Information Links
Related Conferences
Previous Issues Volume 5, Issue 1 - 2021
Sleep Disorders in Patients with Hypertension and Type 2 Diabetes Mellitus
Anna Isayeva, Olena Buriakovska*
Department of Complex risk Reduction of Chronic non-communicable diseases, L.T. Malaya Therapy National Institute of the National Academy of Medical Sciences of Ukraine, Ukraine
Received Date: November 25, 2020
Published Date: March 10, 2021
*Corresponding Author: Olena Buriakovska, Department of Complex risk Reduction of Chronic non-communicable diseases, L.T. Malaya Therapy National Institute of the National Academy of Medical Sciences of Ukraine, Ukraine, E-mail: [email protected]
Copyrights: Isayeva A, et al. @ 2021.
Citation: Isayeva A, and Buriakovska O. Sleep Disorders in Patients with Hypertension and Type 2 Diabetes Mellitus. Mathews J Neurol. 5(1): 18.
Background: Sleep disorders are one of the most frequent non-cardiac complains in hypertensive patients. At the same time insomnia, short sleep duration and obstructive sleep apnea are well-documented risk factors of hypertension and diabetes mellitus. However, information concerning the frequency of different types of sleep disorders in patients with hypertension is limited.
Aim: The aim of this study was to assess the incidence of different types of sleep disorders and the level of daytime sleepiness in patients with hypertension and type 2 diabetes mellitus.
Methods: A cross-sectional study involving 120 patients was conducted. There are 82 patients with combined hypertension and diabetes mellitus type 2; 38 patients with hypertension and 23 healthy participants aged over 45 years. Insomnia was diagnosed according to criteria of International Classification of Sleep Disorders, 3rd Edition. Epworth scale was used to assess the severity day sleepiness. Obstructive sleep apnea was diagnosed with SOMNO check micro.
Results: Insomnia predominated in the group of patients with hypertension, while circadian rhythm sleep disorders as well as obstructive sleep apnea were more frequent in the group of combined hypertension and diabetes mellitus. The patients with hypertension and diabetes mellitus had significantly greater daytime sleepiness on the Epworth scale compared to patients with isolated hypertension and the control group.
Conclusion: The most common sleep disorders in patients with hypertension and diabetes mellitus are insomnia and circadian rhythm sleep-wake disorders. Sleep disorders are more frequent in patients with hypertension and diabetes mellitus than in control group. Insomnia prevalated in patients with hypertension, however circadian sleep disorders prevalated in patients with hypertension and diabetes mellitus.
KEYWORDS: Sleep Wake Disorders; Dyssomnias; Sleep Disorders; Circadian Rhythm; Sleepiness; Hypertension; Diabetes Mellitus; Sleep Apnea; Obstructive
INTRODUCTION
Sleep is an integral part of the high-quality human life. Normal sleep quality depends on the peculiarities of neurophysiological regulation, hormonal profile, genetic predesposity presence of concomitant diseases and social and cultural factors [1-3]. Healthy sleep is necessary for full functioning of the brain, memory, regulation of metabolic processes, muscle recovery. Moreover, most of the repair processes occur during this period. At the same time, all physiological functions of sleep have not been determined yet. Of particular interest is the effect that quality sleep can have on cardiovascular system health. But the majority of studies are focused on the relationship between sleep duration and the state of the cardiovascular system [4]. The American Academy of Sleep Medicine (AASM) and the Sleep Research Society (SRS) recommend that adults of 18-60 years old sleep 7 or more hours per night [5]. In addition, the National Sleep Foundation (NSF) Consensus Report insists that normal sleep duration depends on age, gender, genetic factors. Adults of 18 to 64 years old are advised to sleep 7-9 hours, while 7-8 hours are suggested for people over 65 years old [5]. At the same time, sleep duration is genetically determined and some healthy people have a short sleep without any destructive health effects, including those for the cardiovascular system [6]. While not enough work is devoted to the study of sleep quality. Moreover, according to data obtained by Kocevska D, et al. after analyzing sleep data of more than 200 thousand patients, poor sleep quality (13.3%) and insomnia symptoms (9.6-19.4%) were more common than short sleep duration (6.5% with TST <6 h) in adults (≥18 years old) [7].
Data on the prevalence of various sleep disorders are rather contradictory. In a study of the general population, Ford DE, et al. and Quera-Salva MA, et al. reported the prevalence of insomnia as 10 to 48% [8,9]. According to other authors, the prevalence of insomnia among adults in the world reaches 33-50% [10]. Obstructive sleep apnea in general population occurs much less frequently than insomnia and accounts for 2-4% in men and 1-2% in women out of the general population [11]. Data on the prevalence of circadian rhythm sleep disorders are practically absent.
The aim of this study was to assess the incidence of various sleep disorders and the level of daytime sleepiness in patients with hypertension and diabetes mellitus.
MATERIALS AND METHODS
A cross-sectional study involving 120 patients was conducted. There are 82 patients with combined hypertension and diabetes mellitus type 2 (group 1), 38 patients with hypertension (group 2) and 23 healthy participants (control group). Criteria for inclusion in the study were age over 45 years, presence of hypertension. The diagnosis of hypertension was established in accordance with the recommendations for the management of patients with arterial hypertension of the European Society of Cardiology / European Society of Hypertension (2018) [12]. The diagnosis of diabetes mellitus was established in accordance with the recommendations (2019) ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD [13]. The study did not involve patients with a history of cardiovascular events (myocardial infarction, stroke, were excluded according to ESC), patients with heart failure or ejection fraction less than 45%, and Ear, Throat, and Nose problem, with uncontrolled arterial hypertension or severe hypertension, thyroid dysfunction, gastric ulcer and duodenal ulcer at exacerbation stage, with concomitant diseases that affect the quality of life, patients with degree 3 obesity, with concomitant active cancer diseases, with depressive states (organic depressive disorder (F 06.32), recurrent depressive disorder (F 33.0-3) or depressive episode of any severity (F 31.3-5, F 32.0-3), as well as mixed anxiety and depressive disorder (F 41.2) according to the criteria of the International Classification of Diseases, 10th revision (ICD-10), with insomnia as a symptom of any mental illness, with concomitant therapy with glucocorticosteroids, centrally acting antihypertensive drugs (moxonidine, reserpine), β-adrenergic blocking agents, somniferous agents, patients who confirmed consumption of food at night. All patients in the study received ramipril) or valsartan, a diuretic (indapamide) and a calcium antagonist (amlodipine) to lower blood pressure. All patients were treated by atorvastatin as lipid-lowering therapy. All patients received metformin as antidiabetic therapy.
Assessment of the Presence of Sleep Disorders
The presence of insomnia was diagnosed using the criteria of the International Classification of Sleep Disorders (Issue 3), which was developed by the International Academy of Sleep Disorders (2014): 1 – difficulty initiating sleep or maintaining continuous sleep; 2 – the availability of adequate sleeping conditions; 3 – daytime effects of poor sleep quality [14].
Daytime sleepiness was assessed using Epworth Sleepiness Scale [15]. Degrees of severity on the Epworth scale are distributed as follows, where 0 - 6 points - everything is normal: no signs of excessive daytime sleepiness, 7 - 8 points - moderate daytime sleepiness, 9 - 17 points - significant daytime sleepiness, 17 points or more - sharp daytime drowsiness. The patient was interviewed by a pre-trained study doctor.
To detect sleep apnea syndrome, night monitoring was performed using the SOMNOcheck micro CARDIO cardio-respiratory complex (Germany). The device allows registering the flow of breath, blood saturation, heart rate and is certified for the diagnosis of sleep apnea syndrome [16].
Ethical Aspects
The study was approved by the Committee on Ethics and Deontology of the State Institution “L.T. Malaya Therapy National Institute of the National Academy of Medical Sciences of Ukraine” on 28 April 2017. All participants of the study signed an informed consent; preliminarily they were clearly explained the purpose and objectives of the study.
STATISTICAL ANALYSIS
The type of data distribution was checked by the Kolmogorov-Smirnov (K-S) test. With normal distribution of continuous values, the data are presented as mean values (M) and standard deviation (SD); for quantitative values, the data are presented as the number (n) and frequency (%). Discrepancies were considered statistically significant at p <0.05.
RESULTS
Clinical characteristics of the groups presented in table 1
There were no significant differences between group of hypertension and hypertension + diabetes mellitus in age, ramipril and valsartan use, dose of ramipril, valsartan, amlodipine and indapamid. However, the patients with were hypertension and diabetes mellitus took amlodipine and indapamid more frequently than patients without diabetes mellitus.
|
Index |
Group 1 (Hypertension+ DM 2T) n = 82 |
Group 2 (hypertension) n = 38 |
Control group n = 23 |
p |
|
|
Age, years (M±SD) |
59,15±10,86 |
55,05±9,57 |
53,96±9,06 |
р1-2=0,59 р1-3=0,06 p2-3=0,57 |
|
|
Diabetes mellitus type 2 (n (%)) |
82 (100%) |
0 |
0 |
р1-2<0,05 р1-3<0,05 p2-3 N/A |
|
|
Hypertension |
mild |
0 |
8 (21,05%) |
0 |
р1-2<0,05 р1-3<0,05 p2-3 N/A |
|
moderate |
82 (100%) |
30 (78,95%) |
0 |
р1-2=0,034 р1-3<0,05 p2-3<0,05 |
|
|
Metformin use |
82 (100%) |
0 |
0 |
р1-2<0,05 р1-3<0,05 p2-3 N/A |
|
|
Dose of metformin, mg |
1350,0±732,90 |
0 |
0 |
р1-2<0,05 р1-3<0,05 p2-3 N/A |
|
|
Ramipril use |
32 (39%) |
16 (42%) |
0 |
р1-2=0,23 р1-3<0,05 p2-3<0,05 |
|
|
Dose of ramipril, mg |
6,67±2,73 |
8,64±2,34 |
0 |
р1-2=0,45 р1-3<0,05 p2-3<0,05 |
|
|
Valsartan use |
50 (61%) |
22 (58%) |
0 |
р1-2=0,67 р1-3<0,05 p2-3<0,05 |
|
|
Dose of valsartan, mg |
272,68±98,57 |
268±88,41 |
0 |
р1-2=0,90 р1-3<0,05 p2-3<0,05 |
|
|
Amlodipin use |
49 (60,5%) |
15 (39,95%) |
0 |
р1-2=0,04 р1-3<0,05 p2-3<0,05 |
|
|
Dose of amlodipine |
6,05±2,30 |
6,0±2,64 |
0 |
р1-2=0,94 р1-3<0,05 p2-3<0,05 |
|
|
Indapamid use |
33 (65,85%) |
23 (34,15%) |
0 |
р1-2=0,03 р1-3<0,05 p2-3<0,05 |
|
|
Dose of indapamid, mg |
2,25±1,02 |
2,38±1,75 |
0 |
р1-2=0,92 р1-3<0,05 p2-3<0,05 |
|
Table 1: Clinical characteristics of the groups with hypertension and hypertension + diabetes mellitus type 2.
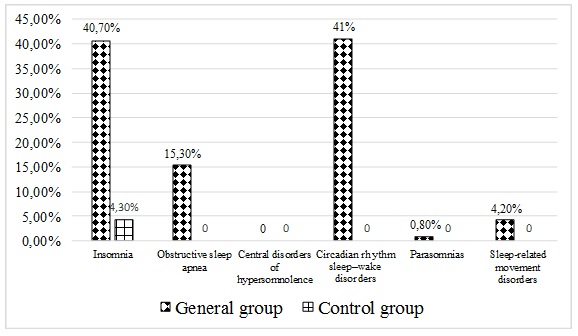
The study analyzed the incidence of all types of sleep disorders in a general group of patients. It was found that circadian rhythm sleep disorders and wakefulness as well as insomnia were observed most frequently (Figure 1). Obstructive sleep apnea was detected in 15.3% of patients. Sleep-associated motor disorders were diagnosed in 4.2% of patients. Parasomnia was detected in 1 patient who was referred for further observation to a neurologist. No sleep disorders of central genesis were detected during the examination of the entire group. In the control group, only insomnia was observed of all sleep disorders – in 4.3% of patients.
Figure 1: Frequencies of occurrence of different types of sleep disorders in studied and control groups.
Thus, circadian rhythm sleep disorders, wakefulness and insomnia dominated in the general group.
All sleep disorders were analyzed separately by groups of patients: group 1 – patients with combined hypertension and type 2 diabetes mellitus, group 2 – patients with isolated hypertension, group 3 – control.
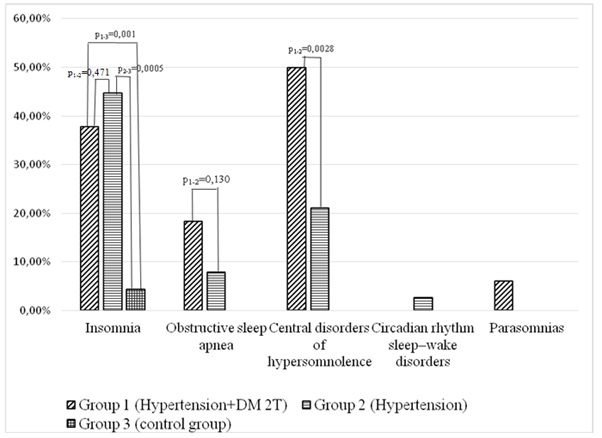
Figure 2: Different sleep disorders in patients with hypertension group, hypertension and diabetes mellitus group and control group.
It was found out that insomnia was significantly more common in patients with isolated hypertension and with its combination with diabetes mellitus compared with the control group (р1-3 = 0.001, р2- = 0.005). When comparing the isolated hypertension and its combination with diabetes mellitus, insomnia was found to be more common in patients with hypertension (p = 0.47). It was observed that obstructive sleep apnea was significantly more common in patients with combined hypertension and diabetes mellitus compared with the control group (р1-3 = 0.01) and the group of patients with isolated hypertension (р1-2 = 0.13). When comparing groups 1 and 2, it was found that circadian rhythm sleep disorders were significantly more common in the group of combined hypertension and type 2 diabetes mellitus than in the group of isolated hypertension. The analysis of sleep-associated motor disorders did not show a significant difference between the groups. Insomnia predominated in the group of patients with hypertension, while circadian rhythm sleep disorders and wakefulness as well as obstructive sleep apnea dominated in the group of combined hypertension and type 2 diabetes mellitus.
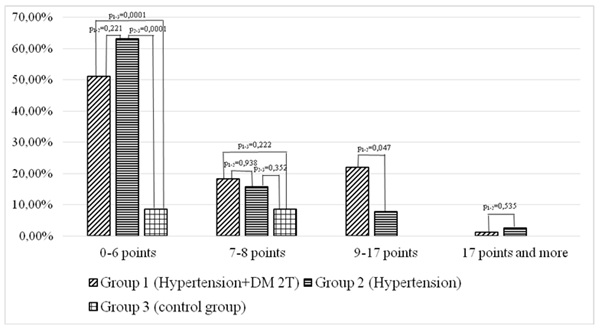
Such a parameter as daytime sleepiness was analyzed according to the Epworth questionnaire. For example, the average score on the Epworth Sleepiness Scale in the general group of examined patients was 6.17 ± 3.73 that corresponded to the normal condition. Analyzing this parameter by groups found out that the daytime sleepiness score on the Epworth scale was 6.53 ± 3.57 in the group 1 (combination of hypertension and type 2 diabetes mellitus) and 5.38 ± 4.02 in the group 2 (isolated hypertension) that was less than in group 1 (p = 0.047).
Figure 3: Daytime sleepiness according to Epworth scale in patients with hypertension group, hypertension and diabetes mellitus group and control group.
Thus, it was detected that patients with combined hypertension and diabetes mellitus had significantly greater daytime sleepiness on the Epworth scale compared with patients with isolated hypertension (p1-2 = 0.047) and the control group (p1-3 = 0.007).
|
|
Insomnia |
Circadian rhythm sleep-wake disorders |
Obstructive sleep apnoea |
p |
|
Average score on the Epworth scale, M±SD |
6,135±3,71 |
6,36±3,77 |
6,24±3,73 |
р1-2=0,89 р2-3=0,96 р1-3=0,78 |
Table 2: Severity of daytime sleepiness in patients with insomnia, circadian rhythm sleep-wake disorders and obstructive sleep apnea.
There were no differences in daytime sleepiness between patients with insomnia, circadian rhythm sleep-wake disorders and obstructive sleep apnea.
DISCUSSION
Good sleep quality is linked to general and cardiovascular health. Although there are enough studies devoted to sleep disorders in patients with cardiovascular pathology, the majority of them concentrated on sleep apnea and sleep duration. However, sleep disorders include different condition and insomnia is one of the most frequent sleep abnormalities. The features of present study are an analysis of all possible sleep disorders according to International Classification of Sleep Disorders in contrast to work investigating only sleep apnea or sleep duration. It should be noted, that sleep disorders are difficult to diagnose and confirm. Although most published studies use polysomnography to assess sleep, this method cannot be optimal because of the inconvenience and its potential negative influence on sleep quality. In our work, we used questionnaires to identify sleep disorders that simplifies the procedure and have no effect on sleep quality.
In 2006, the Behavioral Risk Factor Surveillance System conducted a population-based study of 150,000 Americans, the aim of which was to identify sleep disorders in the general population using self-monitoring diaries. It was shown that general sleep disturbance was most frequently reported in young adults, and rates generally declined with age. It was found out that the prevalence of sleep disorders in men ranged from 13.7% (70–74 years old) to 18.1% (18–24 years old) and that in women ranged from 17.7% (80 years old and older) to 25.1% (18–24 years old). In the whole group, women reported sleep problems more frequently than men [17]. Problems with sleep reported 23.3% of women in the age period 45-49 years; 24.2% in the age group 50-54 years and 23.5% in the age of 55-59 years. In male population the prevalence of sleep disorders corresponds to 16,9% in the age of 45-49 years; 16.1% in the age of 55-59 years and 17.4% in the period of 55-59 years old. These data are consistent with our results. In our study, also, the majority of patients were women. According to National Health and Nutrition Examination Survey difficulties falling asleep, sleep maintenance difficulties, early morning awakenings, and non-restorative sleep were associated with female gender. Moreover, daytime sleepiness was seen in female more frequently. For example, insomnia was registered in 10.6%. At the same time, patients had such symptoms as difficulty falling asleep (11.71%), difficulty maintaining continuous sleep (13.21%), early morning awakenings (10.7%). Obstructive sleep apnea was observed in 31.5% of the adult population [18].
According to Fiorentini A, et al. [20] hypertension and diabetes mellitus are associated with significant comorbidities, as the sleep-disordered breathing, also the sleep's quality disorders. The prevalence of hypertension was 87.1% in "poor sleepers" versus 35.1% in "good sleepers" subjects. Also, the frequency of diabetes mellitus was significantly higher in “poor sleepers”. So, 19,4% subjects with poor sleep quality had diabetes to compare with 8.8% in group with good sleep quality. However, the authors used Pittsburgh Sleep Quality Index for assessment of sleep quality unlike our study, but this method of sleep quality assessment is also subjective [20].
Quite a large number of works are devoted to the relationship between type 2 diabetes mellitus and various disorders of both quality and duration of sleep. This may be due to many pathogenetic mechanisms: activation of the sympatho-adrenal system, the hypothalamic-pituitary axis, increase of the production of counterinsular hormones, eating disorders, and many others [21]. Although, by far, one of the most common sleep disorders in type 2 diabetes is obstructive sleep apnea, the severity of which correlates with the severity of diabetes and the presence of complications, in our study obstructive sleep apnea was a criterion for exclusion [22].
Daytime sleepiness, like in our study, was assessed using the Epworth scale. The most common was mild daytime sleepiness, which occurred in 17.8% of respondents, moderate to severe symptoms occurred in 5.8%, severe – in 10.9% of respondents. However, mild daytime sleepiness according to the Epworth scale dominated in all groups of our study [18].
Another study conducted in New Zealand on 22,389 healthy volunteers of 16-84 years old showed the incidence of sleep disorders such as insomnia, obstructive sleep apnea, and assessed daytime sleepiness according to the Epworth scale. For example, insomnia was observed in 20% of patients, obstructive sleep apnea in 6%, and severe symptoms of daytime sleepiness in 8.8% of respondents [19].
Thus, insomnia and circadian rhythm sleep disorders were more often diagnosed in the study population. At the same time, insomnia was more typical for patients with isolated hypertension, while circadian rhythm sleep disorders and wakefulness were more typical for combined hypertension and type 2 diabetes mellitus. These sleep disorders were accompanied by the same level of daytime sleepiness.
CONCLUSION
- Patients with hypertension and diabetes mellitus type 2 had significantly more frequently sleep disorders than patients in the control group.
- The most common sleep disorder in patients with hypertension and type 2 diabetes mellitus is insomnia, however the most common sleep disorder in patients with hypertension is circadian rhythm sleep disorders.
- There were no differences in daytime sleepiness between patients with insomnia, circadian rhythm sleep-wake disorders and obstructive sleep apnea in patients with hypertension and type 2 diabetes mellitus.
LIMITATIONS OF THE STUDY
This study did not include patients with previous cardiovascular events and severe hypertension, females dominated in study population and study population was not large enough.
CONFLICTs OF INTEREST
The authors declare there are no conflicts of interest.
REFERENCES
- Cajochen C, Chellappa S, Schmidt C. (2010). What keeps us awake?–the role of clocks and hourglasses, light, and melatonin. Int Rev Neurobiol. 93:57–90.
- Fuller PM, Lu J. (2009). Neurobiology of sleep. In Amlaner CJ, Fuller PM, editors. Basics of sleep guide. 2nd edition. Westchester (IL): Sleep Research Society. 53–62.
- Mackiewicz M, Naidoo N, Zimmerman JE, Pack AI. (2008). Molecular mechanisms of sleep and wakefulness. Ann N Y Acad Sci.1129: 335–49. doi: 10.1196/annals.1417.030.
- Bertisch SM, Pollock BD, Mittleman MA, Buysse DJ, Bazzano LA, et al. (2018). Insomnia with objective short sleep duration and risk of incident cardiovascular disease and all-cause mortality: Sleep Heart Health Study. Sleep. 41(6):zsy047.
- Cirelli C, Benca R, Eichler AF. (2017). Insufficient Sleep: Definition, Epidemiology, and Adverse Outcomes. Available from: https://www.uptodate.com/contents/insufficient-sleep-definition-epidemiology-andadverse-outcomes [accessed on 21 November 2018].
- Shi G, Yin C, Fan Z, Xing L, Mostovoy Y, et al. (2020). Mutations in Metabotropic Glutamate Receptor 1 Contribute to Natural Short Sleep Trait. Curr Biol. 31(1):13-24. doi: 10.1016/j.cub.2020.09.071.
- Kocevska D, Lysen TS, Dotinga A, Koopman-Verhoeff ME, Juijk MPCM, et al. (2021). Sleep characteristics across the lifespan in 1.1 million people from the Netherlands, United Kingdom and United States: a systematic review and meta-analysis. Nat Hum Behav. 5(1):113-122. doi: 10.1038/s41562-020-00965-x.
- Ford DE, Kamerow DB. (1989). Epidemiologic study of sleep disturbances and psychiatric disorders. An opportunity for prevention? JAMA. 262:1479-84.
- Quera-Salva MA, Orluc A, Goldenberg F, Guilleminault C. (1991). Insomnia and use of hypnotics: study of a French population. Sleep. 14(5):386-91.
- Johnson EO. (2006). Epidemiology of insomnia: from adolescence to old age. Sleep Medicine Clinics. 1(3):305-317. doi: 10.1016/j.jsmc.2006.06.006.
- Maspero C, Giannini L, Galbiati G, Rosso G, Farronato G. (2015). Obstructive sleep apnea syndrome: a literature review. Minerva Stomatol. 64(2):97-109.
- Williams B, Mancia G, Spiering W, Rosei EA, Azizi M, et al; Authors/Task Force Members. (2018). 2018 ESC/ESH Guidelines for the management of arterial hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension: The Task Force for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. J Hypertens. 36(10): 1953-2041. doi: 10.1097/HJH.0000000000001940.
- Cosentino F, Grant PJ, Aboyans V, Bailey CJ, Ceriello A, et al. ESC Scientific Document Group. (2020). 2019 ESC Guidelines on diabetes, pre-diabetes, and cardiovascular diseases developed in collaboration with the EASD. Eur Heart J. 41(2):255-323. doi: 10.1093/eurheartj/ehz486.
- Sateia MJ. (2014). International classification of sleep disorders-third edition: highlights and modifications. Chest. 146(5):1387-1394. doi: 10.1378/chest.14-0970.
- Doneh B. (2015). Epworth Sleepiness Scale. Occup Med (Lond). 65(6):508. doi: 10.1093/occmed/kqv042.
- Bilgin C, Erkorkmaz U, Ucar MK, Akin N, Nalbant A, et al. (2016). Use of a portable monitoring device (Somnocheck Micro) for the investigation and diagnosis of obstructive sleep apnoea in comparison with polysomnography. Pak J Med Sci. 32(2):471-5. doi: 10.12669/pjms.322.9561.
- Grandner MA. (2017). Sleep, Health, and Society. Sleep Med Clin. 12(1):1-22. doi: 10.1016/j.jsmc.2016.10.012.
- Grandner MA, Petrov MER, Rattanaumpawan P, Jackson N, Platt A, et al. (2013). Sleep symptoms, race/ethnicity, and socioeconomic position. J Clin Sleep Med. 9(9):897–905,905A–D. doi: 10.5664/jcsm.2990.
- Wilsmore BR, Grunstein RR, Fransen M, Woodward M, Norton R, et al. (2013). Sleep habits, insomnia, and daytime sleepiness in a large and healthy community-based sample of New Zealanders. J Clin Sleep Med. 9(6):559-66. doi: 10.5664/jcsm.2750.
- Fiorentini A, Valente R, Perciaccante A, Tubani L. (2007). Sleep's quality disorders in patients with hypertension and type 2 diabetes mellitus. Int J Cardiol. 114(2):E50-2. doi: 10.1016/j.ijcard.2006.07.213.
- Khandelwal D, Dutta D, Chittawar S, Kalra S. (2017). Sleep Disorders in Type 2 Diabetes. Indian J Endocrinol Metab. 21(5):758-761. doi: 10.4103/ijem.IJEM_156_17.
- Siwasaranond N, Nimitphong H, Manodpitipong A, Saetung S, Chirakalwasan N, et al. (2018). The Relationship between Diabetes-Related Complications and Obstructive Sleep Apnea in Type 2 Diabetes. J Diabetes Res. 2018:9269170. doi: 10.1155/2018/9269170.