Information Links
Related Conferences
Previous Issues Volume 7, Issue 2 - 2022
Investigation of Dose-Rate Effects in Pulsed Low Dose Rate Radiotherapy
T Dos-Santos1*, D Cvetkovic1, L Chen1, S Liu2, M Li2, L Yang2, D Wu3, and CM Ma1
1 Fox Chase Cancer Center, Temple University, USA
2 The Third Affiliated Hospital of Qiqihar Medical University, Qiqihar, China
3 The People’s Hospital of Guangxi Zhuang Autonomous Region, Nanning, China
Received Date: June 16, 2022
Published Date: July 26, 2022
*Corresponding Author: Dos Santos Troy, Fox Chase Cancer Center, Temple University, USA. Tel: 215-728-3071 E-mail: [email protected]
ABSTRACT
Purpose: To investigate the radiation dose rate effect for pulsed lowdose-rate (PLDR) radiation therapy using in vitro clonogenic analysis. Materials and Methods: Lung cell line A549 (Adenocarcinomic human alveolar basal epithelial cells) and human prostate cancer cells (PC3) were cultured using Dulbecco's modified Eagle's medium supplemented with 10% fetal calf serum, with penicillin (50 U/ml), and streptomycin (50 µg/ml) at 37°C under 95% humidity and 5% CO2 atmosphere. All experiments used cells in the exponential growth phase by seeding ~200 cells into T-25 flasks, in triplicate, 8-10 hours before use. A clinically calibrated beam from a Varian-2100-iX machine was used to deliver a total dose of 0.25, 0.5, 1, 2 and 4Gy to the cells. The dose rate for the conventional radiation therapy (CRT) group was 500 cGy/min. The effective dose rates (EDR) for PLDR (8.3, 25, 60, 150cGy/min) were determined by varying time between a train of radiation pulses, each 0.25 Gy. After irradiation, cells were incubated for 8 to 9 days, colonies were counted, and the surviving fractions of clonogenic cells were determined. Results: Both cell lines showed comparable responses between CRT and PLDR with different EDRs, where their survival fractions decreased with dose but were unremarkable. All PLDR groups were statistically indistinguishable among each other, and from CRT. Both cell lines were observed to be agnostic towards variation with the dose rate in radiation repair response or low-dose hyper-radiosensitivity in this study. Conclusion: PLDR effect at different EDRs is comparable to that of CRT against two human cell lines. This result adds to the body of research showing PLDR’s clinical efficacy, due its equivalent tumor control and normal tissue sparing properties with decreased EDRs. PLDR may be developed into a clinically viable alternative for treating large tumor masses and/or recurrent cancers with decreased normal tissue tolerances.
Keywords: Pulsed low dose rate (PLDR), radiation therapy, dose rate effect, lung cancer A549, prostate cancer PC3, clonogenic assay, Hyperradiosensitivity (HRS).
INTRODUCTION
Modern external beam radiation therapy has benefited from various and significant technologically driven advancements, such as, the implementation of intensity modulated radiation therapy, which produces highly conformal dose deposition, onboard patient verification imaging systems such as cone beam computed tomography and digital portal imaging, and improvements in the treatment planning algorithms, which guides optimization and dose deposition calculations. The integration of these, and other advances provide new tools for practitioners to escalate dose to a target more confidently while simultaneously minimizing dose to healthy surrounding tissues, and consequently increasing the therapeutic ratio.
While technologically based advances have improved the therapeutic ratio in controlling tumors, there have been comparatively few corresponding advancements in the radiobiological science underpinning the use of radiation therapeutically. One notable recent radiobiology study which supports a modern therapeutic method has been proposed by Song et al [1]. Stereotactic radiosurgery (SRS) and stereotactic ablative radiation therapy (SABR) are two modern treatment techniques whereby high doses are typically delivered in one to five fractions; however, efficacy of these treatment modalities have not been fully supported by classical radiobiology. It is now proposed that ablation of vascular endothelial cells by the SRS/SABR modalities induces ischemic/indirect cell death as the primary mechanism of tumor control, in addition to direct tumor cell death, while indirectly initiating/increasing an increase in anti-tumor immune activity.
Another biologically driven treatment modality relates to the use of the pulsed low dose-rate (PLDR) radiation therapy technique. This technique involves the typical daily fractionation regimen being fragmented, delivered in a series of sub fractions known as ‘pulses’ with a determined time interval between each pulse. The choice of the time interval between each pulse determines the effective dose rate of the daily fraction. A growing consensus postulates that PLDR benefits from two primary mechanisms: the targeting of tumors with radiation pulses below the transition dose, whereby cancer cells exhibit preferential hyper-radiosensitivity (HRS) in contrast to normal healthy tissue, and the superior radiation induced damage repair abilities of normal healthy tissues [2-5]. The ability to target tumors with the PLDR technique while simultaneously allowing healthy tissues to repair, creates a new opening for treatment of tumors which might be near to, or abutting, normal critical structures. PLDR can also prove to be a viable treatment modality against tumors proximal to previously treated healthy tissues which might be near or at its tolerance of received radiation dose. A detailed review of the radiobiology and dosimetric requirements for the implementation of PLDR clinically has been presented by Ma [6].
The PLDR delivery scheme was initially proposed based on a theoretical study on tumor control probability (TCP) for gliomas exhibiting low-dose HRS by Tomé and Howard [7]. Their analyses indicated that delivering therapy in a pulsed fashion by keep the dose per pulse below the transition dose and an effective dose rate of 0.067Gy/min could improve both TCP and normal tissue complication probability (NTCP). However, the effect of the time interval used for the pulsed dose rate scheme was not investigated, and the effective dose rate of 0.067Gy/min was adopted primarily for clinical practicability, e.g., to deliver a daily 2Gy dose in 30 min [6]. A number of in vitro studies were carried out to investigate cell survival for different tumors using similar pulsed dose delivery [8-12]. Particularly, Terashima et al [11] examined the cell survival rates of V79 and A549 cells as a function of time interval and dose rate using clonogenic assays. Cells were irradiated in unit doses of 0.25, 0.5, 1.0 and 2.0 Gy, at dose-rates of 1.0, 1.5 and 2.0 Gy/min at 1 min intervals, and at a dose-rate of 2.0 Gy/min at 10 s, 1 min and 3 min intervals. Both cell lines showed markedly lower survival rates with fractionated exposures (0.25 Gy/pulse) with short time intervals than those for single exposures delivering the same dose.
In this study we utilized the established in vitro clonogenic technique as the method to investigate the radiation dose rate effect in PLDR radiation therapy using in vitro clonogenic analysis. Two human cancer cell lines (lung A549, prostate PC3) were irradiated on a clinical linear accelerator with a total dose of 0.25, 0.5, 1, 2 and 4Gy. The dose rate for the conventional radiotherapy (CRT) group was 200 cGy/min and the effective dose rates (EDR) for PLDR were 8.3, 25, 60, 150cGy/min, which were determined by varying time between a train of radiation pulses, each 0.25 Gy. To study the effect of environmental differences between normally treated cells undergoing PLDR treatments in a clinical linac room at 20°C and cells maintained in ideal growing conditions for the same treatment, some cell groups were maintained at a temperature of 37°C, with a partial pressure of 5% for carbon dioxide and 100% humidity. The results will be analyzed and compared with previous publications.
METHOD AND MATERIALS
Cell preparation
Lung cell line A549 (adenocarcinoma human alveolar basal epithelial cells) and human prostate cancer cell line PC3 were cultured using Dulbecco's modified Eagle's medium supplemented with 10% fetal calf serum, with penicillin (50 U/ml), and streptomycin (50 µg/ml) at 37°C under 95% humidity and 5% CO2 atmosphere. All experiments utilized the clonogenic assay; monolayer cells in the exponential growth phase were collected, counted, and appropriately diluted for seeding ~200 cells into T-25 flasks, in triplicate, 8-10 hours before exposure to irradiation.
Treatment
A clinically calibrated beam from a Varian-2100-iX machine was used to deliver an integral dose of 0, 0.25, 0.5, 1, 2 and 4 Gy to the cells. The dose rate for the conventional radiation therapy (CRT) group was 200 cGy/min. The effective dose rates (EDR) (8.3, 25, 60, 150 cGy/min) for the PLDR groups were determined by varying the time (0.6, 0.3, 1 and 3 minutes) between a train of radiation pulses, where dose to each pulse was 0.25 Gy delivered at 200 cGy/min.
Controlled treatment environment
To compare cellular responses of cells maintained in incubator-like conditions of 37°C and 5% CO2 atmosphere with cells maintained at the normal temperature in the treatment vault of 20°C with standard room atmosphere, it was necessary to replicate similar favorable growing conditions found in the incubator for the duration of PLDR treatments while in the treatment area (see Table 1). For this experiment, a large aquarium was lined with foil on five inner-sides with the top being lined with clinging plastic wrap. A water bath was placed inside of the tank to provide humidity and to raise the temperature within the tank. The temperature within the tank was monitored using a calibrated thermometer and maintained by adjusting the water bath dial appropriately. Partial pressure of CO2 was maintained by tightening special non-filtering caps on T-25 growing flasks immediately after they were removed from the incubator. Four groups of PC3 cells were created, in triplicate, for a total of 6 experiments.
Table 1 Environmental conditions for PC3 cells treated in the linac vault with CRT and PLDR delivery techniques.
|
CO2 pp |
Temperature |
Humidity |
|
|
Incubator |
5% |
37ºC |
95% |
|
Radiation Vault (LINAC) |
0.04% |
20ºC |
Unsure |
Data analysis
After irradiation, cells were incubated for 8 to 9 days, colonies were counted, and the surviving fractions (SF) of clonogenic cells were determined. The plating efficiency (numbers of adherent cells/total cell number for seeding) of control cells was used to normalize the SF of irradiated cells. The SF was determined for all treated groups, which is the total number of colonies divided by the plating efficiency fraction of cells seeded.
RESULTS
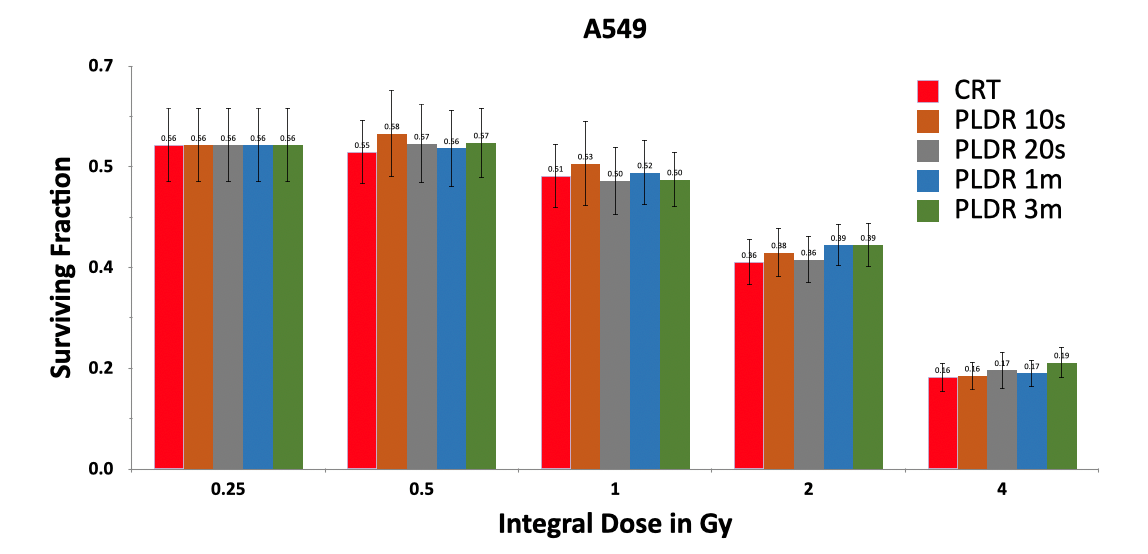
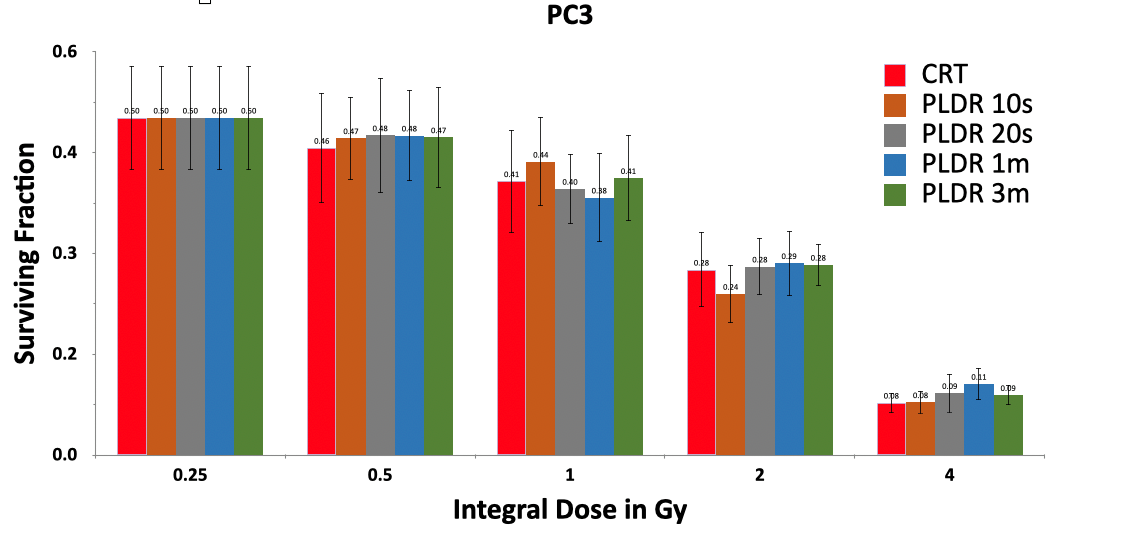
Figures 1 and 2 show the SF of human lung cancer cells (A549) and prostate cancer cells (PC3) treated by CRT and PLDR techniques with different doses and time intervals. As expected, cell survival decreases with the total dose received between 0.25 and 4.0Gy for both cell lines. No significant differences were observed between CRT and PLDR with different time intervals from 10s to 3min (corresponding to effective dose rates 1.5Gy/min to 0.083Gy/min). All PLDR groups were statistically indistinguishable from each other, and from CRT. Both cell lines were observed to be agnostic towards variation with the dose rate in radiation repair response or low-dose HRS.
Figure 1. Survival of human lung cancer cells (A549) irradiated by a total dose of 0.25, 0.5, 1.0, 2.0 and 4.0Gy using CRT and PLDR delivery with a time interval of 10s, 20s, 1min and 3min.
Figure 2. Survival of human prostate cancer cells (PC3) irradiated by a total dose of 0.25, 0.5, 1.0, 2.0 and 4.0Gy using CRT and PLDR delivery with a time interval of 10s, 20s, 1min and 3min.
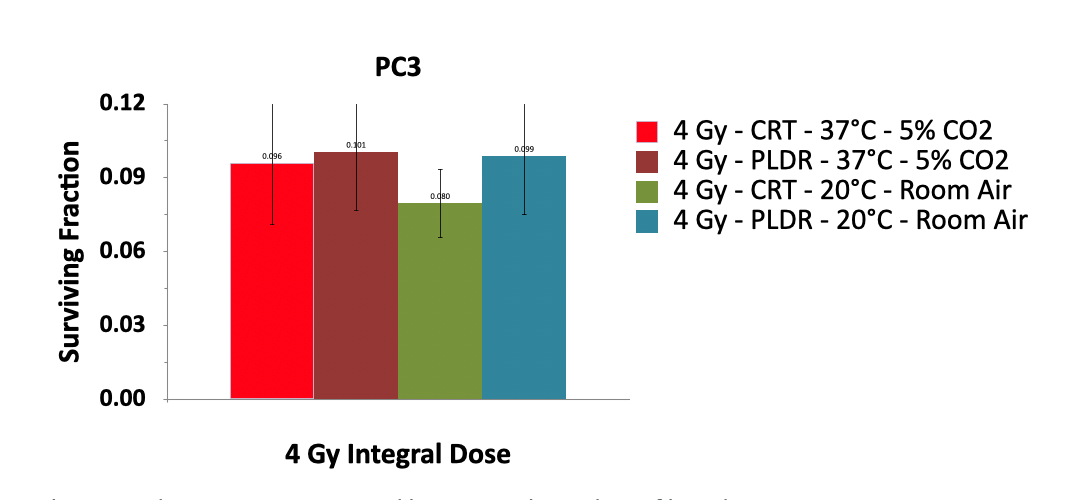
Figure 3 shows the survival fraction of human prostate cancer cells (PC3) irradiated by a total dose of 4Gy using CRT and PLDR delivery with a time interval of 3min under different environmental conditions. There was no statistically significant effect on cell survival for cells treated under ideal conditions, e.g., at temperature between 36°C and 38°C, with a partial pressure of 5% for carbon dioxide at 100% humidity, and for cells treated under normal linac room temperature of about 20°C, 0.04% partial pressure of carbon dioxide and ambient humidity during the course of an extended PLDR treatment.
Figure 3. Survival of human prostate cancer cells (PC3) irradiated by a total dose of 4Gy using CRT and PLDR delivery with a time interval of 3min. Incubator conditions were maintained by using tightened non-filtered caps immediately after leaving the incubator. The temperature was maintained by use of a custom heated and insulated tank.
DISCUSSION
The PLDR dose scheme was initially proposed based on a theoretical study on TCP for gliomas exhibiting low-dose HRS [7]. It was suggested that for malignancies that exhibit low-dose HSR, delivering therapy in a pulsed fashion by dividing the total dose into a number of pulses that are separated by a fixed time interval in which the dose per pulse is below the transition dose of about 0.5Gy could yield an increase in both TCP and normal tissue sparing. However, the effect of the time interval or the effective dose rate was not investigated and the current delivery schedule was chosen for clinical practicality [6]. The low-dose HRS effect may diminish as the total dose accumulates without sufficient time intervals between the pulses, resulting in the eventual accumulation of sufficient damage to induce repair and radiation resistance [12]. In this work, we have studied the PLDR effect at different effective dose rates from 0.083 Gy/min to 1.5 Gy/min for two human cell lines A549 and PC3. No significant differences in survival fractions were observed. This result adds to the body of pre-clinical research showing PLDR’s clinical efficacy, due to its equivalent tumor control and normal tissue sparing properties with decreased effective dose rates [14-18]. PLDR may be developed into a clinically viable alternative for treating large tumor masses and/or recurrent cancers with decreased normal tissue tolerances [2, 5, 6, 19].
Several earlier investigations have reported in vitro experimental results of cytotoxicities of different cell lines irradiated by PLDR dose schemes. Ma et al reported the cytotoxic effect of PLDR and conventional dose rate radiation for human prostate (LNCaP) cell lines with different doses and dose rates [9]. Their results showed greater cytotoxic effects at a low dose rate compared with that extrapolated from the high-dose regions of the cell-survival curves. However, the overall cell survival depended on many factors such as the total prescription dose, fractional dose and dose rate for conventional radiation, and also on partial fractional dose and time sequence (not simply effective dose rate) for PLDR. Schoenherr et al performed an in vitro study to exploit the potential therapeutic advantages of PLDR using 4 glioblastoma multiforme (GBM) cell lines, T98G, U373, U87MG and U138MG [10]. Their results showed that despite the prolonged delivery time, PLDR was equally effective as a single 2Gy dose although PLDR was more effective when given for 5 consecutive days to a total dose of 10 Gy. These results are consistent with our findings. More drastic results were reported by Terashima et al [11], who examined the survival rates of V79 and A549 cancer cells as a function of time interval and dose rate using clonogenic assays. Both cell lines exhibited markedly lower survival rates with pulsed exposures (0.25 Gy/pulse) with short time intervals (10s, 1min and 3min) than those for single exposures delivering the same dose. Todorovic et al investigated the effect of PLDR irradiation in isogenic head and neck squamous cell carcinoma cells with different radiosensitivity [12]. They showed that radioresistant Fadu-RR cells were more sensitive to PLDR irradiation than parental FaDu cells. The variable response of various tumor cell lines to the HRS-formulated dose scheme indicates only a limited success in preventing the early G2 checkpoint activation and downstream DNA repair. The time interval between radiation pulses may not be long enough in some cell lines, resulting in the eventual accumulation of sufficient damage to prematurely trigger the increased radioresistance (IRR) response.
A practical limitation of the in vitro clonogenic technique for this study involved necessary long PLDR treatment times. PLDR treated cells were removed for the optimal growing condition of the incubator; namely, the potential for cells to have their survival influenced due to intracellular changes in response to sub-optimal growing conditions of the radiation treatment space. The repair mechanisms, mediated by temperature and pH sensitive enzymes of cells may have an effect on the response of cells to PLDR. Normal functions of repair enzymes are impaired below 37ºC [20-22], and most DNA repair activity occurs within 2 hours for A549 specifically [23]. To regulate environmental differences between normally treated cells undergoing PLDR treatments, with cells maintained in ideal growing conditions for the same treatment, some PC3 groups in our study were maintained at a temperature between 36°C and 38°C, with a partial pressure of 5% for carbon dioxide and 100% humidity. Interestingly, no significant differences were found in the survival fraction between cells maintained in ideal environmental conditions and at ambient temperature and moisture in a linac room in our study. Another limitation is the difference between the in vitro and in vivo cell environment, which may influence the signaling cascade regulating the early G2 checkpoint. The cell repair process is initiated through ATM activity and maintained by several key kinases/phosphorylation events. Therefore, it is expected that more molecular-based experiments using whole-animal models would offer insight into the HRS/IRR mechanisms for the PLDR-relevant dose-fractionation scheme [14-18].
REFERENCES
- Song CW, Terezakis S, Emami B, Griffin RJ, Sperduto PW, et al. (2020). Indirect cell death and the LQ model in SBRT and SRS. J Radiosurg SBRT. 7(1):1-4.
- Ma CM, Luxton G, Orton CG. (2011). Point/counterpoint: pulsed reduced dose rate radiation therapy is likely to become the treatment modality of choice for recurrent cancers. Med Phys. 38(9):4909-4911.
- Martin LM, Marples B, Lynch TH, Hollywood D, Marignol L. (2014). Exposure to low dose ionising radiation: molecular and clinical consequences. Cancer Lett. 338(2):209-218.
- Joiner MC, Lambin P, Malaise EP, Robson T, Arrand JE, et al. (1996) Hypersensitivity to very-low single radiation doses: its relationship to the adaptive response and induced radioresistance. Mutat Res. 358(2):171-83.
- Chen L. (2021). Clinical Applications of Pulsed Low Dose-Rate Radiation Therapy. Mathews J Cancer Sci. 6(1):22.
- Ma C-M. (2022). Pulsed low dose-rate radiotherapy: radiobiology and dosimetry. Phys Med Biol. 67(3).
- Tomé WA and Howard S P. (2007). On the possible increase in local tumour control probability for gliomas exhibiting low dose hyperradiosensitivity using a pulsed schedule. Br J Radiol. 80(949):32-37.
- Lin PS, Wu A. (2005). Not all 2 Gray radiation prescriptions are equivalent: Cytotoxic effect depends on delivery sequences of partial fractionated doses. Int J Radiat Oncol Biol Phys. 63(2):536-544.
- Ma C-M, Mu ZM, Tafo A, Chen L. (2010). Variation of cytotoxic effect with pulsed dose sequence and low dose rate radiation Int. J. Radiat. Oncol. Biol. Phys. 78(3):S629–630.
- Schoenherr D, Krueger SA, Martin L, Marignol L, Wilson GD, et al. (2013). Determining if low dose hyper-radiosensitivity (HRS) can be exploited to provide a therapeutic advantage: a cell line study in four glioblastoma multiforme (GBM) cell lines Int. J. Radiat. Biol. 89(12):1009-1016.
- Terashima S, Hosokawa Y, Tsuruga E, Mariya Y, Nakamura T. (2017). Impact of time interval and dose rate on cell survival following low-dose fractionated exposures. J Radiat Res. 58(6):782–790.
- Todorovic V, Prevc A, Zakelj MN, Savarin M, Bucek S, et al (2020) Pulsed low dose-rate irradiation response in isogenic HNSCCcell lines with different radiosensitivity. Radiol. Oncol. 54(2):168-179.
- Marples B, Collis SJ (2008). Low-dose hyper-radiosensitivity: past, present, and future. Int J Radiat Oncol Biol Phys. 70(5):1310-1318.
- Park SS, Chunta JL, Robertson JM, Martinez AA, Wong CYO, et al. (2011). MicroPET/CT imaging of an orthotopic model of human glioblastoma multiforme and evaluation of pulsed low-dose irradiation. Int J Radiat Oncol Biol Phys. 80(3):885-892.
- Dilworth JT, Krueger SA, Dabjan M, Grills IS, Torma J, et al. (2013). Pulsed low-dose irradiation of orthotopic glioblastoma multiforme (GBM) in a pre-clinical model: effects on vascularization and tumor control. Radiother Oncol. 108(1):149-154.
- Zhang P, Wang B, Chen X, Cvetkovic D, Chen L, et al. (2015). Local tumor control and normal tissue toxicity of pulsed low dose rate radiotherapy for recurrent lung cancer: an in vivo animal study. Dose Response. 13(2):1–9.
- Meyer K, Krueger SA, Kane JL, Wilson TG, Hanna A, et al. (2016). Pulsed radiation therapy with concurrent cisplatin results in superior tumor growth delay in a head and neck squamous cell carcinoma murine model. Int J Radiat Oncol Biol Phys. 96(1):161-169.
- Wang B, Ren J, Zhang Z, Cvetkovic D, ChenXM, et al. (2019) An in-vivo study on pulsed low-dose-rate radiotherapy for prostate cancer Mathews J. Cancer Sci. 4(2):21.
- Rogacki K, Chao ST, Yu J, Godley A, Balagamwal E, et al. (2018). Review of pulsed reduced dose rate re-irradiation for recurrent tumors. J Cancer Clin. Trials 3:143.
- Hans Bisswanger. (2017). Enzyme Kinetics: Principles and Methods, third ed, Chapter 6: pH and Temperature Dependence of Enzymes. 145-152.
- Oei AL, Vriend LE, Crezee J, Franken NA, Krawczyk PM. (2015). Effects of hyperthermia on DNA repair pathways: one treatment to inhibit them all. Radiat Oncol. 10:165.
- M Samuel & C Rebecca, A Lesley, F Craig. (2019). Cooler temperatures slow the repair of DNA damage in tadpoles exposed to ultraviolet radiation: Implications for amphibian declines at high altitude. Global Change Biology. 26(3):1225-1234.
- Dos Santos T. (2013). Synergistic effects of combining therapeutic radiation and β-Lapachone against tumors. University of Minnesota Digital Conservatory.