Information Links
Related Conferences
Current Issue Volume 11, Issue 1 - 2026
Eosinophilic Esophagitis is GERD against the Background of Allergies: Hypothesis. Diagnosis and Treatment
Michael D Levin*
Dorot. Medical Center for Rehabilitation and Geriatrics, Netanya, Israel
*Corresponding Author: Michael D Levin, MD, Ph.D, Dorot. Medical Center for Rehabilitation and Geriatrics, Department of Pediatric Radiology of the 1-st State Hospital, Minsk, Belarus, Dorot. Medical Center for Rehabilitation and Geriatrics, Amnon veTamar, Netanya, Israel, Tel: 972-538281393, Email: [email protected]
Received Date: January 09, 2026
Published Date: January 28, 2026
Citation: Levin MD. (2026). Eosinophilic Esophagitis is GERD against the Background of Allergies: Hypothesis. Diagnosis and Treatment. Mathews J Gastroenterol Hepatol. 11(1):38.
Copyrights: Levin MD. © (2026).
ABSTRACT
Currently, it is considered that eosinophilic esophagitis (EoE) is an independent disease, which is often combined with gastroesophageal reflux disease (GERD). The clinical picture and histological findings are like GERD, but it should be distinguished from GERD, because these two conditions require different treatment. Based on the analysis of the literature and our own research, it was shown that (1) eosinophilic infiltration can affect other parts of the gastrointestinal tract. (2) PPI treatment is equally effective for both EoE and GERD. (3) Although the frequency of coincidence of EoE with GERD is not precisely determined, it is close to 100%. (4) The use of a lactose-free diet causes a clinical effect and a decrease in the number of eosinophils, both in EoE and GERD. (5) The radiographic picture of EoE indicates a dysfunction (shortening) of the lower esophageal sphincter (LES), which is characteristic of GERD. These facts suggest that EoE is an acid-dependent disorder like GERD, which differs from GERD in that it is an allergic reaction to inflammation caused by refluxate. The proposed diagnostics is based on reliable clinical symptoms (dysphagia, heartburn) combined with allergies, along with radiographic evidence of narrowing of the esophageal lumen and dysfunction of the LES. In the presence of the above symptoms, histological examination to determine the eosinophil count is not necessary, as recent data show that histology is not useful for differential diagnosis between GERD and EoE. Therefore, treatment for EoE involves treating both GERD and allergies. I propose a scientific discussion of the hypothesis of the pathophysiology of EoE and the advisability of modifying my proposed diagnostic and treatment methods for EoE.
Keywords: Eosinophilic Esophagitis, Gastroesophageal Reflux Disease, Allergy, Acid-Related Diseases, Lactose Intolerance, Pathophysiology, Diagnostics, Treatment.
ABBREVIATIONS
EoE: Eosinophilic Esophagitis; GERD: Gastroesophageal Reflux Disease; PAF: Platelet Activating Factor; LES: Lower Esophageal Sphincter; SCJ: Squamo-Columnar Junction; EGIDs: Eosinophilic Gastrointestinal Diseases; RAG: Rigid Antral Gastritis; SR: Schatzki Ring.
INTRODUCTION
EoE was first described as an entity distinct from GERD in 1993 by Attwood and colleagues who observed increased intraepithelial esophageal eosinophils (>20 eos/hpf, mean of 56 eos/hpf) and squamous epithelial hyperplasia in 12 adults with dysphagia in the absence of GERD (that is, with normal findings on endoscopy and 24 h pH testing) [1]. American Gastroenterological Association Institute and the North American Society of Pediatric Gastroenterology, Hepatology and Nutrition defined EoE in 2007 as a clinic-pathological disorder characterized by esophageal symptoms, biopsies showing >15 eos/hpf and the absence of pathological GERD as shown by normal pH testing or lack of response to high-dose PPI treatment. This definition implied that GERD and EoE were mutually exclusive conditions [2]. The consensus definition of EoE (2011) no longer excluded the possibility of combination with GERD [3]. It is believed that EoE and GERD differ from each other and require different treatment. Thirty years have passed since 1993. During this time, physicians dedicated to the study of EoE met twice for conferences. But instead of publishing the discussion materials, as is customary in the scientific world, they published the "arithmetic mean" of their opinions—consensus statements. This may be why it has only recently been found that these two conditions are very similar in clinical manifestations, histological findings, and radiological findings. They allegedly often coexist, and therefore, in many cases, there are serious diagnostic difficulties [4-6].
Difficulties in differentiating between EoE and GERD are due to the loss of diagnostic criteria on which the diagnosis was based by the authors who first described EoE. For example, currently no authors recommend using the pH test, which was the main differential diagnostic method between EoE and GERD in the article by Attwood et al. [1]. This is because the "physiological reflux" is seen in as many as 25% of patients with documented erosive esophagitis [2]. The recommendation not to use pH monitoring is justified, as it contradicts scientific data and common sense. The studies using the preparation of human esophageal mucosa have found that one-time penetration acid stimulates the release of platelet activating factor (PAF), a phospholipid that can attract and activate eosinophils [5]. This means that the penetration of aggressive gastric chyme into the esophagus with a pH <4 to <3.2% in 24 hours (about 45 minutes per day) cannot help but damage the esophagus [7,8].
The widespread promotion of pH monitoring has distorted many ideas about GERD. For example, a review article by Dellon states that "The motility may be altered in these (EoE) patients, and they may have issues with clearing of physiological normal reflux [4]. As shown above, physiological, i.e., normal reflux, cannot exist. It follows that the described EoE patients had as severe GERD, since, firstly, there was refluxate, and it was also poorly removed from the esophagus. In another observation, the authors diagnosed EE with a 1 cm hiatal hernia, presumably GERD. However, what is commonly called a hiatal hernia is a phrenic ampulla, which occurs because of esophageal dilation under the influence of hydrochloric acid [7]. The idea that the ampulla can exist without GERD arose due to the low reliability of pH monitoring [7,8].
The clinical picture also confirms the presence of reflux in EoE, because studies reporting heartburn as a common symptom of EoE [9]. “The primary symptoms of EoE particularly in adolescents and adults are dysphagia and food impaction” [4]. At the same time, all authors claim that: - "At the present time, an adequate biomarker to distinguish these entities is lacking" [4-6,10]. For years it was thought that eosinophils in a biopsy sample were a hallmark of reflux, and it was a finding that was not clinically actionable. In all patients with EoE, eosinophil counts are higher distally than proximally. It has now become clear that even the presence of distal eosinophilia alone does not distinguish between GERD and EoE. In some cases, even a small number of eosinophils can be accompanied by dysphagia, while in others, large numbers of eosinophils can be present without clinical signs of EoE. Therefore, it comes down to synthesizing data on the history, symptoms, endoscopic findings, and biopsies. Sometimes ancillary testing is needed [4].
All articles devoted to EoE describe a limited number of observations, which are assessed based on the consensus decision at the time of publication. It should be acknowledged that there is a lack of theoretical justification for the etiology and pathogenesis of the disease. For example, initially, it was believed that GERD and EoE were distinct and incompatible diseases. Currently, based on numerous observations, it is believed that a person has EoE and GERD simultaneously, and the two conditions are unrelated. However, since this assertion lacks reliable theoretical support, two other options cannot be ruled out. (1). Because the esophagus is inflamed and has decreased compliance in EoE, patients can have secondary reflux. (2). A patient with reflux may have either erosive disease or a microscopic barrier disruption, allowing allergens to penetrate from the esophageal lumen into the mucosa. In this case, a secondary picture like GERD develops, which may be largely due to GERD [4]. In a previous study, I presented a hypothesis about the etiology and pathogenesis of EoE that, when tested, does not contradict existing scientific data [11].
The purpose of this study is to substantiate methods for diagnosing and treating patients with dysphagia based on the proposed hypothesis.
Before settling on the proposed hypothesis, it is necessary to determine reliable diagnostic symptoms and signs of GERD and EoE.
Diagnosis of GERD
The presence of heartburn supports the diagnosis of GERD. Gastroscopy reveals only reflux complications: erosion, ulcers, peptic stenosis, and Barrett's disease. Dilation of intercellular spaces was the most consistently reported histologic change in the esophageal mucosa of patients with nonerosive reflux disease, being observed in 41%-100% of patients and 0%-30% of controls. The mean intercellular space width was at least 2 times greater in patients than in controls, irrespective of esophageal acid exposure. Leukocytes and eosinophils participate in the inflammatory process [12]. Chandrasoma et al., believe that the appearance of cardiac epithelium over the lower esophageal sphincter (LES) occurs due to cardiac metaplasia of the squamous epithelium due to exposure to gastric juice. Thus, cranial displacement of the squamo-columnar junction (SCJ) is indicative of GERD. In the early stages of development, acid damaged only the intra-abdominal portion of the LES. In such cases, it does not yet enter the esophagus and cannot be detected by pH monitoring. This also implies that physiological reflux is an error in using pH monitoring. They also showed that acid exposure to the intra-abdominal portion of the LES leads to its damage. The LES opens, resulting in shortening of its functional portion [13]. X-ray examination with increased gastric pressure reveals shortening of the LES relative to the age norm, dilation of the esophagus, to a greater extent above the LES, i.e., in the phrenic ampulla [8]. (Figure 1).
Diagnosis of ЕоЕ
The main clinical symptom of EoE is dysphagia with intermitted food impactions. EoE is described as a chronic immune-mediated disease and is usually accompanied by allergic disease associated with type 2 inflammation. Normally, i.e., in healthy people, there are no inflammatory cells, including eosinophils, on the wall of the esophagus. Eosinophilia is an allergic reaction to inflammation [14]. Dysphagia is caused by thickening of the esophageal wall and narrowing of its lumen, which is determined by radiographic examination. Radiographic examination also reveals dysfunction of the LES [11]. Many other characteristics, such as heartburn, response to PPI treatment with a decrease in eosinophils in biopsy, do not differ between GERD and EoE.
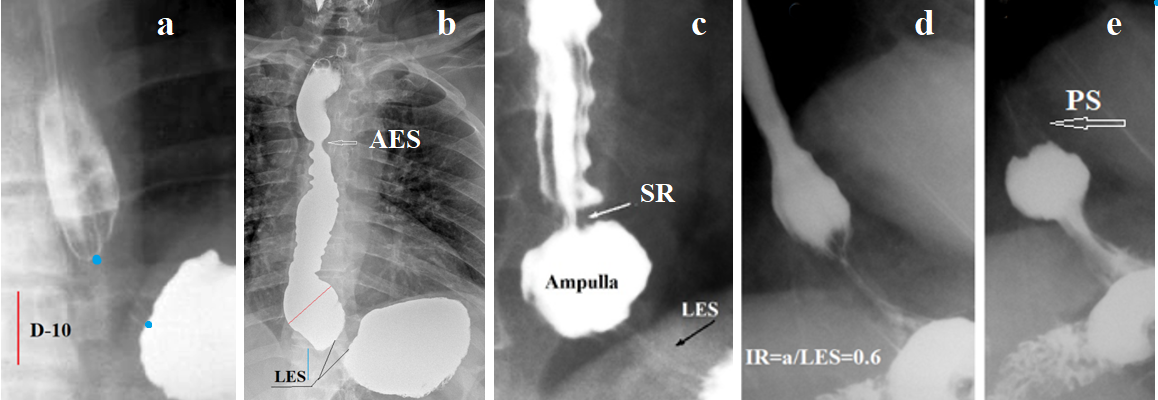
Figure 1. Radiographs of the esophagus and esophagogastric junction taken during high pressure in the stomach. (a) In a patient with mild GERD, the LES is contracted during abdominal compression. Its true length between the two blue dots is 2.4 cm. (The true height of D-10 is 2 cm). (b) A patient with a long history of GERD and non-esophageal symptoms. The LES length is 0.7 cm. Symmetrical narrowing of the esophagus at the level of the aortic arch (aorto-esophageal sphincter - AES) was the cause of the non-esophageal symptoms. (c) Wide esophageal ampulla and narrow esophagus with longitudinal and transverse folds. Between the ampulla and the esophagus, a narrowing at the site of the proximal sphincter - Schatzki ring (SR) is determined. The combination of reflux esophagitis with a narrow esophagus and SR is suggestive of EoE. (d-e) EoE with a narrow lumen and rigid esophageal wall. A small ampulla indicates GERD. To create high pressure, it is closed by a contracted proximal sphincter (PS) and injects barium into the stomach.
None of the hypotheses regarding the etiology and pathogenesis of EoE are supported by scientific data.
Eosinophilic gastrointestinal diseases
For example, the isolation of EoE as an independent disease contradicts studies showing that EoE is part of a group of eosinophilic gastrointestinal diseases (EGIDs) with chronic inflammation of the gastrointestinal tract, which are characterized clinically by symptoms related to the dysfunction of the involved segment(s) of the GI tract and histologically by dense eosinophilic inflammation, in the absence of an identifiable secondary cause.The group of EGIDs comprises EoE, eosinophilic gastritis, eosinophilic gastroenteritis, and eosinophilic colitis. Eosinophilic infiltration can be found in several parts of the digestive tract in one patient [15]. Mahendra et al showed that duodenal eosinophilia was associated with symptomatic erosive GERD [16]. Eosinophilic infiltration of the antrum causes marked thickening of the gastric wall, which is not involved in peristalsis. This type of gastritis is called "rigid antral gastritis" (RAG) [17]. (Figure 2).
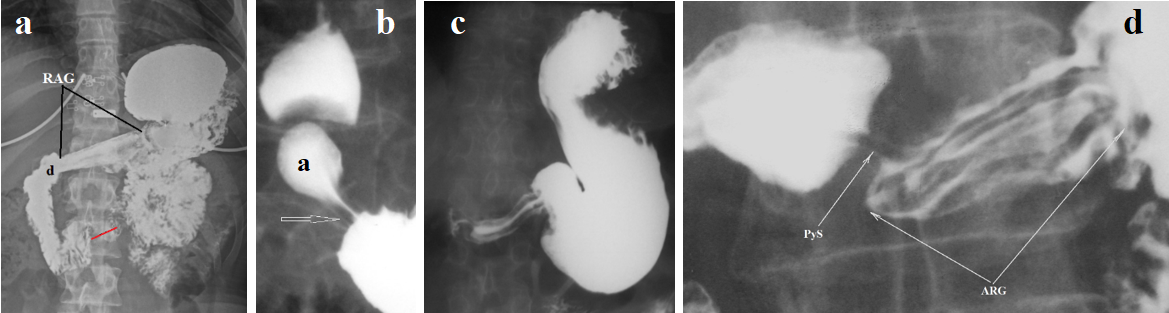
Figure 2. Radiographic appearance of rigid antral gastritis (RAG). (a) A 19-year-old overweight woman complained of abdominal pain. She began vomiting after eating six months ago. During this time, she lost 5 kg. A gastroscopy diagnosed GERD. No pathology was found in the stomach or duodenum. She is allergic to chicken feathers. X-ray examination during high pressure in the stomach revealed: 1. The gastric antrum is narrowed, straightened, with thickened walls and a narrow lumen, consistent with the appearance of rigid antral gastritis (RAG). 2. The duodenal bulb (d) is irregularly shaped. In the third part of the duodenum, contraction of the sphincter of Ochsner (red line) is visible, indicating hypersecretion of hydrochloric acid. (b) A normal gastroduodenal radiograph is shown as an example. Antral peristalsis begins with contraction of the antral sphincter (arrow). The antrum (a) has elastic walls and a wide lumen. The duodenal bulb has a typical arrowhead shape. (c-d) Examples of RAG.
The combination of GERD with impaired gastric emptying due to antral stenosis and thickened walls, along with dyskinesia of the duodenal bulb and Ochsner sphincter, coupled with allergies, allows for a diagnosis of hydrochloric acid hypersecretion with damage to the esophagus, stomach, and duodenum, with eosinophilic infiltration of the walls of the affected segments of the gastrointestinal tract. These data indicate that hydrochloric acid is the primary damaging factor causing the inflammatory process. The body's allergic reaction transforms the inflammation into eosinophilic infiltration.
Response to PPI treatment
PPI use is known to be effective in the treatment of EoE. It is alleviating symptoms and reduces the number of eosinophils. Its effect reaches 33 to 70%, almost the same as for GERD [18-21]. To rule out reflux as a cause of EoE, 66 doctors (2018) in the proceedings of the AGREE conference stated that gastric acid inhibition is not the only important effect of PPIs, suggesting, but without evidence, at the possibility of an anti-inflammatory effect of PPIs [18]. However, what "maybe" but has no scientific evidence should not be considered. The effectiveness of GERD treatment in EoE can be judged by the following case report.
Analysis of the medical history of a 16-year-old girl who first contacted me in 2021 with complaints of abdominal pain, which appeared about 6 years ago. Difficulty swallowing has appeared in the last 2-3 months. During this time, she lost 15 kg.
Anamnesis. All close relatives on my father’s and mother’s sides had acid-related diseases. She was a calm baby but did not sleep at night. Allergic rhinitis and conjunctivitis were diagnosed in early childhood. Since then, has been taking Aerius 5 mg continuously. She was found to be allergic to many substances and foods. She is under supervision by a pediatric gastroenterologist with a diagnosis of GERD. At the age of 13, the test of stool for Helicobacter Pylori was positive. During gastroscopy, lymphoid hypertrophy was found in the duodenal bulb. Eradication of Helicobacter Pylori completed. In the next 3 years, she did not go to a gastroenterologist. From the age of 12, she had low hemoglobin (9.3 g / dl). She received treatment for anemia for two years. At the age of 8, attacks of shortness of breath appeared, which eventually became exceedingly rare. In the family, these attacks were called asthma and the patient used inhales, but doctors did not confirm this diagnosis. At the age of 15, dysphagia appeared. Omeprazole 20 mg was prescribed once a day, but the patient believed that after taking Omeprazole, abdominal pain became more frequent, and she stopped taking it. When she started taking Esomeprazole 20 mg in the morning before meals, there was a significant improvement.
Clinical symptoms (2021). During the first consultation, she complained about pain in the epigastrium and on the right, at night. She used a warm heating pad to relieve pain. During the day, pain occurred behind the breastbone after eating, together with the feeling that food was stuck in the esophagus. She began to eat very slowly, chewing food thoroughly and washing it down with water. Often bitterness in the mouth appears, especially in bed.
Ultrasound of the abdomen. Multiple small stones in the gallbladder without signs of inflammation (Figure 3а). Figure 3 shows radiographs taken at maximum gastric pressure (Figure 3b) and after 5 minutes at rest (Figure 3c).
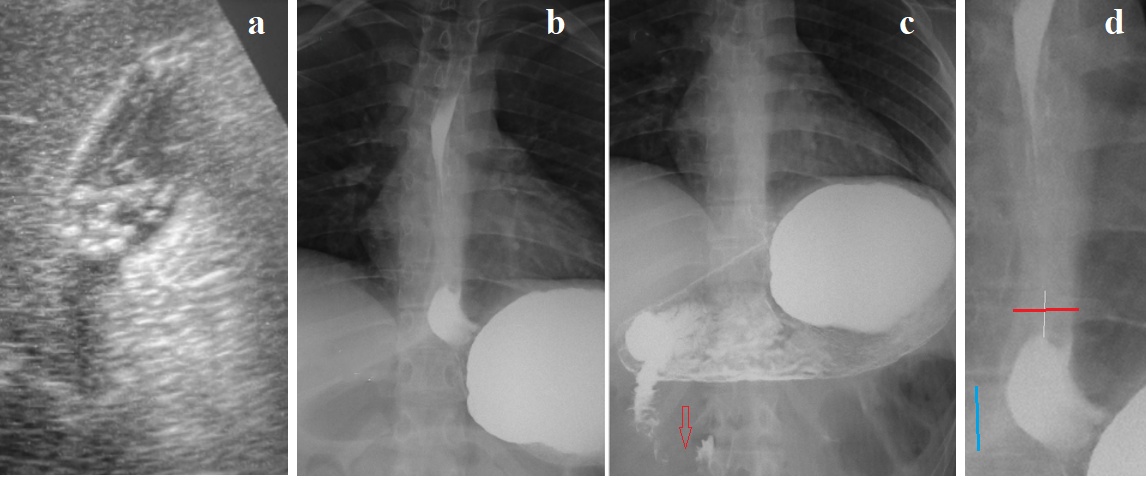
Figure 3. Examination of a 16-year-old girl. (a) Small stones in a contracted gallbladder. (b) Radiograph taken during straight legs raise at the end of a continuous barium drink. Contrast medium is absent between the 6th and 8th thoracic vertebrae above the phrenic ampulla. A significant amount of barium is in the stomach, indicating good fluid permeability through the esophagus. The diagram of this radiograph (Figure d) shows thickening of the walls in the lower part of the esophagus and shortening of the LES (the distance between the ampulla and the stomach). (c) After 5 minutes at rest, finely wavy contours of the second part of the duodenum and contraction of the sphincter of Ochsner (red arrow) are visible.
Based on medical history, clinical presentation (dysphagia, abdominal pain, GERD, allergy), and radiological findings (gallstones, lower esophageal wall thickening, shortening of the LES, and duodenal dyskinesia, including contraction of the Ochsner sphincter), a diagnosis of hydrochloric acid hypersecretion, GERD, and duodenal dyskinesia was made. Taken together, these findings suggested the possibility of EoE. Endoscopy with a lower esophageal biopsy was recommended. Endoscopy revealed no esophageal pathology, diagnosed nodular gastritis, and found some erosions in the duodenal bulb. Histological examination revealed no esophageal or gastric pathology. However, no specific eosinophil count was performed. I recommended continuing the PPI and antiallergic medication, lying down only on an empty stomach, and avoiding lactose-containing products. From then (2021) to today (2025), the patient has been feeling well. She is studying at university and has not seen any more doctors.
The idea that a lack of response to PPI treatment rules out GERD has no scientific basis. This is due to the low diagnostic accuracy of pH monitoring and the misconception that physiological reflux is possible. Normal secretion, or even hyposecretion, of hydrochloric acid during PPI use does not prevent esophageal damage if the LES is weakened and opens when gastric pressure increases. Gastric chyme, which has low acidity but breaks down dietary proteins, also breaks down proteins in the esophageal wall.
Dietary intervention
Dietary intervention studies provide significant information regarding the pathogenesis of GERD and EoE. For example, pediatric patients with refractory GERD who had failed prior medical and surgical treatments directed at GERD found that treatment with an elemental formula resulted in marked reduction in intraepithelial esophageal eosinophils and improvement in symptomatology, introducing the concept that a food-protein-induced, allergic mechanism was responsible for the pathogenesis of EoE [4]. Kliewer et al. compared the effectiveness of a milk elimination nutrition program (1FED) with a 4-ingredient elimination nutrition program (4FED), including milk. Although 4FED moderately improved symptoms compared with 1FED, the histologic, endoscopic, QoL, and transcriptomic outcomes were similar in both groups. They concluded that 1FED is a reasonable first-choice therapy for pediatric EoE, given its effects, tolerability, and relative simplicity [3]. These studies show that EoE and GERD respond similarly to milk exclusion. However, there is no evidence that milk protein is the chemical element that causes pathology. Moreover, an allergy to milk protein is very rare (2-3%) and is accompanied by a severe reaction, even death [22]. Høst et al. state that no single laboratory test is diagnostic of milk protein allergy [22]. Gastrointestinal manifestations after milk ingestion are often not mediated by IgE, and a clinical response to a cow's milk-free diet does not prove immune system involvement [22,23]. They may be a consequence of GERD [23]. My research has shown that in patients with lactose intolerance, eliminating lactose-containing dairy products from diet leads to significant improvement symptoms of both GERD and EoE [24,25]. This is because undigested lactose causes an increase in mast cells in the small intestinal mucosa, which leads to hypersecretion of hydrochloric acid [24]. The exclusion of all products containing lactose enhances the therapeutic effect of the diet. This is especially important for patients over 50 years of age, because the older the patient, the less lactose quantity causes an exacerbation of acid-related problems.
On the coincidence of GERD and EoE
Literary analysis indicates that EoE is associated with GERD. It is well known that GERD and EoE may be accompanied by eosinophilia in mucosal biopsies. Therefore, the question of a possible connection between these diseases has repeatedly arisen [18]. Monnerat and Lemme, using pH monitoring, found pathological reflux in 25% of patients with EoE [19]. Pesce et al found that Higher esophageal acid exposure time and lower baseline impedance values were significantly associated with eosinophilic infiltration (P < .05 and P < .01, respectively) [10]. Frazzoni et al, using impedance-pH monitoring, concluded that reflux plays a role in the pathogenesis of EoE [20]. We should not be confused by the low percentage of reflux detection (25%), because it is known that pH monitoring detects only severe forms of GERD. Currently, pH monitoring is not recommended for the diagnosis of EOE because the "physiological reflux" is seen in as many as 25% of patients with documented erosive esophagitis [2]. This means, firstly, that the coincidence rate is at least 50%. Secondly, pH monitoring, which was considered the only accurate method for diagnosing GERD, has ceased to be effective. At the same time, as shown in numerous studies, the number of eosinophils in a special histological study also turned out to be unreliable in the differential diagnosis of GERD and EOE [4-6,10]. These results suggest that reflux of aggressive gastric contents into the esophagus is observed in all patients with EOE.
X-ray image of the esophagus and LES in EoE
All authors pay attention to the narrow lumen of the esophagus but do not compare it to any normal values. The presence of erosions and ring-like indentations is considered characteristic of EoE. Since patients with GERD always shortens the LES, I searched the literature for radiographic studies of patients with EoE where the EGJ was captured. In each of the 10 such cases, LES damage was found, characteristic of GERD (Figure 4).
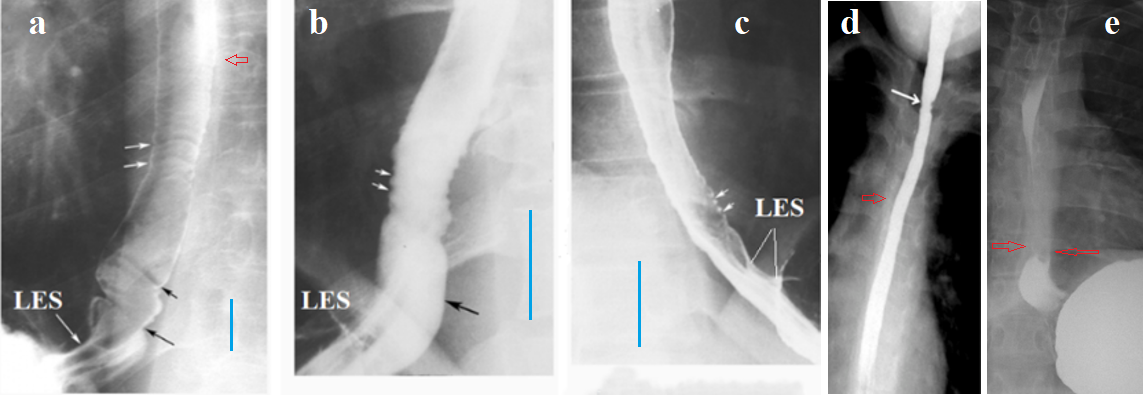
Figure 4. Radiographs of the esophagus and LES in patients with EoE. (a, b, c) from the article by Zimmerman et al. [26]. (a) In addition to ring-like indentations, the authors describe a smooth tapered structure located between the esophagus and the stomach. This is a short LES with several longitudinal folds (my designation), which is indicative of GERD. (b) I have shown the intra-abdominal, sharply dilated part of the LES (cardiac chalasia) located under the diaphragm. (c) Shortening of the LES due to the opening of its intra-abdominal (under the diaphragm) part. On the radiographs, the blue line marks the height of the 7th or 8th thoracic vertebrae, which is approximately 1.5 cm in adults. Based on this, the width of the esophageal lumen in patient (a) is clearly not narrowed, but in patients (b-c) there is a significant narrowing of the lumen. (d) From the article by Al-Hussaini et al. [27]. A sharp narrowing of the esophageal lumen is determined without signs of peristalsis. (e) From my observation (Figure 3d). The distance between the esophagus and the stomach is sharply shortened, indicating a shortening of the LES. Thickening of the esophageal walls forms a white line that obscures the bony elements of the thoracic vertebrae. The red arrows indicate this obscuration on other radiographs.
An analysis of radiographs of patients with EoE that captured the EGJ revealed significant shortening of the LES in all patients, suggesting GERD. We can conclude that significant narrowing of the esophageal lumen is compelling evidence of esophageal wall thickening and a high probability of EoE.
EoE is a cause of Schatzki ring
Schatzki first reported a ring-like structure at the esophagogastric mucosal junction in 1953. There is still no uniform agreement as to its exact location, etiology, or clinical importance. Johnson et al. found Schatzki ring (SR) in 15% to 18% of 22,368 patients having routine upper gastrointestinal examinations [28]. SR is commonly associated with hianal hernia (96%), EoE (40%) and GERD (40%) [29]. Sarbinowska et al found the increase in TGF-β1 and MBP concentrations, which indicates the inflammatory and probably fibrostenotic pathogenesis of SR. Obtained results do not allow for an unequivocal classification of SR as a complication typical only for GERD or EoE [30]. The relationship between the localization of SR and the width of the esophagus and the state of the LES is shown in Figure 3. I have selected typical cases SR where peptic stenosis could be excluded (Figure 5).
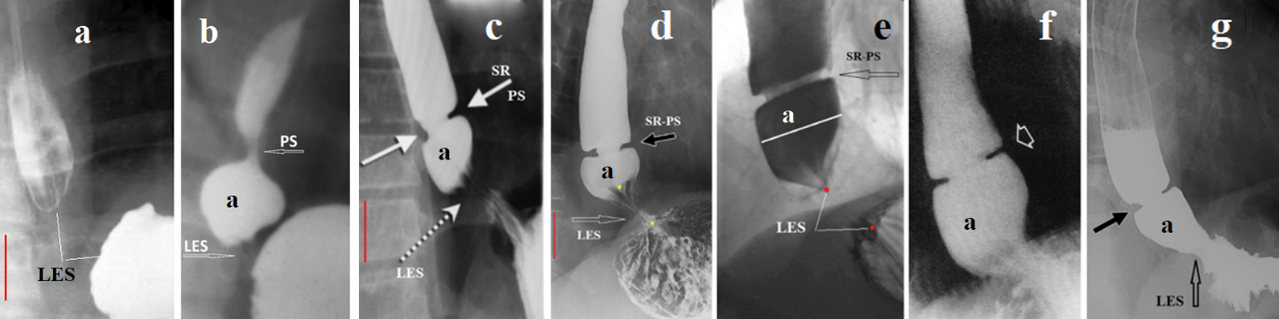
Figure 5. Radiographs of the EGJ explaining the location and pathogenesis of the Schatzki ring (SR). (a) Early-stage GERD. The contracted LES (without barium) is located between the esophagus and stomach. (b) In severe GERD, the esophagus dilates and the LES shortens. To create high pressure, the peristaltic wave in the wide esophagus is closed proximally by the functional proximal sphincter (PS). Continuing to contract, the ampulla creates high pressure between the PS and LES, which leads to opening of the LES, and the ampulla injects its contents into the stomach. Radiographs (c-g) show that the SR is in the location where the PS typically functions in GERD.
Shortening of the LES (d, e), with chalasia of the EGJ (f, g) and the presence of longitudinal folds (c) indicate GERD. Narrowing of the esophagus and ampulla relative to the height of the thoracic vertebra (red line) and the absence of peristalsis confirm the diagnosis of EoE. The proximal sphincter is not an anatomical sphincter, but a functional sphincter that occurs only in GERD. Over time, it transforms into a wide fibrous ring, which is mistaken for the edge of the stomach that has penetrated the chest. Infiltration with eosinophils leads to a sharp decrease in the diameter of the PS. Thus, the SR is a fibrous ring of the PS, which becomes narrow because of eosinophilic infiltration.
Hypothesis of the pathogenesis of EoE
The above analysis shows that all patients with EoE have GERD. If we discard assumptions that have no scientific evidence, as well as statements that contradict known scientific facts, then the pathophysiology of EoE is as follows. Eosinophilic esophagitis is a reflux esophagitis in individuals with an allergic reaction to various allergens. Hypersecretion of hydrochloric acid disrupts the integrity of esophageal mucosa and causes an inflammatory reaction. Most often or always the trigger for hypersecretion is lactose in patients with lactose intolerance. In EE and GERD, an increase in the number of eosinophils, basal cell hyperplasia, intercellular edema, and elongation of epithelial papillae are observed. There is no absolute histological criterion allowing distinction between EoE and GERD, and cutoff values for numbers of eosinophils vary according to studies and authors. Only the exclusion of foods containing lactose from food leads to clinical and histological improvement. Esophageal narrowing that leads to dysphagia does not occur suddenly but is the culmination of a process that began long before dysphagia and was manifested by GERD symptoms. Since hypersecretion of hydrochloric acid affects all parts of the digestive tract, it can lead to eosinophilic infiltration of other parts and cause a violation of their functions.
Diagnosis of EoE from the point of view of the proposed hypothesis of pathogenesis
Thirty-two years have passed since Attwood and colleagues first discovered eosinophilia in the esophageal wall in patients with dysphagia in 1993 [1]. During this time, numerous studies have uncovered important patterns. It turned out that clinical, histological, and pH-metric studies, which were initially considered valuable for the differential diagnosis of EoE from GERD, are ineffective. This is since EoE is GERD that develops against the background of an allergic reaction, which leads to eosinophilic infiltration of the esophageal wall. It follows that the combination of clinical symptoms (dysphagia, heartburn) with an allergic reaction to any substance (not only food), with radiographic symptoms of GERD (shortened LES, presence of phrenic ampulla) and esophageal stenosis is sufficient to diagnose EoE. There is no point in performing a histological examination, which, as it turns out, does not allow differentiation of these diseases. When diagnosing EoE, we must keep in mind that the cause of the disease is the hypersecretion of hydrochloric acid, which leads to damage to the upper gastrointestinal tract. Lactose intolerance may be the primary cause of hypersecretion. The scheme of the pathogenesis of EoE is outlined below.
Treatment of EoE from the point of view of the proposed hypothesis of pathogenesis
Treatment for EoE includes:
1. Antiallergic treatment.
2. PPIs are used to suppress hydrochloric acid secretion, 20 mg in the morning 30 minutes before meals.
3. PPIs will be ineffective unless the intake of factors that trigger hydrochloric acid hypersecretion is stopped. Avoiding lactose-containing foods has a significant therapeutic effect.
4. Taking gastrointestinal mucosal protectors if pain in any location and/or heartburn is present, including bismuth salts or Esoxx.
5. Preventive measures for reflux include: eating small meals, limiting physical activity after meals, avoiding wearing a tight belt, and only lying down on an empty stomach [25].
CONCLUSION
Based on a literature review and our own research, the hypothesis of the pathogenesis of EoE is substantiated. It suggests that EoE is a GERD in patients with allergies. Hypersecretion of hydrochloric acid impairs the function of the LES and causes inflammation in the esophageal wall. The allergic reaction is accompanied by eosinophilic infiltration of the esophageal wall, leading to lumen narrowing. This hypothesis resolves all the contradictions discussed in the literature. The proposed diagnostics is based on reliable clinical symptoms (dysphagia, heartburn) combined with allergies, along with radiographic evidence of narrowing of the esophageal lumen and dysfunction of the LES. Since there is no linear relationship between dysphagia and the number of eosinophils, histological studies are of no practical value. Based on this hypothesis, treatment for EoE involves treating both GERD and allergies. I propose a scientific discussion of the hypothesis of the pathophysiology of EoE and the advisability of modifying my proposed diagnostic and treatment methods for EoE.
REFERENCES
- Attwood SE, Smyrk TC, Demeester TR, Jones JB. (1993). Esophageal eosinophilia with dysphagia. A distinct clinicopathologic syndrome. Dig Dis Sci. 38(1):109-116.
- Furuta GT, Liacouras CA, Collins MH, Gupta SK, Justinich C, Putnam PE, et al. (2007). Eosinophilic esophagitis in children and adults: a systematic review and consensus recommendations for diagnosis and treatment. Gastroenterology. 133(4):1342-1363.
- Liacouras CA, Furuta GT, Hirano I, Atkins D, Attwood SE, Bonis PA, Burks AW, et al. (2011). Eosinophilic esophagitis: updated consensus recommendations for children and adults. J Allergy Clin Immunol. 128(1):3-20.e6.
- Dellon ES. (2024). Challenges in Differentiating and Diagnosing Gastroesophageal Reflux Disease vs Eosinophilic Esophagitis. Gastroenterol Hepatol (N Y). 20(1):46-49.
- Kia L, Hirano I. (2015). Distinguishing GERD from eosinophilic oesophagitis: concepts and controversies. Nat Rev Gastroenterol Hepatol. 12(7):379-386.
- Cheng E, Souza RF, Spechler SJ. (2014). Eosinophilic esophagitis: interactions with gastroesophageal reflux disease. Gastroenterol Clin North Am. Jun;43(2):243-256.
- Levin M. (2025). Functional Heartburn is Gastroesophageal Reflux. Mathews J Gastroenterol Hepatol. 10(1):28.
- Levin MD. (2025). X-Ray Imaging of the Esophagus and Lower Esophageal Sphincter and Its Role in the Diagnosis of Gastroesophageal Reflux Disease. Mathews J Gastroenterol Hepatol. 10(2):31.
- Inage E, Furuta GT, Menard-Katcher C, Masterson JC. (2018). Eosinophilic esophagitis: pathophysiology and its clinical implications. Am J Physiol Gastrointest Liver Physiol. 315(5):G879-G886.
- Pesce M, Krishnan U, Saliakellis E, Lopez R, Lindley KJ, Thapar N, et al. (2019). Is There a Role for pH Impedance Monitoring in Identifying Eosinophilic Esophagitis in Children with Esophageal Atresia? J Pediatr. 210:134-140.
- Levin MD. (2025). Eosinophilic Esophagitis: Etiology, Pathogenesis, Diagnosis and Treatment. Hypothesis. Available at; https://doi.org/10.20944/preprints202501.1947.v1
- Dent J. (2007). Microscopic esophageal mucosal injury in nonerosive reflux disease. Clin Gastroenterol Hepatol. 5(1):4-16.
- Chandrasoma P, DeMeester T. (2016). A New Pathologic Assessment of Gastroesophageal Reflux Disease: The Squamo-Oxyntic Gap. Adv Exp Med Biol. 908:41-78.
- Inage E, Furuta GT, Menard-Katcher C, Masterson JC. (2018). Eosinophilic esophagitis: pathophysiology and its clinical implications. Am J Physiol Gastrointest Liver Physiol. 315(5): G879-G886.
- Koutri E, Papadopoulou A. (2018). Eosinophilic Gastrointestinal Diseases in Childhood. Ann Nutr Metab. 73(Suppl 4):18-28.
- Mahendra Raj S, Ravindran S, Kaur M, Braganza MC, Kunnath AP. (2022). Duodenal eosinophilia is associated with symptomatic erosive gastro-oesophageal reflux disease, presence of co-morbidities, and ethnicity but not undifferentiated functional dyspepsia: A retrospective Malaysian study. Med J Malaysia. 77(4):494-499.
- Novofastovskaia LR. (1959). Rentgenodiagnostika rigidnogo antral'nogo gastrita [X-ray diagnosis of rigid antral gastritis]. Vestn Rentgenol Radiol. 34(1):16-22.
- Katzka DA. (2014). The complex relationship between eosinophilic esophagitis and gastroesophageal reflux disease. Dig Dis. 32(1-2):93-97.
- Monnerat MM, Lemme EM. (2012). Eosinophilic esophagitis: manometric and pHmetric findings. Arq Gastroenterol. 49(2):113-117.
- Frazzoni M, Penagini R, Frazzoni L, de Bortoli N, Mauro A, Tolone S, et al. (2019). Role of Reflux in the Pathogenesis of Eosinophilic Esophagitis: Comprehensive Appraisal With Off- and On PPI Impedance-pH Monitoring. Am J Gastroenterol. 114(10):1606-1613.
- Molina-Infante J, Ferrando-Lamana L, Ripoll C, Hernandez-Alonso M, Mateos JM, Fernandez-Bermejo M, et al. (2011). Esophageal eosinophilic infiltration responds to proton pump inhibition in most adults. Clin Gastroenterol Hepatol. 9(2):110-117.
- Høst A, Jacobsen HP, Halken S, Holmenlund D. (1995). The natural history of cow's milk protein allergy/intolerance. Eur J Clin Nutr. 49(Suppl 1):S13-S18.
- Salvatore S, Agosti M, Baldassarre ME, D'Auria E, Pensabene L, Nosetti L, et al. (2021). Cow's Milk Allergy or Gastroesophageal Reflux Disease-Can We Solve the Dilemma in Infants? Nutrients. 13(2):297.
- Levin MD. (2025). Hydrochloric acid hypersecretion is the cause of pathology of the upper digestive tract. Literature analysis. Preprint v1 · CC-BY 4.0. Available at: https://www.qeios.com/read/4QXH2Q/pdf
- Levin MD. (2025). Prevention and Treatment of Esophageal Reflux Disease: Literature Analysis. Mathews J Gastroenterol Hepatol. 10(2):33.
- Zimmerman SL, Levine MS, Rubesin SE, Mitre MC, Furth EE, Laufer I, et al. (2005). Idiopathic eosinophilic esophagitis in adults: the ringed esophagus. Radiology. 236(1):159-165.
- Al-Hussaini A, AboZeid A, Hai A. (2016). How does esophagus look on barium esophagram in pediatric eosinophilic esophagitis? Abdom Radiol (NY). 41(8):1466-1773.
- Johnson AC, Lester PD, Johnson S. (1992). Esophagogastric ring: why and when we see it, and what it implies: a radiologic-pathologic correlation. South Med J. 85(10):946-952.
- Towbin AJ, Diniz LO. (2012). Schatzki ring in pediatric and young adult patients. Pediatr Radiol. 42(12):1437-1440.
- Sarbinowska J, Wiatrak B, Waśko-Czopnik D. (2021). Association of eosinophil-mediated inflammatory biomarkers with the presence of the Schatzki ring. Adv Med Sci. 66(2):279-283.