Information Links
Related Conferences
Previous Issues Volume 4, Issue 1 - 2020
Metarteriole and Mesenchyme - Cellular Angiofibroma
Anubha Bajaj
Corresponding Author: Anubha Bajaj, Consultant Histopathologist, A.B. Diagnostics, Delhi, India, E-mail: [email protected] Received Date: September 11, 2020 Published Date: October 12, 2020 Copyright ©: Anubha B. ©2020. Citation: Anubha B. (2020). Metarteriole and Mesenchyme - Cellular Angiofibroma. Mathews J Cytol Histol. 4(1): 15. ABSTRACT Cellular angiofibroma is a contemporary, benign, mesenchymal neoplasm comprised of dual cellular components of spindle-shaped cells and prominent, stromal blood vessels. The neoplasm is commonly delineated in middle aged adults of 40 years to 60 years and typically incriminates subcutaneous tissue of vulvovaginal region in females and inguinoscrotal region in males. An asymptomatic, painless or painful, firm, gradually progressive soft tissue mass is usually discerned. Minimally to moderately cellular neoplasm displays an abundant myxoid stroma intermixed with tumour cells fascicles. Characteristically, bundles of bland spindle- shaped cells encompassing well developed vascular articulations and an intervening collagenous or myxoidstroma with numerous, miniature to medium sized blood vessels with perivascular hyaline fibrosis is exemplified. Tumour cells are immune reactive to vimentin, CD34, CD99, CD31, smooth muscle actin (SMA), oestrogen receptors (ER), progesterone receptors (PR) and are immune non reactive to S100 protein, desmin, epithelial membrane antigen (EMA), caldesmon and cytokeratin. The neoplasm can be subjected to surgical resection. KEYWORDS: Mesenchymal; Fibroblastic; Vascular; Stromal INTRODUCTION Cellular angiofibroma is a distinctive, exceptional, contemporary, benign, mesenchymal neoplasm comprised of dual cellular components of spindle-shaped cells and prominent, stromal blood vessels. The tumour is additionally nomenclated as angiomyofibroblastoma-like tumour (AMF). Of obscure genesis, the benign neoplasm is histologically classified as an angiomyxoid tumour. The neoplasm was initially scripted by Nucci MR, et al. in 1997 and subsequently elaborated upon by Laskin WB, et al. in 1998. An accurate preoperative diagnosis is crucial although the tumefaction can be challenging to ascertain upon cogent clinical symptoms or imaging features, thus appropriate pathological and immune histochemical concordance is paramount [1,2]. DISEASE CHARACTERISTICS The neoplasm is commonly delineated in middle aged adults of 40 years to 60 years although age of disease emergence extends up to eighth decade. Typically, subcutaneous tissue of genital areas such as vulvovaginal region in females and inguinoscrotal region in males is incriminated. Vulvar lesions can be misinterpreted as a Bartholin gland cyst. Commonly, inguinal region is incriminated in males followed by scrotum and perineum. Tumefaction can additionally be discerned within the head and neck, nasopharynx, oral mucosa, male pelvic tissue, retroperitoneum, perineum, subcutaneous tissue of thoracic cavity or superficial soft tissue of trunk. A specific gender predilection is absent [3,4]. Characteristically, proliferation of spindle-shaped cells intermingled with miniature to medium- sized, hyalinised blood vessels is discerned. Intra-tumoural adipose tissue is discerned within 24 to 65% of neoplasms. Pertinent genomic deletion of RB1 gene at chromosome region 13q14 is documented, thus indicating a concurrence between spindle cell lipoma, mammary- type myofibroblastoma and atypical spindle cell lipomatous tumour. Cellular angiofibroma genetically simulates spindle cell lipoma as the neoplasms depict aberrant karyotypes with heterozygous deletion of long arm of chromosome 13. Although genesis of tumour is unexplained, it possibly is derived from perivascular stem cells endowed with a potential to differentiate into adipose and myo-fibroblastic tissue [3,4]. CLINICAL ELUCIDATION A well circumscribed lesion of enhanced vascularity with a distinct adipose tissue component in indicative of cellular angiofibroma. However, macroscopic foci of adipose tissue are absent. The neoplasm manifests as an asymptomatic, painless or painful, firm, gradually progressive soft tissue mass situated within the subcutaneous tissue with a superimposed cutaneous epithelial layer. Nasopharyngeal tumours manifest frequent epistaxis or persistent nasal obstruction. Retroperitoneal tumours delineate pain within the iliac fossa. Morphological variations are contingent to anatomic location of the neoplasm. Extra-genital neoplasms demonstrate morphological features simulating genital neoplasms [3,4]. Neoplasms arising in female subjects can arise from hormone- receptor containing mesenchymal cells situated within lower genital tract [4]. HISTOLOGICAL ELUCIDATION Grossly, a smooth, pale, mobile, firm to rubbery, elliptical tumefaction encompassed within a pale-pink, smooth fibrous tissue capsule is enunciated. The neoplasm can be well circumscribed, un-encapsulated, soft and miniature, usually below <3 centimetres magnitude. Variable pseudo-capsule is observed. Cut surface is greyish-white or pink [5]. Tumefaction discerned in males is enlarged, can extend up to 14 centimetres in magnitude and display an ill defined tumour perimeter. Focal areas of tumour extension into circumscribing soft tissue can ensue [5]. The neoplasm is minimally to moderately cellular with abundant myxoid stroma and configures fascicles or a haphazard tumour pattern. Characteristically, bland spindle- shaped cells encompassing well developed vascular articulations are observed. Mitosis is exceptional. Intervening stroma is collagenous or myxoid with numerous, miniature to medium sized blood vessels with perivascular hyaline fibrosis. Uniform, spindle- shaped cells with scant, faintly eosinophilic cytoplasm, elliptical to fusiform nuclei and indistinct cytoplasmic boundaries are discerned. An epithelioid cell component can be exhibited. Tumour cells are intermingled with numerous miniature to medium-sized vascular configurations with hyalinization and fibrosis of vessel wall [4,5]. Vascular configurations depict degenerative alterations, fibrin thrombi, intramural inflammation and deposits of hemosiderin pigment. Encompassing stroma is myxoid or collagen-rich with fine fibrils. Spindle-shaped cells are diffusely disseminated within a loosely configured, extracellular, fibrous interstitial tissue with scattered vascular articulations [5]. As the cellular neoplasm is composed of uniform, bland, miniature spindle-shaped cells configuring short fascicles enmeshed with wispy collagen fibres and an abundance of miniature or medium- sized, spherical blood vessels with intramural hyalinization, several vascular articulations can display thick, hyalinised vessel walls [4,5]. Lesions appearing in females composed of spindle-shaped cells with uniform, oval to fusiform nuclei, pale eosinophilic cytoplasm and indistinct cellular boundaries can display a brisk mitotic activity. Prominent mitotic figures up to 11/ 10 high power fields can be discerned although atypical mitosis is absent. Although infrequent, focal areas of significant cellular atypia and sarcomatous transformation are documented [6]. Tumour periphery can depict lobules of mature adipocytes. Stromal mast cells are frequent and occasional, miniature aggregates of mature lymphocytes can be observed. Lymphocytic infiltration with formation of lymphoid follicles can ensue. An estimated 50% of neoplasms depict a minimal component of mature adipose tissue (<5%). Areas of haemorrhage or tumour necrosis are absent [5,6].
.gif) Figure 1: Cellular angiofibroma demonstrating uniform spindle-shaped cells commingled within a collagenous stroma and miniature, hyalinised vascular articulations [Source: BMC Clinical Pathology].
Figure 1: Cellular angiofibroma demonstrating uniform spindle-shaped cells commingled within a collagenous stroma and miniature, hyalinised vascular articulations [Source: BMC Clinical Pathology].
.jpg) Figure 2: Cellular angiofibroma enunciating uniform spindle-shaped cells intermixed within a collagen-rich stroma and several, medium sized, hyalinised vascular configurations [Source: Semantic Scholar].
Figure 2: Cellular angiofibroma enunciating uniform spindle-shaped cells intermixed within a collagen-rich stroma and several, medium sized, hyalinised vascular configurations [Source: Semantic Scholar].
.jpg) Figure 3: Cellular angiofibroma with uniform, spindle-shaped cells encompassed in a collagenous stroma and miniature vascular articulations with peripheral hyalinisation [Source: Radiopedia].
Figure 3: Cellular angiofibroma with uniform, spindle-shaped cells encompassed in a collagenous stroma and miniature vascular articulations with peripheral hyalinisation [Source: Radiopedia].
.jpg) Figure 4: Cellular angiofibroma exemplifying bland, spindle-shaped cells with several, patent, medium sized vascular arrangements and an encompassing collagenous tomyxoidstroma [Source: Histopathology.india.net].
Figure 4: Cellular angiofibroma exemplifying bland, spindle-shaped cells with several, patent, medium sized vascular arrangements and an encompassing collagenous tomyxoidstroma [Source: Histopathology.india.net].
.jpg) Figure 5: Cellular angiofibroma enunciating bland spindle-shaped cells with commingled collagenous stroma and hyalinised, miniature vascular articulations.
Figure 5: Cellular angiofibroma enunciating bland spindle-shaped cells with commingled collagenous stroma and hyalinised, miniature vascular articulations.
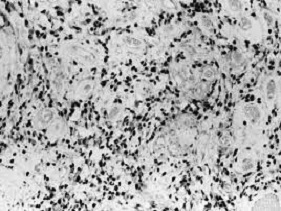 Figure 6: Cellular angiofibroma exhibiting abundant, spindle-shaped cells intermixed within a collagenous stroma and numerous, hyalinised, medium sized vascular configurations [Source: Gynaecologic oncology].
Figure 6: Cellular angiofibroma exhibiting abundant, spindle-shaped cells intermixed within a collagenous stroma and numerous, hyalinised, medium sized vascular configurations [Source: Gynaecologic oncology].
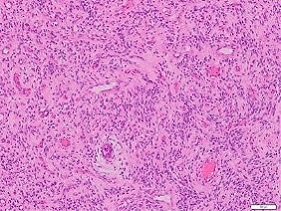 Figure 7: Cellular angiofibroma delineating abundant spindle-shaped cells within a collagen-rich stroma and several medium sized blood vessels with peripheral hyalinisation [Source: Pathology Outlines].
Figure 7: Cellular angiofibroma delineating abundant spindle-shaped cells within a collagen-rich stroma and several medium sized blood vessels with peripheral hyalinisation [Source: Pathology Outlines].
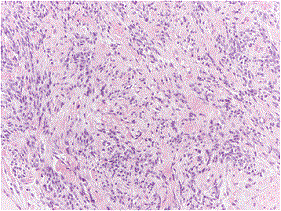 Figure 8: Cellular angiofibroma depicting bland spindle-shaped cells entangled in a collagenous stroma with numerous miniature, hyalinised blood vessels [Source: Diagnostic Pathology].
Figure 8: Cellular angiofibroma depicting bland spindle-shaped cells entangled in a collagenous stroma with numerous miniature, hyalinised blood vessels [Source: Diagnostic Pathology].
IMMUNE HISTOCHEMICAL ELUCIDATION Tumour cells are immune reactive to vimentin, CD34 (30 to 60%), CD99, CD31, smooth muscle actin (SMA-21%), oestrogen receptors (ER) and progesterone receptors (PR). Tumour cells are immune non reactive to S100 protein, desmin, epithelial membrane antigen (EMA), caldesmon and cytokeratin although focal immune reactivity to desmincan be discerned. Ki-67 proliferation index is below< 2 % within the cell cycle [3,4]. Differential Diagnosis Cellular angiofibroma requires histological separation from vessel- rich neoplasms of myofibroblastic or fibroblastic origin such as solitary fibrous tumour (SFT), spindle cell lipoma, angiomyofibroblastoma (AMF) and superficial angiomyxoma and aggressive angiomyxoma [6,7]. • Superficial angiomyxoma manifests singular tumour nodules which are minimally to moderately cellular with enveloping, copious, basophilic, interstitial substance. The tumour is constituted by scattered, spindle-shaped and stellate cells intermixed with a sparse vascular network. • Aggressive angiomyxoma is an enlarged, hypo-cellular neoplasm situated within deep-seated soft tissue with an infiltrative tumour perimeter and abundant myxoid matrix. The neoplasm is immune reactive to desmin [5,6]. • Angiomyofibroblastoma is a well circumscribed, subcutaneous neoplasm commonly discerned in the vulvovaginal region. The tumour is composed of epithelioid and spindle-shaped mesenchymal cells configuring cords. Tumour cells preferentially nest around several attenuated, miniature to medium- sized blood vessels with ectasia. Angiomyofibroblastoma is a minimally cellular lesion composed of spherical, myoid tumour cells immune reactive to desmin [6,7]. • Spindle cell lipoma is common in males, is situated within the neck or upper back and demonstrates ropey, refractive fascicles of collagen with a lack of innumerable or hyalinised blood vessels. Spindle cell lipoma is a neoplasm which consists of miniature, cylindrical, spindle-shaped cells, mature adipocytes admixed with collagen fibres and a matrix composed of mucinous substances. The neoplasm is immune reactive to CD34, Vimentin and B cell lymphoma 2 (Bcl-2) although S100 protein is immune non reactive [7,8]. • Perineurioma depicts a whorled, cellular pattern, an absence of thick walled blood vessels and is immune reactive to epithelial membrane antigen (EMA) [7]. • Leiomyoma is a neoplasm composed of spindle-shaped cells. Tumour cells are immune reactive to actin and desmin. Subcategories of leiomyomas such as pilo-leiomyoma, angio-leiomyoma, lipo-leiomyoma or genital leiomyoma can delineate features of smooth muscle differentiation such as intersecting fascicles of spindle-shaped cells, blunt- ended nuclei and eosinophilic cytoplasm. Tumours are immune reactive to diverse myogenic markers [7,8]. • Leiomyosarcoma wherein a cellular angiofibroma can be misinterpreted as a sarcoma on account of brisk mitotic activity. • Solitary fibrous tumour is discovered incidentally, with imaging or can initiate localized tumour compression upon abutting anatomic structures. Computerized tomography (CT) and magnetic resonance imaging (MRI) demonstrate homogenously enhanced tumefaction with definitive tumour margin [7,8]. Solitary fibrous tumour demonstrates intervening hyper-cellular and hypo-cellular zones, areas of prominent, staghorn-shaped blood vessels and encompassing hyalinised collagen. Microscopically, the neoplasm denominates a pattern- less, haphazard, spindle-shaped cellular articulation admixed with amorphous collagenous areas and lack of prominent endothelium. Solitary fibrous tumour displays a haemangiopericytoma- like vascular pattern with stromal hyalinization akin to a keloid. Tumour cells are immune reactive to CD34, CD99 and B cell lymphoma 2 (Bcl-2) [7,8]. Tumefaction such as fibro-epithelial stromal polyp, well differentiated liposarcoma, myxoidliposarcoma, glomangiopericytoma, pleomorphic hyalinising angiectatic tumour (PHAT) and mammary type myofibroblastoma also require segregation [8]. Investigative Assay Typically, cellular angiofibroma delineates diverse pathological and imaging manifestations. Various haematological and serum biomarkers are inconclusive. Ultrasonography of tumour site depicts an enlarged, heterogeneous, soft tissue mass. Colour Doppler delineates a predominantly vascular lesion [9]. Magnetic resonance imaging (MRI) of incriminated soft tissue is a valuable adjunctive modality of tumour assessment. The technique provides adequate structural and functional information and can be employed for cogent preoperative characterization of tumour histology. Conventional magnetic resonance imaging (MRI) is reliably adopted for segregating diverse intra-visceral and extra-visceral soft tissue masses. Adequate preoperative categorization of histological elements of diverse lesions abutting organs and soft tissue, contingent to morphological data and tissue signal intensity can be achieved. However, imaging features can be non specific [8,9]. Magnetic resonance imaging (MRI) exhibits a nodule abutting and displacing adjacent soft tissue. The neoplasm is in-homogenous and enunciates signal intensity identical or slightly enhanced to adjacent normal viscera upon T1 weighted and T2 weighted images. Restricted diffusion on transverse diffusion weighted (DW) images is absent. Aforesaid features are indicative of a benign vascular tumour. Imaging features of cellular angiofibroma is concurrent with tumour histology and is contingent to quantifiable spindle-shaped cells and adipose tissue component with myxoid and collagenous stroma. On MRI, tumour is sharply defined with a heterogeneous, enhanced signal intensity upon T2 weighted images and signal intensity akin to skeletal muscle upon T1 weighted images. Heterogeneous image enhancement is observed following administration of gadolinium contrast. An abundant, myxoid stroma and innumerable intra- tumoural blood vessels engender hyper-intensity of lesion upon T2 weighted images and patterns of contrast enhancement, respectively [8,9]. Discernment of tumefaction and lesion characterization upon diffusion weighted (DW) images pertains to tissue cellularity wherein enhanced cellularity with restricted diffusion and declining apparent diffusion coefficient accompanies malignant neoplasms. However, cellular angiofibroma is associated with intense heterogeneous enhancement of the tumour with subsequent plateau within the delayed phase, thereby enunciating a type II curve, which concurs with a benign neoplasm. Fluorescent in situ hybridization (FISH) demonstrates a mono-allelic deletion of RB1 gene [9]. Therapeutic Options Benign cellular angiofibroma without atypia can be adequately alleviated with localized surgical extermination along with broad, excised, tumour free perimeter. Recommended therapeutic strategy is comprehensive surgical extermination of the neoplasm. Extended follow up is required as localized tumour reoccurrence is documented. However, tumour reoccurrence is minimal and the neoplasm is devoid of distant metastasis [8,9]. CONCLUSION Benign cellular angiofibroma is constituted by dual cellular components of spindle-shaped cells and predominant, stromal blood vessels. The neoplasm requires histological segregation from myofibroblastic or fibroblastic neoplasms such as solitary fibrous tumour (SFT), spindle cell lipoma, angiomyofibroblastoma (AMF), superficial angiomyxoma, aggressive angiomyxoma, perineurioma, leiomyoma or leiomyosarcoma. Also, fibro-epithelial stromal polyp, well differentiated liposarcoma, myxoidliposarcoma, glomangiopericytoma, pleomorphic hyalinising angiectatic tumour (PHAT) and mammary type myofibroblastoma require a demarcation. Ultrasonography, colour Doppler and magnetic resonance imaging (MRI) can be adopted for segregating intra-visceral and extra-visceral soft tissue neoplasms. Cellular angiofibroma can be adequately alleviated with localized surgical extermination along with excision of a broad, tumour free perimeter. REFERENCES 1. Nucci MR, Granter SR, Fletcher CD. (1997). Cellular angiofibroma- a benign neoplasm distinct from angiomyofibroblastoma and spindle cell lipoma. Am J Surg Pathol. 21(6):636-44. 2. Laskin WB, Fetsch JF, Mostofi FK. (1998). Angiomyofibroblastoma-like tumour of the male genital – analysis of 11 cases with comparison to female angiomyofibroblastoma and spindle cell lipoma. Am J Surg Pathol. 22(1);6-16. 3. Chien YC, Mokanszki A, Huang HY, Silva Jr. RG, Chen CC, et al. (2020). First glance at molecular profile of atypical cellular angiofibroma/ cellular angiofibroma with sarcomatous transformation by next generation sequencing. Diagnostics (Basel). 10(1); 35. 4. Liu Y, Xu Y, Wang Q, Chen Q. (2019). Cellular angiofibroma in the hypopharynx. Medicine. 98(50):e18385. 5. Ding G, Yu Y, Jin M, Xu J, Zang Z. (2014). Angiomyofibroblastoma- like tumour of the scrotum- a case report and literature review. Onco Lett. 7(2):435-438. 6. Ntorkou AA, Tsili AC, Giannakis D, Batistatou A, Stavrou S, et al. (2016). Magnetic resonance imaging findings of a cellular angiofibroma of the tunica vaginalis of the testis- a case report. J Med Case Rep. 10:71. 7. Erdur ZB, Yener HM, Yilmaz M, Karaaltin AB, Inan HC, et al. (2017). Cellular angiofibroma of the nasopharynx. J Craniofac Surg. 28(8):720-22. 8. Brandao A, Campos S, Fraga J, Silva T, Aguas F, et al. (2017). [Retroperitoneal cellular angiofibroma- a rare gynaecological entity]. Acta Med Port. 30(12):882-886. 9. Panagopoulos I, Gorunova L, Bjerkehagen B, Andersen K, Lund-Iversen M, et al. (2017). Loss of chromosome 13 material in cellular angiofibromas indicates pathogenetic similarity with spindle cell lipoma. Diagn Pathol. 12:17.