Information Links
Related Conferences
Previous Issues Volume 8, Issue 1 - 2024
Review on Toxoplasmosis and Its Status in Ethiopia
Gemechu Negesa1, Isayas Asefa Kebede2,*
1School of Veterinary Medicine, Wolaita Sodo University, Wolaita Sodo, Ethiopia
2School of Veterinary Medicine, Ambo University, Guder, Ethiopia
*Corresponding author: Isayas Asefa Kebede, School of Veterinary Medicine, Ambo University, P.O. Box 19, Guder, Ethiopia, Tel: +251-(09)-11-89-49-73; E-mail: [email protected].
Received Date: February 02, 2024
Published Date: March 18, 2024
Citation: Negesa G, et al. (2024). Review on Toxoplasmosis and Its Status in Ethiopia. Mathews J Vet Sci. 8(1):39.
Copyrights: Negesa G, et al. © (2024).
ABSTRACT
Toxoplasmosis is a zoonotic infection of animals’ caused by the protozoan parasite Toxoplasma gondii, which is an obligate intracellular protozoan parasite and cats have a major influence on the epidemiology of the disease. Accordingly, T. gondii is a coccidian parasite with cats as the definitive host, and all warm-blooded animals can be used as intermediate hosts. T. gondii readily infects human beings, and infection is relatively common depending on environmental conditions, age, and immune status. After birth, humans are usually infected with T. gondii by ingestion of oocysts in food or water that have been contaminated with cat feces or by ingestion of tissue cysts. However, before birth, human toxoplasmosis can result from a congenital or acquired infection. The life cycle of T. gondii involves two phases. The sexual phase takes place in the definitive host (Felidae family) and the asexual phase in any warm-blooded animal, including humans and birds. The diagnosis of toxoplasmosis was performed by the isolation of the parasite from patients and by serological tests such as indirect fluorescent antibody (IFA) and enzyme-linked immunosorbent assay (ELISA). The major importance of toxoplasmosis in farm animals is its zoonotic potential. Since cats are key to the transmission of T. gondii as they are the only definitive hosts that shed the oocysts, prevention and control methods should target the Felidae family. Generally, domestic and barn cats should be restricted in farm environments from nesting and defecating in hay, straw mows, grain stores, or other loose piles of livestock feed.
Keywords: Toxoplasma gondii, Toxoplasmosis, Zoonotic importance.
LIST OF ABBREVIATION
AC: Acetone; AIDS: Acquired Immune Deficiency Syndrome; DNA: Deoxyribonucleic Acid; ELISA: Enzyme-Linked Immunosorbent Assay; IFA: Indirect Fluorescent Antibody; MAT: Modified Agglutination Test; PCR: Polymerase Chain Reaction; OT: Ocular Toxoplasmosis.
INTRODUCTION
Toxoplasmosis is among the most important protozoan parasites of worldwide zoonotic disease caused by an obligate intracellular parasite Toxoplasma gondii that can infect almost all warm-blooded animals, including humans and is considered one of the most successful eukaryotic pathogens as reviewed by [1]. The parasite causes abortion in animals and humans [2]. During different periods of the parasite life cycle, individual parasites convert into various cellular stages. These stages include the sporozoites, bradyzoites, and tachyzoites. And also consists of two stages: a sexual stage that appears in cat (the definitive host) and an asexual stage that occur in all warm-blooded animals (intermediate host), including humans .it can affect both humans and animals, including camels, cattle, sheep, goats, poultry, and wild animals [3]. The preferred hosts are felids, although a recent study [4] developmental success in mice treated with specific enzyme inhibition and diet modification [5].
The parasite is a cosmopolitan protozoon with no host specificity in the asexual stage, whereas, in the sexual stage, it is specific to felids. Cats are the definitive host for Toxoplasmosis while cattle, shoats, poultry, pigs, and camel are reservoir hosts and facilitate the transmission of this disease to humans [6,7]. Toxoplasma infection in livestock leads to significant economic losses because of reproductive failure, i.e., abortion, fetal resorption, and barrenness. Humans are not the primary host, so the zoonotic parasite will act differently in humans and it causes health problems that range from those that are minor to the very serious [8]. Of infected humans, very few have symptoms because a healthy individual immune system usually keeps the parasite from causing illness. However, pregnant women who have compromised immune systems should be cautious, as the disease is abortion fanciest [9].
T. gondii infection is substantially more likely to occur in pregnant and immunocompromised people than in immunocompetent people [7]. Intermediate hosts such as rodents become infected after ingesting soil, water, or plant material contaminated with oocysts [10]. Despite its complex yet simple transmission patterns, epidemiological research on Toxoplasmosis is lacking in sub-Saharan Africa [11]. Given the geographical distribution of Toxoplasmosis and the 30% seropositivity rate worldwide [12].
Oocysts, the environmentally robust stage of T. gondii, play an important role in the epidemiology of this zoonotic parasite. Attributes of oocysts' biology may further explain the global distribution of T. gondii and how it has evolved to be one of the most prevalent infectious agents of animals and humans. While oocysts are exclusively deposited on land due to definitive hosts being solely terrestrial animals, the prevalent nature of infections observed in aquatic animals demonstrates a significant role in waterborne transmission. High prevalence of T. gondii exposure in marine species (up to 100% in some populations [13] further suggests that oocyst transport to, and accumulation in nearshore or open ocean habitats is possible and epidemiologically significant [14].
The diagnosis of toxoplasmosis can be established by serologic tests, amplification of specific nucleic acid sequences by PCR, histologic demonstration of the parasite and/or its antigens by immune peroxidase stain, or isolation of the organism [15]. Globally, caprine animals are susceptible to toxoplasmosis as a result of social contact [16]. It is known to result in reproductive miscarriage [17]. Due to abortion or the production of weak offspring in food animals, this may result in significant economic losses [18]. Due to inbreeding, the infection incidence varies greatly from herd to herd; an average rate of 30% has been observed. A significant factor in disease transmission from domesticated animals to people is the high prevalence of toxoplasmosis in cattle, sheep, and goats [19]. The socioeconomic impact of this disease on people suffering from it and the cost of care for sick children, especially those with mental retardation and blindness, are enormous [20]. In Ethiopia, the incidence of toxoplasmosis is increasing the disease is neglected and infestation in humans of children-bearing age is unknown. Therefore, the objectives of this paper are to review the available literature on the etiology, epidemiology, diagnosis, and control of Toxoplasmosis in animals and humans, and its zoonotic importance and status in Ethiopia.
TOXOPLASMOSIS
History of Toxoplasmosis
Nicolle and Manceaux isolated Toxoplasma gondii from the African rodent Chenodactylus gundi in 1908 and used that isolation to name the parasite. The condition was identified as Toxoplasma gondii in 1909 after being recognized as leishmania [15].
About the organism's crescent-shaped morphology, the name of the genus is derived from the Greek word toxon, which means crescent. A serological dye test created in 1948 by Sabin and Feldman provided the foundation for particular diagnostic assays [21].
Etiology
Toxoplasma gondii, the causative agent, is a systemic coccidian, a universal parasite, a sporozoan, and a member of the suborder Eimeriina. It is a particular parasite of the final host (members of the felidae family), although it has a variety of intermediate hosts [21].
Epidemiology
Geographical distribution
Toxoplasma gondii is a widespread microbe that can be found throughout the world, although tropical regions have greater rates of infection because humidity and warmer temperatures promote the maturation of oocysts in the soil. Around the world, there are significant regional differences in the seroprevalence of T. gondii. The parasite is notably more common in Western European, South American, and African nations [22].
Cats play an important role in maintaining T. gondii in nature because they are the definitive hosts for this protozoan parasite and rarely develop clinical disease as a result of infection. Toxoplasmosis has been discovered to be absent from islands that are geographically remote and don't contain cats [23]. This result can be explained by the group's hunting techniques and diet, which include wild birds, rodents, and, occasionally, placentas infected with Toxoplasma and stillborn fetuses [24]. The global prevalence of T. gondii infection in humans and animals varies depending on the country's geographic location. The causes of these variations are not yet known [25]. Infections with T. gondii in cats are usually asymptomatic, and vertical transmissions occur only infrequently. However, latent infections with T. gondii are common in domestic cats and wild felines throughout the world [26].
Predisposing Factors
Environmental variables may affect how naturally T. gondii infections spread. Warm, low-lying areas are more susceptible to infection than cold, mountainous areas, while humid regions are more susceptible to infection than dry ones. This is most likely connected to elements that support sporulation and oocyst survival [27].
Human Health Risk Factors
Humans are easily infected by T. gondii, and while clinical sickness is not common (roughly 30% of the population depending on age and environment), infection is rather common (about 30% of the population depending on age and environment). In pregnant women, the parasite can pose a serious threat to the fetus if the mother contracts it for the first time while carrying it, as well as immunosuppressed people, such as those undergoing tissue transplants, those with AIDS, certain types of cancer patients, and those receiving certain cancer therapies, are at a higher risk of developing clinical illness. If untreated, these people run the risk of getting an infection that might kill them quickly [28].
Transmission of Toxoplasmosis
Fecal-oral transmission is the most common pathway through which T. gondii is spread from the primary host to secondary hosts. Only felids can transmit oocysts (although oocysts can also be produced in the lab) [29].
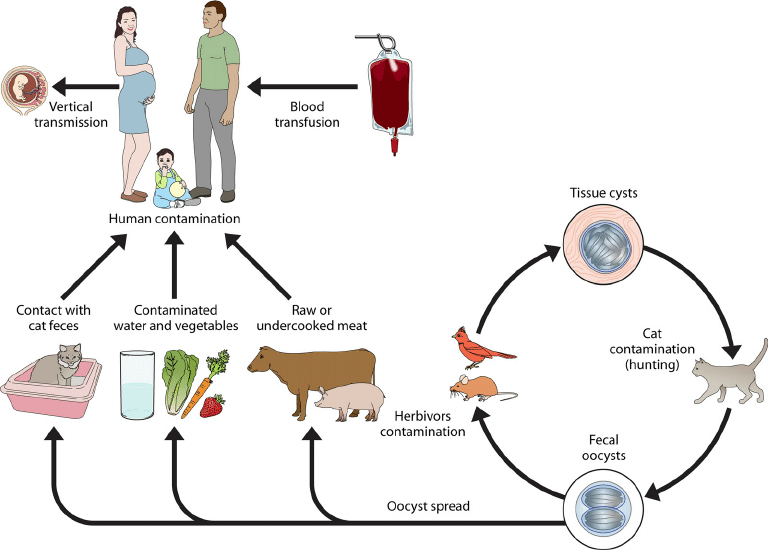
Toxoplasmosis is mostly spread by cats. Epidemiologic statistics show that the majority of cats get an infection in the wild shortly after weaning, either by sharing food supplied by the dam or by consuming raw pet food. Consequently, wild cats are more likely than domestic cats to get infected with T. gondii. Humans are one of the intermediary host species when Toxoplasma gondii is transmitted vertically. Transplacental vertical transmission is facilitated by tachyzoites, usually after the primary and during the acute phases of infection [28]. T. gondii can also be transmitted via blood or leucocytes from infected donors [30]. T. gondii tissue cysts have been found in the edible tissues of most food animals [20].
Besides its vertical transmission through T. gondii tachyzoites passed from the placenta to the fetus, a horizontal transmission can occur by drinking water, meat, milk, fruits, or vegetables contaminated with bradyzoites or oocytes released from cats [31].
Toxoplasma gondii can also spread through semen in cases where the secondary host is a man and through congenital transmission in cases where the secondary host is a pregnant woman. The connection between these two routes is possible. T. gondii has been linked to sex-transmitted infections in rats [32], dogs [33], sheep [34], and even humans [35].
Transmission in Animals
Toxoplasma gondii is a tissue cyst-forming protozoan microorganism that is vertically transmitted to puppies if a female is infected during pregnancy. Infected dogs usually remain clinically normal, but the puppies are severely affected. Toxoplasmosis is known to cause fetal death and is a rare cause of abortion in dogs. Premature birth, stillbirth, and the birth of weak puppies are common in toxoplasmosis [21]. In the environment, oocysts are distributed through wind, rain, surface water, or harvested feeds. They may also be spread via earthworms, coprophagous invertebrates, or manure. Hay, straw, and grain that are contaminated with cat feces have been identified as sources of infection for livestock [35].
The prevalence of infection is highest in young cats hunting for the first time. For the infection of cats, the period of excretion of oocysts is short, approximately 2 weeks. The number of cats excreting oocytes in their feces at any point in time is likely to be quite small, but the contamination of the environment over time is significant. Domestic and barn cats in farm environments tend to nest and defecate in hay, straw mows, grain stores, or other loose piles of commodities that the livestock feeds with manure and bedding from buildings that contain cat feces. Oocysts can survive in the winter in cold climates but are less viable in arid environments [21].
Infection of farm animals occurs as the result of feed or water contaminated with cat feces, which contain infective oocysts, via the ingestion of contaminated stored feeds, contaminated pastures, or contaminated water supplies [36].
Transmission to human
The major modes of transmission of Toxoplasmosis to humans include the consumption of undercooked meat containing Toxoplasma cysts, Fecal-oral transfer of Toxoplasma cysts from cat feces directly or in contaminated food, water, or soil Vertical transmission from mother to fetus if primary infection occurs during pregnancy [37].
In adults, the incubation period for T. gondii infection ranges from 10 to 23 days after the ingestion of undercooked meat and from 5 to 20 days after the ingestion of oocysts from cat feces [38].
Sources of Infection
Tissue cysts of T. gondii contained in the meat of livestock are an important source of infection for humans. In meat-producing animals, tissue cysts of T. gondii are most frequently observed in the tissues of infected pigs, sheep, and goats, and less frequently in infected poultry, rabbits, dogs, and horses. Tissue cysts are found only rarely in beef or buffalo meat, although antibodies in up to 92% of cattle and up to 20% of buffaloes are evidence of past exposure to the parasite [39].
Cat feces are the sole source of infection for sheep, cattle, and horses. Cats become infected as a result of ingesting tissues of intermediate hosts infected with the parasite; commonly, these are rodents and small birds, but all animals can be IH for T. gondii. Rodents have served as reservoirs of infection for a long time. Feeds of animals contaminated with cat feces due to nests of cats, Actions of earthworms, and other soil inhabitants bring superficially buried feral cat feces to surfaces, which contaminates pastures. Ingestion of meat, dead rodents, cannibalized piglets, and blood while tail- or ear-biting by pigs [21].
Life Cycle of T. gondii
Toxoplasma gondii is a parasite with a distinct life cycle that involves both asexual and sexual reproduction. The sexual cycle of the only species in the genus Toxoplasma, T. gondii, is exclusive to felines [40]. T. gondii can infect a wide variety of warm-blooded animals due to its asexual life cycle, including various species of mammals and birds. T. gondii replicates asexually in the intermediate host and develops into permanent tissue cysts that include the bradyzoite stage [41]. Felids ingest T. gondii by eating encysted bradyzoites on infected intermediate hosts. Bradyzoites are released from cysts and enter the small intestine's epithelial cells under the action of digesting enzymes and acid. Although the parasite may spread throughout the body of the final host and induce clinical signs, this is not typical [42].
Feline felids that have consumed one of the three infectious forms—tachyzoites, bradyzoites, or oocysts—will shed unpolluted oocysts. The longevity and infectiousness of unpolluted oocysts, which are incapable of infecting hosts, are influenced by climatic factors, specifically temperature extremes and a drop in relative humidity [43]. The sporulated oocysts become infective and environmentally resistant for periods depending on local climatic conditions, up to 12–18 months, and remain viable when stored for at least 54 months at 4 °C in water [44].
Tachyzoites and bradyzoites are two separate kinds of asexually or clonally reproducing forms that grow in warm-blooded intermediate hosts. Tachyzoites are a type of parasite that proliferates rapidly and is frequently linked to acute infection, reactivation, and vertical transmission. The bradyzoites, on the other hand, are a dormant, encysted form that is slow-growing and metabolically active. Bradyzoites are frequently linked to persistent toxoplasmosis, immunological evasion, and resistance to presently existing anti-toxoplasmosis pharmacotherapies [45].
In immunoprotein people, tissue cysts develop as a result of these protozoans spreading throughout the body via the blood or lymph, which results in a chronic stage of the asexual cycle. Cysts are primarily found in the hosts' brains, skeletal muscles, and cardiac muscles. The cysts are broken down by proteolytic enzymes after they are consumed by intermediary hosts like dogs, pigs, rats, or humans. Then, bradyzoites are discharged and transformed back into tachyzoites as they infect the epithelium of the intestinal lumen and quickly spread within leukocytes, infecting their hosts' bodies [27].
Evasion is mediated by cell type tropism and sequestration to immune-privileged sites such as the brain and skeletal muscle, as well as the process of encystation. Both tachyzoites and bradyzoites follow a cell division scheme known as endodyogeny, consisting of a single round of DNA replication by semi-closed nuclear mitosis. The internal assembly of two daughter cells occurs concomitantly with nuclear mitosis inside the mother cell. Tachyzoite cell division is rapid, generating two new cells per mother cell every 6–8 h. Bradyzoites instead divide slower but can assemble and sustain between 1000 and 2000 bradyzoites per cyst [46].
Figure 1. Toxoplasma gondii life cycle; Source: (Esch and Petersen, 2013).
Pathogenesis of Toxoplasmosis
Infection with Toxoplasma gondii is acquired by carnivory, ingestion of feces containing oocytes, or congenitally [47]. In an unexposed cat after ingestion of uncooked meat containing tissue cysts. T. gondii initiates enteroepithelial replication. Bradyzoites are released from tissue cysts by digestion in the stomach and small intestine and invade the intestinal epithelium by undergoing sexual replication, culminating in the release of oocytes in the feces. Oocysts are first seen in the feces three days after infection and are released for up to 20 days after infection. After exposure to air for 24 hours, oocysts sporulate, become infective, and may persist in the environment for up to one year. Cats generally develop immunity to T. gondii after the initial infection and therefore only shed oocysts once in their lifetime [48].
In animals
The extent of T. gondii infection in cats depends on the availability of infected birds and small mammals. Toxoplasmosis has been confirmed in some 200 species of vertebrates, including primates, ruminants, swine, equine, carnivores, rodents, marsupials, insectivores, and numerous avian species. T. gondii causes abortion and neonatal mortality in sheep worldwide. T. gondii has been recognized as one of the main causes of infective ovine abortion in New Zealand, Australia, the United Kingdom, Norway, and the United States [27].
In human
In humans, clinical disease is common in immunocompromised individuals or results from an acute infection of the expectant mother. The severity of congenital infections depends on the stage of pregnancy when the acute infection occurs, spontaneous abortions, or neurological disorders [49].
Clinical Manifestations
The clinical manifestations of toxoplasmosis vary depending on parasite characteristics such as the virulence of the strain and inoculum size, as well as host factors such as genetic background and immune status [7]. There are at least three genetic types of T. gondii: types I, II, and III. They differ in virulence and epidemiological patterns of occurrence [50].
In animals
The main clinical symptoms are abortion and newborn mortality, but adult goats can also get toxoplasmosis with symptoms like liver, kidney, and brain damage [27]. The People's Republic of China, the West Indies, Norway, and the Czech Republic all reported having suspected cases of clinical caprine toxoplasmosis. According to Masala et al. [51], T. gondii was found in 6.4% of 362 fetuses and 3.0% of 211 aborted tissues in Italy.
In human
The most prevalent illnesses were tuberculosis (8.7%), meningeal cryptococcosis (21%), and brain toxoplasmosis (32.2%). More research conducted in Mexico revealed that 47% of AIDS patients' neurological symptoms in the state of Yucatan are caused by toxoplasmosis [52]. The reactivation of a chronic or latent infection brought on by the loss of cellular immune surveillance accounts for more than 95% of toxoplasmic encephalopathy in AIDS patients. According to a study conducted in Thailand [53], among HIV-positive and T. gondii antibody-positive groups, 43.2% displayed symptoms and signs of acute toxoplasmosis affecting the eye and/or the central nervous system.
Ocular toxoplasmosis used to be attributed to congenital infection, but recently, researchers compared prenatal and postnatal toxoplasmosis. They concluded that at least two-thirds of ocular toxoplasmosis is caused by postnatal infection, which has major public health implications [54].
Diagnostic Technique
Human toxoplasmosis can be diagnosed by serological, molecular, or histological methods. Sometimes, these techniques are combined to improve the effectiveness of diagnosis [55]. The diagnosis of T. gondii infection or toxoplasmosis may be made using serologic tests, polymerase chain reaction (PCR) amplification of specific nucleic acid sequences, immunoperoxidase staining of tissue, or isolation of the organism [56]. The main test that is advised for the diagnosis of T. gondii in both people and various animals is the MAT [27].
Indirect fluorescent antibody test
Patient serum is tested for action against dead organisms as part of the IFAT process. The patient's antibody (IgG) recognizes fixed tachyzoites, and then a fluorescent-labeled anti-human antibody (IgG) is added to enable fluorescence microscopy identification of the antibody. No fluorescence is seen in the absence of an antibody against the T. gondii tachyzoite [57].
Modified Agglutination Test
The development of a simple direct agglutination test has greatly aided in the serological diagnosis of toxoplasmosis in humans and other animals. In this test, no special equipment or conjugates are needed. The MAT has been extensively used for the diagnosis of toxoplasmosis in animals. The sensitivity and specificity of MAT have been validated by comparing serologic data and the isolation of the parasite from naturally and experimentally infected pigs [23].
In the MAT, sera are treated with 2-mercaptoethanol to remove non-specific IgM or IgM-like substances. This test detects only IgG antibodies; therefore, it may give false-negative results during the early stages of acute infection. The MAT is commercially available (Toxo-Screen DA, bioMerieux, Charbonnieres-Beins, France). The Toxo-Screen DA is the same test as the MAT. The results obtained with MAT differ depending on the preservative used to prepare the antigen. Jones (2003) reported that using acetone (AC test) in place of formalin (HS test) can detect IgG present during acute infection. The AC test has been very us
Enzyme-linked immunosorbent assay
The ELISA system usually includes the solid phase antigen or antibody, enzyme-labeled antigen or antibody, and the substrate of the enzyme reaction, which can be modified to test both antibodies and antigens. ELISA can be automated so that a large number of samples can be simultaneously tested. There have been different types of ELISA developed to detect T. gondii antibodies or antigens, such as indirect ELISA, and sandwich ELISA. In the indirect ELISA, the antigen is coated onto the solid phase and the sample containing antibodies is added, the antigen-antibody reaction is enhanced by the addition of a secondary enzyme-linked antibody, and the reaction can be evaluated by quantification of the color that develops [1].
Detection of T. gondi DNA
Toxoplasma gondi DNA can be detected by PCR. There are many different markers, but the 35-copy B1 gene and the 300-copy 529-bp element are most frequently used. The disadvantage of using a single-copy gene target is that the sensitivity is compromised compared to highly repetitive sequences. However, this can be compensated for to some extent by nested PCR. The 529-bp repeat marker is 10–100 times more sensitive than the B1 marker. The advantage of the recently developed real-time PCR assay is that it also quantifies the parasite DNA. Overall, PCR is very sensitive (it can detect DNA from one tachyzoite), specific, and can provide a rapid diagnosis. It is most suitable for clinical specimens [39].
Treatments of Toxoplasmosis
To treat Toxoplasmosis, dihydropteroate synthetase, and dihydrofolate reductase inhibitors were suggested. One of the efficient dihydrofolate reductase inhibitors used to treat this condition is pyrimethamine. Additionally, medications including sulfamethoxazole and trimethoprim were utilized to treat this illness [58]. Depending on where you obtain medical care, the course of treatment for toxoplasmosis during pregnancy may change. Spiramycin may be prescribed if the infection occurs before the 16th week of pregnancy [59]. Congenital toxoplasmosis can cause neurological issues in newborns, although using this medication may lessen that risk [60].
Prevention and Control
Although preventive interventions can considerably lower the chance of contracting T. gondii infection, they cannot always ward off infection [61]. Accordingly, various agencies, including the World Health Organization, have recommended techniques for the surveillance and management of toxoplasmosis in humans due to the significant influence that T. gondii has on the quality of human life [62].
To get rid of the cysts, the meat needs to be frozen at -20°C for at least 24 hours or boiled at 60°C for at least 15 minutes. Avoid coming into contact with cat waste at all costs. To prevent illness, one should always wash their hands after coming into contact with raw meat, cats, or potentially cat-contaminated dirt. Consumption of raw eggs and nonpasteurized milk, particularly goat’s milk, should be avoided. Fruits and vegetables should be adequately washed before ingestion. Daily cleaning of the cat litter box is also essential to prevent sporulation. For females, only nonpregnant women should perform this duty. Blood transfusions and organ transplants from seropositive individuals should be avoided if the recipient is seronegative [63].
Wearing gloves when cleaning a litter box is essential because it is possible to come into contact with sand or soil that has been contaminated by cat feces and may contain toxoplasmosis. After the litter box is cleaned, the person who cleaned the important litter box must wash their hands with soap and warm water. The frequency or severity of planned serological screening of pregnant women can be greatly reduced or lessened in the event of fetal infection, helping to identify any women who are at risk of contracting the illness or any women who are already infected. Cats should be kept indoors to avoid contracting toxoplasmosis or shedding oocysts into the environment, which will help prevent toxoplasmosis in cats [64].
ZOONOTIC IMPORTANCE
Toxoplasmosis is considered the most prevalent parasitic zoonotic disease worldwide. Since it affects up to one-third of the world’s population, Toxoplasma gondii infection is important both in veterinary and human medicine. It can produce a wide range of clinical manifestations or, in most cases, progress asymptomatically [65]. Humans acquire the infection through the oral route through the consumption of undercooked meat contaminated with cysts, food products, or water contaminated with oocysts [66,39].
Although seroprevalence research suggests that farm animals are infected at reasonably high rates, the infection is subclinical, and T. gondii is mostly insignificant as a cause of clinical illness in farm animals—except those linked to miscarriage and neonatal sickness in sheep. Toxoplasmosis in farm animals is significant because of its potential to spread to humans [67].
Toxoplasma gondii, an obligate intracellular protozoan parasite, is the cause of toxoplasmosis, a water-borne zoonotic disease [68]. By consuming tainted food or water that contains oocysts from cat feces that have been infected, humans can contract this protozoon [66]. Fever, lymphadenopathy, and convulsions are all brought on by Toxoplasma gondii. Fetal mortality and early birth may result from infected pregnant mothers spreading the virus to the fetus through the placenta. In cats, toxoplasmosis is largely asymptomatic. It primarily results in stillbirth in sheep, goats, and pigs [69].
Toxoplasmosis is one of the most widespread parasitic zoonoses. According to Santoro et al. [70], bradyzoites from infected tissues or sporulated oocysts in contaminated food and water can cause foodborne infections in humans. The consumption of raw or undercooked meat from live animals that contains tissue cysts is thought to be a significant source of infection for humans, according to Gazzonis et al. [71]. Still, the increased consumption of game meat, including wild boar and red deer, is expected to be measured as an emerging risk factor for T. gondii infection in humans [72]. Furthermore, monitoring T. gondii infection has been indicated as a high priority, not only in domestic animals but also in large game [73].
TOXOPLASMOSIS STATUS IN ETHIOPIA
Toxoplasmosis is a disease that is neglected in Ethiopia, and it is unknown whether it affects pregnant women. According to a few studies conducted in Ethiopia, the prevalence of toxoplasmosis in the general population ranged from 20.2% to 97.7% [74]. With approximately 82 million people and a high AIDS prevalence, Ethiopia is the second-most populated country in the Horn of Africa. Notably, Adem and Ame [75] found that T. gondii antibodies were present in HIV patients with a seroprevalence of 93.3%. The current study showed a strong association between the average monthly income of pregnant women and seropositivity for T. gondii. This finding agrees with the previous study conducted in Zambia [76].
In contrast, a study conducted in central Ethiopia showed the absence of a significant association between average monthly income and seropositivity for T. gondii. More seropositivity was found among pregnant women with low socio-economic status than high socioeconomic. This significant association could be due to pregnant women of low socio-economic status being more prone to live and work in highly dense areas with poor sanitary conditions and a lack of good education on good hygienic practices [77]. Studies conducted in different geographical locations of Ethiopia from 2007 to 2020 indicated a high seroprevalence of T. gondii infection in humans, sheep, goats, and pigs. Hydrocephalus, retinochoroiditis, convulsion, and intracerebral calcifications in fetuses, and lymphadenitis and encephalitis in immunocompromised groups are the major findings of toxoplasmosis [15].
Table 1. Status of Toxoplasmosis in human
|
Study Area |
Population surveyed |
No. |
Serological test |
% |
References |
|
Adama, Hospital |
Peopleaged15 days-65years |
65 |
MAT |
60.0 |
[78] |
|
Butajira, Addis Ababa |
Patients aged 15–49 years |
456 |
ELISA-VI |
95.1 |
[74] |
|
Mettu Karl Hospital, Ethiopia |
HIV/AIDS Patients |
120 |
Different model checking and model diagnostic test |
60 |
[79] |
|
Addis Ababa area |
Hospitalized patients |
330 |
ELISA-BC |
93. 3 |
[80] |
|
Jimma town, Southwestern Ethiopia |
Pregnant women |
201 |
ELISA |
83.6 |
[81] |
Table 2. Status of Toxoplasmosis in animal
|
Host |
Year |
Population surveyed |
No. |
Method |
% |
Reference |
|
Cat |
2011 |
Central Ethiopia |
36 |
MAT |
91.67 |
[84] |
|
Sheep |
1999 |
Nazareth |
116 |
MDAT |
52.6 |
[85] |
|
Goat |
2010-2011 |
Central Ethiopia |
927 |
ELISA |
19.7 |
[86] |
|
Sheep |
2010-2011 |
Central Ethiopia |
1130 |
ELISA |
31.59 |
[82] |
|
Chicken |
2012–2013 |
Central Ethiopia |
601 |
MAT |
30,45 |
[87] |
Toxoplasmosis is a zoonotic parasitic disease caused by the protozoa T. gondii. It infects a wide range of animals including mammals and birds. While seroprevalence studies indicate relatively high rates of infection in animals subclinical T. gondii has virtually no importance as a cause of clinical disease in farm animals except that associated with abortion and neonatal disease in sheep. The major importance of Toxoplasmosis in farm animals is its zoonotic potential as it is the cause of hydrocephalus, retinochoroiditis, convulsions, and intracerebral calcifications in fetuses, lymphadenitis, and encephalitis in immunocompromised patients. For this reason, the epidemiology of Toxoplasmosis in pregnant women immunocompromised patients, and farm animals is important. Thus, health education for women of childbearing age should include information about preventing T. gondii transmission from food, soil, litter boxes, and contact with infected cats; Awareness creation should be given to pregnant women about food hygiene and avoiding exposure to cat feces; Domestic and barn cats should be restricted in farm environments not nesting and defecating in the hay, straw mows, grain stores, or other loose piles of commodity livestock feeds, and The government and meat industry should continue efforts to reduce the presence of T. gondii in meat.
DECLARATION
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
Not applicable.
CONSENT FOR PUBLICATION
Not applicable.
AVAILABILITY OF DATA AND MATERIALS
All the datasets generated or analyzed during this study are included in this manuscript.
COMPETING INTERESTS
All authors have nothing to disclose in this work.
FUNDING
The current study was not funded by any institution.
AUTHORS' CONTRIBUTIONS
All authors contributed to data collection, study design, data interpretation, reference search, manuscript writing and editing, and all authors have approved the submission of the final manuscript.
REFERENCES
- Liu Q, Wang ZD, Huang SY, Zhu XQ. (2015). Diagnosis of toxoplasmosis and typing of Toxoplasma gondii. Parasites & vectors. 8:1-4.
- Mohamed K. (2020). Toxoplasmosis in humans and animals in Saudi Arabia: A systematic review. J Infect Dev Ctries. 14(8):800-811.
- Tonouhewa AB, Akpo Y, Sessou P, Adoligbe C, Yessinou E, Hounmanou YG, et al. (2017). Toxoplasma gondii infection in meat animals from Africa: Systematic review and meta-analysis of sero-epidemiological studies. Vet World. 10(2):194-208.
- Attias M, Teixeira DE, Benchimol M, Vommaro RC, Crepaldi PH, De Souza W. (2020). The life-cycle of Toxoplasma gondii reviewed using animations. Parasit Vectors. 13(1):588.
- Galeh TM, Sarvi S, Hosseini SA, Daryani A. (2022). Genetic diversity of Toxoplasma gondii isolates from rodents in the world: A systematic review. Transbound Emerg Dis. 69(3):943-957.
- Dubey JP, Su C. (2009). Population biology of Toxoplasma gondii: what's out and where did they come from. Mem Inst Oswaldo Cruz. 104(2):190-195.
- Mose JM, Kagira JM, Kamau DM, Maina NW, Ngotho M, Karanja SM. (2020). A Review on the Present Advances on Studies of Toxoplasmosis in Eastern Africa. Biomed Res Int. 2020:7135268.
- Rahman T, Rahman A, Chakraborty S. (2018). Infection of Toxoplasma gondii in humans and livestock animals: an emerging silent threat for Bangladesh. Open Journal of Medical Microbiology. 8(4):109-117.
- Fisk EA, Cassirer EF, Huggler KS, Pessier AP, White LA, Ramsay JD, et al. (2023). Abortion and Neonatal Mortality Due To Toxoplasma Gondii in Bighorn Sheep (Ovis Canadensis). J Wildl Dis. 59(1):37-48.
- González-Parra G, Arenas AJ, Chen-Charpentier B, Sultana S. (2023). Mathematical modeling of toxoplasmosis with multiple hosts, vertical transmission and cat vaccination. Computational and Applied Mathematics. 42(2):88.
- Hotez PJ, Kamath A. (2009). Neglected tropical diseases in sub-saharan Africa: review of their prevalence, distribution, and disease burden. PLoS Negl Trop Dis. 3(8):e412.
- Marín-García PJ, Planas N, Llobat L. (2022). Toxoplasma gondii in foods: prevalence, control, and safety. Foods. 11(16):2542.
- Shapiro K, Bahia-Oliveira L, Dixon B, Dumètre A, de Wit LA, VanWormer E, et al. (2019). Environmental transmission of Toxoplasma gondii: Oocysts in water, soil and food. Food Waterborne Parasitol. 15:e00049.
- Miller M, Shapiro K, Murray MJ, Haulena M, Raverty S. (2018). Protozoan parasites of marine mammals. In: CRC Handbook of marine mammal medicine. ISBN9781315144931. United States: CRC Press. pp. 425-470.
- Pal M, Bulcha MR, MitikuBune W, Koliopoulos T. (2021). Toxoplasmosis: Public health importance and its status in Ethiopia. Available at: https://www.telegeco.gr/eISSN2623-4882.html.
- Gazzonis AL, Marino AM, Garippa G, Rossi L, Mignone W, Dini V, et al. (2020). Toxoplasma gondii seroprevalence in beef cattle raised in Italy: A multicenter study. Parasitol Res. 119:3893-3898.
- Vilares A. (2019). Genetic study of Toxoplasma gondii strains isolated from humans and animals. Available at: http://hdl.handle.net/10451/43871.
- Nayeri T, Sarvi S, Moosazadeh M, Daryani A. (2021). Global prevalence of Toxoplasma gondii infection in the aborted fetuses and ruminants that had an abortion: A systematic review and meta-analysis. Vet Parasitol. 290:109370.
- Pereira DC, Dubey JP, Da Mata A, Neto H, Cardoso L, Lopes AP. (2020). Seroepidemiology of Toxoplasma gondii in domestic cattle, sheep, goats and pigs from São Tomé and Príncipe. Rev Bras Parasitol Vet. 29(1):e014819.
- Hill DE, Dubey JP. (2016). Toxoplasma gondii as a Parasite in Food: Analysis and Control. Microbiol Spectr. 4(4):227-247.
- Ibrahim N. (2017). Review on Toxoplasmosis and Its Zoonotic Importance. Jimma University Open Access Institutional Repository. 5(2):21-29.
- Elsheikha HM, Marra CM, Zhu XQ. (2020). Epidemiology, Pathophysiology, Diagnosis, and Management of Cerebral Toxoplasmosis. Clin Microbiol Rev. 34(1):e00115-e00119.
- Acha PN, Szyfres B. (2001). Zoonoses and Communicable Diseases Common to Man and Animals: Volume 3: Parasitoses. Pan American Health Org.
- Hill SL, Cheney JM, Taton-Allen GF, Reif JS, Bruns C, Lappin MR. (2000). Prevalence of enteric zoonotic organisms in cats. J Am Vet Med Assoc. 216(5):687-692.
- Radostits OM, Gay C, Hinchcliff KW, Constable PD, editors. (2006). Veterinary Medicine E-Book: A textbook of the diseases of cattle, horses, sheep, pigs, and goats. Elsevier Health Sciences.
- Pal M, Alem B, Gari G, Tuli G. (2014). Toxoplasmosis in animals and humans, its diagnosis, epidemiology and control. Int J Livest Res. 4(2):1-0.
- Dubey JP. (2010). Toxoplasma gondii infections in chickens (Gallus domesticus): prevalence, clinical disease, diagnosis, and public health significance. Zoonoses Public Health. 57(1):60-73.
- Dubey JP, Jones JL. (2008). Toxoplasma gondii infection in humans and animals in the United States. Int J Parasitol. 38(11):1257-1278.
- Johnson SK, Johnson PTJ. (2021). Toxoplasmosis: Recent Advances in Understanding the Link Between Infection and Host Behavior. Annu Rev Anim Biosci. 9:249-264.
- Castagnini M, Bernazzali S, Ginanneschi C, Marchi B, Maccherini M, Tsioulpas C, et al. (2007). Fatal disseminated toxoplasmosis in a cardiac transplantation with seropositive match for Toxoplasma: should prophylaxis be extended? Transpl Immunol. 18(2):193-197.
- Asiyabi Aghdam S, Hajipour N, Moosavy MH. (2023). Use of PCR to determine Toxoplasma gondii in milk samples from camels (Camelus dromedarius), cattle (Bos taurus) and buffalos (Bubalus bubalis) in East Azarbaijan province, Iran. Vet Med Sci. 9(1):400-404.
- Dass SA, Vasudevan A, Dutta D, Soh LJ, Sapolsky RM, Vyas A. (2011). Protozoan parasite Toxoplasma gondii manipulates mate choice in rats by enhancing the attractiveness of males. PloS one. 6(11):e27229.
- Arantes TP, Lopes WD, Ferreira RM, Pieroni JS, Pinto VM, Sakamoto CA, et al.( 2009). Toxoplasma gondii: Evidence for the transmission by semen in dogs. Exp Parasitol. 123(2):190-194.
- Gutierrez J, O'Donovan J, Williams E, Proctor A, Brady C, Marques PX, et al. (2010). Detection and quantification of Toxoplasma gondii in ovine maternal and foetal tissues from experimentally infected pregnant ewes using real-time PCR. Vet Parasitol. 172(1-2):8-15.
- Tenter AM. (2009). Toxoplasma gondii in animals used for human consumption. Mem Inst Oswaldo Cruz. 104(2):364-369.
- Stelzer S, Basso W, Benavides Silván J, Ortega-Mora LM, Maksimov P, Gethmann J, et al. (2019). Toxoplasma gondii infection and toxoplasmosis in farm animals: Risk factors and economic impact. Food Waterborne Parasitol. 15:e00037.
- Singh B, Debrah AY, Debrah LB, Djameh G. (2022). Prenatal Diagnosis and Genetic Characterization of Toxoplasmosis in Immunocompetent and Immunocompromised Pregnant Women and the Risk of Congenital Toxoplasmosis: A Prospective Study in Ghana. Journal of Science and Technology (Ghana). 41(2):18-42.
- Wellington AO, Oladipo OO, Chimere OA, Oladele TO, Anunobi CC, Soyebi K. (2009). Congenital Toxoplasmosis: A Review of its Pathology, Immune Response, and Current Treatment Options. Sierra Leone J Biomed Res. 1(1):9-20.
- Tenter AM, Heckeroth AR, Weiss LM. (2000). Toxoplasma gondii: from animals to humans. Int J Parasitol. 30(12-13):1217-1258.
- Martorelli Di Genova B, Wilson SK, Dubey JP, Knoll LJ. (2019). Intestinal delta-6-desaturase activity determines host range for Toxoplasma sexual reproduction. PLoS Biol. 17(8):e3000364.
- Al-Malki ES. (2021). Toxoplasmosis: stages of the protozoan life cycle and risk assessment in humans and animals for an enhanced awareness and an improved socio-economic status. Saudi J Biol Sci. 28(1):962-969.
- Calero-Bernal R, Gennari SM. (2019). Clinical Toxoplasmosis in Dogs and Cats: An Update. Front Vet Sci. 6:54.
- Meerburg BG, Kijlstra A. (2009). Changing climate-changing pathogens: Toxoplasma gondii in North-Western Europe. Parasitol Res. 105(1):17-24.
- Yan C, Liang LJ, Zheng KY, Zhu XQ. (2016). Impact of environmental factors on the emergence, transmission and distribution of Toxoplasma gondii. Parasit Vectors. 9:137.
- Cerutti A, Blanchard N, Besteiro S. (2020). The Bradyzoite: A Key Developmental Stage for the Persistence and Pathogenesis of Toxoplasmosis. Pathogens. 9(3):234.
- Tomasina R, Francia ME. (2020). The Structural and Molecular Underpinnings of Gametogenesis in Toxoplasma gondii. Front Cell Infect Microbiol. 10:608291.
- Lopez A, Dietz VJ, Wilson M, Navin TR, Jones JL. (2000). Preventing congenital toxoplasmosis. MMWR Recomm Rep. 49(RR-2):59-68.
- Conrad PA, Miller MA, Kreuder C, James ER, Mazet J, Dabritz H, et al. (2005). Transmission of Toxoplasma: clues from the study of sea otters as sentinels of Toxoplasma gondii flow into the marine environment. Int J Parasitol. 35(11-12):1155-1168.
- Pleyer U, Gross U, Schlüter D, Wilking H, Seeber F. (2019). Toxoplasmosis in Germany: Epidemiology, diagnosis, risk factors, and treatment. Dtsch Arztebl Int. 116(25):435-444.
- Dardé ML. (2008). Toxoplasma gondii, "new" genotypes and virulence. Parasite. 15(3):366-371.
- Masala G, Porcu R, Daga C, Denti S, Canu G, Patta C, Tola S. (2007). Detection of pathogens in ovine and caprine abortion samples from Sardinia, Italy, by PCR. J Vet Diagn Invest. 19(1):96-98.
- Castro-Sansores CJ, Góngora-Biachi RA, González-Martínez P. (2004). HIV-encephalopathy as initial manifestation of acquired immunodeficiency syndrome in Yucatán State, Mexico. Arch Med Res. 35(1):49-53.
- Sukthana Y. (2006). Toxoplasmosis: beyond animals to humans. Trends Parasitol. 22(3):137-142.
- Gilbert RE, Stanford MR. (2000). Is ocular toxoplasmosis caused by prenatal or postnatal infection? Br J Ophthalmol. 84(2):224-226.
- Hill D, Dubey JP. (2002). Toxoplasma gondii: transmission, diagnosis and prevention. Clin Microbiol Infect. 8(10):634-640.
- Montoya JG. (2002). Laboratory diagnosis of Toxoplasma gondii infection and toxoplasmosis. J Infect Dis. 185(Suppl 1):S73-S82.
- Karthikeyan R, Anbazhagan S, Srinivas K, Angappan M, Agri H, Yadav A. (2021). Toxoplasmosis: Mysterious Disease with Paradigm for One Health. Int J Livest. Res. 11:1-2.
- Antczak M, Dzitko K, Długońska H. (2016). Human toxoplasmosis-Searching for novel chemotherapeutics. Biomed Pharmacother. 82:677-684.
- Dunay IR, Gajurel K, Dhakal R, Liesenfeld O, Montoya JG. (2018). Treatment of Toxoplasmosis: Historical Perspective, Animal Models, and Current Clinical Practice. Clin Microbiol Rev. 31(4):e00057-17.
- Kravetz JD, Federman DG. (2005). Prevention of toxoplasmosis in pregnancy: knowledge of risk factors. Infect Dis Obstet Gynecol. 13(3):161-165.
- Elsheikha HM. (2008). Congenital toxoplasmosis: priorities for further health promotion action. Public Health. 122(4):335-353.
- De Berardinis A, Paludi D, Pennisi L, Vergara A. (2017). Toxoplasma gondii, a Foodborne Pathogen in the Swine Production Chain from a European Perspective. Foodborne Pathog Dis. 14(11):637-648.
- Das D, Islam S, Bhattacharjee H, Deka A, Yambem D, Tahiliani PS, et al. (2014). Parasitic diseases of zoonotic importance in humans of northeast India, with special reference to ocular involvement. Eye Brain. 6:1-8.
- Dabritz HA, Conrad PA. (2010). Cats and Toxoplasma: implications for public health. Zoonoses Public Health. 57(1):34-52.
- Remington JS, Klein JO. (2001). Infectious diseases of the fetus and newborn infant. London: WB Saunders. Available at: https://doi.org/10.1136%2Fadc.2006.102566.
- Dubey JP. (2004). Toxoplasmosis - a waterborne zoonosis. Vet Parasitol. 126(1-2):57-72.
- Kanková S, Sulc J, Nouzová K, Fajfrlík K, Frynta D, Flegr J. (2007). Women infected with parasite Toxoplasma have more sons. Naturwissenschaften. 94(2):122-127.
- Npankaew T, Thi Thuy N, Nimsuphan B, Kengradomkij C, Kamyingkird K, Chimnoi W, et al. (2021). Seroprevalence of Toxoplasma gondii infection from water buffaloes (Bubalus bubalis) in northeastern and southern Thailand. Folia Parasitol (Praha). 68:1-6.
- Aguirre AA, Longcore T, Barbieri M, Dabritz H, Hill D, Klein PN, et al. (2019). The one health approach to toxoplasmosis: epidemiology, control, and prevention strategies. Ecohealth. 16(2):378-390.
- Santoro M, Viscardi M, Sgroi G, DʼAlessio N, Veneziano V, Pellicano R, et al. (2019). Real-time PCR detection of Toxoplasma gondii in tissue samples of wild boars (Sus scrofa) from southern Italy reveals high prevalence and parasite load. Parasit Vectors. 12(1):335.
- Gazzonis AL, Villa L, Riehn K, Hamedy A, Minazzi S, Olivieri E, et al. (2018). Occurrence of selected zoonotic food-borne parasites and first molecular identification of Alaria alata in wild boars (Sus scrofa) in Italy. Parasitol Res. 117(7):2207-2215.
- Slany M, Reslova N, Babak V, Lorencova A. (2016). Molecular characterization of Toxoplasma gondii in pork meat from different production systems in the Czech Republic. Int J Food Microbiol. 238:252-255.
- Abrantes AC, Vieira-Pinto M. (2023). 15 years overview of European zoonotic surveys in wild boar and red deer: A systematic review. One Health. 16:100519.
- Tedla Y, Shibre T, Ali O, Tadele G, Woldeamanuel Y, Asrat D, et al. (2011). Serum antibodies to Toxoplasma gondii and Herpesvidae family viruses in individuals with schizophrenia and bipolar disorder: a case-control study. Ethiop Med J. 49(3):211-220.
- Adem D, Ame M. (2023). Toxoplasmosis and its significance in public health: a review. Journal of Biomedical and Biological Sciences. 2(1):1-20.
- Frimpong C, Makasa M, Sitali L, Michelo C. (2017). Seroprevalence and determinants of toxoplasmosis in pregnant women attending antenatal clinic at the university teaching hospital, Lusaka, Zambia. BMC Infect Dis. 17(1):10.
- Adugna B, Tarekegn ZS, Damtie D, Woldegebreal SN, Raju RP, Maru M, et al. (2021). Seroepidemiology of Toxoplasma gondii Among Pregnant Women Attending Antenatal Care in Northwest Ethiopia. Infect Drug Resist. 14:1295-1303.
- Negash T, Tilahun G, Medhin G. (2008). Seroprevalence of Toxoplasma gondii in Nazaret town, Ethiopia. East Afr J Public Health. 5(3):211-244.
- Yesuf KM, Melese ZT. (2015). Prevalence of toxoplasmosis in HIV/AIDS patients in Mettu Karl hospital. American Journal of Health Research. 3(3):183-188.
- Shimelis T, Tebeje M, Tadesse E, Tegbaru B, Terefe A. (2009). Sero-prevalence of latent Toxoplasma gondii infection among HIV-infected and HIV-uninfected people in Addis Ababa, Ethiopia: A comparative cross-sectional study. BMC Res Notes. 2:213.
- Zemene E, Yewhalaw D, Abera S, Belay T, Samuel A, Zeynudin A. (2012). Seroprevalence of Toxoplasma gondii and associated risk factors among pregnant women in Jimma town, Southwestern Ethiopia. BMC Infect Dis. 12:337.
- Gebremedhin EZ, Gizaw D. (2014). Seroprevalence of Toxoplasma gondii infection in sheep and goats in three districts of Southern Nations, Nationalities and Peoples’ Region of Ethiopia. World Appl Sci J. 31(11):1891-1896.
- Kebeta MM. (2014). Toxoplasma Gondii In Selected Sites Of Central Ethiopia: Seroprevalence, Risk Factors, And Bioassay In Pigs (Doctoral dissertation, MSc Thesis, College of Veterinary Medicine and Agriculture of Addis Ababa, Addis Ababa University. pp. 20-25. Available at: http://etd.aau.edu.et/handle/123456789/4984.
- Dubey JP, Darrington C, Tiao N, Ferreira LR, Choudhary S, Molla B, et al. (2013). Isolation of viable Toxoplasma gondii from tissues and feces of cats from Addis Ababa, Ethiopia. J Parasitol. 99(1):56-58.
- Negash T, Tilahun G, Patton S, Prevot F, Dorchies PH. (2004). Serological survey on toxoplasmosis in sheep and goats in Nazareth, Ethiopia. Revue de medecine veterinaire. 155(10):486-487. Available at: http://www.biomedcentral.com/1746-6148/9/117.
- Zewdu E, Agonafir A, Tessema TS, Tilahun G, Medhin G, Vitale M, et al. (2013). Seroepidemiological study of caprine toxoplasmosis in East and West Shewa Zones, Oromia Regional State, Central Ethiopia. Res Vet Sci. 94(1):43-48.
- Gebremedhin EZ, Tesfamaryam G, Yunus HA, Duguma R, Tilahun G, DI Marco V, et al. (2015). Seroepidemiology of Toxoplasma gondii infection in free-range chickens (Gallus domesticus) of Central Ethiopia. Epidemiol Infect. 143(3):608-617.