Previous Issues Volume 2, Issue 1 - 2017
Initial Response to Therapy with Fulvestrant and Cyclin-Dependent Kinase 4/6 Inhibitor in a Male with Stage IV Breast Cancer
Aurelio B. Castrellon*, Steven M. Nguyen, Adriana M. Milillo Naraine, Michel Velez, Luis E. Raez
Memorial Cancer Institute, Breast Center, Florida, USA.
Corresponding Author: Aurelio B. Castrellon, Memorial Healthcare System, Memorial Cancer Institute, Breast Center, Director of Breast Cancer Research, Florida USA, Tel: (954) 265-4325; E-Mail: [email protected]
Received Date: 21 Feb 2017 Accepted Date: 27 Feb 2017 Published Date: 01 Mar 2017
Copyright © 2017 Castrellon AB
Citation: Castrellon AB, Nguyen SM, Milillo Naraine AM, Velez M, et al. (2017). Initial Response to Therapy with Fulvestrant and Cyclin-Dependent Kinase 4/6 Inhibitor in a Male with Stage IV Breast Cancer. Mathews J Cancer Sci. 2(1): 012.
ABSTRACT
Male breast cancer (MaBC) accounts for less than 1% of breast cancers (BC). Based on the low incidence of MaBC, information from randomized clinical trials is not available. Therefore, the same guidelines for palliative endocrine therapy in women with advanced BC are generally followed for men. Endocrine therapy (ET) represents the first line of treatment for male patients with stage IV disease in the absence of a visceral crisis. However, resistance to ET in hormone receptor positive advanced breast cancer is common, and given sufficient time, most patients are faced with disease progression. In this report we describe the case of a male that experienced systemic recurrence of BC following adjuvant therapy for locally advanced disease. First line endocrine treatment was combined with a cyclin-dependent kinase 4/6 inhibitor, leading to a dramatic radiographic and clinical response.
INTRODUCTION
Male breast cancer (MaBC) accounts for less than 1% of breast cancers (BC) [1]. In the United States, males were expected to account for only 2600 of the estimated 249,260 cases of breast cancer during 2016 projections [1]. The worldwide variation of MaBC resembles that of BC in women, with higher rates in North America and Europe and lower rates in Asia [2]. Although the epidemiologic literature on female breast cancer (FBC) is extensive, little is known about the etiology of MaBC; it appears to be associated with marital status, previous breast and testicular pathology, gynecomastia and liver diseases [3]. Due to its rarity, information from randomized clinical trials is not available. Men tend to be diagnosed with locally advanced or metastatic disease more frequently than women; more than 40% of patients have stage III or IV disease at diagnosis [4, 5].They also have a proportionately higher mortality, although outcomes for male and female patients with breast cancer are similar when survival is adjusted for age at diagnosis and stage of disease [4-7]. The association between estrogen levels and breast cancer in men is of interest because estrogen-related risk factors have been strongly implicated in the etiology of FBC [8]. Obesity has been implicated in the etiology of MaBC due to higher circulating estrogen levels and has consistently been associated with an increased risk of MaBC [9].Men with a mutation in the BRCA2 gene have an increased risk of breast cancer with a lifetime risk of about 6 in 100. BRCA1 mutations can also cause breast cancer in men, but the risk is lower - about 1 in 100 [10-13]. Histologically, the majority of breast cancers in men are infiltrating ductal carcinomas, but the entire spectrum of histological variants of breast cancer has been seen [14]. Papillary and lobular carcinoma are seen in less than 2% of the cases. About 80% of MaBC are hormone receptor positive (HR+), 15% overexpress human epidermal growth factor receptor 2 (HER2), and 4% are triple negative (estrogen receptor (ER), progesterone receptor (PR), and HER2/neu negative) [15, 16]. In terms of gene expression profile, recent studies by Johansson and colleagues have revealed two subgroups: luminal M1 and luminal M2. These subgroups demonstrated differences in tumor biological features and outcome, and differed from the intrinsic subgroups described in FBC. Luminal M2 tumors were characterized by high expression of immune response genes and genes associated with ER signaling. On the other hand, luminal M1 tumors, despite being ER positive (ER+) by immunohistochemistry, showed a lower correlation to genes associated with ER signaling and displayed a more aggressive phenotype and worse prognosis [17-19]. Since gene expression is typically related to protein expression, examination of the differences between MaBC and FBC were studied at the hormone receptor (HR) immunohistochemical level in a large series of 514 matched cases [20]. Strikingly, hierarchical clustering in FBC revealed ERa clustered together with PR, while in MBC, ERa clustered with the androgen receptor (AR) suggesting a clinically actionable difference between genders in HR biology. Although MaBC is typically considered ERa driven, there are distinct molecular features found in MaBC, which could be used as targets for currently available or yet undeveloped therapies [20]. Prospective trials evaluating the benefits of palliative endocrine therapy (ET) specifically for men with MaBC have not been performed. Therefore, the same guidelines for palliative ET in women with advanced BC are generally followed for men [21]. Given their proven efficacy and generally favorable toxicity profile, with the exception of patients with advanced visceral disease, most patients with advanced BC will receive ET, particularly those with metastatic ER+/HER2 negative BC. Resistance to ET is common, and given sufficient time, most patients are faced with disease progression [22]. The mechanisms underlying disease progression and ET-resistance development are complex and not fully understood [23]. Nevertheless, over the past several years, insights into several pathways of resistance have grown and have led to increased understanding of the clinical value of sequential lines of therapy and co-targeting strategies.
Cell Cycle Control and Proliferation Cell cycle regulation is identified as an attractive target for targeted drug therapy, in particular, the cyclin-dependent kinase (CDK) enzymes [24]. CDK4 and CDK6, together with cyclin D, promote G1-to-S phase transition by phosphorylating the retinoblastoma protein (Rb), which releases the E2F transcription factor and activates downstream target genes [25]. CDK4/6 and Cyclin D are activated in ER+ BC via the ER, along with other oncogenic signaling pathways [26, 27]. Earlier generation, non-specific CDK inhibitors demonstrated low monotherapy efficacy and high toxicity [28], but preclinical and subsequent clinical data of selective inhibitors of CDK 4/6 have shown promising results in HR+/ HER2 negative advanced BC treatment with more acceptable toxicity [29-32]. CDK 4/6 inhibitors prevent the cyclin D-CDK4/6 complex phosphorylation of Rb required for the commitment to S-phase and ultimately, cellular mitosis [31, 32]. Selective inhibition of CDK 6 has been reported to have a role in anti-angiogenesis as well [33]. An additional suggested mechanism of action for the novel CDK 4/6 inhibitor, palbociclib, is decreasing the expression of cyclooxygenase-II (COX-II), an enzyme associated with the epithelial-mesenchymal transition (EMT) in metastasis [34]. CDK 4/6 inhibition results in un-phosphorylation of Forkhead Box M1 (FOXM1), a transcription factor involved in the expression of genes that upregulate proliferative capacity [35]. These potent, ATP-competitive CDK 4/6 selective inhibitors are orally administered and have little to no function on other CDK enzymes, even at clinical doses. Selectivity for the CDK 4/6 proteins has been shown to be important in mitigating cytotoxic effects that were highlighted in the pan-CDK inhibitor predecessors [35]. In 2015, the FDA approved palbociclib, a highly selective inhibitor of CDK4/6 kinase, in combination with letrozole as an initial therapy for postmenopausal women with advanced ER+/ HER2 negative BC. The approval was based on results from the phase II PALOMA-1 clinical trial. [36]. The confirmatory phase III PALOMA-2 study randomized 666 patients in a 2:1 letrozole alone daily versus letrozole plus oral palbociclib 125 mg given once daily for 3 weeks followed by 1 week off over a 28-day cycle. Patients in the palbociclib containing arm experienced a progression-free survival (PFS) of 24.8 months while the control arm demonstrated a PFS of 14.5 months (p< 0.000001). The objective response rate (ORR) was also higher in the palbociclib group; 55.3% of the patients who had measurable disease experienced a reduction in size vs 44% in letrozole monotherapy group (P=0.013). In terms of side effects, neutropenia (79.5 vs 6.3%), fatigue (37.4 vs. 27.5%), and nausea (35.1% vs 26.1%) were more noticeable in the investigational arm; neutropenic fever was only seen in 2.5 % of the patients [37]. Similarly, in the second line setting, the PALOMA-3 doubleblind phase III clinical trial randomized 427 patients with ER+/ HER2 negative advanced BC who experienced disease progression either within 12 months of adjuvant therapy or within one month of prior ET. The study compared palbociclib plus fulvestrant versus fulvestrant plus placebo. In conjunction with study treatment, premenopausal and perimenopausal women were required to take goserelin. The study met the primary endpoint, PFS, which was 9.2 months in the palbociclib and fulvestrant arm versus 3.8 months in the fulvestrant and placebo arm (p< 0.001) [38]. The activity of CDK 4/6 inhibitors in the treatment of advanced MaBC is not well known. In this report we present the initial response to fulvestrant and palbociclib in a male, who developed metastatic disease to the lung and bone, while undergoing adjuvant ET with tamoxifen.
CASE PRESENTATION
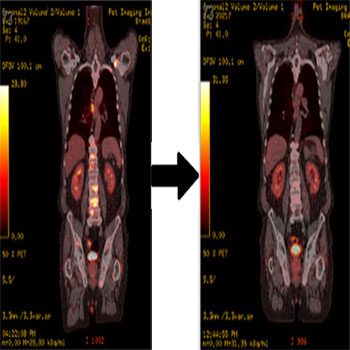
We present a 71-year-old male who was diagnosed with BC in March 2012. The patient underwent a left modified radical mastectomy with the finding of a 2.2 cm high grade invasive ductal carcinoma ER+, PR+, HER 2/neu negative, KI 67 25%, with 2 out of 24 axillary lymph nodes involved by carcinoma. He received adjuvant chemotherapy with dose-dense Adriamycin and Cyclophosphamide followed by 12 weeks of paclitaxel. He received adjuvant radiation therapy to the left chest wall and regional lymphatics (5040 cGy delivered in 28 fractions). Patient started adjuvant ET with tamoxifen in November 2012. Genetic testing revealed absence of deleterious mutations for the BRCA1 or BRCA2 genes. After doing clinically well for nearly four years, he developed progressive back and pelvic pain. Based on these findings, systemic recurrence of disease was suspected. 18F-fluorodeoxyglucose-positron emission tomography-computed tomography (FDG-PET/CT) scan (Figure 1A) demonstrated new multiple FDG avid mediastinal and right hilar adenopathy, mixed lytic and sclerotic bone metastases scattered in the axial and appendicular skeleton involving sternum, bilateral ribs, scapula, spine, pelvis and bilateral proximal femora. Tumor markers demonstrated elevation in CA 15-3 level to 109 U/mL (reference range: < 32 U/mL) and CEA level to 25 ng/mL (reference range: < 2.5 ng/mL). Bone biopsy of the right pubic area, revealed metastatic carcinoma consistent with a breast primary (ER +, PR+, HER 2/neu negative). The patient was started on therapy for stage IV ER+/HER2 negative breast cancer. His treatment consisted of a fulvestrant loading dose followed by a monthly 500 mg intramuscular (IM) dose in addition to oral palbociclib 125 mg given once daily for 3 weeks followed by 1 week off over a 28-day cycle. He also received intravenous zolendronic acid 4 mg monthly for the prevention of skeletal related events. Soon after starting therapy, the patient demonstrated symptomatic improvement of his bone pain. FDG-PET/CT scan following 3 months of systemic therapy is shown on Figure 1B. Tumor markers decreased in levels; CA 15-3 level was 49 U/mL and CEA level was 4.4 ng/mL. Treatment was well tolerated with minimum toxicity. Absolute neutrophil count remained above 1.5 1000/ uL throughout therapy.
Figure 1A and 1B: Radiographic response following 3 months of systemic therapy. A) FDG-PET/CT demonstrating new multiple FDG avid mediastinal and right hilar adenopathy, mixed lytic and sclerotic bone metastases scattered in the axial and appendicular skeleton. B) FDG-PET/CT demonstrating significant improvement with decreased FDG uptake of pulmonary, nodal and osseous metastases.
DISCUSSION
The case presented in this report demonstrates the effect of combining fulvestrant with palbociclib in the treatment of advanced MaBC. Fulvestrant works as a competitive inhibitor by interfering with the binding of estradiol to the ER in addition to downregulating the ER pathway [39].Fulvestrant was initially approved at the dose of 250 mg IM monthly following progression on an antiestrogen therapy such as tamoxifen [40]. However, pharmacokinetic findings from the phase III EFECT trial prompted researchers to explore a 500 mg dose [41]. Two clinical trials have explored fulvestrant in the front line treatment of advanced FBC. The FIRST phase II open-label study randomized postmenopausal patients with ER+/HER2 negative advanced BC to receive fulvestrant (n = 102) or anastrozole (n = 103). The study demonstrated an impressive time to progression of 23.4 versus 13.1 months favoring fulvestrant (p = 0.01) [42]. Overall survival was also improved to 54.1 months with fulvestrant compared to 48.4 months with anastrozole (p = 0.041) [43]. The confirmatory FALCON phase III clinical trial randomized 462 postmenopausal patients with advanced breast cancer in a 1:1 to receive the same dose and frequency of fulvestrant or anastrozole. The study met the primary end point PFS. Patients receiving fulvestrant had a PFS of 16.6 vs. 13.8 months for those receiving anastrozole (p = 0.0486) [44]. Based on these two studies and considering the impressive results of adding CDK 4/6 inhibitor to front line endocrine therapy in stage IV breast cancer, we chose to treat our patient with fulvestrant in combination with palbociclib. It is important to note that this patient was not considered to be ET refractory as metastatic disease was developed after 2 years of receiving adjuvant ET with tamoxifen. Therefore, he could have been treated with letrozole and palbociclib. Data demonstrates, however, that aromatase inhibitors could be inferior to tamoxifen in the adjuvant treatment of MaBC [45-48]. The data presented in this report could guide clinicians in need of selecting palliative therapies for HR+ advanced MaBC. No information is currently known on the effect of CDK 4/6 inhibitors in males with metastatic disease, as clinical trials of these agents have excluded males from participating. Of interest is the fact that the ET study MONALEESA-3 trial is evaluating ribociclib in combination with fulvestrant compared to fulvestrant alone in men and postmenopausal women with HR+/HER2 negative advanced BC in the second line endocrine therapy setting (NCT02422615).
REFERENCES
- Siegel RL, Miller KD and Jemal A. (2016). Cancer statistics. CA Cancer J Clin. 66(1): 7-30.
- Kreiter E, Richardson A, Potter J and Yasui Y. (2014). Breast cancer: trends in international incidence in men and women. Br J Can. 110(7): 1891-1897.
- Sasco AJ, Lowenfels AB and Pasker-de JP. (1993). Review article: epidemiology of male breast cancer. A meta-analysis of published case-control studies and discussion of selected aetiological factors. Int J Can. 53(4): 538-549.
- Anderson, WF, Jatoi I, Tse J, et al. (2010). Male breast cancer: population-based comparison with female breast cancer. J Clin Oncol. 28(2): 232-239.
- Leone JP, Zwenger AO, Iturbe J, Bhargava R, et al. (2016). Prognostic factors in male breast cancer: a populationbased study. Breast Cancer Res Treat. 156(3): 539-548.
- Hong JH, Ha KS, Jung YH and Woo IS. (2016). Clinical Features of Male Breast Cancer: Experiences from Seven Institutions Over 20 Years. Cancer Res Treat. 48(4): 1389-1398.
- Adami HO, Holmberg L, Malker B and Ries L. (1985). Longterm survival in 406 males with breast cancer. Br J Can. 52(1): 99-103.
- Brinton LA, Key TJ, Kolonel LN, Cook MB, et al. (2015). Prediagnostic Sex Steroid Hormones in Relation to Male Breast Cancer Risk. J Clin Oncol. 33(18): 2041-2050.
- Brinton LA, Richesson DA, Gierach, GL, Schatzkin A, et al. (2008). Prospective evaluation of risk factors for male breast cancer. J Natl Cancer Inst. 100(20): 1477-1481.
- Biesma HD, Schouten PC, Lacle MM, Linn SC, et al. (2015). Copy number profiling by array comparative genomic hybridization identifies frequently occurring BRCA2-like male breast cancer. Genes Chromosomes Cancer. 54(12): 734-744.
- Basham VM, Lipscombe JM, Ward JM, Ponder BAJ, et al. (2002). BRCA1 and BRCA2 mutations in a populationbased study of male breast cancer. Breast Cancer Res. 4(1): R2.
- Tai YC, Domchek S, Parmigiani G and Chen S. (2007). Breast Cancer Risk Among Male BRCA1 and BRCA2 Mutation Carriers. J Natl Cancer Inst. 99(23): 1811-1814.
- Zhou R, Yu L, Zhou S, Shui R, et al. (2014). Male breast carcinoma: a clinicopathological and immunohistochemical characterization study. Int J Clin Exp Pathol. 7(10): 6852-6861.
- Sharif MA, Mamoon N, Arif A, Khadim MT, et al. (2009). Histological and immuno-histochemical study of male breast carcinoma in Northern Pakistan. J Pak Med Assoc. 59(2): 67-71.
- Sanchez-Munoz A, Roman-Jobacho A, Parez-Villa L, Alba E, et al. (2012). Male Breast Cancer: Immunohistochemical Subtypes and Clinical Outcome Characterization. Oncology. 83(4): 228-233.
- Deb S, Lakhani SR, Ottini L and Fox SB. (2016). The cancer genetics and pathology of male breast cancer. Histopathology. 68(1): 110-118.
- Johansson I, Nilsson C, Berglund P, Hedenfalk I, et al. (2011). High-resolution genomic profiling of male breast cancer reveals differences hidden behind the similarities with female breast cancer. Breast Cancer Res Treat. 129(3): 747-760.
- Johansson I, Nilsson C, Berglund P, Hedenfalk I, et al. (2012). Gene expression profiling of primary male breast cancers reveals two unique subgroups and identifies Nacetyltransferase-1 (NAT1) as a novel prognostic biomarker. Breast Cancer Res. 14(1): R31.
- Johansson I, Killander F, Linderholm B and Hedenfalk I. (2014). Molecular profiling of male breast cancer - lost in translation? Int J Biochem Cell Biol. 53: 526-535.
- Shaaban AM, Ball GR, Brannan RA, Speirs V, et al. (2012). A comparative biomarker study of 514 matched cases of male and female breast cancer reveals gender-specific biological differences. Breast Cancer Res Treat. 133(3): 949-958.
- Giordano SH. (2005). A review of the diagnosis and management of male breast cancer. Oncologist. 10(7): 471-479.
- Lumachi F, Luisetto G, Basso SM, Camozzi V, et al. (2011). Endocrine therapy of breast cancer. Curr Med Chem. 18(4): 513-522.
- Osborne CK and Schiff R. (2011). Mechanisms of endocrine resistance in breast cancer. Annu Rev Med. 62: 233-247.
- Shapiro GI. (2006). Cyclin-dependent kinase pathways as targets for cancer treatment. J Clin Oncol. 24(11): 1770-1783.
- Miller TW, Balko JM, Fox EM, Arteaga CL, et al. (2011). ERa-dependent E2F transcription can mediate resistance to estrogen deprivation in human breast cancer. Cancer Discov. 1(4): 338-351.
- Dean JL, Thangavel C, McClendon AK, Knudsen ES, et al. (2010). Therapeutic CDK4/6 inhibition in breast cancer: key mechanisms of response and failure. Oncogene. 29: 4018-4032
- Klein EA and Assoian RK. (2008). Transcriptional regulation of the cyclin D1 gene at a glance. J Cell Sci. 121: 3853- 3857.
- Santo L, Siu KT and Raje N. (2015). Targeting Cyclin-Dependent Kinases and Cell Cycle Progression in Human Cancers. Semin Oncol. 42(6): 788-800.
- Turner NC, Bartlett CH and Cristofanilli M. (2015). Palbociclib in Hormone-Receptor-Positive Advanced Breast Cancer. N Engl J Med. 373(3): 209-219.
- Toogood PL, Harvey PJ, Repine JT, Fry DW, et al. (2005). Discovery of a potent and selective inhibitor of cyclindependent kinase 4/6. Journal of medicinal chemistry. 48(7): 2388-2406.
- Fry DW, Harvey PJ, Keller PR, Toogood PL, et al. (2004). Specific inhibition of cyclin-dependent kinase 4/6 by PD 0332991 and associated antitumor activity in human tumor xenografts. Mol Cancer Ther. 3(11): 1427-1438.
- Finn RS, Dering J, Conklin D, Kalous O, et al. (2009). PD 0332991, a selective cyclin D kinase 4/6 inhibitor, preferentially inhibits proliferation of luminal estrogen receptor-positive human breast cancer cell lines in vitro. Breast Cancer Res. 11(5): R77.
- Ehab M, Elbaz M. (2016). Profile of palbociclib in the treatment of metastatic breast cancer. Breast Cancer (Dove Med Press). 8: 83-91.
- Qin G, Xu F, Qin T, Shi D, et al. (2015). Palbociclib inhibits epithelial-mesenchymal transition and metastasis in breast cancer via c-Jun/COX-2 signaling pathway. Oncotarget. 6(39): 41794-41808.
- VanArsdale T, Boshoff C, Arndt KT, and Abraham RT. (2015). Molecular Pathways: Targeting the Cyclin DCDK4/6 Axis for Cancer Treatment. Clin Cancer Res. 21(13): 2905-2910.
- Anders L, Ke N, Hydbring P, Choi YJ, et al. (2011). A systematic screen for CDK4/6 substrates links FOXM1 phosphorylation to senescence suppression in cancer cells. Cancer cell. 20(5): 620-634.
- Finn RS, Martin M and Rugo HS. (2016). PALOMA-2: Primary results from a phase III trial of palbociclib (P) with letrozole (L) compared with letrozole alone in postmenopausal women with ER+/HER2- advanced breast cancer. J Clin Oncol. 34: 507.
- Turner NC, Ro J and Andre F. (2015). PALOMA3: A doubleblind, phase III trial of fulvestrant with or without palbociclib in pre- and post-menopausal women with hormone receptor-positive, HER2-negative metastatic breast cancer that progressed on prior endocrine therapy. J Clin Oncol. 33: LBA502.
- Cristofanilli M, Turner NC, Bondarenko I, Ro J, et al. (2016). Fulvestrant plus palbociclib versus fulvestrant plus placebo for treatment of hormone-receptor-positive, HER2- negative metastatic breast cancer that progressed on previous endocrine therapy (PALOMA-3): final analysis of the multicentre, double-blind, phase 3 randomised controlled trial. Lancet Oncol. 17(4): 425-439.
- Bross PF, Cohen MH, Williams GA and Pazdur R. (2002). FDA drug approval summaries: fulvestrant.Oncologist. 7(6): 477-480.
- Chia S, Gradishar W, Mauriac L, Bines J, et al. (2008). Double-blind, randomized placebo controlled trial of fulvestrant compared with exemestane after prior nonsteroidal aromatase inhibitor therapy in postmenopausal women with hormone receptor-positive, advanced breast cancer: results from EFECT. J Clin Oncol. 26: 1664-1670.
- Robertson JF, Lindemann JP, Llombart-Cussac A, Ellis MJ, et al. (2012). Fulvestrant 500 mg versus anastrozole 1 mg for the first-line treatment of advanced breast cancer: follow-up analysis from the randomized 'FIRST' study. Breast Cancer Res Treat. 136(2): 503-511.
- Robertson JF, Llombart-Cussac A, Feltl D, Dewar JA, et al. (2015). Fulvestrant 500 mg versus anastrozole as first-line treatment for advanced breast cancer: Overall survival from the Phase II 'FIRST' study. Cancer Research. 75(suppl): S6-S4.
- Ellis MJ, Bondarenko I, Trishkina E, Panasci L, et al. (2016). FALCON: A phase III randomised trial of fulvestrant 500 mg vs. anastrozole for hormone receptor-positive advanced breast cancer. Annals of Oncology. 27(suppl 6): LBA14_PR.
- Eggemann H, Ignatov A, Smith BJ, Altmann U, et al. (2013). Adjuvant therapy with tamoxifen compared to aromatase inhibitors for 257 male breast cancer patients. Breast Cancer Res Treat. 137(2): 465-470.
- Doyen J, Italiano A, Largillier R, Thyss A, et al. (2010). Aromatase inhibition in male breast cancer patients: biological and clinical implications. Ann Oncol. 21: 1243-1245.
- Finn RS, Crown JP, Lang I, Fowst C, et al. (2015). The cyclin-dependent kinase 4/6 inhibitor palbociclib in combination with letrozole versus letrozole alone as first-line treatment of oestrogen receptor-positive, HER2-negative, advanced breast cancer (PALOMA-1/TRIO-18): a randomised phase 2 study. Lancet Oncol. 16(1): 25-35.
- Osborne CK, Wakeling A and Nicholson RI. (2004). Fulvestrant: an oestrogen receptor antagonist with a novel mechanism of action. Br J Cancer. 90(1 Suppl): S2-S6.