Information Links
Related Conferences
Current Issue Volume 11, Issue 1 - 2026
Sexual Dimorphism in Prognosis and Treatment of Spontaneous Dermatophytosis in Mus Musculus
Alicia Garcia-Falgueras1,2,*
1Spanish University, Madrid, Spain, 28019
2Netherlands Institute for Neuroscience, Meibergdreef 47, 1105 BA Amsterdam, The Netherlands
*Corresponding author: Alicia Garcia-Falgueras, Spanish University, Madrid-28019, Spain, Netherlands Institute for Neuroscience, Meibergdreef 47, 1105 BA Amsterdam, The Netherlands, Email: [email protected]
Received Date: February 28, 2026
Published Date: April 10, 2026
Citation: Garcia-Falgueras, Alicia. (2026). Sexual Dimorphism in Prognosis and Treatment of Spontaneous Dermatophytosis in Mus Musculus. Mathews J Psychiatry Ment Health. 11(1):61.
Copyrights: Garcia-Falgueras, Alicia. (2026).
ABSTRACT
A spontaneous outbreak of dermatophytosis was detected in a domestic rural mouse strain, “Mus musculus” of 24 specimens. This research article presents the possible causes of the outbreak (wounds from fights and/or mite bites), along with the presence of insects (Musca domestica, Xylocopa, Apis) as mechanical vectors for the transmission of fungal spores. The treatment process and the variables involved are described, revealing that only four males of the mice colony (male N =11; females N = 13) presented this skin condition, which was resolved completely with the use of various topical antifungals (Clotrimazol 10 mg/g and Bifonazol 10 mg/gr). In humans itches by skin inflammatory disease goes together with psychological absence of well being (anxiety, depression, suicidal ideation, and substance misuse). The rodents observed with the skin disease showed similar body weight, but also displayed a prolonged scratching behavior, facial expression of intense discomfort, a vehement reaction to surprising or frightening stimuli and a loss of appetite. The potential barrier of the immune system in rodents is considered a possible explanation for the absence of other infections in specimens that shared the same cage. Similarly, the possible effect of their aggressive behavior in captivity under laboratory conditions are considered variables to contemplate in fungal proliferation and the ease with which these infections spread.
Keywords: Fur, Scab, Itches, Mites, Sexual Dimorphism, Chronic Wound, Antifungal, Immune System, Common Fly, Bumblebee, Rural Environment, Mus musculus.
INTRODUCTION
Insects play a significant role in the mechanical transmission of trichophytosis (dermatophytosis/ringworm), particularly in rural and agricultural settings [1]. Flies, including house flies (Musca domestica) and stable flies (Stomoxys calcitrans), have been shown to transport Trichophyton spores and mycelial elements on their bodies, spreading them from infected animals to healthy ones [2,3]. Acting as mechanical vectors, flies act as passive carriers, picking up fungus from contaminated environments or directly from lesions, often aggregating on the faces of animals and spreading infections like T. verrucosum. Studies have shown that flies exposed to dermatophytic cultures can transfer mycelial elements onto the coats of laboratory animals and other parasites (mites), but also bees might carry mites in their fur or cuticle.
In humans itches by skin inflammatory disease (psoriasis) goes together with psychological absence of well being (anxiety, depression, suicidal ideation, and substance misuse) [4] and also shame, disgust, insomnia, social anxiety, stigmatization, compulsive scratching and neuropsychiatric conditions [5]. Other skin conditions in humans involve the presence of a fungus that feeds on some component of the skin, as is the case with dandruff and Malassezia. Although the fungus may differ, and even if it is an inflammatory condition instead of a fungus, the topical treatments are very similar: various types of antifungal ointments or oral treatment (triazole, itraconazole) or, in extreme cases, corticosteroids [6]. These skin pathologies might not be independent because the immune memory responses directed against skin fungi can directly promote skin inflammation, leading to comorbidity between skin pathologies and exacerbating one another [7].
Some persons do also present the diagnosis as psychosomatic itches, that are felt as real but they might be produced by the delusional itches brain areas activation, similar to those for pain or phantom syndromes, without external stimuli [8,9]. The perception of itch is not restricted to the activation of brain sensory areas but it also requires the sensorial integration and cooperation of sensory, motor, and affective areas together with the precuneus cortex in the parietal lobe more evolved in human beings [8]. Scratching the skin due to psychosomatic itching can produce bleeding wounds, as much or even more than if the cause were a real fungus.
Aggressive behavior is having a meaning in evolution as a strategy for gaining access to resources such as territory, food, and potential mates, and it happens in a huge animal range from crustaceans to primates [10]. However, captivity in rodents under laboratory conditions might also have an effect on aggressive behavior when mice are having access to sufficient food, resources, territory and agreed-upon mates. In this article we suggest that aggression is not only a source of pain, destruction of specimens with unrepeatable DNA but also a source of bacterial infections, parasitic insects, and fungal growth.
Recent studies have proved this in rodents: that being socialized or not are relevant variables to activate aggressive behavior. Classically the ventro medial hypothalamus activation (VMH) alone was proved to override factors that are classically essential for aggression, such as the need for testosterone and the ability to smell. However in recent studies it was proved the aggressivity happened even in castrated or anosmic resident males. But even more relevant than the hypothalamic activation was the previous social boundaries of the male: they found that ventro medial hypothalamus (VMH) activation in socially bonded males inserted as intruders was not enough to induce aggression [10-12].
Fungi reproduce on skin but also on plants and some porous surfaces by capillary action (soil moisture) or condensation (air moisture), preferring damp and shady environments, just like mites. Home remedies for eliminating them are mainly based on I) temperature (boiling infected surfaces for several minutes), II) mechanical contact with the application of copper, which acts as a fungicide (like on hospital handrails and applied to plants copper sulfate or oxychloride), and III) chemical solution which causes asphyxiation or suffocation for the fungus through the application of oxygen and/or oxidation with sodium hypochlorite (bleach) (NaClO), ozone (O₃), white vinegar or acetic acid (C2H4O2) and potassium permanganate (KMnO4). Used alone or in combination (foam bleach, H₂O₂ + O₂), these products disinfect surfaces because they suffocate and oxidize fungi and aerobic bacteria. But on skin, nails, or mucous membranes due to their different pH, other chemical compounds are required. Antifungal ointments and creams are used, however dermic studies in this field are still developing because trying to boil the patient in hot water to completely kill the fungus seems to be not very ethical.
METHOD
In a colony of 24 house mice (Mus musculus, females N = 13 and males N = 11), only the males were fed wild insects and this became the main independent variable. These insects included bumblebees, carpenter bees, cockroaches, beetles, flies, etc., captured in the wild. The goal was to provide protein nutrients from the animal kingdom they might have hunted in the wild by themself and consequently satisfy their potential hunting instinct, thus reducing intragender territorial fights which might cause open, bleeding wounds on tails and fur. These territorial fights are quite common in male rodents even if their cages are large enough and quite spacious with plenty of nests for everyone. Especially among ages, the older mice bullied the younger ones. The intimidation of conspecifics by threats usually displays submissive responses like freezing, passive coping or escape [10]. One young mouse stood out for his successfully escaping behavior due to these attacks. Named “Houdini” because of this ability, on numerous occasions and after it was verified that the cage was secure, Houdini escaped when placed in a common cage with the unfriendly older mice. After freeing, he wandered for a few days, but the researcher never. Soon, whenever he was hungry or thirsty, he fell back into one of the rodent traps that were strategically placed along his routes to capture him without injuring or harming him. Despite the frustrated attempts to seal the cages properly, Houdini escaped at least three times: due to his small size and avoiding older male aggressivity, he always managed to find and squeeze through the only bars of the cage he discovered as possible ways out. Aggression from older males was a major trigger, if not the primary cause, of this escape behavior. Other younger males, such as Houdini, were also attacked, but they did not escape and instead they chose to fight, causing injuries and suffering from infection as explained below.
On the contrary the females did not try to escape, nor were provided by any wild insects in their diet to consume at all. Moreover, females barely display any aggressive behavior toward intragender specimens and no injuries have ever been observed on their tails or fur as a result of territorial fights. Females Mus musculus were noticeably cleaner and tidier with respect to their excrescences compared to the males of the same genus and species. None of the female rodents showed any signs of itching or injuries from excessive scratching, nor fungal lesions. However, females displayed other behaviors contrary to the proliferation of their own species or to health, including cannibalizing the offspring of other mothers and eating the corpses of adults.
After observing the possible toxic consequences of ingesting wild insects, we were able to closely explore the anatomy of a common bee and detected the presence of mites on its face and legs (Figure 1), thus allowing this parasite to be transmitted to our male rodents unintentionally but experimentally becoming an independent variable.

Figure 1. Pictures A, B and C are depicting the microscopic nature of a bee. With a red arrow is marked something that might be mites, around the eye and in a leg. In pictures D a fly with many things attached (might be pollen or dust but also mites or other micellial elements) and the same in picture E, an antenna of a beetle with white particles.
All mice were housed at 25ºC, under standard 12 h day/night cycle, in comfortable cages separated by sex and provided with beds of finely ground Spanish pine sawdust, fresh hay, straw and rodent litter. All the cages were cleaned, changed and dewormed using antiparasitic spray, sepiolite and diatomite litters at least once every week. The standard food provided was carefully chosen in a healthy variety following nutritional instructions for little rodent necessities: natural seeds without additives, lettuce, nuts (pumpkin seeds, black and white/black sunflower seeds, wheat, walnuts, peanuts in shell, peanut butter, oatmeals, etc.), pasta (raw macaroni), toasted bread crumbs or bread peaks (Spanish regañás and picos), fresh and dried proteins [dried tenebrio molitor coleoptera and dried larvae of hermetia illucens (black soldier fly)]. The males were occasionally fed with wild insects while they were 6 to 12 months of age. This distinction in feeding between males and females unintentionally and casually established the experimental design of this study, which is detailed in the Figure 2.
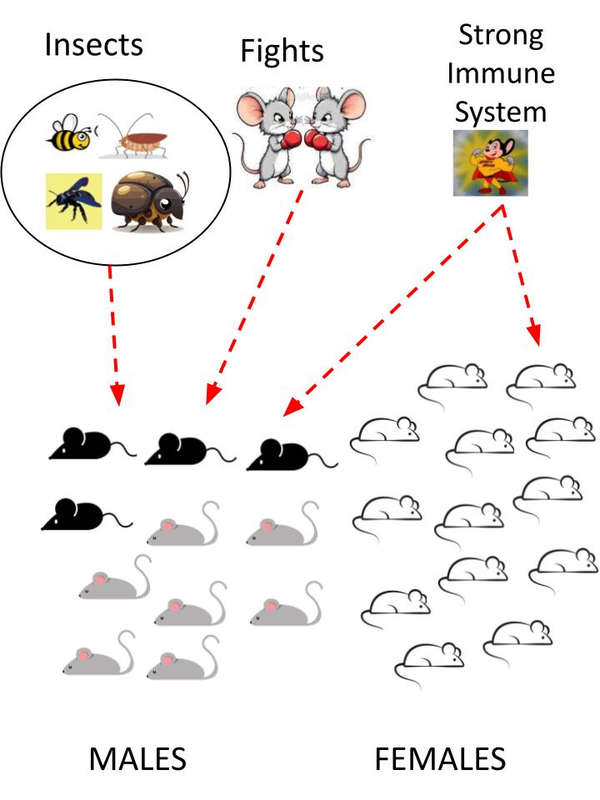
Figure 2. Drawing despite the experimental design. The independent variables were the insects to eat, provided by the researcher, and the fights between males which occurred repeatedly at the beginning of cohabitation between young and adult males. The four affected males for the fungus in their fur are dark in the picture. Another variable could have been the strong immune system, present in both groups, due to caregiving, brain stimulation, diet, or proper hydration that could have prevented the further spread of the fungus to other hosts [13, 14].
At 18 months of age, we detected in a male mouse a persistent wound on the lower back, at the base of the tail (Figure 3). We attributed it to fighting between males without knowing its exact diagnosis, but it could also have happened because of mites (parasites) which came from the insects. Mites can cause scabies, because they feed on tissue beneath the skin's surface and mainly flakes of dead skin. Therefore, the tail is a preferred location for mites in rodents, as its lack of fur facilitates access to dead skin cells to eat. No other male presented a similar wound in their tails, although later on three other males had their fur affected with the fungus (Figure 3).
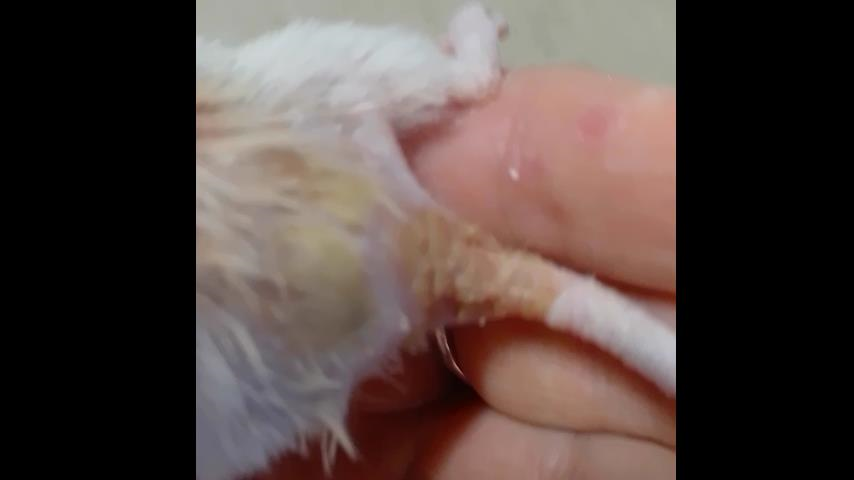
Figure 3. Picture shows the fungal lesion and the wound at the end of the tail and the beginning of the back that presented the first case of dermatophytosis. The different texture and moisture of the hairless skin was noticeable, as well as the formation of a scab that fell off repeatedly due to the fungus, which was completely eradicated after antifungal treatment.
The weight in grams of the male with the wound and the other three which resulted in infection were within the parameters for mice the same age (Table 1).
Table 1. In this list of weights is shown how the affected by the dermatophytosis were located in the male group, having similar body weights than their cage mates with no infections
|
MALE |
(gr) |
FEMALE |
(gr) |
|
1.- Persistent wound |
40,3 |
Big white |
57,2 |
|
2.- Infected siamese |
37,13 |
Big white II |
58,4 |
|
3.- Infected white |
38 |
White |
38 |
|
4.- Infected younger |
39 |
White II |
40 |
|
5.- White |
38 |
White III |
24,8 |
|
6.- Dark |
46,03 |
White IV |
26,03 |
|
7.- Cinnamon I |
37,06 |
Grey I |
30,06 |
|
8.- Cinnamon II |
53,11 |
Grey II |
33,11 |
|
9.- White II |
37 |
Grey III |
34,4 |
|
10.- Younger II |
38 |
Mother |
56,06 |
|
11.- Houdini |
37,2 |
Cinnamon |
40 |
|
Little |
19,04 |
||
|
One armed |
22 |
All experiments were approved by local authorities and were in accordance with the guidelines for animal experiments of the EU (Directive 2010/63/EU). We also strictly follow the general protocols established for mice by the American Institutional Animal Care & Use Committee (IACUC) concerning the Policy on Investigation Noncompliance and Animal Welfare and the Environmental Enrichment for Animals Standard Operating Procedures (SOP).
Treatment
Before the correct diagnoses and the antifungal treatment, several inefficient remedies were applied till we realized it was dermatophytosis. The mouse with the wound in his back was isolated for at least 6 weeks. At the beginning the injury was considered as a minor skin condition and subsequently we applied: a) aloe vera cream (one week), b) healing silver spray (one week) and c) dry healing powder (one week). Nevertheless, these treatments only provided temporary relief and the wound frequently recurred and crusted over. After that and due to its persistence we tried to clean and heal “a common wound” using: d) diluted chlorhexidine (0.1%) for one week, e) iodine (one week), f) chlorhexidine + iodine (one week). But these treatments only added humidity to the wound and provided very little improvement; instead, they seemed to create a moist environment that allowed the fungus to proliferate.
Then, because we observed a similar pathology in wild cats and without possible access to veterinarians specializing in behavioral laboratory rodents, we could arrive at the correct diagnosis: the dermatophytosis condition caused by a fungus that had taken hold in some of the existing skin wounds, as a result of fights between males or mite bites and which were transmitted by wild insects, both happening only in the male cage.
Once the fungal infection was diagnosed, we proceeded to search all pharmacies for antifungal medications with proven scientific efficacy. We search for several antifungals ointments and oral treatments: Triazole, Itroconazole [8,15], terbinafine [16,17]. Oral treatment of fungal infections in dermatology has also become a preferred modality for the management of these very common conditions [6]. But in Spain these chemical remedies are not available to buy in veterinarian medicine shops nor in pharmacies. Currently the only antifungal drugs available to obtain are Clotrimazol (10 mg/gr.) and Bifonazol (10 mg/gr.). Moreover, for this research, we attempted to purchase an itch-relieving product online, based on traditional Chinese medicine which contains phellodendron, radix arnebla root, sophora flavescens and aloe. Unfortunately, the product was held up in the Spanish Customs House and to obtain it, ten times its market price would have to be paid. Consequently we were not able to include this product into the research. The antifungal treatment started: Clotrimazol (10 mg/gr.) and Bifonazol (10 mg/gr.) every day, twice a day for consecutive 15 days.
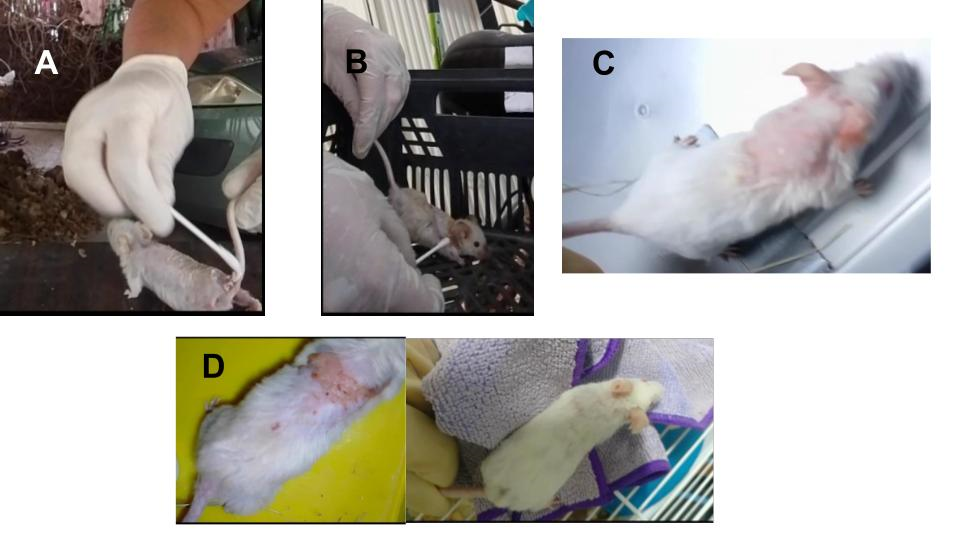
Figure 4. The picture is showing the skin disease in the four males affected. A) presents the male with the primary wound in his back, the same male as in fig. 3. B) is showing the Siamese one infected. C) shows the white male affected and D) depicts the younger male affected which was cured and his hair re-grown (picture in the right). The affected area of the younger's skin was smaller. All these animals were cured and the fungus eradicated. The rodent colony was safe and completely recovered.
RESULTS
Finally after the antifungal treatment for 15 days consecutively, the result was successful: the itching subsided, the body infected hair fell out, and the wound on their backs and low tail began to heal. Immediately, after applying the ointment with a cotton swab and wearing gloves, the mice´s appetite improved and it began eating peanut butter (a good nutritive option when it was difficult to gnaw and swallow).
When the first mice infected started to cure (Figure 2 and A in Figure 3), he was restored to the common cage. These other three were similarly treated and isolated together. This grouping of sick specimens may have facilitated the survival of those affected, since hair loss would cause a drop in their body temperature in these altricial animals and being in a group could have helped them to warm up among themselves.
The four males infected were all healed and the fungus was completely eradicated without any further infection appearing in the male nor female colony. The three older males (in Figure 4, pictures A, B and C), at the age of 20 months, almost 2 years old, were cured of the fungus but did not live long enough for their hair to grow back. The male of 6 months (in Table 1, infected younger, in Figure 4 picture D and in Figure 5 the A) was completely recovered and his hair fully grown back. In Figure 5 all the surviving males to the fungal infection are depicted while all retain their intact and healthy fur. The three remaining rodents in the C group are the adults from the previous generation where the initial wounded tail mouse belonged and they are all approximately two to two and a half years old. All these animals, except the A, were infected free, probably for their strong immune system. The animals mentioned in Table 1 (Cinnamon II and White I) are not visible in this picture because they were deceased of natural causes due to their age (almost 3 years old).
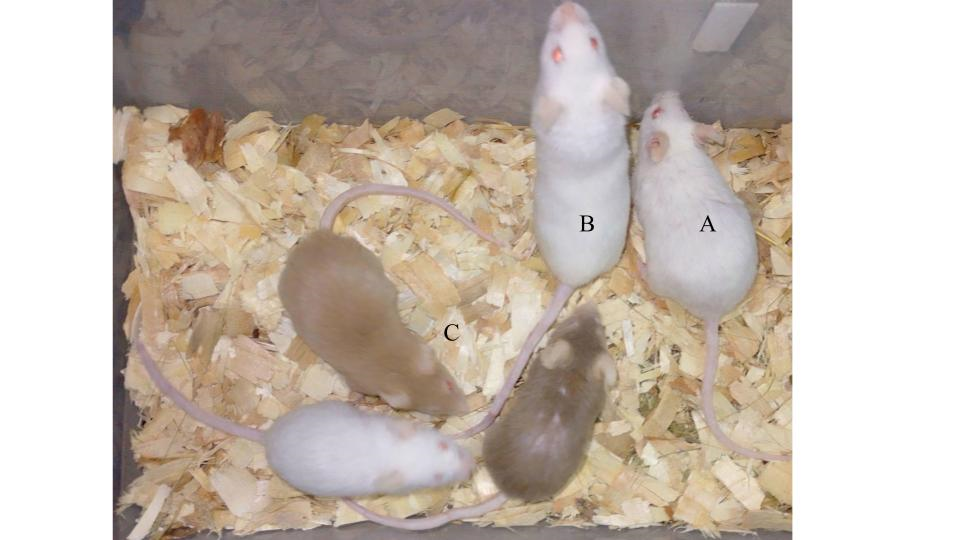
Figure 5. In this image, it is clear how the current males that survived the fungal infection retain their intact and healthy fur. Rodent A is the recovered juvenile, distinguishable by a cut on its ear caused during a fight. Rodent B is Houdini, distinguishable by its bent tail, fractured at one end. The three remaining rodents in group C are adults from the previous generation, approximately two to two and a half years old. All these animals, except the A, were infected free, probably for their strong immune system.
In the autopsy and post-mortem analysis of the infected males and cured but bold males, no internal organs affected by the fungus were found, having the autopsy material a predictable and within normal limits (WNL) anatomy, without any visible possible misfunction, unexpected change or alteration due to the the fungus. That autopsia exploration might be demonstrating that this particular fungus strain only affects the superficial skin. Moreover the antifungal treatments applied were specifically recommended for superficial fungus and they completely cured the animals and extinguished the fungus.
DISCUSSION
Standard experiments for skin treatments are generally performed on animal eroded skin that has been cutaneously inoculated with different strains of dermatophytes [18]. However in this experimental observation, the lesion appears spontaneously, giving the chance of collecting more natural explanatory variables. This experimental approach would provide information about what normally happens in nature without artificial human intervention. Although the variables were less controllable, the remedy proved was just as effective as in artificially created injuries.
Solutions and medications against fungi must be implemented, taking into account the current trade censorship and the foreseeable increase of these fungi in nature. Due to heavy rains and global warming these types of skin conditions could proliferate in the following years, causing losses in psychological well-being.
It would be worthwhile to explore the clinical applications of copper, a metal already used in plants, hospital railings, and sprays for skin conditions, due to its antifungal and antimycotic properties. Currently a diluted solution of copper sulfate in spray (0.1% 250 ml) is used in humans to protect the skin in cases of herpes, chickenpox, eczema or superficial irritations. The idea of copper for animals and pets would not be so far-fetched, since the aluminum spray is already marketed to treat, disinfect, and help heal pets' wounds. Copper and aluminium are both non-ferrous metals and their healthy properties are well proved. The same applies to Chinese medicine and its wise ancestral remedies.
While exploring remedies for eliminating fungi, we discovered that effervescent tablets, commonly sold for cleaning dentures, dental prostheses, orthodontic appliances, or dental dressings, are very useful and effective for eliminating fungi and sticky biofilms simply by immersing them in contaminated water from animal drinking troughs. The oxygen provided by these tablets, along with alkaline peroxides (potassium carotene), effervescent agents (citric acid), and surfactant enzymes (sodium lauryl sulfate and sodium carbonate), helps to break down and smother the bacterial or fungal biofilm and cleanse the surface of toxic agents.
The four males infected might have been involved in fights to have open wounds and /or lesion by mites transmitted via insects (attached to the fur or cuticle). The exact reason can not be elucidated and fights might not be erased (Figure 5 shows a dark male with a puncture wound in his fur, probably due to a fight injury). However with these pioneering observational experiments, the complete absence of fungal infection in the female animal group might be settling a line of further experimental reasoning for subsequent research using more natural variables.
CONCLUSION
This spontaneous domestic experiment highlights some of the skin conditions currently affecting the animal kingdom, specifically domestic mammals. Our experimental approach has allowed us to completely correct and cure the condition, thus setting a precedent for the treatment of similar ailments in the future. Since this condition is common among mammals, including humans, this minimally invasive and highly respectful experiment has provided us with relevant information for the cure and treatment of these uncomfortable and distressing conditions.
ACKNOWLEDGEMENTS
We would like to thank Prof. Dick Swaab from the Netherlands Institute for Neuroscience for his valuable and essential alliance. Moreover we would like to express our deepest gratitude to the reviewers for their praise and insightful comments that led to improvements. This article reports the result of research only. Mention of trade names or commercial over-the-counter pharmaceutical products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by any private or public institution.
CONFLICTS OF INTEREST
The Author declares that there are no conflicts of interest.
REFERENCES
- Ysquierdo CA, Olafson PU, Thomas DB. (2017). Fungi Isolated From House Flies (Diptera: Muscidae) on Penned Cattle in South Texas. J Med Entomol. 54(3):705-711.
- Pinetti P, Lostia A, Tarantino F. (1974). The role played by flies in the transmission of the human and animal dermatophytic infection. Mycopathologia et Mycologia Applicata. 54:131-134.
- Belluco S, Bertola M, Montarsi F, Di Martino G, Granato A, Stella R, et al. (2023). Insects and Public Health: An Overview. Insects. 14(3):240.
- Blackstone B, Patel R, Bewley A. (2022). Assessing and Improving Psychological Well-Being in Psoriasis: Considerations for the Clinician. Psoriasis (Auckl). 12:25-33.
- Courtney A, Su JC. (2024). The Psychology of Atopic Dermatitis. J Clin Med. 13:1602.
- Odds F, Ausma J, Van Gerven F, Woestenborghs F, Meerpoel L, Heeres J, et al. (2004). In vitro and in vivo activities of the novel azole antifungal agent r126638. Antimicrob Agents Chemother. 48(2):388-391.
- Hurabielle C, Link VM, Bouladoux N, Han SJ, Merrill ED, Lightfoot YL, et al. (2020). Immunity to commensal skin fungi promotes psoriasiform skin inflammation. Proc Natl Acad Sci U S A. 117(28):16465-16474.
- Misery L, Dutray S, Chastaing M, Schollhammer M, Consoli SG, Consoli SM. (2018). Psychogenic itch. Transl Psychiatry. 8(1):52.
- Pandolfi M, Scaia MF, Fernandez MP. (2021). Sexual Dimorphism in Aggression: Sex-Specific Fighting Strategies Across Species. Front Behav Neurosci. 15:659615.
- Yang T, Yang CF, Chizari MD, Maheswaranathan N, Burke KJ Jr, Borius M, et al. (2017). Social Control of Hypothalamus-Mediated Male Aggression. Neuron. 95(4):955-970.e4.
- Carvalho VMA, Nakahara TS, Papes F. (2017). The Strange Case of Aggression and the Brain. Neuron. 95(4):734-737.
- Garcia-Falgueras Alicia. (2024a). Cognitive Map is Earned with Experience, Effort and Physical Activity: Possible Disfigurement of Memory with Early GPS Use. Psychol Psychology Res Int J. 9(4):000438.
- Garcia-Falgueras Alicia. (2024b). Aging Might Increase Efficiency in Behavioral Performance for Risk Situations. Psychol Behav Sci Int J. 22:556087.
- Tanio T, Ichise K, Nakajima T, Okuda T. (1990). In vivo efficacy of SM-8668 (Sch 39304), a new oral triazole antifungal agent. Antimicrob Agents Chemother. 34(6):980-984.
- Sharma B, Kumar P, Joshi SC. (2011). Topical Treatment of Dermatophytic Lesion on Mice (Mus musculus) Model. Indian J Microbiol. 51(2):217-222.
- Nabeela S, Date A, Ibrahim AS, Uppuluri P. (2022). Antifungal activity of alexidine dihydrochloride in a novel diabetic mouse model of dermatophytosis. Front Cell Infect Microbiol. 12:958497.
- Van Cutsem J, Janssen PA. (1984). Experimental systemic dermatophytosis. J Invest Dermatol. 83(1):26-31.