Previous Issues Volume 3, Issue 1 - 2018
Progress of Chemical Components and Biological Activities of Lonicera Maackii
Yang Yang1,3, Olagoke Zacchaeus Olatunde2 , Jianping Yong1 *, Canzhong Lu1,2*
1Xiamen Institute of Rare-earth Materials, Haixi Institute, Chinese Academy of Sciences, Xiamen. 2Fujian Institute of Research on the Structure of Matter, Haixi Institute, Chinese Academy of Sciences, Fuzhou. 3ShanghaiTech University, Shanghai. Corresponding Author: Jianping Yong and Canzhong Lu, Xiamen Institute of Rare-earth Materials, Haixi Institute, Chinese Academy of Sciences, Xiamen, Tel: +86-591-63173162; E-Mail: [email protected], [email protected] Received Date: 26 Nov 2018 Accepted Date: 28 Nov 2018 Published Date: 30 Nov 2018 Copyright © 2018 Yong J and Lu C Citation: FYang Y, Olatunde OZ, Yong J and Lu C. (2018). Progress of Chemical Components and Biological Activities of Lonicera Maackii. M J Canc. 3(1): 016. ABSTRACT Lonicera Maackii is a shrub belonging to the family Caprifoliaceae. Its fruits, leaves and flowers contain many kinds of biological compounds together with a wide spectrum of biological activities. Some chemical components together with biological activities have been studied and reported. This review briefly describes the progress of the chemical components and biological activities of Lonicera Maackii, for providing a reference for the researchers. KEYWORDS Lonicera Maackii; Chemical Components; Biological Activities. INTRODUCTION
Lonicera Maackii (Rupr.) Maxim (Amur honeysuckle in English, gold and silver wood in Chinese, Figure 1) is a kind of deciduous shrub belonging to the family Caprifoliaceae and is native to temperate Asia in northern and western China, Mongolia, Japan, Korea, and Southeastern Russian. In China, Lonicera maackii is an important ornamental plant and street trees, widely planted because of its fragrant flowers, long flowering period and attractive red fruits. The leaves and flowers of Lonicera Maackii possess distinct antibacterial activity and the roots of Lonicera Maackii also possess antibacterial activity and been used for treatment for malaria disease [1]. The aqueous extraction of the roots of Lonicera Maackii exhibits effective activities of anti-inflammatory[2], removing fever and immune system, granules can be used to treat pulmonary fever bronchitis in children caused by bacteria [3]. In this minireview, we briefly describe the progress of the components and biological activities of Lonicera Maackii to provide a reference for the researchers.
Chemical components of Lonicera Maackii It is reported that there are many kinds of bioactive compounds in fruits, flowers and leaves of Lonicera Maackii,which contain iridoids, triterpenes, volatileoils,flavonoids,phenylpropanoids, etc..
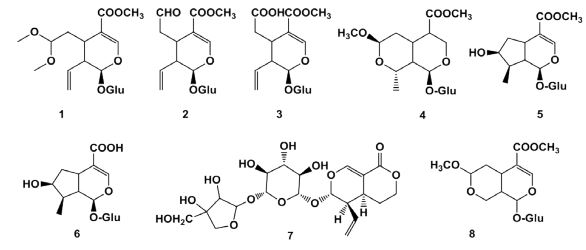
Iridoids Guangshu Wang et al. [4] obtained cleaved seloganin dimethylacetal (1), seloganin (2) and secoxyloganin (3) from the fruits alcohol extract of Lonicera Maackii. Shengjie Yuan [5] isolated 7S-O-methylmorroniside (4), loganin (5), loganin acid (6), 6’-O-β-D-apiofuranosyl sweroside (7) from the fruits of Lonicera Maackii. Heng Zhu et al. [6] identified morronside,(8) loganin and secoxyloganin from the flowers of Lonicera Maackii. The structures of the related compounds listed in Figure 2.
Figure 2: The structures of the related iridoids
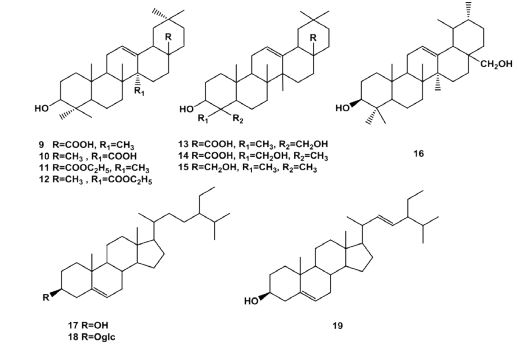
Triterpenes Some triterpenes were isolated from the fruits of Lonicera maackii, which were oleanolic acid (9), 3β-hydroxyolean-12- en-27-oic acid (10), ethyl ursolic acetate (11), 3β-hydroxyolean12-en-27-oic acid ethylester (12), isohederagenin (13), hederagenin (14), erythrodiol (15), urusol (16), β-sitosterol (17), β-daucosterol (18), stigmasterol (19) [7-9]. The structures listed in Figure 3.
Volatile oils Xiaoling Ji et al. [10] identified thirty-two compounds from the essential oil extracted from the fruits of Lonicera macckii by GC-MS., and the methylhexyl hydroperoxide was in the highest amount of all detected compounds and accounting for 1.31% of the volatile oil. Some complex molecular structure such as digitoxin, 9-[2’,2’-dimethyl propionyl hydrazone]3,6-dichloro-2,7-bis[2-[diethylamino]ethoxy]fluorine, cholest-22-en-21-ol, 3, 5-dehydro-6-methoxy valerate were also detected in this study. Some simple compounds: such as hexane, 2,4-dimethylpentane, 2-dimethyl-3-methylenebicyclo[2,2,1]hexane, 7,7-trimethyl- bicyclo[4,1,0]hex3-ene, 1-methyl-4-isopropylbenzene, heptadecyl-trichlorosilane, 1-iodo-2-methylolane, O-alkenylhydroxylamine, 1,1,3α-7-tetramethyl- 1α,2,3,3α,4,5,6,7β-octahydro-1-hydrocyclopropanol[α]decene, (2E)-2-dodecanoic acid, 2,6,10-trimethyltetradecane, pentadecane, methyl 12,15-octadecadienoate, 11,14-octadecadienoic acid methyl ester, 12,15-octadecadienoic acid methyl ester, 2-(7-acetylheptadecanyloxy)- tetrahydropyran, 9,12-heptadecacadienoic acid glycerol (2,3-ditrimethylsilyl)ether, [2Z,5Z]-2,5-pentadecanol, 7-methyl-Z-hexadecyl carbonate, [2E,6E]-10,11- epoxy-7-ethyl-3,11-dimethyl-2,6-tetradecanoic acid methyl ester, 3-heptyloxymethyl-7-methoxy-benzofuran-2-one, 2-methyl-2-pentadecanethiol, methylene bis[6-tert-butyl] p-cresol, 2-ethanol octadecyl ether, 2-[(trimethylsilyl)oxy] -1-[[(trimethylsilyl)oxy]methyl]ethyl[9E,12E,15E]9,12,15- octa-decatrienoic acid ester, 10-octadecenoic acid, 3,7-Dimethyl-3-ethyl-7-tert-butyl-hexadecane, 2-bromo- octadecanoic acid, phthalic acid mono[2-ethylhexyl] ester were also detected . Jinying Li et al.[11] identified 16 components from mature fruits of Lonicera Maackii,including aldehydes(2-hexenal, phenylacetaldehyde, nonanal, decanal, β-cyclocitral), alcohols [ trans-2-hexen-1-ol, linalool, phenethyl alcohol, (-)-terpinen-4-ol], terpenes (3-heptene), alkanes (dodecane, tridecane, hexadecane), ester [(2,6,6-Trimethyl- 2-hydroxycyclohexylidene)acetic acid lactone] , acid(trans-2-hexenyl-hexanoate)and ketones(damascenone, geranylacetone), the first two classes of compounds accounted for a high mass fraction of 55.80% and 28.48%, respectively. Guangshu Wang et al. [12] obtained the essential oil from the dry buds of Lonicera macckii and confirmed 18 components by GC-MS, the detected compounds were: 1-(2,6,6-trimethyl-1,3-cyclohexanedienyl)-2-butene-1-one, 1,1,6-trimethyl- 1,2,3,4-tetrahydro-naphthalene, tetradecane, 1-ethyl-2,3-dimethylbenzene, (-) farnesene, pentadecane, hexadecane, octadecane, phytane, nonadecane, hexadecanoic acid, eicosane, heneicosane, docosane, methyl behenate, nonacosane, methyl lignocerate, which account for 70.4% of the total volatile oil. Xinyan Gao et al. [13] obtained the volatile components from the flowers and fresh leaves of Lonicera macckii using the Soxhlet extraction method, and confirmed their components by GC-MS. The detected compounds were: ethanedioic acid dimethyl ester, 6-chloro6-methyl-bicyclo[3.1.0] hexane, (Z)-3-methyl-4-undecene, (E)-ethyl-3-hexenyl carbonate, p-xylene, cyclohexanol, 2,3-dimethyl-3-hexanol, benzaldehyde, cis-octahydro-pentalene, hexanoic acid-4-hexenyl-1-ester, 5-ethyl-2-furaldehyde, benzeneacetaldehyde, 5-(1-methyl-propyl)-nonane, 2,7,10-trimethyl-dodecane, 1,2,3,4-tetramethyl-benzene, naphthalene, 2-(1-methyl-ethyl)-cyclohexanol, cantharidin, 3,7-dimethyl-2,6-nona- dien-1-ol, (6-hydroxy-methyl-2,3-di-methyl-phenyl)-methanol, 3,5-dimethyl-benzoic acid methyl ester, 3,5-bis(1,1-dimethyl-ethyl)-phenol, 3-(2,6,6-trimethyl1-cyclohexenyl)-2-propen-1-ol, 2,3-dihydro-4,4-dimethyl-indole-4-ol-2-one, (-)-isolongifolol methyl ether, (-)-isolongifolol, 2-hydroxy-4,4,8-trimethyl-5-tricyclo [6.1.1.0(2,6)]-2(6)-decene, imiprothrin, D-bicuculline, scoparone, picrotoxin, 2,4,7,14-tetramethyl-4-vinyl-tricyclo [5.4.3.0(1,8)] tetradecan-6-ol, dibutyl phthalate. Xinyan Gao et al [14] also obtained the following components from the fresh fruits of Lonicera macckii: 3-(2-methoxyethoxymethoxy)-2-methyl-1-pentaol, 2,7,10-trimethyl-dodecane, 2,4-bis(1,1-dimethylethyl)-phenol, benzoic acid-2,6-bis[(trimethylsilyl)oxy]-timethylsilyl ester, 7-chloro-1,3-dihydro-5-phenyl- 1-(trimethylsilyl)-3- [(trimethylsilyl)oxy]-2H-1,4-benz-odiazepin-2-one, benzoic acid-2,5-bis(trimethylsiloxy)-trimethylsilyl ester, benzeneacetic acid-α-3,4-tris [(trimethylsilyl)oxy] -trimethylsilyl ester, pentacosane, tetracosane, dot riacontane, (Z,Z)- 9-hexadecenoic acid-9-octadecenyl ester, octadecanoic acid2-(hexadecyloxy) ethyl ester, 1,2-cinnolinedicarboxylic acid1,2,3,5,6,7,8,8a-octahydro- 4-trimethylsilyloxy-diethyl ester
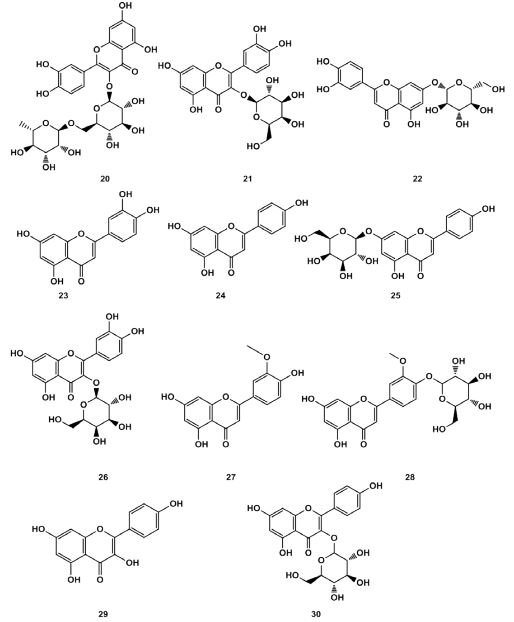
Flavonoids Heng Zhu et al. [6] identified flavonoids such as rutin (20), hyperoside (21), luteolin (22) from the flowers of Lonicera macckii. Luteolin (23), apigenin (24), apigenin-7-glucoside (25), hyperoside (26), chrysoderol (27), chrysoderol-4`-glucoside (28), kaempferol (29) and kaempferol-3-glucoside (30) were isolated from the leaves of Lonicera macckii by Junli Ma [15,16]. The Lonicera macckii fruit is one of the main parts where total flavonoids exist in. The structures of the related compounds listed in Figure 4.
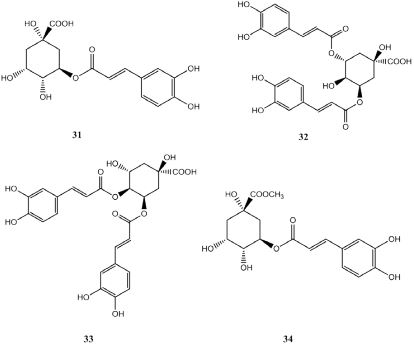
Phenylpropanoids Heng Zhu et al.[6] identified phenylpropanoid compounds such as chlorogenic acid (31), isochlorogenic acid A (32), isochlorogenic acid C (33) from the flowers of Lonicera macckii by HPLC-DAD-ESI-Q-TOF/MS. Junli Ma [15,16] isolated chlorogenic acid and methyl chlorogenic acid (34) from the leaves of Lonicera macckii. The structures of the related compounds listed in Figure 5.
Other components In addition to the above-mentioned components, other compounds have also been discovered in the fruits of Lonicera macckii, such as benzy -O-β-D-xylopyranosyl-(1→6)-β-Dglucopyranoside [4], 1,3-dihydroxylpropyl (9Z, 12Z) -octadeca-9, 12-dienate, 1,3-dihydroxylpropyl (9E, 12E)-octadeca-9, 12-dienate, 3, 7-dimethyl-3, 8-dihydrooctene, (E)-2, 6-dimethylocta-2, 7-dien-1-ol, 1-acetyl-2-methyl-5-(2-vinyloxiran-2-yl) pentanester ͵ tripalmitin, glucose, sucrose [8, 9].
It was reported that the extractions from the leaves of Lonicera maackii exhibited antibacterial, antipyretic-analgesic effects and improved the immunity. The extractions of the roots of Lonicera Maackii possess higher activities with antibacterial, anti-inflammatory, removing the fever, treatment of malarial disease, and improving the immunity. The extractions of fruits of Lonicera Maackii exhibited higher anticancer, protective liver, reducing blood sugar and treatment of cardiovascular disease.
Antioxidant Activity Yingchen Wang [17] reported that the polysaccharides in fruits of Lonicera Maackii can effectively scavenge hydroxyl radicals, superoxide anion radicals and DPPH radicals, the scavenging ability is positively correlated with the concentrations, which indicated that the polysaccharides in the fruits of Lonicera Maackii possess the antioxidant activity. Shumei Li et al.[18] reported that the flavonoids in fruits of Lonicera maackii possess distinct antioxidant activity and the antioxidant ability is more superior to vitamin E. Chlorogenic acid in Lonicera Maackii is also a good in vitro antioxidant[19].
Antitumor activity Duowu Chai [20] reported that the rough extractions in fruits of Lonicera Maackii exhibited higher antitumor activity, and the ethyl acetate extraction is more active than other extractions. Oleanolic acid exhibited higher anticancer activity against liver cancer cell lines 7721 and leukemia HL-60 with the inhibition of 57.35% and 55.23% respectively. The antitumor effect of oleanolic acid is expressed in various stages of tumor development, including inhibition of tumor development, inhibition of tumor growth, and induction of tumor cell differentiation. It can also effectively inhibit angiogenesis, tumor cell invasion and metastasis. Kasai et al. [21] found that chlorogenic acid can inhibite the increase of 8-OH-dG in DNA induced by 4-nitroquinoline-1-oxide (a carcinogen formed by oxygen free radicals) through constructing a rat carcinogenesis model. Rutin components in Lonicera Maackii can induce the apoptosis of HepG2, and also inhibit the colon cancer and colorectal cancer [22].
Antibacterial activity Qian Ren et al. [23] reported that the extractions of Lonicera maackii exhibited higher antibacterial activity, the antibacterial sequence of the extractions was leaf > bud > stem bark. Xinyan Gao et al.[11]reported that the extractions from the fresh leaves of Lonicera Maackii exhibited higher antibacterial activity against E. coli, Bacillus subtilis and Staphylococcus aureus with the IC50s:0.039, 0.099 and <0.0119g/mL. Yanwu Hu et al. [24] reported that all different solvents extractions of Lonicera Maackii contain antibacterial ingredients and the antibacterial effect is synergistic action of all antibacterial ingredients.
Protective liver The oleanolic acid and iridoid in fruits of Lonicera Maackii possess protective effects on the liver. Xinjiang Gong et al. [25] constructed a cyclophosphamide-induced liver injury model and carried out experiments. It was found that oleanolic acid could inhibit the activities of alanine aminotransferase, aspartate aminotransferase and lactate dehydrogenase in mice serum caused by liver injury, and increases the MTT value of hepatocytes, indicating that oleanolic acid had anticyclophosphamide-induced hepatocyte injury activity in rats. Reisman et al. [26] reported that oleanolic acid can activate the nuclear accumulation of Nrf2 and induce Nrf2-dependent genes, thus protecting the liver from acetaminophen-induced hepatotoxicity. Hui Su [27] established a model of liver injury induced by carbon tetrachloride in mice and conducted experiments, the results showed that the total iridoid glycosides in Lonicera Maackii fruits could significantly reduce the elevated alanine aminotransferase and aspartate aminotransferase in the serum of mice with liver injury and revealed that the total iridoid glycosides of Lonicera Maackii fruits had significant hepatoprotective activity.
Reducing blood sugar Su Hui [27]constructed alloxan-induced hyperglycemia model and adrenaline model, by comparing normal and hyperglycemia model mice, it was found that high dose group (4 g/kg total iridoid glycosides) had significant hypoglycemic effect, while the high dose group (4 g/kg total iridoid glycosides) and middle dose group (2 g/kg total iridoid glycosides) could significantly antagonize the increase of blood sugar induced by adrenaline. The monomeric substance loganin which obtained from the Lonicera Maackii fruit was studied and found to significantly alleviate the increase of blood glucose in alloxan-induced hyperglycemia mice, but not as good as the treatment of total iridoid glycosides. Linpu Xie [28] constructed a streptozotocin-induced hyperglycemia rat model to study the effect of oleanolic acid on insulin and hepatic glycogen content in hyperglycemic rats,it was found that high dose group (100 mg/kg) had significant hypoglycemic effect, not only reduced fasting blood glucose in hyperglycemic rats, but increased serum insulin and liver glycogen content as well. From the above review, we found that although some studies about the chemical components and biological activities of Lonicera Maackii have been carried out, the results are simple and further studies need to be carried out. These years, our research group carried out the drug design, synthesis, isolation of the plant medicines and their biological evaluation. The fruits of Lonicera Maackii are attractive and eaten by birds, whether it can be eaten by human? So we will carry out the further studies about the chemical components and biological activities of the fruits, leaves and flowers of Lonicera maackii in subsequent work. ACKNOWLEDGEMENTS
This work was financially supported by the National Natural Science Foundation of China (21875252) and the Project of The Plan of Xiamen Science and Technology (3502ZCQ20171000).
- Zhu YC. (1989). Medicinal plants in Northeast China. Harbin: Heilongjiang Science and Technology Press. 1061.
- Liu BQ, Ji YP, He SL, Duan WH, et al. (1992). Review of North Honeysuckle (Lonicera Maackii). Jilin Journal of Traditional Chinese Medicine. 14 (3): 41- 42.
- Jiao YC and Lu Z. (1986). Observation on Curative Effect of “Lonicera Maackii Granule” in Treating 100 Cases of Infantile Pneumonia. Heilongjiang Journal of Traditional Chinese Medicine. 8(5): 38.
- Wang GS, Zhou XP, Yuan SJ and Yang XH. (2010). Chemical Constituents in the Fruits of Lonicera macckii. Chinese Journal of Medicinal Chemistry. 20(3): 211-213.
- Yuan SJ. (2007). The Isolation and Structure Elucidation of Iridoid Compounds from the Fruits of Lonicera macckii (Rupr.) Maxim. Changchun: Jilin University.
- Zhu H, Cui L, Liu Q, Jiang JJ, et al. (2017). Determination of chemical constituents in Lonicera Maackii with HPLCDAD- ESI-Q-TOF/MS. Chinese Traditional and Herbal Drugs. 48(11): 2300-2305.
- Wang YL, Yang L, Luo GA and Wang YM. (2007). Study on Nonirodoids from Fruits of Lonicera Maackii. Nat Prod Res Dev. 19(1): 51-54
- JP Yong, XY Wu and CZ Lu. (2014). Chemical Constituents Isolated from the Fruits of Lonicera Maackii. Chemistry of Natural Compounds. 50(4): 765-766.
- JP Yong, CZ Lu and SJ Huang. (2014). Chemical Constituents of Lonicera Maackii. Chemistry of Natural Compounds. 50(5): 945-947
- Ji XL, Yong JP and Wang L. (2011). GC-MS Analysis of Volatile Components in Lonicera Maackii Fruits. Chinese Journal of Spectroscopy Laboratory. 28(6): 3110-3112.
- Li JY, Liu HZ, Liu SY and Zhao CL. (2015). Composition of Volatile Organic Compounds in Lonicera maacki Maxim and Lonicera caerulea L. Journal of Northeast Forestry University. 43(7): 133-135.
- Wang GS, Liu LJ, Sun W and Yang XH. (2009). Study on the Chemical Constituents of Essential Oil in the Buds of Lonicera macckii (Rupr.) Maxim. Special Wild Economic Animal and Plant Research. 3: 60-61.
- Gao XY, Wang HY, Liu ZM, Duan XL, et al. (2018). Analysis of Volatile Components and Antibacterial Activity of Lonicera macckii (Rupr.) Maxim. Extracts. Biomass Chemical Engineering. 51(1): 10-16
- Gao XY, Wang HY and Liu ZM. (2018). GC-MS Analysis of Fresh Fruits Extracts of Lonicera macckii. Chinese Wild Plant Resources. 37(4): 25-28.
- Ma JL. (2013). Isolation and Identification of Chemical Constituents from Lonicera macckii Leaves. Asia-Pacific Traditional Medicine. 9(2): 33-34.
- Ma JL and Li JS. (2013). Chemical Constituents from Leaves of Lonicera macckii. Drugs & Clinic. 28(4): 476-479.
- Wang YC. (2012). Extraction of Polysaccharides from Lonicera Maackii and Its Anti-oxidation. Guizhou Agricultural Sciences. 40(7): 92-93.
- Li SM, Fu ZS, Li L and Sheng XL. (2012). Research on Extraction and activity of Flavonoid Compounds from Lonicera maackii Fruits. Journal of Inner Mongolia Agricultural University. 33(5-6): 237-240.
- Rodriguez de Sotillo DV and Hadley M. (2002). Chlorogenic Acid Modifies Plasma and Liver Concentrations of : Cholesterol, Triacylglycerol, and Minerals in (fa/fa) Zucker Rats. The Journal of Nutritional Biochemistry. 13(12): 717-726.
- Chai DW. (2010). Screening of the Natural anti-tumor Substances in fruits of Lonicera Maackii (Rupr.) Maxim. Lanzhou: Lanzhou University
- Kasai H, Fukada S, Yamaizumi Z, Sugie S, et al. (2000). Action of Chlorogenic Acid in Vegetables and Fruits as an Inhibitor of 8-Hydroxydeoxyguanosine Formation In Vitro and in A Rat Carcinogenesis Model. Food and Chemical Toxicology. 38 (5): 467-471.
- Volate SR, Davenport DM, Muga SJ and Wargovich MJ. (2005). Modulation of Aberrant Crypt Foci and Apoptosis by Dietaryherbal Supplements (Quercetin, Curcumin, Silymarin, Ginseng Andrutin). Carcinogenesis. 26(8): 1450- 1456.
- Ren Q, Li Q, Li WB, Chen GL, et al. (1991). Study on Antibacterial Activity of 15 Kinds of Lonicera Linn Medicinal Plants. Territory & Natural Resources Study. 2: 68-70.
- Hu YW, Li ML, Wang J and Wang D. (2010). Determination of Chlorogenic Acid in the Stems or Leaves of Lonicera maackii (Rupr.) Maxim. in Changbai Mountain and its Antibacterial Activities. Northern Horticulture. 9: 172-173.
- Gong XJ, Ding H, Qiu YS, Jia ZR, et al. (2006). Toxicity of Cyclophosphamide on Primary Cultured Hepatocytes of Rat and Protective Effects of Oleanolic Acid. Herald of Medicine. 25(11): 1114-1116.
- Reisman SA, Aleksunes LM and Klaassen CD. (2009). Oleanolic Acid Activates Nrf2 and Protects from Acetaminophen Hepatotoxicity via Nrf2-dependent and Nrf2-independent Processes. Biochemical Pharmacology. 77(7): 1273-1282.
- Su H. (2008). The Studies of Active Iridoid Constituents in the Fruits of Lonicera Maackii (Rupr.) Maxim. Changchun: Jilin University.
- Xie LP. (2012). Effects of the Oleanolic Acid on Insulin and Liver Glycogen Store in Diabetic Rats. Chinese Archives of Traditional Chinese Medicine. 30(7): 1669-1670.