Information Links
Related Conferences
Previous Issues Volume 5, Issue 1 - 2020
Literature Review: Post Traumatic Headache Interventions in the Pediatric Population
Mary Kelley, Megan Berry, Viviane Ugalde*
2200 NE Neff d, Bend, OR 97701, USA
Received Date: Feb 24, 2020 Published Date: Jun 03, 2020 Corresponding Author: Viviane Ugalde* 2200 NE Neff Rd, Bend, OR 97701, USA Copyright: Ugalde V, et al. ©2020 Citation: Ugalde V. (2020). Literature Review: Post Traumatic Headache Interventions in the Pediatric Population. Mathews J Pediatr. (5) 1: 01
ABSTRACT
Objective: To report on the current level of knowledge and understanding of post traumatic headache (PTH) in the pediatric population, as well as current best practice for treatment and early interventions.
Methods: A review of literature pertaining to the topic was conducted using PUBMED.
Results: We identify support for the use of ibuprofen and acetaminophen in combination, occipital nerve block, and craniosacral therapies. Many of the other available therapies are limited by lack of supporting evidence or relevant studies in pediatric populations.
Conclusion: PTH is a common problem affecting millions of patients each year. The pathophysiology is still incompletely understood, although risk factors have been identified. Currently, research focused on pediatric patients with PTH is extremely limited, and no treatment guidelines exist. Much of the current treatment in a clinical setting is based on studies of primary headache disorders or adult patients with PTH. Therefore, additional research in pediatric populations is needed to determine a gold standard of care in this common childhood condition.
Keywords: Post traumatic headache; Post concussive syndrome; Traumatic brain injury; Migraine; Cervicogenic headache
Abbreviations: Post Traumatic Headache (PTH); Post Concussive Syndrome (PCS); Traumatic Brain Injury (TBI); Mild Traumatic Brain Injury (mTBI); Tricyclic Antidepressants (TCAs); Anti-Epileptics (AEDs); Cognitive Behavioral Therapy (CBT); Craniosacral Therapy (CST); Visceral Manipulation (VM); Neural Manipulation (NM)
BACKGROUND
a. Methods: All literature referenced in this review was identified using PUBMED. Keywords for the search included:post traumatic headache treatment, pediatric post traumatic headache, post concussive syndrome, pediatric mild traumatic brain injury, pediatric migraine treatment, occipital nerve blocks, and individual treatments along with pediatric.
b. Epidemiology: Post traumatic headache (PTH) is considered a secondary headache disorder with a clinical phenotype described by its main features. PTH typically has features resembling one of the primary headache disorders: tension, migraine, or migraine-like-cluster. Headache is the most common symptom in the acute and chronic phases in traumatic brain injury (TBI) of any severity [1]. Post concussive syndrome (PCS) is the development of chronic symptoms from a mild traumatic brain injury (mTBI) and occurs in 10-20% of mTBI patients [1]. A traumatic brain injury is classified as mild when loss of consciousness and/or confusion and disorientation is less than 30 minutes in duration and may also be referred to as a concussion. The most common symptom in PCS is headache, occurring in up to 88% of sports related concussions, followed closely by photophobia. In the United States 1.8 million people develop acute PTH and 400,000 develop chronicPTH annually [1]. In a prospective study of pediatric patients in the emergency room and brain injury clinic, those with persistent headaches 3 months after injury, 55% met the criteria for migraine [2].
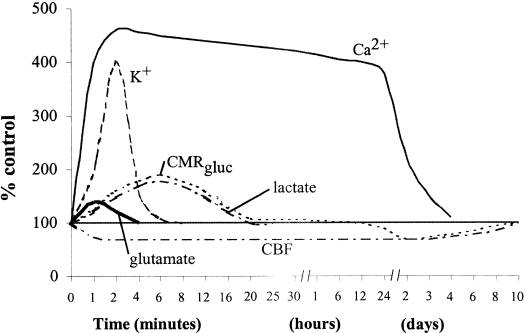
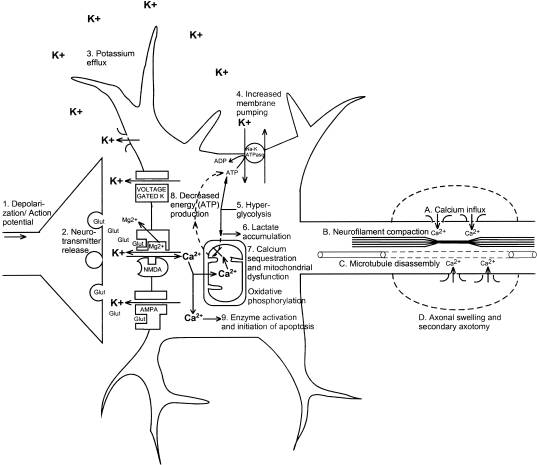
C. Pathophysiology: The exact mechanisms for PTH are currently unknown. It is inferred that the pathophysiology of concussions and mTBI may cause or contribute to PTH [1]. The neurometabolic cascade that occurs after injury has been extensively described (Figure 1). Traumatic biomechanical forces disrupt cell membrane integrity and can distort axons. This disruption and distortion then leads to dysfunction in axonal transport and connectivity. A decrease in cerebral blood flow and brain energy dysfunction is also observed (Figure 2). It is thought that excitotoxic cell damage, inflammation, genetics and psychologic influences also contribute to the development and heterogeneity of posttraumatic clinical syndromes [3]. PTH may also have a cervicogenic etiology, in which case the headache is due to referred pain perceived in the head from a source in the neck. Cervicogenic headache is caused by a disorder of the cervical spine and may involve bone, disc and soft tissue damage that were a resulting from the trauma that caused the mTBI. It is important to evaluate for cervicogenic causes in persistent headaches so as to select the most beneficial treatment.
Figure 1: Neurometabolic cascade following experimental concussion. K+, potassium; Ca2+, calcium; CMRgluc, oxidative glucose metabolism; CBF, cerebral blood flow [9].
Figure 2: Neurometabolic cascade following traumatic injury. (1) Nonspecific depolarization and initiation of action potentials. (2) Release of excitatory neurotransmitters (EAAs). (3) Massive efflux of potassium. (4) Increased activity of membrane ionic pumps to restore homeostasis. (5) Hyperglycolysis to generate more adenosine triphosphate (ATP). (6) Lactate accumulation. (7) Calcium influx and sequestration in mitochondria leading to impaired oxidative metabolism. (8) Decreased energy (ATP) production. (9) Calpain activation and initiation of apoptosis. A, Axolemmal disruption and calcium influx. B, Neurofilament compaction via phosphorylation or sidearm cleavage. C, Microtubule disassembly and accumulation of axonally transported organelles. D, Axonal swelling and eventual axotomy. K+, potassium; Na+, sodium; Glut, glutamate; Mg2+, magnesium; Ca2+, calcium; NMDA, N-methyl-D-aspartate; AMPA, d-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid [9].
Other proposed mechanisms of headache are visual changes and vestibulo-ocular impairments [4]. It is postulated that frontal lobe injury from coup-contracoup injury can lead to frontal gaze center disruption and subtle functional visual changes with convergence insufficiency [5]. Alnawmasi MM, et al. found adults with “mild TBI affects processing in both the dorsal and ventral cortical processing streams equally compared to normal controls” [6]. There is preservation of visual acuity for distance. Leddy’s work at the University of Buffalo has led to a classification of persistent concussive symptoms including headache in adults separating headaches triggered by physiological responses to exercise intensity, vestibulo-ocular changes, and cervicogenic [7]. Cordingley D, et al. used a graded treadmill exercise test in concussed patients <20 years of age and diagnosed physiologic post concussive disorder in 58/106 patients tested [8].
D. Risk Factors: In a 2018 review of post traumatic headaches in pediatrics, the following were identified as risk factors for PTH persistence: female gender, adolescence, prior concussion with prolonged recovery, history of prior headache/migraine, and high number of acute symptoms, particularly migrainous symptoms, following concussion [10,11]. Consideration of these factors is useful for follow-up planning [11]. A family history of migraine, particularly maternal has also been identified as a risk factor for the development of PTH. In a prospective study, children with persistent PTH at three months post injury, 56% had a prior history of headaches or migraines and 82% had a family history of migraines [2].
E. Associated Factors: Overlap with other conditions should also be considered. Depression is common after mild TBI. Chrisman and Richardson found a 3.3 fold increase in depression with pediatric concussion. Headache is a common symptom related to depression [12].
TREATMENTS
A. Current standards: A 2018 review of pharmacotherapy for persistent PTH in children and adolescents determined that no guidelines exist to aid in the pharmacological treatment of PTH in the pediatric population [13]. Additionally, there were no placebo-controlled trials available and most evidence was derived from a few uncontrolled trials and studies conducted in the adult population.Evidence was found to suggest that PTH can be classified as the primary headache disorder that it most closely resembles and can be treated accordingly. Many of the agents used to treat persistent PTH, such as: triptans, NSAIDS, tricyclic antidepressants (TCAs), anti-epileptics (AEDs), anti-hypertensives and melatonin, have supporting data for the management of migraine or chronic migraine. Few have been studied for the treatment of persistent PTH in a systematic manner, including TCA’s, AED’s, beta-blockers and nutraceuticals [13].
In an article published in 2018 in the Pediatric Annals, a multifaceted and individualized treatment approach was recommended, including lifestyle modifications, preventative and abortive medications, and rehabilitation strategies for persistent PTH [3]. Lifestyle modifications include addressing sleep hygiene, stress, headache triggers, hydration, regular meals, and comorbid conditions. Preventative medications can include riboflavin, melatonin, magnesium, anti-epileptic drugs, and TCA’s. These should be considered for headaches which are debilitating, occur more than twice per week, or are unresponsive to abortives. Abortive medications including NSAIDs and Tylenol should be used no more than twice per day on three days per week to avoid medication overuse headache. Triptans are another option for abortive treatment, and should be used no more than twice per week, and less than 9 times per month. Complete rest is no longer recommended; in fact, it may postpone recovery [10]. Graded return to activity should begin as soon as possible [3]. For persistent PTH not responding to the suggested lifestyle and medication approaches, cognitive behavioral therapy and physical rehabilitation should be considered.
A 2016 report from the Pediatric Mild TBI Guideline Workgroup offered level B evidence for the use of acetaminophen and ibuprofen for PTH pain [14]. The workgroup found poor evidence for the use of amantadine for persistent headache. They also report insufficient evidence to recommend use of hypertonic saline in the ED for acute headache treatment in pediatric patient. This set of guidelines can be useful in a clinical setting for diagnosing patients with concussion and estimating their risk of chronic PTH; however, guidelines related to treatment of those headaches are lacking.
EXPERIMENTAL INTERVENTIONS
i. Nerve Blocks: 7 pediatric publications have reported excellent therapeutic effectiveness of bilateral greater occipital nerve blocks on primary chronic headaches as well as PTH in pediatric populations [15]. Lidocaine is preferred and the value added by concomitant steroids remains to be determined. It was also determined that complete paresthesia of the blocked nerves maximizes effectiveness [15].
ii. Behavioral therapies and mind body interventions: A systematic review published in 2018 concluded that heterogeneity across studies made assessment of efficacy difficult; thus, the findings were inconclusive. Interventions included group cognitive behavioral therapy (CBT), telephonic counseling, individual CBT, cognitive training, psychoeducation and computer-based or therapist directed cognitive rehabilitation. Many of the negative studies were done at a date that was greater than one-year post injury. The authors identified considerations to improve the quality of future studies on behavioral therapies. They determined that the sooner the intervention, the better the outcome. Supportive, therapeutic relationships may facilitate improved outcomes. Studies incorporating telephone support suggested that face-to-face interventions may not be necessary and may be population dependent [16].
iii. Ibuprofen and Acetaminophen: In a four-arm open label randomized controlled trial pilot study, 80 people aged 8-18 years were studied. Participants were sequentially randomized to one of the four treatment arms: (i) acetaminophen for 72 hours (10 mg/kg to 15 mg/kg, every 4 hours, maximum 75 mg/kg/day or 4 g/day); (ii) ibuprofen for 72 hours (10 mg/kg, every 8 hours, maximum 3200 mg/day); (iii) both acetaminophen and ibuprofen (dosed as per arms (i) and (ii)); and (iv) standard treatment for headache (no recommendation of routine analgesia). In patients with headache for less than 48 hours following a concussive event it was found that patients who received medication had significantly fewer headache days, fewer episodes of headache and lower headache intensity than control. Patients who received the combination of ibuprofen and acetaminophen (79%) and ibuprofen alone (61%) were more likely to be back at school 1-week post injury as compared with acetaminophen alone (33.3%) and the control group (21.1%) [17]. These findings suggest benefit to combination treatment and, given the limitations of a pilot study, would warrant conducting a similar trial on a larger scale. In addition, the Pediatric Mild TBI Workgroup Guidelines support the use of ibuprofen and acetaminophen for use following acute mTBI [14]. Patients and parents should be counseled on the risk of analgesic overuse and rebound headache if this strategy is employed.
iv. Gabapentin and Tricyclics: A retrospective cohort study of 277 adult patients clinically diagnosed with concussion found decrease in both headache and symptom scores over time in each medication group as well as the group not receiving medication. Patients in the medication groups showed significantly higher headache and symptom scores. Neither medication showed significant longitudinal improvements in outcome scores. Piecewise regression showed short term improvements with gabapentin and more sustained improvements with TCAs. Gabapentin and TCAs have immediate effects on improving symptom burden but long-term outcomes similar to those without medication [18]. Given the retrospective review, one question whether the group with higher severity score was also in the medication group, causing a bias that would affect the analysis. Although this study demonstrates a promising short-term role for TCA’s and gabapentin in treating PTH in adults, it cannot be assumed that pediatric patients will respond similarly. Further research is needed focusing on a pediatric population.
v. Craniosacral therapy and visceral manipulation: A single blind case series of 11 male retired NFL/CFL players diagnosed with PCS concluded that after ten sessions of specific craniosacral therapy, visceral manipulation, and neural manipulation (CST/VM/NM) therapy resulted in statistically greater improvements in perceived pain intensity, range of motion, memory, cognition, and duration of sleep [19]. Given the statistically significant improvements seen in this study and the low risk associated with this intervention craniosacral therapy and visceral manipulation may be promising treatment options. Further research in more diverse patient populations is warranted.
vi. Exercise therapy: A review of observational and experimental data presented evidence for subthreshold aerobic exercise as a tool to safely evaluate concussion and speed recovery for patients with PCS [20]. Additionally, the article states that there is emerging evidence for the effectiveness of controlled aerobic exercise in the acute phase after adolescent sports-related concussion. This study did not specifically look at PTH but highlights the positive effects of controlled exercise. In another study by Cordingly D, et al. use of tailored submaximal exercise treatment for physiologic post-concussive disorder in pediatric patients with sports-related concussion led to clinical improvement in 90% of patients, with 80% successfully returning to sports participation [8]. Further research into its potential effects on PTH in the pediatric population is warranted [20,21].
vii. Vision therapy: There is improved understanding of the pathophysiology of visual impairments after mild TBI [6]. However, there are no randomized controlled trials of different vision therapies. Empirically, vision therapy is used for patients with abnormal Visual Oculo-Motor Screening examinations. [22]. Our concussion clinical experience indicates that much more study is needed to understand those patients who may benefit versus those whose symptoms are worsened by the therapy.
viii. Cefaly: Schoenen found that transcutaneous supraorbital electrical stimulation of the trigeminal nerve reduced migraine days, headache days and amount of migraine medications needed in the group using electrical stimulation versus a sham group [23]. Supraorbital electrical stimulation used daily has also demonstrated utility as a prophylactic agent for episodic migraines [24]. It is FDA approved for 18 years of age or greater. This device needs further study to demonstrate effectiveness in a pediatric population.
xi. Amantadine: In a 2013 retrospective case controlled trial, 25 adolescent athletes referred to a sports concussion clinic after 3-4 weeks of persistent concussion symptoms were treated with amantadine 100mg twice daily for 3-4 weeks [25]. They were matched with 25 control cases that were not treated with medication, and symptoms were compared in the two groups using ImPACT scores. The study showed improvement in reported symptoms (z = 0.79, 95% CI, 0.22-1.37) in the treatment group compared to the control group. The authors acknowledge the limitations of their study due to small sample size and the retrospective case-control design. Further research using a double-blinded randomized-control trial is recommended before any conclusion can be drawn as to the effectiveness of amantadine. In addition, the researchers did not specify which post-concussive symptoms had improved, limiting the reliability of this study in treatment of PTH specifically.
CONCLUSION
PTH affects millions of people each year, including many child and adolescent athletes, but relatively few high-quality studies exist detailing the best treatment options for pediatric patients. Most current treatment options have been extrapolated from studies on the treatment of migraine and other primary headache types, despite our understanding of the unique pathophysiology of PTH. Furthermore, much of the research pertaining to treatments has been conducted in adult populations and may therefore not be applicable in the pediatric population. Although solid recommendations against absolute rest and cocooning in concussion exist, further research is needed on the best treatment options for pediatric patients who experience ongoing headaches. The best current evidence exists for lidocaine nerve blocks, a combination of ibuprofen and acetaminophen, and craniosacral therapy; although all three need further study through randomized controlled trials in a pediatric population. Behavioral therapies are potentially promising, although they can be difficult to study in isolation, and they may be a helpful addition to pharmacologic therapies. Gabapentin and TCA’s have shown little long-term benefit to adults, andthey have not been studied in pediatric patients. Finally, aerobic exercise may be a promisingtreatment option for PTH and other long-term concussion symptoms. Randomized controlled trials are sorely lacking in the treatment of pediatric patients with PTH. Further research could reduce the symptom burden on millions of children and families struggling with these chronic headaches. This review highlights the paucity of strong data to support a standard of care in pediatric PTH. Given the prevalence of this condition and it’s potential for negatively effecting the lives of children it is important that more research be done on potential treatments so that clear guidelines can be established for their care.
CONFLICT OF INTEREST
All listed authors have approved the submitted version of the manuscript. The authors do not have any competing interests to disclose. This manuscript is an original work and has not been submitted or published elsewhere. All figures have been used with permission from their original creators, as sited in the text.
REFERENCES 1. Mares C, Dagher JH, Harissi-Dagher M. (2019). Narrative Review of the Pathophysiology of Headaches and Photosensitivity in Mild Traumatic Brain Injury and Concussion. Can J Neurol Sci. 46(1):14-22. doi: 10.1017/cjn.2018.361. 2. Kuczynski A, Crawford S, Bodell L, Dewey D, et al. (2013). Characteristics of post-traumatic headaches in children following mild traumatic brain injury and their response to treatment: a prospective cohort. Dev Med Child Neurol. 55(7):636-41. doi: 10.1111/ dmcn.12152. 3. Langdon R, Taraman S. (2018). Posttraumatic Headache. Pediatr Ann. 47(2):e61-e68. doi: 10.3928/19382359-20180131-01. 4. Master CL, Master SL, Wiebe DJ, Storey EP, et al. (2018). Vision and Vestibular System Dysfunction Predicts Prolonged Concussion Recovery in Children. Sport Med. 28(2):139-145. doi: 10.1097/ JSM.0000000000000507. 5. Wright WG, Tierney RT, McDevitt J. (2017). Visualvestibular processing deficits in mild traumatic brain injury. J Vestib Res. 27(1):27-37. doi: 10.3233/VES- 170607. 6. Alnawmasi MM, Chakraborty A, Dalton K, Quaid P, et al. (2019). The effect of mild traumatic brain injury on the visual processing of global form and motion. Brain Inj. 33(10):1354-1363. doi: 10.1080/02699052.2019.1641842. 7. Ellis MJ, Leddy JJ, Willer B. (2015). Physiological, vestibulo-ocular and cervicogenic post-concussion disorders: an evidence-based classification system with directions for treatment. Brain Inj. 29(2):238-48. doi: 10.3109/02699052.2014.965207. 8. Cordingley D, Girardin R, Reimer K, Ritchie L, et al. (2016). Graded aerobic treadmill testing in pediatric sports-related concussion: safety, clinical use, and patient outcomes. J NeurosurgPediatr. 25(6):693-702. 9. Giza CC, Hovda DA. The Neurometabolic Cascade of Concussion.J Athl Train. 2001;36(3):228-235. 10. Blume HK. (2018). Posttraumatic headache in pediatrics: an update and review.CurrOpinPediatr. 30(6):755-763. doi: 10.1097/MOP.0000000000000691. 11. Choe M, Barlow KM. (2018). Pediatric Traumatic Brain Injury and Concussion.Continuum (MinneapMinn). 24(1, Child Neurology):300-311. doi: 10.1212/ CON.0000000000000569. 12. Chrisman SP, Richardson LP. (2014). Prevalence of diagnosed depression in adolescents with history of concussion. J Adolesc Health. 54(5):582-6. doi: 10.1016/j.jadohealth.2013.10.006. 13. Kacperski J. (2018). Pharmacotherapy for Persistent Posttraumatic Headaches in Children and Adolescents: A Brief Review of the Literature. Paediatr Drugs. 20(5):385-393. doi: 10.1007/s40272-018-0299- 8. 14. Lumba-Brown A, Yeates KO, Gioia G, Turner M, et al. (2016). Report from the Pediatric Mild Traumatic Brain Injury Guideline Workgroup: Systematic Review and Clinical Recommendations for Healthcare Providers on the Diagnosis and Management of Mild Traumatic Brain Injury Among Children. CDC.gov. Aug 15. Dubrovsky AS. (2017). Nerve Blocks in Pediatric and Adolescent Headache Disorders.Curr Pain Headache Rep.21(12):50. doi: 10.1007/s11916-017-0650-8. 16. Minen M, Jinich S, Ellett GV. (2019). Behavioral Therapies and Mind-Body Interventions for Posttraumatic Headache and Post-Concussive Symptoms: A Systematic Review. Headache. 59(2):151-163. doi: 10.1111/head.13455. 17. Petrelli T, Farrokhyar F, McGrath P, Sulowski C, et al. (2017). The use of ibuprofen and acetaminophen for acute headache in the postconcussiveyouth: A pilot study.PaediatrChild Health.22(1):2-6. doi: 10.1093/ pch/pxw011. 18. Cushman DM, Borowski L, Hansen C, Hendrick J, et al. (2018). Gabapentin and Tricyclics in the Treatment of Post-ConcussiveHeadache, a Retrospective Cohort Study.Headache. doi: 10.1111/head.13451. 19. Wetzler G, Roland M, Fryer-Dietz S, DettmannAhern D. (2017). CranioSacral Therapy and Visceral Manipulation: A New Treatment Intervention for Concussion Recovery.Med Acupunct.29(4):239-248. doi: 10.1089/acu.2017.1222. 20. Anderson V, Manikas V, Babl FE, Hearps S, et al. (2018). Impact of Moderate Exercise on Post-concussive Symptoms and Cognitive Function after Concussion in Children and Adolescents Compared to Healthy Controls.Int J Sports Med. 39(9):696-703. doi: 10.1055/ a-0592-7512 21. Leddy JJ, Haider MN, Ellis M, Willer BS. (2018). Exercise is Medicine for Concussion.Curr Sports Med Rep. 17(8):262-270. doi: 10.1249/JSR.0000000000000505. Review. 22. Fox SM, Koons P, Dang SH. (2019). Vision Rehabilitation after Traumatic Brain Injury. Phys Med RehabilClin N Am. 30(1):171-188. doi: 10.1016/j.pmr.2018.09.001. 23. Schoenen J, Vandersmissen B, Jeangette S, Herroelen L, et al. (2013). Migraine prevention with a supraorbital transcutaneous stimulator: a randomized controlled trial. Neurology. 80(8):697-704. doi: 10.1212/ WNL.0b013e3182825055. 24. Robbins MS, Lipton RB. (2017). Transcutaneous and Percutaneous Neurostimulation for Headache Disorders. Headache. 57 Suppl 1:4-13. doi: 10.1111/ head.12829. 25. Reddy CC, Collins M, Lovell M, Kontos AP. (2013). Efficacy of amantadine treatment on symptoms and neurocognitive performance among adolescents following sports-related concussion. J Head Trauma Rehabil. 28(4):260-265.