer Institute Thyroid Fine-Needle Aspiration State of the Science Conference. Diagn Cytopathol. 36(6): 400-406.
- Pitman MB, Abele J, Ali SZ, Duick D, et al. (2008). Techniques for thyroid FNA: a synopsis of the National Cancer Institute Thyroid Fine-Needle Aspiration State of the Science Conference. Diagn Cytopathol. 36(6): 407-424.
- Su HK, Dos Reis LL, Lupo MA, Milas M, et al. (2014). Striving toward standardization of reporting of ultrasound features of thyroid nodules and lymph nodes: a multidisciplinary consensus statement Thyroid : official journal of the American Thyroid Association. 24(9): 1341-1349.