Information Links
Related Conferences
Current Issue Volume 11, Issue 1 - 2026
Prebiotic Supplementation with Partially Hydrolyzed Guar Gum and Inulin Improves Bowel Habits and Gut Functional Markers: A Case Series Study
Vania Mattoso Ribeiro¹, Ana Beatriz Baptistella², Silvana Paiva Orlandi³, Katherine Guerra4, Nathalia Laganaro4, Valeria Abrahao Schilling Rosenfeld4,*
1Federal University Fluminense, Brazil
2Independent Researcher, Brazil
3Postgraduate Program in Nutrition and Foods, University Hospital/EBSERH, Federal University of Pelotas, Brazil
4Nestle Health Science, Brazil
*Corresponding Author: Valeria Abrahao Schilling Rosenfeld, Nestle Health Science, São Paulo, Brazil, ORCID: 0000-0003-2372-9649, Email: [email protected]
Received Date: March 13, 2026
Published Date: March 30, 2026
Citation: Ribeiro VM, et al. (2026). Prebiotic Supplementation with Partially Hydrolyzed Guar Gum and Inulin Improves Bowel Habits and Gut Functional Markers: A Case Series Study. Mathews J Gastroenterol Hepatol. 11(1):41.
Copyrights: Ribeiro VM, et al. © (2026).
ABSTRACT
Prebiotic fibers play a crucial role in regulating intestinal motility and contribute to improvements in gastrointestinal symptoms associated with altered transit and fermentative activity. The aim of this exploratory case series was to evaluate the effects of a fiber blend containing inulin and partially hydrolyzed guar gum on bowel habits, gastrointestinal symptoms, and biochemical markers in adults reporting constipation and/or diarrhea. Two women received supplementation for 36 days, including a 6-dayramp-up phase until reaching 22g/day. Clinical symptoms, stool characteristics, biochemical markers, and functional stool parameters were assessed at baseline and post-intervention. Both cases showed improvements in bowel frequency and/or stool consistency, with progression on the Bristol Stool Form Scale. Reductions were observed in fecal calprotectin, zonulin, and fecal fat, suggesting improved intestinal inflammatory status and digestive function. Systemic metabolic and hepatic biomarkers remained stable, reinforcing the safety of the intervention. These findings support the potential benefits of prebiotic fiber supplementation for bowel function and gut-related biomarkers, warranting further studies with larger samples.
Keywords: Gut Health, Constipation, Soluble Fiber, Prebiotic Fiber Supplementation.
ABBREVIATIONS
WHO: World Health Organization; DF: Dietary Fiber; MIF: Formalin Embedding Medium; ESR: Erythrocyte Sedimentation Rate; SGOT: Glutamic Oxaloacetic Transaminase; SGPT: Glutamic Pyruvic Transaminase; GGT: Gamma-Glutamyl Transpeptidase; TSH: Thyroid-Stimulating Hormone; CRP: C-Reactive Protein; TMAO: Trimethylamine N-Oxide.
INTRODUCTION
The World Health Organization (WHO) defines dietary fiber (DF) as complex carbohydrates that are neither digested nor absorbed in the small intestine, reaching the large intestine intact. These components contribute to the regulation of digestion and bring several health benefits [1,2]. Epidemiological studies have consistently demonstrated the positive effects of dietary fiber on gastrointestinal health, especially from whole foods, which exert beneficial effects on the gastrointestinal tract and microbiota and have therapeutic potential in gastrointestinal disorders [3].
Dietary fibers, present mainly in foods of plant origin, are classified as soluble and insoluble. Soluble fibers have viscous properties and are fermented by bacteria in the colon, which contributes to adequate intestinal transit and reduces pH, in addition to generating by-products with beneficial local and systemic effects. These fibers are considered prebiotics because they serve as substrates for bacterial fermentation, promoting a favorable composition and metabolic activity of the intestinal microbiota [4,5]. Insoluble fibers, in turn, mainly perform the function of increasing the volume of the fecal bolus, facilitating elimination [6].
The gut microbiota is a complex ecosystem of bacteria, fungi, viruses, and protozoa that colonize the intestine. Fermentation produces byproducts, such as short-chain fatty acids, which benefit digestive, immune, and metabolic health. Diets rich in vegetables and dietary fiber promote a diverse and healthy microbiota, but prebiotic fiber supplementation may be necessary to achieve the optimal amount of DF, favoring immune and metabolic responses [7-9].
In addition, prebiotic fibers in the individual's diet, intestinal transit time is another factor that affects the composition and diversity of the microbiota [10]. Intestinal transit time depends on intestinal motility, which determines the characteristics of feces during the evacuation process and aims to eliminate undigested and unabsorbed food constituents, as well as cells and components of the microbiota. A bowel movement frequency between three times a week and three times a day is considered “normal”. Thus, a change in this pattern is recognized as a gastrointestinal complication and can be grouped into constipation, diarrhea, or irritable bowel syndrome [11].
Constipation is defined by reduced frequency of bowel movements, lumpy or hard stools, and a sensation of incomplete evacuation [12]. The prevalence ranges from 7% to 10%, and is higher in women [11,12]. The pathophysiology of this disorder is not yet fully understood, but it is multifactorial influenced by age, sex, medication use, comorbidities, and lifestyle factors, such as insufficient hydration, inadequate nutrition, and low physical activity [13,14]. Among the main recommendations for management increased in dietary fiber intake [6,13,15].
Like constipation, chronic diarrhea, characterized by increased bowel movement frequency and reduced stool consistency for more than four weeks, can occur for different reasons, such as medications and surgical procedures, structural changes in the intestine, secondary to diseases, irritable bowel syndrome, malabsorption, infections, and microbiota imbalances, among others [16,17]. The prevalence in adults ranges from 3% to 5%, and it is difficult to diagnose due to multiple primary or secondary causes [18].
Prebiotic fiber supplementation can significantly benefit health and intestinal microbiota, acting directly as a substrate and indirectly by improving intestinal function [6]. Among the prebiotics studied for this purpose are inulin and partially hydrolyzed guar gum.
Inulin, a fructan present in fruits and vegetables such as wheat, onion, banana, garlic, artichoke, chicory, and leek, favors the colonic environment by stimulating the growth of beneficial bacteria and improving intestinal function, helping to reduce cases of constipation [19,20]. Similarly, partially hydrolyzed guar gum, extracted from the seeds of Cyamopsis tetragonolobus, promotes the production of bacterial byproducts and also improves intestinal function. This supplement is safe and effective in optimizing metabolic and intestinal parameters, benefiting both cases of constipation and diarrhea [21,22].
Therefore, given the evidence on the efficacy of prebiotics, this study aims to evaluate the effects of supplementation with a fiber mixture containing guar gum and inulin on intestinal motility in individuals with complaints of constipation and/or diarrhea. Improvements in gastrointestinal symptoms will be analyzed, along with markers of intestinal health and biochemistry, including inflammation, liver function, thyroid function, and nutrient levels.
MATERIALS & METHODS
Study design and participants
This is a series of two cases in which the effects of supplementation with a fiber mix (Fiber Mais® Suco Verde) on the intestinal motility of women with a history of constipation were evaluated. After selection participants underwent a Merthiolate–Iodine–Formalin (MIF) parasitological exam to rule out intestinal parasites, a condition that can directly interfere with the protocol´s application.
Application of the protocol
The supplementation protocol lasted 36 days. During the first six days, as an adaptation phase, the participants followed a ramp protocol for supplement consumption until reaching the desired dose. Thus, during the first two days, they consumed 5.5g of the fiber supplement, doubling the dose every two days until reaching the final dose of 22g per day. After the 6 days of adaptation, the participants consumed 22g per day for 30 days. During the supplementation period, the patients maintained their usual eating pattern, without receiving any intervention and/or dietary guidance.
Assessment of symptoms, biochemical tests, and intestinal microbiota composition
At the beginning and end of the period (T0 and T30), participants were assessed for the occurrence of gastrointestinal symptoms using the Gastrointestinal Symptom Rating Scale, which contains questions about the occurrence of gastrointestinal symptoms in the last 7 days [23] and also regarding the characteristics of the feces according to the Bristol Scale – a validated scale that contains photos of seven types of feces with precise descriptions of their appearance and consistency [24,25].
In order to detect possible changes in biochemical parameters related to inflammation, at the beginning and end of the study (T0-T30), analyses of the following biochemical parameters were performed: complete blood count (including erythrocyte sedimentation rate – ESR), glycemic profile (fasting blood glucose, fasting insulin + HOMA-IR and glycated hemoglobin), lipid profile (total cholesterol and fractions, triacylglycerols), liver profile (glutamic oxaloacetic transaminase – SGOT, glutamic pyruvic transaminase (SGPT), gamma-glutamyl transpeptidase (GGT), ferritin, homocysteine, uric acid, amylase, lipase, thyroid-stimulating hormone (TSH), triiodothyronine (T3), thyroxine (T4), cobalamin (B12), folic acid (B9), zinc (Zn), 25-hydroxyvitamin D (vitamin D), magnesium (Mg), protein C-reactive protein (CRP), fibrinogen and trimethylamine N-oxide (TMAO). To alleviate changes in stool pattern, digestive function, and intestinal health, the functional stool test was performed at T0 and T30.
RESULTS
Case 1 – Functional Constipation with Mild Inflammation and Good Clinical Response
A 32-year-old woman (BMI 23.7) with functional constipation reported irregular bowel movements and Bristol type 2 stools. After 30 days of fiber supplementation, she achieved daily bowel movements and improved stool form to Bristol type 3, accompanied by a reduction in abdominal distension and gas.
Stool analysis demonstrated a notable reduction in calprotectin (from 87 to <25 mcg/g) and zonulin (from 130 to 69 ng/mL), indicating reduced intestinal inflammation and improved gut barrier function. Fecal fat decreased from 20% to 10%, suggesting better lipid absorption. A marked increase in fecal histamine occurred without clinical symptoms, and a low elastase value likely reflected sampling variability rather than clinical insufficiency.
All hepatic, glycemic, thyroid, and inflammatory markers (CRP, ESR, liver enzymes, glucose, insulin, homocysteine, TSH) remained within normal ranges throughout the intervention. TMAO decreased from 2.9 to 1.8 µM, suggesting possible modulation of the microbiota and reduced cardiometabolic risk.
Pancreatic elastase decreased from 174 to 25 mcg/g, a value consistent with exocrine insufficiency phenotype; however, no gastrointestinal symptoms were observed to corroborate this finding, suggesting possible analytical variability or pre-analytical error, although functional alteration cannot be ruled out.
Case 2 – Lifelong Constipation with Significant Gastrointestinal Improvement
A 37-year-old woman (BMI: 19.4 kg/m²) with long-standing constipation, infrequent bowel movements, and Bristol type 2 stool form. After supplementation, she reported increased bowel frequency to every other day, although stool form remained unchanged. She also described less effort to evacuate, reduced gas, and improved well-being.
Laboratory analysis revealed a substantial decrease in calprotectin (from 368 to 32 mcg/g), fecal fat (from >30% to 5%), and histamine (from high to 99 ng/g). These findings are consistent with reduced intestinal inflammation and enhanced digestive capacity. Zonulin remained stable (125 to 122 ng/mL), and pancreatic elastase increased from 398 to 636 mcg/g. TMAO rose from 1.6 to 4.8 µM but remained within the expected range.
All other laboratory parameters, including liver enzymes, glycemia, insulin, CRP, ESR, and thyroid markers, were within normal ranges before and after the intervention.
Both cases demonstrated improved bowel patterns and favorable changes in functional stool markers, with stable metabolic, hepatic, and endocrine profiles, supporting the intervention's safety.
Table 1. Evolution of clinical, laboratory and coprological parameters after fiber supplementation (22g/day for 30 days)
|
CASE 1 |
CASE 2 |
|||
|
Parameter |
Pre |
Post |
Pre |
Post |
|
Bristol Scale |
2 |
3 ↑ |
2 |
2 |
|
Evacuation Frequency |
Irregular |
Daily |
Constipation |
Alternate days |
|
Calprotectin (mcg/g) |
87 |
<25 ↓ |
368 |
32 ↓ |
|
Zonulin (ng/mL) |
130 |
69 ↓ |
125 |
122 ↓ |
|
Pancreatic elastase (mcg/g) |
174 |
25 ↓ |
398 |
636 ↑ |
|
Fecal fat (%) |
20 |
10 ↓ |
>30 |
5 ↓ |
|
Fecal histamine (ng/g) |
234 |
4437 ↑ |
- |
99 |
|
TMAO (µM) |
2.9 |
1.8 ↓ |
1.6 |
4.8 ↑ |
|
ESR (mm) |
5 |
8 ↑ |
9 |
7 ↓ |
|
CRP (mg/dL) |
0.03 |
0.03 → |
0.03 |
0.03 → |
|
Fasting glucose (mg/dL) |
84 |
82 ↓ |
87 |
86 ↓ |
|
Fasting insulin (µU/mL) |
6 |
4.1 ↓ |
9.8 |
7 ↓ |
|
↑ Increase | ↓ Decrease | → No change. TMAO: Trimethylamine N-Oxide. ESR: Erythrocyte Sedimentation Rate. CRP: C-Reactive Protein |
||||
DISCUSSION
The human gut hosts trillions of bacteria, primarily from the Firmicutes and Bacteroidetes phyla, which interact with intestinal epithelial cells to exert metabolic, structural, and protective functions [26]. A balanced gut microbiota plays a fundamental role in modulating intestinal motility, immune function, and overall health [7,27].
In the present study, daily supplementation with 22 g of a prebiotic fiber mix (partially hydrolyzed guar gum and inulin) for 30 days was associated with improvements in bowel habits and gastrointestinal symptoms in both cases. Notably, participants achieved daily or alternate-day evacuation patterns, accompanied by improved stool consistency on the Bristol scale—features consistent with enhanced intestinal motility.
These clinical benefits are supported by the fermentative action of the fibers used: guar gum and inulin are prebiotic substrates that are metabolized by colonic bacteria, leading to the production of short-chain fatty acid (SCFAs), increased peristalsis, and pH modulation [4-6,28]. This mechanism aligns with the literature describing improved colonic transit and stool quality in constipated patients treated with these fibers [21,29].
Beyond symptom relief, the cases showed biochemical improvements, particularly reductions in calprotectin, zonulin, and fecal fat, markers indicative of lower intestinal inflammation and improved barrier function [30-32]. These results suggest a potential role of fiber supplementation in the recovery of gut mucosal integrity, possibly mitigating features of leaky gut syndrome, which has been implicated in functional constipation [33].
Pancreatic elastase levels increased in Case 2, suggesting an enhancement in exocrine pancreatic function. This aligns with studies linking microbiota modulation to improved digestion and enzyme secretion [34].
The increase in TMAO in the Case 2 participant, although remaining within normal limits, highlights the complex interplay between microbial metabolism and host responses [10,35]. TMAO is a metabolite derived from microbial metabolism of dietary substrates and has been associated with cardiovascular risk, although its short-term fluctuations lack clinical relevance outside dietary context [9].
Systemic metabolic markers inlcuding glycemia, liver enzymes, TSH, and CRP remained within normal ranges for all participants, reinforcing the safety and local effects of the intervention on intestinal function.
From a functional perspective, this case series corroborates existing literature on the use of soluble prebiotic fibers as a safe and effective strategy for managing chronic constipation and restoring gut function [6,13,15,36-39].
LIMITATIONS
The small sample size (n=2) and short duration (30 days at full dose) limit generalizability and statistical inference. There was no dietary standardization, which may influence stool and microbiota outcomes. Stool analyses reflect functional status but are subject to biological variability. Nonetheless, these findings offer valuable insights for hypothesis generation and future clinical trials.
CONCLUSION
This exploratory case series demonstrated that daily supplementation with a prebiotic fiber blend improved bowel habits and functional stool markers in adults with chronic constipation. The intervention was safe and well-tolerated, with stability in biochemical and metabolic markers. Larger controlled trials are warranted to validate these findings and elucidate underlying mechanisms.
AUTHOR’S CONTRIBUTION
V.A.S.R. and N.L. designed the study. V.M.R. and K.G. collected the data; A.B.B. wrote the paper and all authors critically reviewed the final manuscript.
ACKNOWLEDGEMENTS AND FUNDING
Nestle Health Science provided the supplements.
V.A.S.R. and N.L. are employees of Nestlé Health Science. The other authors have declared no conflicts of interest.
REFERENCES
- Food and Agriculture Organization. (2010). World Health Organization Codex Alimentarius Commission. Codex Alimentarius: Guidelines on Nutrition Labelling CAC/GL 2-1985.
- Zielinski G, DeVries JW, Craig SA, Bridges AR. (2013). Dietary fiber methods in Codex Alimentarius: current status and ongoing discussions. Cereal Food World. 58(3):148-152.
- Gill SK, Rossi M, Bajka B, Whelan K. (2021). Dietary fibre in gastrointestinal health and disease. Nat Rev Gastroenterol Hepatol. 18(2):101-116.
- Roberfroid M, Gibson GR, Hoyles L, McCartney AL, Rastall R, Rowland I. (2010). Prebiotic effects: metabolic and health benefits. British Journal of Nutrition, [S.L.]. 104(s2):1-63. Cambridge University Press (CUP). DOI: 10.1017/s0007114510003363.
- Lee S, You H, Lee Y, Baik H, Paik J, Lee H, et al. (2021). Intake of MPRO3 over 4 Weeks Reduces Glucose Levels and Improves Gastrointestinal Health and Metabolism. Microorganisms. 10(1):88.
- Bernaud FS, Rodrigues TC. (2013). Fibra alimentar--ingestão adequada e efeitos sobre a saúde do metabolismo [Dietary fiber--adequate intake and effects on metabolism health]. Arq Bras Endocrinol Metabol. 57(6):397-405.
- Jandhyala SM, Talukdar R, Subramanyam C, Vuyyuru H, Sasikala M, Nageshwar Reddy D. (2015). Role of the normal gut microbiota. World J Gastroenterol. 21(29):8787-803.
- Rinninella E, Raoul P, Cintoni M, Franceschi F, Miggiano GAD, Gasbarrini A, et al. (2019). What is the Healthy Gut Microbiota Composition? A Changing Ecosystem across Age, Environment, Diet, and Diseases. Microorganisms. 7(1):14.
- da Silva TF, Casarotti SN, de Oliveira GLV, Penna ALB. (2021). The impact of probiotics, prebiotics, and synbiotics on the biochemical, clinical, and immunological markers, as well as on the gut microbiota of obese hosts. Crit Rev Food Sci Nutr. 61(2):337-355.
- Vandeputte D, Falony G, Vieira-Silva S, Tito RY, Joossens M, Raes J. (2016). Stool consistency is strongly associated with gut microbiota richness and composition, enterotypes and bacterial growth rates. Gut. 65(1):57-62.
- Rollet M, Bohn T, Vahid F, On Behalf Of The Oriscav Working Group. (2021). Association between Dietary Factors and Constipation in Adults Living in Luxembourg and Taking Part in the ORISCAV-LUX 2 Survey. Nutrients. 14(1):122.
- Li Y, Tong WD, Qian Y. (2021). Effect of Physical Activity on the Association Between Dietary Fiber and Constipation: Evidence From the National Health and Nutrition Examination Survey 2005-2010. J Neurogastroenterol Motil. 27(1):97-107.
- Duncan PI, Enters-Weijnen CF, Emami N, McLean P, Nunes T, Beaumont M, et al. (2018). Short-Term Daily Intake of Polydextrose Fiber Does Not Shorten Intestinal Transit Time in Constipated Adults: A Randomized Controlled Trial. Nutrients. 10(7):920.
- Yurtdaş G, Acar-Tek N, Akbulut G, Cemali Ö, Arslan N, Beyaz Coşkun A, et al. (2020). Risk Factors for Constipation in Adults: A Cross-Sectional Study. J Am Coll Nutr. 39(8):713-719.
- Bharucha AE, Pemberton JH, Locke GR. (2013). American Gastroenterological Association technical review on constipation. Gastroenterology. 144(1):218-238.
- Arasaradnam RP, Brown S, Forbes A, Fox MR, Hungin P, Kelman L, et al. (2018). Guidelines for the investigation of chronic diarrhoea in adults: British Society of Gastroenterology, 3rd edition. Gut. 67(8):1380-1399.
- Schiller LR, Pardi DS, Sellin JH. (2017). Chronic Diarrhea: Diagnosis and Management. Clin Gastroenterol Hepatol. 15(2):182-193.e3.
- Gómez-Escudero O, Remes-Troche JM. (2021). Approach to the adult patient with chronic diarrhea: A literature review. Rev Gastroenterol Mex (Engl Ed). 86(4):387-402.
- Amadieu C, Coste V, Neyrinck AM, Thijssen V, Leyrolle Q, Bindels LB, et al. (2022). Restoring an adequate dietary fiber intake by inulin supplementation: a pilot study showing an impact on gut microbiota and sociability in alcohol use disorder patients. Gut Microbes. 14(1):2007042.
- Reimer RA, Soto-Vaca A, Nicolucci AC, Mayengbam S, Park H, Madsen KL, et al. (2020). Effect of chicory inulin-type fructan-containing snack bars on the human gut microbiota in low dietary fiber consumers in a randomized crossover trial. Am J Clin Nutr. 111(6):1286-1296.
- Chan TC, Yu VMW, Luk JKH, Chu LW, Yuen JKY, Chan FHW. (2022). Effectiveness of Partially Hydrolyzed Guar Gum in Reducing Constipation in Long Term Care Facility Residents: A Randomized Single-Blinded Placebo-Controlled Trial. J Nutr Health Aging. 26(3):247-251.
- Yasukawa Z, Inoue R, Ozeki M, Okubo T, Takagi T, Honda A, et al. (2019). Effect of Repeated Consumption of Partially Hydrolyzed Guar Gum on Fecal Characteristics and Gut Microbiota: A Randomized, Double-Blind, Placebo-Controlled, and Parallel-Group Clinical Trial. Nutrients. 11(9):2170.
- Souza GS, Sarda FA, Giuntini EB, Gumbrevicius I, Morais MB, Menezes EW. (2016). Translation and validation of the brazilian portuguese version of the Gastrointestinal Symptom Rating Scale (GSRS) questionnaire. Arq Gastroenterol. 53(3):146-151.
- Martinez AP, Azevedo GR. (2012). The Bristol Stool Form Scale: its translation to Portuguese, cultural adaptation and validation [Tradução, adaptação cultural e validação da Bristol Stool Form Scale para a população brasileira]. Rev Lat Am Enfermagem. 20(3):583-589.
- Lewis SJ, Heaton KW. (1997). Stool form scale as a useful guide to intestinal transit time. Scand J Gastroenterol. 32(9):920-924.
- Ahlawat S, Asha, Sharma KK. (2021). Gut-organ axis: a microbial outreach and networking. Lett Appl Microbiol. 72(6):636-668.
- Mayer EA, Savidge T, Shulman RJ. (2014). Brain-gut microbiome interactions and functional bowel disorders. Gastroenterology. 146(6):1500-1512.
- Rao SSC, Brenner DM. (2021). Efficacy and Safety of Over-the-Counter Therapies for Chronic Constipation: An Updated Systematic Review. Am J Gastroenterol. 116(6):1156-1181.
- Polymeros D, Beintaris I, Gaglia A, Karamanolis G, Papanikolaou IS, Dimitriadis G, Triantafyllou K. (2014). Partially hydrolyzed guar gum accelerates colonic transit time and improves symptoms in adults with chronic constipation. Dig Dis Sci. 59(9):2207-2214.
- Khalif IL, Quigley EM, Konovitch EA, Maximova ID. (2005). Alterations in the colonic flora and intestinal permeability and evidence of immune activation in chronic constipation. Dig Liver Dis. 37(11):838-849.
- Chae YR, Lee YR, Kim YS, Park HY. (2024). Diet-Induced Gut Dysbiosis and Leaky Gut Syndrome. J Microbiol Biotechnol. 34(4):747-756.
- González R, Ceacero-Heras D, Tena-Garitaonaindia M, Álvarez-Mercado A, Gámez-Belmonte R, Chazin WJ, et al. (2024). Intestinal inflammation marker calprotectin regulates epithelial intestinal zinc metabolism and proliferation in mouse jejunal organoids. Biomed Pharmacother. 174:116555.
- Fasano A, Not T, Wang W, Uzzau S, Berti I, Tommasini A, et al. (2000). Zonulin, a newly discovered modulator of intestinal permeability, and its expression in coeliac disease. Lancet. 355(9214):1518-1519.
- Löhr JM, Dominguez-Munoz E, Rosendahl J, Besselink M, Mayerle J, Lerch MM, et al. (20170. United European Gastroenterology evidence-based guidelines for the diagnosis and therapy of chronic pancreatitis (HaPanEU). United European Gastroenterol J. 5(2):153-199.
- Kaur K, Zubair M, Adamski JJ. (2024). Fecal Occult Blood Test. In: StatPearls [Internet]. Treasure Island (FL), USA: StatPearls Publishing.
- Sun D, Lo KM, Chen SC, Leung WW, Wong C, Mak T, et al. (2024). Consistency of Feces Affects Defecatory Function. J Neurogastroenterol Motil. 30(3):373-378.
- Teige ES, Hillestad EMR, Steinsvik EK, Brønstad I, Lundervold A, Lundervold AJ, et al. (2024). Fecal bacteria and short-chain fatty acids in irritable bowel syndrome: Relations to subtype. Neurogastroenterol Motil. 36(9):e14854.
- Wu L, Wu H, Huang F, Li XY, Zhen YH, Zhang BF, et al. (2023). Causal association between constipation and risk of colorectal cancer: a bidirectional two-sample Mendelian randomization study. Front Oncol. 13:1282066.
- Mitrou P. (2022). Is lifestyle Modification the Key to Counter Chronic Diseases? Nutrients. 14(15):3007.
SUPPLEMENTARY MATERIAL
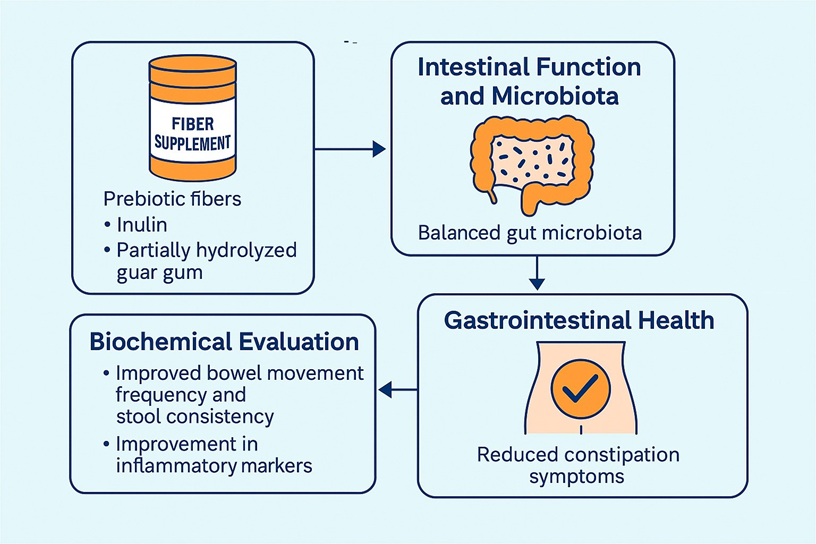
Figure. Fiber Supplementation and Gut Outcomes (graphical summary).