Information Links
Related Conferences
Previous Issues Volume 6, Issue 1 - 2021
Kratom in America: Legal Differences across Federal, State, and Local Jurisdictions
Ethan Ramirez1*, Bardia Adibmoradi1, Marie Bourgeois2
1University of South Florida, Morsani College of Medicine, Florida, USA
2University of South Florida, College of Public Health, Florida, USA
*Corresponding Author: Ethan Ramirez, University of South Florida, Morsani College of Medicine, Florida, USA, E-mail: [email protected].
Received Date: December 04, 2020
Published Date: January 27, 2021
Copyright: Ramirez E, et al. (2021).
Citation: Ramirez E, et al. (2021). Kratom in America: Legal Differences Across Federal, State, and Local Jurisdictions. Mathews J Psychiatry Ment Health. (6)1:28.
ABSTRACT
Kratom (Mitragyna speciosa) is a tropical plant species that is indigenous to Southeast Asia. The leaves of the kratom plant have traditionally been consumed for the stimulant and opioid-like effects it can produce. In the United States, kratom is gaining popularity as an herbal supplement, and a natural alternative to traditional prescription opioids. Kratom is a controversial substance in America as it is not currently regulated on a federal level in the United states and can be legally obtained and used in many areas of the country without a prescription. The Drug Enforcement Agency (DEA) has listed kratom on a list of drugs of concern, but kratom has yet to be scheduled under the Controlled Substances Act. On the state level, kratom laws are constantly changing and vary greatly from state-to-state with some states completely banning the substance and other states adopting legislation that protects kratom consumers by regulating its manufacturing and sale. Most states have no kratom laws and have ultimately left the decision up to the federal government. There are even instances where local counties and cities have abolished kratom in opposition to their own state laws. The majority of readily available legal information on kratom is dispersed amongst individual state government databases. Most of the consolidated legal information on kratom can only be found on websites supported by pro-kratom lobbying organizations which are inherently biased in their representation of the laws in place. The purpose of this article is to provide a complete, current, and unbiased review of federal, state, and local kratom laws in the United states while exploring state and local rationales for such legal variance. This article concludes by discussing possible implications of these differing laws on legislators, consumers, and the healthcare providers moving forward.
INTRODUCTION
Kratom is the common name given to the leaves of a tropical evergreen tree (Mitragyna speciosa) that is native to Southeast Asia [1]. The alkaloid compounds mitragynine and 7-hydroxymitragynine give the leaves of the kratom tree psychotropic and opioid-like properties, which has led to kratom’s use as an alternative self-treatment for chronic pain, opioid withdrawal, and anxiety [2]. Kratom is also used as a recreational drug by a portion of users. These combined aspects make kratom a substance of both considerable potential and controversy [3].
Countries in Southeast Asia have had a tumultuous relationship with kratom for centuries. The Association of Southeast Asian Nations (ASEAN) made its sale, purchase, and consumption illegal in 2013 [4]. However, enforcement of this ruling varies depending on the country. This is evident in the fact that Indonesia and Vietnam are amongst the largest suppliers of kratom in the world. Meanwhile, while production in Thailand and Malaysia can result in a prison sentence up to 4 years [5].
The United States seems to be following a similar pattern of legal variance as kratom polices are markedly different throughout the country. There are currently no federal regulations on the production, sale, purchase, or use of kratom in the US after failed attempts to schedule it under the Controlled Substances Act (CSA). In 2017, a letter was sent from the Department of Health and Human Services (HHS) to the Drug Enforcement Agency (DEA) suggesting that kratom should be included as a Schedule I controlled substance under the CSA. The letter also included recommendations from the Food and Drug Administration (FDA) and National Institute on Drug Abuse (NIDA) in which both entities concluded that kratom should be listed under Schedule I of the CSA [6]. However, kratom merely remains classified as a Drug and Chemical of Concern which only describes kratom as a substance of potential abuse and danger by the DEA [7].
Despite the legal status of kratom federally, there are significant differences across city, county, and state governments. While some states have adopted policy to protect consumers by regulating the production of the kratom supplements, other states are banning kratom and scheduling kratom under the CSA themselves. With that said, kratom is currently legal in most states and a growing number of states are adopting policy aimed at regulating the production of kratom supplements and protecting consumers [8]. This is often attributed to lobbying efforts by pro-kratom organizations that intend to maintain the legal status of kratom in America.
The disparity in kratom legality across the US does not come without consequence. The novelty of kratom in the US and the Federal government’s stance on kratom has resulted in disorganized rulings on kratom policy which provides an unsound foundation for future legislation to be based upon. It is apparent that more research needs to be done on the pharmacological properties of kratom in order to draw conclusions on its impact to medicine and public safety. The dilemma is that federal, state, and local policies limit where kratom research can be conducted. Yet policy makers can benefit greatly from research data when deciding future action to take regarding kratom. Any inability to conduct research will inevitably have a negative impact on educating healthcare professionals. This in turn can leave healthcare professionals unprepared to advise patients about kratom use as an alternative medication amidst the opioid crisis.
This article aims to provide a complete, current, and unbiased review of all federal, state, and local kratom laws in the United states while exploring the rationales for these laws. In addition, this paper will examine the possible implications that can arise from the current state of kratom policy in America.
1. Kratom on a National Level
Kratom (Mitragyna speciosa) has been used for centuries across Southeastern Asia as a natural treatment for pain. The leaves of a kratom tree are traditionally picked from the tree, dried, and made into an herbal tea to achieve this pain mitigating effect. It is also documented that farmers and other physical laborers chew the leaves of the kratom tree as a means to boost energy levels and relieve fatigue [9]. Allopathic medicine as a discipline holds skepticism toward many herbal medicines based on varying levels of evidence and a paucity of randomized controlled trials for certain interventions. These views may explain why kratom did not gain much attention in the United States from government officials and healthcare workers alike. This status of kratom has changed within the last decade as research now shows that the leaves of kratom contain many psychoactive compounds. Mitragynine and 7-hydroxymitraginie are the two most well-researched compounds in kratom and they have been shown to bind to opioid receptors in the central nervous system [10]. Medications that bind to these opioid receptors are aptly classified as opioids. Opioids include illegal drugs such as heroin, very potent synthetic opioids (i.e. fentanyl), and many pain relievers available legally by prescription, such as oxycodone, hydrocodone, codeine, and morphine [11]. Additional biological effects of kratom remain largely unknown, and more research needs to be done to fully elucidate all chemical properties.
Under the Federal Food Drug and Cosmetic Act (FD&C Act), the manufacture, sale, and marketing of kratom fall under the purview of the FDA [12]. Seizure of kratom and other substances considered adulterated or misbranded is warranted under §334 of the FD&C Act. Large-scale seizures of kratom shipments first began in September 2014, when the FDA determined that kratom was being marketed as a dietary supplement without undergoing the proper New Dietary Ingredients (NDI) Notification Process as described in §413 of the FD&C Act. Over 25,000 pounds of raw kratom, seized from Rosefield Management, Inc. in Van Nuys, California, was worth an estimated $5 million [13,14]. Over 121,000 pounds of kratom materials were seized by law enforcement between September 2014 to July 2016. Shortly after these seizures began, a report from the Centers for Disease Control and Prevention (CDC) cited a ten-fold increase in kratom-related calls to U.S. poison centers from 2010-2015 [15].
In addition to oversight by the FDA, a variety of substances fall under the jurisdiction of the US Drug Enforcement Administration (DEA). In August 2016, the DEA announced its intent to place mitragynine and 7-hydroxymitragynine under Schedule I of the CSA, citing the increase in poison center calls as justification for scheduling [16]. Substances in Schedule I are deemed to have no accepted medical use in the US, have a high potential for abuse, and are a threat to public safety. The announcement to schedule kratom was met with protest from both the public and Congress. Kratom advocacy groups, such as the American Kratom Association (AKA), fronted petition campaigns, which included a White House petition that received over 140,000 signatures from individuals who reported using kratom as a natural method to wean off of prescription opioid addiction and/or cope with chronic pain [17]. Constituents also enlisted the aid of their state representatives. Fifty-one members of the US House of Representatives and 9 members of the U. S. Senate sent letters to acting DEA administrator, Chuck Rosenburg, asking the DEA to reconsider or at least delay the ban on kratom products [18]. In October 2016, the DEA revoked their announcement to schedule kratom and opened an online forum for public comments. In October 2017, the US Department of Health and Human Services (DHHS) sent a letter to the new acting DEA administrator, Robert Patterson, urging the DEA to reconsider making kratom a schedule I controlled substance since there were no current medicinal uses for kratom [6]. Kratom still remains on the DEA’s ‘Drugs of Concern’ list as a substance that poses risk to those who abuse it [7].
Despite present DEA disputes relating to potential scheduling of kratom, the FDA continues to enforce current regulations pertaining to kratom use in the US. In 2018, the FDA continued to advise companies selling kratom to remove their products from the market. The former commissioner of the FDA, Scott Gottlieb, released a statement on the possibly dangerous opioid-like properties of kratom, stating it should not be used medicinally or recreationally [19]. This remains the official stance FDA stance on kratom usage. In May 2019, the US House of Representatives Committee on Appropriations issued Report 116-62 which included a request for the National Institute of Health (NIH) to expand research requests that NIH expand research on kratom and its constituent compound. The House Committee stated they were ‘aware of the potential promising results of kratom for acute and chronic pain patients who seek safer alternatives to sometimes dangerously addictive and potentially deadly prescription opioids [20].
Notwithstanding federal restrictions, some stakeholders seek to facilitate patient access to kratom. Despite clear federal regulations governing the manufacture, sale, and advertisement of kratom, not all manufacturers comply with relevant law. As a result, multiple manufacturers continue to sell kratom by labeling it ‘not for human consumption.’ Many companies also avoid making any health claims about their product [21].
In addition to federal law, several states have passed additional laws regulating the manufacture, sale and use of kratom. These laws are vastly different from state-to-state. The next section of this article will discuss some of these differences and how states came to these conclusions.
2. Kratom State and Local Levels
State laws on kratom manufacture, sale, and consumption differ in the US to a degree that is largely unmatched in the nation’s history. The most recent parallel one could make would be the disparity in marijuana legislation that has occurred in the US over the past decade. However, marijuana laws continue to grow more homogenous and the same cannot be said with regards to kratom.
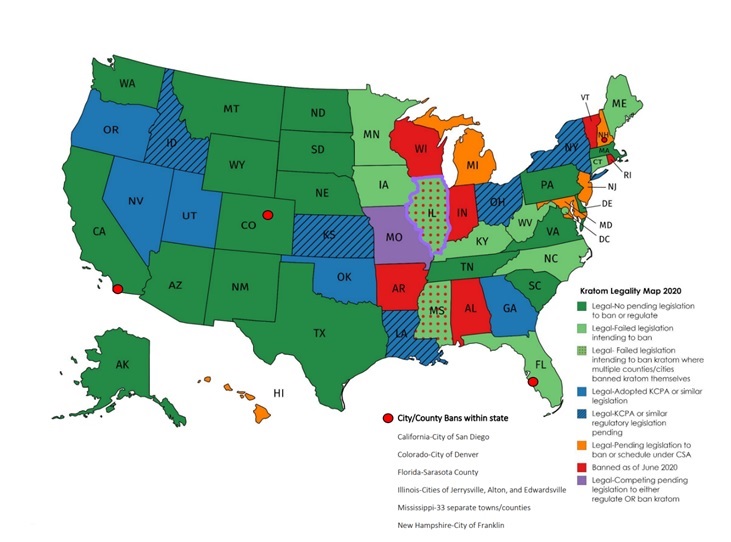
Kratom is currently legal in 44 states and is banned in six states (see Figure 1). This simple categorization fails to represent the truly tumultuous status of kratom policy across these states. For example, of the 44 kratom-legal states, six states (CA, CO, IL, FL, NH, MS) have cities or counties where kratom is not legal to possess, sell, or consume [22-27]. There are also eight kratom-legal states (HI, IL, MD, MI, MS, MO, NH, NJ) with pending legislation that if passed would either ban kratom or schedule it under the CSA [28-35]. Mississippi’s Senate Bill (SB) 2084 if passed would make kratom a schedule I drug [32] Maryland would also make kratom a schedule I drug if SB 147 passed [30]. Michigan’s SB 0443 would classify kratom as a schedule II drug [31]. Hawaii’s SB 3064 would classify kratom as a schedule V drug if passed [28]. Illinois and Missouri seem particularly conflicted as these states have competing legislation pending that will either ban or regulate kratom use. Illinois HB 4681 introduced February 6 2020 aims to regulate kratom and HB 5657 introduced February 14 2020 aims to ban kratom in Illinois [28,36]. In Missouri, HB 2061 passed March 20, 2020. This bill is essentially the KCPA and its passing has led to increased regulation on the production and sale of kratom products in the state [37]. However, SB 765 introduced January 1, 2020 if passed would supersede HB 2061 and make kratom a schedule I controlled substance under the CSA [38].
In contrast to the states moving to ban kratom, five states (AZ, GA, NV, UT, OK) have adopted legislation within the last three years that regulates kratom production and ensures quality control measures for consumers in their state [39-43]. The legislation being adopted is known as the Kratom Consumer Protection Act (KCPA). The act’s specific goal is to “ to regulate the preparation, distribution, and sale of kratom products; to prohibit the preparation, distribution, and sale of adulterated or contaminated kratom products; to prescribe fines and penalties and allow remedies; and to provide for the powers and duties of certain state governmental officers and entities” [44]. The KCPA is a piece of legislation that is lobbied by the American Kratom Association (AKA). The AKA is a non-profit corporation registered in Virginia that was launched in 2015. Their mission statement is to support kratom consumers in both in America and globally through education and advocacy of kratom’s benefits as a natural alternative to prescription drugs and maintain kratom supply through practicing sustainability and environmental protection. The AKA is very politically active, and their website states their intention to pass the KCPA in an as many states as possible [45].
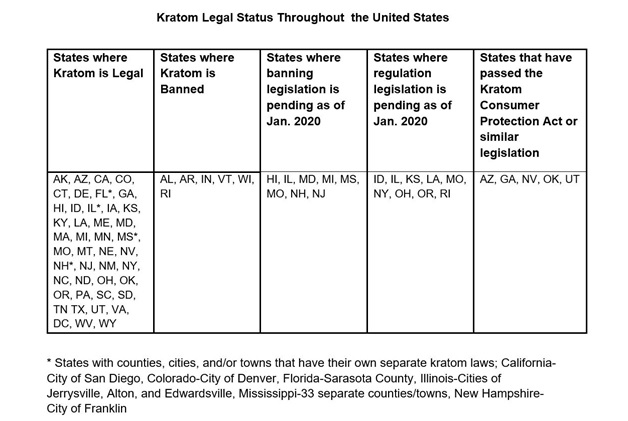
Oregon lawmakers proposed a particularly unique piece of legislation on January 9, 2017 when SB 518 was introduced. If passed, the bill would grant the Oregon State Board of Pharmacy to conduct research studies for purpose of determining whether kratom and its derivatives should be scheduled as controlled substance. This bill was referred to a senate subcommittee and no further action has been made on the bill since 2017 [46]. However, Oregon HB 4013 introduced on February 3, 2020 aimed at regulating kratom products in the state and penalizing any unlawful preparation, distribution, sale or offer for sale of kratom products with a $1250 fine and/or 30 days imprisonment [47]. Table 1 summarizes the legal status of kratom in all 50 states in addition to listing the states with pending kratom legislation as of January 2020.
Figure 1: Kratom Legality Map 2020 (last updated October 2020).
Table 1: United States kratom legality table categorizing states based upon having current or pending legislation to legalize or ban kratom (last updated October 2020).
2.1 Rationales for State Law Disparity
As seen above, the variance in kratom legislation from state-to-state is astounding. The question that comes to mind when studying this topic is why do the laws differ so much? State and local jurisdictions seem to base their kratom policies on a wide spectrum of reasons including reports of citizen fatality, pro-kratom lobbying efforts, and increased federal pressure on drug policy amidst the opioid crisis. For example, Alabama chose to ban kratom sale, possession, and consumption in 2016 and cited their rampant issues with opioids as a reason to ban kratom [48]. At the time of making kratom a schedule I substance under the CSA , Alabama was the highest per capita opioid prescribing state with a rate of 121 prescriptions per 100 persons, which is equivalent to 1.2 prescriptions for every man, woman and child [49]. In Arkansas, kratom was also made a schedule I substance in 2016 following an annual medical examiner report linking kratom to three citizen deaths the year prior. Although legal in Colorado, kratom is banned specifically in Denver following similar reports of death due to kratom toxicity. It is important to note that multidrug ingestion was implicated in the majority of reported cases and research on kratom toxicity and mortality is currently very limited [50].
Indiana’s rationale for banning kratom in unique in that the state banned kratom over 16 years ago after labeling the plant a ‘synthetic drug.’ This labeling has been protested by kratom advocates on the basis that kratom is actually not derived synthetically, but the law was revised in 2014 when SB 305 made kratom a schedule I controlled substance without an option for reform or repeal. Several states including Maine, Kansas, Oregon, and Connecticut have attempted to ban kratom within the past decade but failed to do so after public objection from kratom consumers and lobbying groups such as the AKA.
Another isolated incident that impacted the perception of kratom in the eyes of lawmakers was a mass salmonella outbreak in 2018 that was linked to contaminated kratom products by the CDC. The CDC reports that 199 people infected with the outbreak strains of Salmonella were reported from 41 states with 50 people requiring hospitalization. Fortunately, there were no fatalities reported by the CDC. Six unique strains of Salmonella were identified during testing and over 65 unique kratom products were confirmed to be contaminated. The outbreak led many states to release public warnings urging the public not to use kratom undoubtably affected the reputation of kratom as a safe alternative to prescription medications. In response to the outbreak, the AKA released a statement in February 2018 where they voiced their concern about the CDC recommending that kratom should not be consumed after their investigation. The AKA stressed that kratom should be supported by federal and local agencies to ensure quality control for consumers.
3. Conclusions: Implications of Kratom Policy Disparity
What are some possible implications and potential consequences of kratom’s current legal status in America? The disparity in US kratom laws has had a negative impact on research, public awareness, and quality control, all of which can potentiate possible dangers of kratom while limiting its potential as a beneficial therapeutic. In this final section we will discuss the effects of kratom legal disparity on the consumer population, scientific community, and future legislators.
3.1 Implications for Consumers
Kratom consumers arguably suffer the most from the kratom policy in place today. In states such as Florida, Mississippi, Illinois, and Colorado, kratom sale and possession may be completely legal in one town and could result in hefty fines or possible jail time in the next town over. This is analogous to alcohol sale and possession laws in many southern U.S. states such as Arkansas and Mississippi where “dry” and “wet” counties abut one another [53]. One could argue that consumer awareness of kratom laws is less than that of alcohol as kratom awareness in general is still relatively low. Unintentional violations of local and state kratom laws have resulted in arrests and these violations will likely continue to occur with current kratom policies across the nation [54].
The lack of federal kratom regulation also puts consumers at an increased risk of becoming harmed from kratom product consumption. Without quality assurance taking place by a governing agency such as the FDA, kratom consumers are at an increased risk for consuming contaminated and even adulterated kratom products. The 2018 salmonella outbreak discussed previously is one of the more recent and substantial instances where a lack of quality control measures led to serious adverse events for consumers across the country. Kratom adulteration is a more insidious prospect that can and has occurred due to a lack of federal oversight. Several toxicology researchers have published findings of kratom being adulterated with added amounts of 7-hydroxymitragynine as well as phenethylamine which is another “mood-boosting” compound available commercially without a prescription [55,56]. There have even been reports of kratom being adulterated with hydrocodone which is a federally regulated opioid requiring a prescription for legal consumption [57]. The final implication of kratom’s legal disparity on consumers is that it has severely limited the public’s awareness and understanding of what kratom is. Without proper public understanding of kratom, consumers may be more likely to use it with other psychotropic agents such as prescription opioids or alcohol which can result in potentially fatal synergism. There are no available studies that have investigated consumer understanding of what kratom is, but it can be postulated that some percentage of active kratom consumers are unaware that kratom is an opioid. Federal regulation of kratom would likely include appropriate labeling of kratom products to warn consumers of possible contraindications in an effort to reduce kratom-related adverse events including fatality.
3.2 Implications for the Scientific Community
The degree to which kratom laws vary across the United States have very profound implications on the scientific community. Research that aims to further decipher the pharmacological properties of kratom has been steadily advancing. However, the overall progress of kratom research can be slowed when potential researchers are limited to conducting experiments in areas where kratom is legal. The prospects of synthesizing a safer opioid therapeutic from kratom for the treatment of chronic pain is also limited as the psychotropic compounds in kratom have been made illegal in six states and many other cities/counties across the nation. The 2019 U.S. House of Representatives Committee on Appropriations request that the NIH expand kratom research on has currently not yielded an increase in kratom research publications [20]. This can be attributed to several factors including the COVID-19 pandemic which halted many new research initiatives in both academic and private sectors.
Additionally, healthcare professional’s education about kratom is also limited and its possible that many healthcare providers do not know how to advise their patients who consume kratom. There is very little formal introduction to kratom in many medical training programs. Healthcare professionals training and working in kratom legal states may also be more familiar with kratom when compared to states where kratom is banned. This lack of kratom education could cause physicians to prescribe medications that are contraindicated with kratom use which can result in adverse events in patient populations.
3.3 Implications for Future Legislators
The final topic of discussion for this article will be the possible implications of current kratom legislation on future legislation. As previously stated, kratom is currently legal in forty-four states and is illegal in six states. Federal kratom regulation seems less likely to occur as more states continue to propose bans on kratom. It seems possible that the future legal status of kratom will resemble that of marijuana in which it is federally illegal, but legally regulated in many states.
Considering state laws, any future state decisions on kratom policy will be predicated on the policy decisions currently in place. This can be problematic in states that have banned kratom and included verbiage in their legislation that eliminates the possibility of reversing the ban. If a potential therapeutic containing mitragynine or 7-hydroxymitragynine were synthesized, these states cannot adopt its use due to their unwavering bans of these compounds. The idea of this occurring is not beyond the realm of possibility as several schedule I substances including methylenedioxymethamphetamine (MDMA) and psilocybin are now being revisited for their potential therapeutic effects on victims of PTSD and other mental disorders[58].
In conclusion, kratom is a plant with the possibility of helping people alleviate physical and mental ailments, but still has the potential to be dangerous like most therapeutics. The current stratification between federal, state, and local kratom laws has only yielded a muddled understanding of kratom which must be remedied if we are to minimize the problems associated with kratom in America today. Federal regulation of kratom sale, distribution, and consumption would seem to be the most appropriate change to kratom law in terms of ensuring the safety of consumers across the nation, but these decisions must be made after more research has been conducted and after we gain a better understanding of kratom’s role in society as a whole. Possible future directions of this research would be to conduct a survey study that characterizes consumer’s understanding of what kratom is in addition to studying the consumer perspective on kratom policy and how it affects their perceptions of kratom.
REFERENCES
- Kratom. In: Liver Tox: Clinical and Research Information on Drug-Induced Liver Injury. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012.
- Meireles V, Rosado T, Barroso M, Soares S, Goncalves J, et al. Mitragyna speciosa: Clinical, Toxicological Aspects and Analysis in Biological and Non-Biological Samples. Medicines (Basel). 2019;6(1).
- Sethi R, Hoang N, Ravishankar DA, McCracken M, Manzardo AM. (2020). Kratom (Mitragyna speciosa): Friend or Foe? Prim Care Companion CNS Disord. 22(1).
- Kratom: Miracle treatment or dangerous drug? Digital Media Nusantara. (2018). https://theaseanpost.com/article/kratom-miracle-treatment-or-dangerous-drug. Accessed 2020.
- A Guide to Kratom Legality: Where Is Kratom Legal? https://speciosaguide.com/guide-kratom-legality-kratom-legal/#Kratom_Legality_Asia. Published March 2020. Accessed 2020.
- R. W. Patterson, Department of Human Health Services: 18. (2017).
- "Drugs of Concern List." 2020, from https://www.dea.gov/taxonomy/term/311.
- Kratom Consumer Protection Act: What Is It and Why We Need It. (2019, April19, 2019). Retrieved from https://kraoma.com/kratom-consumer-protection-act/
- Jansen KL, Prast CJ. (1988). Ethnopharmacology of kratom and the Mitragyna alkaloids. J Ethnopharmacol. 23(1):115-9.
- Kruegel AC, Gassaway MM, Kapoor A, Varadi A, Majumdar S, et al. (2016). Synthetic and Receptor Signaling Explorations of the Mitragyna Alkaloids: Mitragynine as an Atypical Molecular Framework for Opioid Receptor Modulators. J Am Chem Soc. 138(21):6754-6764.
- Summary of Issue: Opioids. NIDA. National Institute of Health. (2020). from https://www.drugabuse.gov/drugtopics/opioids#:~:text=Opioids%20are%20a%20class%20of,%2C%20morphine%2C%20and%20many%20others.
- 21 U.S. Code CHAPTER 9—FEDERAL FOOD, DRUG, AND COSMETIC ACT. Legal Information Institute, Cornell Law School.
- US Food & Drug Administration. (12/19/2019). "New Dietary Ingredients (NDI) Notification Process". Dietary Supplements 2020, from https://www.fda.gov/food/dietary-supplements/new-dietary-ingredients-ndi-notification-process.
- Commons KM. (2018). Cracking Down on Kratom: FDA Investigation, Enforcement, Seizure, and Recall of Products Reported to Contain Kratom. From https://www.fdli.org/2018/08/update-cracking-down-on-kratom-fda-investigation-enforcement-seizure-and-recall-of-products-reported-to-contain-kratom/#:~:text=According%20to%20FDA%2C%20imported%20kratom,not%20present%20a%20significant%20or.
- Anwar, Mehruba; Law, Royal; Schier, Josh (2016-01-01). Notes from the Field: Kratom (Mitragyna speciosa) Exposures Reported to Poison Centers – United States, 2010–2015. MMWR. Morbidity and Mortality Weekly Report. 65(29):748–49.
- National Media Affairs Office. DEA announces intent to schedule Kratom. [Web page] 2016, August 30; Available from: https://www.dea.gov/press-releases/2016/08/30/dea-announces-intent-schedule-kratom.
- Ingraham C. (2016). DEA defies senators’ appeal to reconsider ‘unprecedented’ kratom ban. 2020, from https://www.washingtonpost.com/news/wonk/wp/2016/09/30/dea-defies-senators-appeal-to-reconsider-unprecedented-kratom-ban/.
- Nelson, S. (2016, 10/3/2016). Dozens of Congressmen Ask DEA Not to Ban Kratom Next Week. from https://www.usnews.com/news/articles/2016-09-23/45-congressmen-ask-dea-not-to-ban-kratom-next-week.
- Chappell, B. (2018, 4/8/18). FDA Orders An Unprecedented Recall After Kratom Company Ignored Its Requests. The Two-Way. From https://www.npr.org/sections/thetwo-way/2018/04/04/599443476/fda-orders-an-unprecedented-recall-after-kratom-company-ignored-its-requests.
- (2019). DEPARTMENTS OF LABOR, HEALTH AND HUMAN SERVICES, AND EDUCATION, AND RELATED AGENCIES APPROPRIATIONS BILL, 2020 H.R. 2740. H. o. Representatives, 116th Congress.
- (2017). "Terms & Conditions." from https://kratomoflife.com/terms-and-conditions/.
- San Diego’s Synthetic and Psychoactive Drug Laws. In: Council SDC, ed. sandiego.gov. 2016.
- Roberts M. Denver Bans Controversial Herbal Drug Kratom for Human Consumption. In West word; 2017.
- Title 7 Chapter 16 Kratom Prohibited. In: Dept. ACP, ed. Title Number 7. American Legal Publishing: Sterling Codifiers 2018.
- (2014). CODE OF ORDINANCES OF SARASOTA COUNTY, FLORIDA. Sarasota County Board of County Commissioners.
- Regulating products containing kratom. In. Sen. Feltes DSFC, Dist 21; Sen. Hennessey, Dist 5; Sen. Dietsch, Dist 9; Sen., Kahn D, trans. 2020 SESSION ed2020.
- Apel T. Authorities: Legal opioid substitute is dangerous, even deadly. Gray Television inc. Published 2019. Accessed2020
- Hawaii Senate Bill 3064. In. Sharon Moriwaki RB, Lorraine Inouye, trans2020.
- House Bill 4681 In. Evans M, trans. LRB101 15173 2020.
- House Bill 283. Criminal Law Kratom Prohibited Purchase, Distribution, or Sale. In. Young R, trans. CF HB 283. Regular Session 2020.
- Senate Bill 0433. In. John Bizon MD, trans 2019.
- Senate Bill 2084 In. Younger, trans. 2020 Regular Session ed2020.
- Makes kratom a Schedule I controlled substance. In. Onder, trans. 3137S.01I. SECOND REGULAR SESSION ed2020.
- Senate Bill 758: Regulating products containing kratom. In. Sen. Feltes DSFC, Dist 21; Sen. Hennessey, Dist 5; Sen. Dietsch, Dist 9; Sen., Kahn D, trans. 2020 Session.
- New Jersey Assembly Bill 2236. In. Dancer R, trans. 219th Legislature ed2020.
- House Bill 5657. CONTROLLED SUB-KRATOM-SCHED II. In. Moylan M, trans. 101 General Assembly ed2020.
- House Bill No. 2061. In. Anelli C, trans. 3383H.02P. First Regular Session ed2020.
- Senate Bill: 765 Makes kratom a Schedule I controlled substance. In. Onder, trans. 3137S.01I. Second Regular Session.ed2020.
- House Bill 2561: Appropriation; drug diversion programs; Pima. In: Representatives SoAHo, ed. 54 ed2020.
- House Bill 551. In. Hill D, trans. 16-13-120. 2019.
- Assembly Bill No. 303–Assemblymen Wheeler and Yeager. In. 80th ed2019.
- Senate bill 58. Kratom Consumer protection Act. In. Bramble CS, trans2019.
- House Bill: 2846 Creating the Oklahoma Kratom Consumer Protection Act. In. Pae D, trans2020.
- Kratom Consumer Protection Act: What Is It and Why We Need It. Kraoma. Published 2019. Updated April 19 2019. Accessed 7/8/2020.
- Our Mission American Kratom Association About AKA Web site. Published 2020. Accessed.
- Senate Bill 518: Relating to kratom; declaring an emergency. In. 79th Session ed2017
- House Bill 4013: Establishes regulations for kratom products. In. Post C, Barker, trans. 80th ed2020.
- Senate Bill 226: Controlled substances, additional compounds included in Schedule I. In. Orr A, trans. 173673-1. Alabama 2016.
- Understanding the Opioid Crisis. Alabama Department of Mental Health Understanding Opioids Web site. https://mh.alabama.gov/understanding-the-opioid-crisis. Accessed2020.
- Gershman, K. (2019). Deaths in Colorado Attributed to Kratom. New England Journal of Medicine. 380: 97-98.
- Multistate Outbreak of Salmonella Infections Linked to Kratom (Final Update). Center for Disease Control. https://www.cdc.gov/salmonella/kratom-02-18/index.html. Published 2018. Updated May 24 2018. Accessed 2020.
- AKA ADDRESSES CDC SALMONELLA REPORT, FDA RELEASE. www.americankratom.org: American Kratom Association February 22, 2018.
- Staff E. 'Dry Towns' in the USA. American Addiction Centers. https://www.alcohol.org/statistics-information/dry-towns/. Published 2019. Updated 05/2019. Accessed 11/20, 2020.
- News 5 Investigates: The legal status of the drug Kratom after two recent arrests [press release]. wkrg.com: Nexstar Media Group, 11/25/2019 2019.
- Lydecker AG, Sharma A, McCurdy CR, Avery BA, Babu KM, et al. (2016). Suspected Adulteration of Commercial Kratom Products with 7-Hydroxymitragynine. J Med Toxicol. 12(4):341-349.
- Nacca N, Schult RF, Li L, Spink DC, Ginsberg G, et al. (2020). Kratom Adulterated with Phenylethylamine and Associated Intracerebral Hemorrhage: Linking Toxicologists and Public Health Officials to Identify Dangerous Adulterants. J Med Toxicol. 16(1):71-74.
- McCurdy C. Kratom (Mitragyna Speciosa) as a Potential Therapy for Opioid Dependance Paper presented at: ESPD 50 Conference Ethnopharmacological Search for Psychoactive Drugs; June 7 2017, 2017; Tyringham Hall, UK.
- Mithoefer MC, Grob CS, Brewerton TD. (2016). Novel psychopharmacological therapies for psychiatric disorders: psilocybin and MDMA. Lancet Psychiatry. 3(5):481-488.