Previous Issues Volume 4, Issue 1 - 2019
Colorectal Cancer in Australian Young Adults
Reger R. Mikaeel1,2,3,4 , Timothy J. Price1,3, Eric Smith1,5, Paul A. Drew5, Wendy Uylaki1,6, Mehgan Horsnell1, Joanne P. Young1,2,3
1Department of Haematology and Oncology, The Queen Elizabeth Hospital, Australia 2SAHMRI Colorectal Node, Basil Hetzel Institute, Australia. 3Faculty of Health and Medical Sciences, University of Adelaide, Australia 4Department of Biology, College of Science, University of Duhok, Duhok, Kurdistan 5University of Adelaide Department of Surgery, The Queen Elizabeth Hospital, Woodville South 5011, South Australia, Australia 6Department of Gastroenterology, The Queen Elizabeth Hospital, Australia
*Corresponding Author: Reger Romi Mikaeel, Department of Haematology and Oncology, School of Medicine, Basil Hetzel Institute, University of Adelaide, Australia.
Received Date: May 09, 2019
Published Date: Jun 17, 2019
Citation: Mikaeel RR. (2019). Colorectal Cancer in Australian Young Adults. Mathews J Cancer Sci. 4(1): 18.
ABSTRACT
Purpose of Review: Although the overall incidence and mortality rates of colorectal cancer (CRC) have been decreasing in Australia over the last 20 years, there has been a significant increase in the incidence of early-onset CRC (EOCRC) without a clear explanation. In this review, we have outlined the epidemiology, suggested risk factors, clinicopathological and molecular features, survival and prognosis, and treatment approach for Australian young adults with CRC.
Recent findings: There has been a shift in the epidemiology of bowel cancer incidence and mortality across different age groups, and there are clinicopathological and molecular feature differences between EOCRC and late onset CRC (LOCRC). EOCRC is more common in the distal colon and rectum. Young patients are diagnosed at more advanced stages of the disease with increased prevalence of aggressive pathologic features and tend to receive more aggressive chemotherapies. EOCRC patients present with a predominance of symptoms in the left side of the colon, and importantly, cases do not have clinically known risk factors. The microbiota may play a significant role in the CRC pathogenesis through an impact on host metabolism and through the transmission of metabolic and even CRC risk in non-Mendelian familial aggregation.
Summary: The increase in the incidence and mortality rates of young adults with CRC has been significant. Thus, more collaborative research is needed to explain the reasons behind the increase in CRC incidence, and to identify young adults in the population at an increased risk of developing CRC at an early age.
Keywords: Colorectal cancer, Early-onset CRC, Late-onset CRC, Incidence and mortality rates, Risk factors
Abbreviations: CRC: Colorectal Cancer; EOCRC: Early-Onset CRC; LOCRC: Late-Onset CRC; LS: Lynch Syndrome
INTRODUCTION
In Australia, colorectal cancer (CRC) was estimated to be the most frequently diagnosed digestive-tract cancer in 2018 and was Australia’s most frequent digestive-tract cancer killer [1]. CRC is the third most commonly diagnosed cancer and the second leading cause of cancer death worldwide [2]. The age distribution, however, has altered with an increase in incidence of early-onset CRC (EOCRC) being reported in Australia, the USA and many other developed countries [3-9]. There is a difference in the literature regarding the exact definition of early onset as different researchers have used various age groups including individuals under 30, 35, 40, 50 or 55 years, and therefore findings of various studies are difficult to be compared and interpreted [10-24]. However, in general these studies show the increase in the incidence of CRC in young adults. A study published in 2019 found that found that CRC incidence has increased by almost 10% in individuals under 50 years of age since 1990 in Australia [25]. Between 1990 and 2010, the incidence of CRC increased by 85% to 100% in Australian aged 20-29 years and by 35% in the age group 30-39 years [26]. In 2018, as many as 14% of all new CRC cases and 10% of all Australian who died from CRC were <55 years [27]. In 2019, CRC is estimated to be the third most commonly diagnosed cancer in Australians aged 25-49 years [28]. In contrast, there has been a remarkable decrease in the overall prevalence of CRC in individuals above the age of 50 years (LOCRC) since the 1990s [29-32]. The increase in EOCRC incidence is not well explained; some researchers recommend that colonoscopy screening age for people with normal risks should be lowered from 50-45 years. However, those young adults who are under the age of 45 years will not benefit from this recommendation and this does not address efforts to explain why the incidence and mortality rates of CRCs have been increasing in young adults [33]. This review will explore the epidemiology, suggested risk factors, clinicopathological and molecular features, survival and prognoses, and treatment approach for Australian young adults with CRC.
Epidemiology of EOCRC
CRC is largely a disease of older patients, with an average age at diagnosis in Australia of 69 years [34]. Screening not only detects cancers but also detects advanced pre-cancerous polyps in the ratio of four to five lesions for every one cancer detected, thus facilitating both prevention and early detection [35]. In the USA, colonoscopy screening is now recommended at 45 instead of 50 years of age for individuals at normal risk for developing bowel cancer [36]. In Australia, under the guidelines individuals under the age of 50 years are not included in the target age range for the National Bowel Cancer Screening Program (NBCSP) [37]. This program, increasing awareness of the need to screen for bowel cancer, and advances in cancer therapy, have likely caused a significant decrease in both the incidence and mortality rates of LOCRC [32,38]. The global analysis of CRC temporal trends shows a decrease in incidence and mortality rates of this disease in all ages combined over time in Australia. In contrast, the incidence and mortality rates of EOCRC have been increasing, when the analysis is stratified into specific age groups, over the last two decades in Australia, the USA, and other countries with similar overall trends of this disease (Figure 1) [32,38]. The incidence of CRC has increased 186% in individuals between the ages of 15-24 years in the last three decades [27] and it is considered to be the most common cancer-related cause of death for those aged 25-29 [15]. In addition, Feletto et al., recently analysed over 375,000 cases of colon and rectal cancers from 1982 to 2014 in Australia. This study confirms that the incidence rates of colon cancer has increased in young adults under the age of 50 years since the mid-2000s, with the increase in the annual percentage changes (APCs) ranging from 1.7% to 9.3% per annum depending on specific age group. The incidence rates of rectal cancer has also increased among the same group of ages from the 1990s, with the increase in the APCs ranging from 0.9% - 7.1% per annum [32]. The data shows that early-onset CRC is a current public health concern in the USA, Australia [23,27,39] and in other countries[3-9] (Table 1).
Figure 1: Age-adjusted SEER incidence rates of CRC from 1990-2015 among people under and above 50 years of ages in the USA. Graph (A) shows the incidence of CRC in people under the age of 50 years. Graph (B) shows the incidence of CRC in people above the age of 50 years. (https://seer.cancer.gov/faststats/selections.php?#Output).
Table 1: The prevalence of early-onset CRC in different countries.
|
Country |
Early-onset CRC |
Age of onset |
Reference |
|
Pakistan |
52% |
Under 40 years |
[40] [41] |
|
India |
39% |
Under 40 years |
[42] |
|
Iran |
36% |
Under 40 years |
[43] |
|
Nigeria |
31.50% |
Under 40 years |
[44] |
|
Nepal |
28.60% |
Under 40 years |
[45] |
|
Egypt |
25% |
Under 40 years |
[46] |
|
Korea |
22.4% |
Under 50 years |
[47] |
|
Jordon |
20.2% |
Under 40 years |
[48] |
|
Turkey |
20% |
Under 45 years |
[49] |
|
Sri Lanka |
19.70% |
Under 40 years |
[50] |
|
Taiwan |
18.8% |
Under 50 years |
[51] |
|
Saudi Arabia |
17% |
Under 40 years |
[52] |
|
Australia |
14% |
Under 55 years |
[27] |
|
Singapore |
14% |
At or under 55 years |
[53] |
|
Canada |
10% |
Under 50 years |
[54] |
|
USA |
10% |
Under 50 years |
[55] |
|
UK |
5% |
Under 50 years |
[56] |
The prevalence of CRC is not uniform across all racial and ethnic groups, though there is an increase in the prevalence of this disease across the entire population. This variation in proportion of CRC occuring at an earlier age may reflect the expected lifespan of these sub-group in particular countries, as well as genetic background and a greater sensitivity to Western lifestyle risk factors (Table 1). For example, in the USA, incidence rates were reported to be higher among African Americans aged between 20-40 years than non-Hispanic Whites [57]. In New Zealand, even though the death rates of young-onset CRC were similar among Pacific, non-Mâori non-Pacific and Mâori people by 1996-99, the death rates had accelerated up to 10-fold among Pacific people, by 50% among Mâori and decreased by 10% to 20% among non-Mâori non-Pacific people during 1981 to 1995 [58].
Among Australians aged 15-24 years from 2010 to 2014, CRC ranked as the fifth and fourth most commonly diagnosed cancer in males and females, respectively [27]. The incidence of CRC is slightly higher in males which accounts for 53% of cases under the age of 55 years in Australia [15]. Weir et al., reported that Aboriginals are significantly younger than non-aboriginals when diagnosed with CRC (17% compared to 6% of EOCRC) and are more likely to have diabetes or other chronic diseases [59]. It is notable that studies vary in the exact proportion of anatomical distribution of the primary site of CRC (sometimes described by exact site or right vs left vs rectum). However, regarding EOCRC, there does seem to be a consensus that there is an increased prevalence in the distal colon and rectum [22,60-65]. By contrast, proximal colon tumours are more predominant within LOCRC patients [23,63,64,66]. This variation in incidence by anatomical sites have made scientists suggest to separate risk factors for colon versus rectal cancer. These anatomical subistes are different in terms of embryological origins, level of oxygenation, concentration of bile salts and the microbial enviroment [67]
Hereditary and Family High-Risk of CRC Cases
It is reported that up to 30% of bowel cancer patients have a strong family history of the disease, and 5% of those are due to a demonstrable inherited genetic abnormality [68]. Compared to individuals without a family history of CRC, people with a first-degree family history of CRC have two to four times the risk of developing this cancer [69]. In Australia, 16% of CRC patients have at least one first-degree relative with bowel cancer [70]. However, this figure is lower in Sweden and Finland as ~11% to 13% of all CRC cases have at least one first-degree relative with CRC [70-72]. Hereditary and family history risk factors can induce the development of EOCRC and form a group of people with higher risk relative to the general population who should be under closer cancer surveillance [73]. However, given that hereditary CRC is under closer surveillance in current times, it is unlikely that this group of patients are responsible for the increase in incidence in EOCRC.
Lynch syndrome (LS) is the most frequently diagnosed of the EOCRC inherited syndromes, accounting for up to 17% of all CRC cases [74] (Tables 1 and 2). It is defined as an autosomal dominant cancer predisposition syndrome which is caused by germline mutation in one of the four mismatch repair (MMR) genes (MSH2, MLH1, MSH6, and PMS2) (Table 2). Germline mutations in the MLH1 and MSH2 account for approximately 70% to 90% of the LS cases [75,76] while mutations in MSH6, PMS2 are detected in 10% to 20% of LS cases [77]. With the loss of activity of DNA MMR enzyme, there is accumulation of multiple mutations which facilitate cancer growth and metastasis, as well as leading to manifestations of microsatellite instability (MSI). An alternative silencing cause for MSH2 and leading to LS is a germline deletion in the epithelial cell adhesion molecule (EPCAM) gene which is located upstream of MSH2 [78]. EPCAM germline mutations are reported to account for 6.3% of all LS cases [79]. This germline deletion results in silencing the transcription of MSH2 by causing its allele-specific methylation. Therefore, the risk of bowel cancer in patients with EPCAM germline mutations (75%) is similar to those with MSH2 mutations (77%) by the age of 70 years [80]. The clinicopathological features associated with CRCs in LS include poor tumour differentiation, proximal location, mucinous histology, lymphocytic reactions, and synchronous and metachronous lesions [81,82]. People with LS have a 70% lifetime risk of susceptibility to CRC [83] (Table 2). In addition, a recently identified Lynch-like syndrome (LLS) has been described where there is an MMR-deficient colorectal tumour without germline mutations and/or MLH1 promoter methylation, and it can be as frequent as 70% of suspected LS patients [84].
Approximately 35% to 45% of LS cases are diagnosed with CRC before the age of 40-45 years, and this syndrome accounts for approximately one-third of EOCRC in people under the age of 30 years [74,82,85,86]. However, the average age at bowel cancer diagnosis among people with LS syndrome is 42-45 years (Table 2). Rarey, bi-allelic deleterious mutations of MMR genes result in constitutional mismatch repair deficiency. This condition predisposes patients to CRC, among other cancers, with an average age of 16 years at the time of diagnosis which is much earlier than LS [87-90].
Familial Adenomatous Polyposis (FAP) is the second most frequently diagnosed autosomal dominant inherited syndrome (less than 1%). Clinically, classic FAP is characterized by the development of numerous (hundreds to thousands) adenomatous polyps in the bowel, appearing from the age of 10-12 years (Table 2). Patients with this syndrome have a 100% lifetime risk CRC by the age of 40 years if prophylactic colectomy is not performed. However, the median age at CRC diagnosis among people with FAP syndrome is around 39 years [91] (Table 2). Germline mutations in adenomatous polyposis coli (APC) gene is an early event in the progression of FAP and is the main cause of the classic and attenuated FAP (AFAP) syndromes [92] (Table 2).
Table 2: Summary of the main hereditary and familiarly high risk CRC syndromes.
|
Syndrome |
Inheritance |
Gene |
The average age of CRC diagnosis |
CRC lifetime risk factor |
Incidence in EOCRC |
Clinicopathological features |
|
LS |
Dominant |
MLH1, MSH2, MSH6, PMS2, EPCAM |
40-45 |
70% |
17% |
Poor differentiation carcinoma, mucinous carcinoma, tumour-infiltrating lymphocytes and more commonly found in the right colon |
|
FAP |
Dominant |
APC |
39 |
100% |
< 1% |
Good differentiation carcinoma, no mucinous carcinoma, no lymphocytic reaction and more commonly found in the distal colon |
|
MAP |
Recessive |
MUTYH |
48 |
43% - 100% |
<1% |
Development of 10 to 100 adenomatous polyps in the colon and rectum |
|
SPS |
Unknown |
Unknown |
55-65 |
<50% |
<1% |
Multiple and/or large serrated polyps throughout the colon and rectum |
|
LS: Lynch syndrome, FAP; Familial Adenomatous Syndrome, MAP: MUTYH-Associated Polyposis, SPS: Serrated Polyposis Syndrome |
||||||
MUTYH-Associated Polyposis (MAP) is another subtype of polyposis which has an autosomal recessive transmission, and is related to bi-allelic germline mutations in the mutY DNA glycosylase (MUTYH) gene [93]. MAP patients have a lifetime risk of developing CRC of 43% to 100%, at an average age of 48 years, with about 50% of cases presenting with cancer at the time of diagnosis [94,95]. The phenotypes of AFAP and MAP syndromes are often indistinguishable. There are currently, no exact phenotypes of the MAP condition. Some reports show one single CRC and no or less than ten polyps, or cases presenting with mostly hyperplastic/serrated polyps [96], while proximal adenomas are also commonly seen. Moreover, the cancers related to MAP syndrome are more likely to be found in the proximal side of the colon in comparison to the AFAP related tumours [95]. Germline mutations in the MUTYH have also been reported in patients with LLS (Table 2).
Another syndrome of CRC predisposition is serrated polyposis syndrome (SPS). SPS is under recognized and is characterized by the presence of many serrated polyps in the colon and rectum [97]. Boparai et al., found CRC in 35% of individuals with SPS, with the majority of cases (94.5%) being diagnosed with CRC at the time of SPS diagnosis. However, 6.5% of patients were under surveillance for SPS while diagnosed with CRC [98]. Currently, 40% to 60% of SPS patients demonstrate a family history of bowel cancer rather than of polyposis. Nevertheless, it is worth noting that the exact patterns of inheritance of this syndrome are still not clear, and autosomal recessive alleles and autosomal dominant alleles are suggested [99,100] (Table 2). SPS accounts for a currently unknown proportion of EOCRC.
Risk Factors and Microbiota
Different lifestyle-related risk factors including obesity and diabetes have been associated with the significant increase in the prevalence of EOCRC (Table 3). In Australia, the prevalence of obesity and overweight have increased [101], and from 1985-2012, childhood obesity has also grown among Australian’s children [102]. The proportion of Australians aged 18 years and over, who were obese or overweight, has increased since 1995, and 46% of adults aged 18-24 were obese or overweight in 2017-2018, and this had increased to 68.7% by age 35-44 years [103]. According to the available data, though red meat consumption has decreased since the 1970s in Australia, it remains above 1 kg per week (32). Also, adults in Australia consumed over 11 g of processed meat daily in 2011-2012 (32). Australia is amongst the highest in the OECD (95 kg in 2016) in terms of meat consumption per capita [104]. Smoking and lack of physical activity have also been associated with a higher risk of developing CRC (32). However, in Australia, smoking has deceased across all age groups since 1995 with the largest decline in individuals aged between 18-34 years [103]. Nevertheless, though some risk factors for CRC in young adults have increased in parallel to the increase in EOCRC, and an independent relationship is yet to be clearly demonstrated. Nguyen et al., has recently reported a significant association between increased risk of EOCRC and sedentary TV watching time after adjusting the persumed risk factors such as lack of exercise and obesity [105]. Additionally, there is a consistent relationship between type 2 diabetes and CRC, and this is likely to be more pronounced in young adults under 55 years [106]. In summary, lifestyle factors involving activity level and metabolism issues are the most liekly contenders for the increase in the incidence of EOCRC.
Table 3: Some factors associated with the risk of colorectal cancer (CRC).
|
Risk factor |
Higher risk percentage |
Author |
|
Inflammatory bowel diseases (IBDs) |
People with IBDs have a two to threefold higher risk of developing CRC. |
[107] |
|
Diabetes mellitus |
Higher risk of developing CRC compared to general population (HR, 1.3, 95%CI 1.2-1.5). |
[106] |
|
Solid Organ Transplantation |
The increase of proximal colon tumour (SIR=1.69, 95%CI: 1.53-1.87) |
[108] |
|
Appendicitis |
Appendicitis patients have a higher risk of CRC (SIR 4.6, 95% CI 4.0-5.2; SIR 3.5, 95% CI 2.9-4.1) |
[109] |
|
Physical Inactivity |
Least active individuals are by 27% at higher risk of susceptibility to CRC |
[110] |
|
Obesity |
Obese or overweight women have double the risk of developing bowel cancer under age 50 years compared to healthy body weight women |
[111] |
|
Meat Consumption |
Red meat and processed meat increase the risk of CRC by 30% and 20%, respectively |
[112] |
|
Fibre |
Swedish women with a high intake of vegetables and fruits had 32% risk reduction for bowel cancer |
[113] |
|
Calcium |
T risk of CRC was about 70% lower in people with the highest consumption of Ca+2 compared to people with the lowest calcium intake |
[114] |
|
Vitamin D |
There was a considerably lower risk of developing CRC in people with a high blood level of vitamin D (1,25(OH)2D3) and high lymphocyte accounts |
[115] |
|
Alcohol |
A modest increased risk of colon tumour (45%) and rectal tumour (49%) with regular high intake of alcohol (>45 g/day) in both genders combined in relative non-drinkers |
[116] |
|
Smoking |
Smokers were 2-fold at higher risk of advanced neoplasia than non-smokers, similar to or higher than those patients that have the first-degree relative with this disease |
[117,118] |
The microbiota may play a role through impacting onhost metabolism, and through the transmission of metabolic and even CRC risk factors in non-Mendelian familial aggregation, as has been shown in co-housed preclinical animal models [119]. Obesity and diabetes, and CRC itself have been linked to changes in the gingival and gut microbiota in humans. Though there have been multiple findings suggesting causation in animal models, a number of confounding factors such as genetic background, and stress may have played a role. It is currently not definitively known whether these observations are readily translated into human settings and whether human studies suggest direct causation or setting-associated colonisation in a predisposed host. For example, findings in humans of insulin sensitivity being improved in obese subjects after faecal transplantation into the small intestine from lean donors [120], indicate that microbiota in the obese do not necessarily cause obesity, but that a certain element of the microbiota from lean individuals lacking in the obese can modify insulin sensitivity. Similarly, obese patients who experienced weight loss had improved response to periodontal therapy over those who remained obese with persisting gum disease [121,122], could be interpreted as setting-associated. The composition of bacteria chronologically alters as people age, as well as depending on location within the bowel [123,124]. Some species of bacteria have been recognized to have a role in the investigating bowel cancer pathogenesis such as Bacteroides fragilis, Fusobacterium nucleatum, Streptococcus bovis, and some strains of Escherichia coli. Findings of studies in older CRC patients showed a significant role of F. nucleatum in the pathogenesis of CRC, particularly in the right-side cancers [124]. Other studies found that F. nucleatum travels as bowel cancer metastasizes in mice, and murine cancers with this bacterium respond to the metronidazole antibiotic [125]. However, while it can be postulated that microbiota might be involved, there is not yet much convincing evidence in an area which is difficult to research. Also, it is currently not known whether the microbiota has a role in EOCRC.
Survival rate, Prognosis and Clinical Features of EOCRC
The overall 5-year survival rate of CRC for all stages combined from diagnosis is approximately 69% in Australia [126] and 60% in the USA [30,127]. Considering all stages of the disease the 5-year survival rate of CRC has increased by 18% between 1985-1989 and 2010-2014 in Australia [126]. In 2007, Quah et al., reported that the outcomes (recurrence rates and 5-year survival) of CRC stages I, II, III were similar, if not better, between EOCRC and LOCRC patients. In particular, there were no noticeable differences in the local and distant recurrence rates between the groups (17% vs 18%). Moreover, there were no differences in the 5-year recurrence-free survival between EOCRC (80%) and older patients (79%) after four years and eight months of following up. Finally, the overall relative survival rate in older patients (73%) was worse than in EOCRC patients (84%) [14,128]. In 2004, O'Connell et al., reported that the 5-year survival rate, when matched for stage, was the same for stage I and II between EOCRC and LOCRC patients, but it was substantially higher in younger individuals for stage III and IV disease [12]. In contrast, a retrospective analysis, including nine phases III trials, reported that progression-free survival- but not response rate or overall survival- was higher among EOCRC patients (<40) compared to LOCRC patients [129].
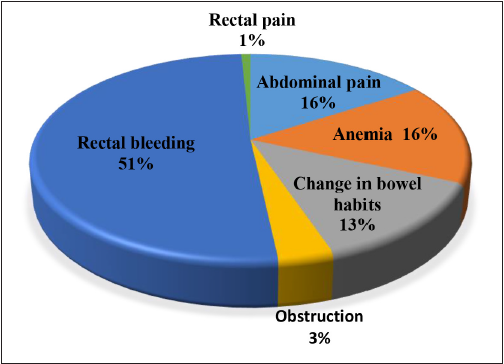
It has been reported that approximately 86% of EOCRC cases are symptomatic at the time of diagnosis [60]. Symptoms are often non- specific, such as abdominal pain, fatigue or weight loss; but EOCRC patients also tend to have a predominance of the symptoms in the left side including a change in the bowel habits and rectal bleeding [12] (Figure 2). In addition, EOCRC compared to LOCCRC patients more frequently present with symptoms of abdominal pain (41.2 vs 27.2%) and hematochezia (28.8 vs 23.2%) (130). However, although CRC in young adults is often symptomatic, diagnosis is generally delayed possibly because of the lack of awareness of the increasing incidence of early-onset CRC [131]. It has reported that the majority of EOCRC cases at the time of diagnosis were already distantly metastatic or regionally advanced, as they tend to present with more aggressive pathological features [10,14,22,61,64-66,132-143]. In addition, mucinous and signet ring features are more common in EOCRC patients than in LOCRC patients [64,66,135,140,144-146]. Chang et al., reported that CRC patients at and under the age of 40 years tend to have higher signet features compared to late-onset CRC patients ( 13% vs 1%), higher perineural invasion (29% vs 11%) and higher venous invasion ( 22% vs 6%) [147]. Studies have shown that tumours with mucinous and signet ring features are associated with a negative prognostic factor, poor differentaition, and more advanced tumour stages [148-153].
Figure 2: Presenting symptoms of CRC patients aged under 50 years old (Adapted from (154).
Molecular Characteristics of Sporadic EOCRC
Approximately 65% of CRC is sporadic with no apparent hereditary genetic predispositions or family history [154,155]. Though EOCRC raises the likelihood of a hereditary predisposition, only the minority of CRC cases in young adults (15% to 20%) are due to the hereditary syndromes [60,156], which is slightly higher than all CRC cases (2% to 8%) [157]. Among EOCRCs and in the general population, the adenoma-carcinoma pathway contributes to the development of approximately 85% of all CRCs. In contrast, evidence has shown that about 15% to 30% of CRCs exhibit the features of an alternative serrated neoplasia pathway [158-160]. There are three underlying molecular mechanisms which have been described in the development of CRC: chromosomal instability (CIN), microsatellite instability (MSI) and CpG methylator phenotype (CIMP) [161]. These three mechanisms are not mutually exclusive and may overlap in some subsets of CRC [158-162].
CIN, which accounts about 85% of sporadic CRCs [163], is characterized by continuing errors in chromosomal segregation including a high rate of gains or losses of whole chromosomes or large fractions of chromosomes [164]. This results in aneuploidy, rearrangement of chromosomes, copy number variations, as well as mutations in tumour suppressor genes and oncogenes such as APC, TP53, KRAS and BRAF which subsequently contribute to CRC carcinogenesis [156-166]. In general, the genome of EOCRC patients is more frequently euploid and hypermethylated than LOCRC cases [167,168]. Somatic mutations of the KRAS gene are found in about 35% to 45% of CRCs [169] and predict a lack of response to anti-EGFR targeted therapy [170-172]. In EOCRC, the incidence of KRAS mutations remains questionable, with the incidence of these mutations ranging from 4% to 54% in EOCRCs [144,146,147,173-176]. Similarly, BRAF mutations in EOCRC are reported to range from 0% to 14.3% [143,144,177-181]. In addition, loss of the chromosomal regions coding for loci, where APC, SMAD4 and DCC genes, is more common in LOCRCs than EOCRCs [182-184]. In contrast, EOCRCs lose the chromosomal regions which code for CRC markers (TJP2) [185-187] and FOX transcriptional factors [188], and gain regions coding for AMP-kinase regulatory subunit and BMPR1A [185]. Puccini et al., reported that mutations in genes such as KDM5C, KMT2A, KMT2D and SETD2 which are involved in the modification of histones are higher in EOCRCs than LOCRC.
MSI-H, which represents ~21% of the EOCRC, is characterized by the inability of the MMR system to maintain the DNA structure or to correct errors during the process of DNA replication as well as by accumulation of point mutations and changes in the repetitive microsatellite nucleotide sequences [146,147,173,175]. MSI-H cancers in EOCRC are mostly linked to LS, with some cases having epigenetic inactivation of MLH1 and wild-type BRAF which are categorized as epimutation-type LS. CIMP has been shown to contribute to approximately 40% of all CRCs and is involved in the alternative serrated neoplasia pathway [155,189]. This pathway is categorized by high methylation of CpG islands and early BRAF mutations. Hypermethylation of the MMR gene MLH1 is also frequently reported in this pathway, and can result in diploid CRCs that are MSI. Tumours which are CIMP-high are usually found in the right-side colon, have high-MSI, a higher rate of BRAF mutations and are poorly differentiated. CRC with CIMP and BRAF that are MSI-H are mostly observed in LOCRC. Tumours which are CIMP-low are also observed in young adults with CRCs [190].
Studies have consistently shown that the prevalence of KRAS/RAS and BRAF mutations, as well as MSI, is higher in right-side colon tumours than left-side [191-194]. This is clinically relevant given survival benefits with selective anti-EGFR inhibitors are higher in patients with left-side RAS wild-type colon tumours compared to the individuals with proximal colon tumours. The differing prevalence of primary site may lead to survival implications based on age. For female EOCRC estrogen may play a role as some studies have suggested that estrogen might be a protective factor for the development of CRC in the proximal colon. As its level decreases with age, this may result in the increasing prevalence of proximal colon tumours in female adults above the age of 50 years [195]. Evidence for this hypothesis comes from reports that women with higher estrogen exposure were more protected against high-MSI cancers which are very often found in the proximal colon [196]. However, a tumour mutational burden (TMB) is more frequent in young adults with left-sided colon tumour than their older counterparts (9.7% vs 2.8%, P<0.001). TMB may have relevance to immunotherapy options for this group of patients [197]. For example, although RAS WT is higher in left-sided cancer, there is a higher rate of HER2 amplification and NF1muattions in young adults with left-sided colon tumour than older individuals [197] which may have clinical relevance. Therefore, though there is a difference in the proportion of mutations between EOCRC and LOCRC, this may reflect, at least in part, the different site distrubution of CRC between the two age groups.
Treatment
CRC patients are generally treated in a standardized way based on current guidelines [198,199]. Sporadic CRC in individuals younger than 50 years may have a different molecular profile, and treatment may differ based on this [136,197,200,201], but ultimately the therapeutic strategy will be guided by the exact profile rather than age. There are subtle but real differences that may reflect age. For example, guidelines allow choice of first-line systemic chemotherapy schedules, which may vary from single-agent fluoropyrimidine to triplet therapy (FOLFOXIRI), although in general doublet chemotherapy is recommended [202]. However, data from registries do suggest that young adults with CRC tend to receive more aggressive chemotherapies compared to older patients with this disease. This is probably because these patients can tolerate more aggressive regimens and the misconception that EOCRC patients have worse treatment outcomes. EOCRC patients more commonly receive adjuvant therapy more often with multi-agent adjuvant regimens. Despite this trend, there is little evidence this improves outcome significantly. For example, Kneuertz et al., showed no survival gain in their analysis for patients diagnosed with stage II CRC (RR, 0.90; 95% CI, 0.69-1.17). A minor survival benefit may exist for those diagnosed at stages III-IV (RR, 0.89; 95CI, 0.81-0.97) [136]. However, further evidence is required to which subgroups may benefit most [150]. Surgical intervention does appear to differ by age, with resection of primary cancer more commonly performed in young adults with metastatic CRC (mCRC) compared to older mCRC patients (70.8% versus 66.6%; P < 0.001) [14,203-205]. Resection of primary may impact on the outcome by preventing future complications [204-206] and may impact on survival [145,204,205,207-210].
EOCRC patients are also more likely to undergo radiation therapy in the setting of metastatic rectal cancer than their older counterparts [14]. Radiation therapy for the rectal primary in metastatic disease is used to control the local recurrence rate. There are few studies regarding the recurrence rate of rectal cancer in young adults compared to their older counterparts after radiation therapy. However, You et al., reported that the recurrence incidence of the tumour was higher in young adults with rectal cancer, especially distant metastasis than their older counterparts after a similar length of following up [211]. In addition, Fossum et al., conducted a retrospective review comparing patients with synchronous resectable lung or liver metastasis who did not receive neoadjuvant therapy versus those who received neoadjuvant therapy. It was found that none of the patients who received neoadjuvant therapy had a local recurrence after follow up of 43 months while 26% of patients without neoadjuvant therapy had a local recurrence (P<0.001) [212].
CONCLUSION
EOCRC incidence is increasing in Australian and has some different clinicopathological and molecular features in comparison to LOCRC, largely related to the higher prevalence of genetic predisposition, and the increased prevalence of distal colon and rectal primary site. While the American Cancer Society has lowered the screening age to 45 years, Australians under 50 years of age are still not included in the target age range for the NBCSP. EOCRC is a heterogeneous disease, the majority of the cases are sporadic and there are no identified risk factors, though various lifestyle-related factors and gut microbiota might have contributed to this significant increase. While the pathogenesis of inherited CRC syndromes in young adults is well explored, the molecular features of sporadic EOCRC are not robustly explained. More collaborative research is needed for a better explanation of the molecular features of EOCRCs. Molecular features may play a role in understanding for development of EOCRC as they may reflect the sentinel risk factors which trigger this condition. A better understanding of the molecular characteristic differences between EOCRC and LOCRC may also facilitate personalized medicine in the treatment of EOCRC in the future.
REFERENCES
- https://www.aihw.gov.au/getmedia/892d43f7-ab5d-48fe-9969-129f138687f3/aihw-can-117.pdf.aspx?inline=true
- Dozois EJ, Boardman LA, Suwanthanma W, Limburg PJ, et al. (2008). Young-onset colorectal cancer in patients with no known genetic predisposition: can we increase early recognition and improve outcome? Medicine (Baltimore). 87(5): 259-263.
- Abou-Zeid AA, Jumuah WA, Ebied EF, Abd El Samee Atia KS, et al. (2017). Hereditary factors are unlikely behind unusual pattern of early - Onset colorectal cancer in Egyptians: A study of family history and pathology features in Egyptians with large bowel cancer (cross-sectional study). Int J Surg. 44: 71-75.
- Hessami Arani S and Kerachian MA. (2017). Rising rates of colorectal cancer among younger Iranians: is diet to blame? Curr Oncol. 24(2): e131-e137.
- Troeung L, Sodhi-Berry N, Martini A, Malacova E, et al. (2017). Increasing Incidence of Colorectal Cancer in Adolescents and Young Adults Aged 15-39 Years in Western Australia 1982-2007: Examination of Colonoscopy History. Front Public Health. 5: 179.
- Gandhi J, Davidson C, Hall C, Pearson J, et al. (2017). Population-based study demonstrating an increase in colorectal cancer in young patients. Br J Surg. 104(8): 1063-1068.
- Brenner DR, Ruan Y, Shaw E, De P, et al. (2017). Increasing colorectal cancer incidence trends among younger adults in Canada. Prev Med. 105: 345-349.
- Vuik F, Nieuwenburg S, Bardou M, Dinis-Ribeiro, et al. (2018). Increasing Incidence of colorectal cancer in young adults in Europe over the last 25 years. Gut. doi: 10.1136/gutjnl-2018-317592
- Malekzadeh R, Bishehsari F, Mahdavinia M and Ansari R. (2009). Epidemiology and molecular genetics of colorectal cancer in iran: a review. Arch Iran Med. 12(2): 161-169.
- Teng A, Lee DY, Cai J, Patel SS, et al. (2016). Patterns and outcomes of colorectal cancer in adolescents and young adults. J Surg Res. 205(1): 19-27.
- Khan SA, Morris M, Idrees K, Gimbel MI, et al. (2016). Colorectal cancer in the very young: a comparative study of tumor markers, pathology and survival in early onset and adult onset patients. J Pediatr Surg. 51(11): 1812-1817.
- O'Connell JB, Maggard MA, Liu JH, Etzioni DA, et al. (2004). Do young colon cancer patients have worse outcomes? World J Surg. 28(6): 558-562.
- Gryfe R, Kim H, Hsieh ET, Aronson MD, et al. (2000). Tumor microsatellite instability and clinical outcome in young patients with colorectal cancer. N Engl J Med. 342(2): 69-77.
- Abdelsattar ZM, Wong SL, Regenbogen SE, Jomaa DM, et al. (2016). Colorectal cancer outcomes and treatment patterns in patients too young for average-risk screening. Cancer. 122(6): 929-934.
- https://www.bowelcanceraustralia.org/never-too-young
- Naidoo R, Tarin M and Chetty R. (2000). A comparative microsatellite analysis of colorectal cancer in patients< 35 years and> 50 years of age. Am J Gastroenterol. 95(11): 3266-3275.
- Fu J, Yang J, Tan Y, Jiang M, et al. (2014). Young patients (≤ 35years old) with colorectal cancer have worse outcomes due to more advanced disease: a 30-year retrospective review. Medicine (Baltimore). 93(23): e135.
- Cusack JC, Giacco GG, Cleary K, Davidson BS, et al. (1996). Survival factors in 186 patients younger than 40 years old with colorectal adenocarcinoma. J Am Coll Surg. 183(2): 105-112.
- Palmer ML, Herrera L and Petrelli NJ. (1991). Colorectal adenocarcinoma in patients less than 40 years of age. Dis Colon Rectum. 34(4): 343-346.
- Griffin PM, Liff JM, Greenberg RS and Clark WS. (1991). Adenocarcinomas of the colon and rectum in persons under 40 years old: a population-based study. Gastroenterology. 100(4): 1033-1040.
- McKay A, Donaleshen J, Helewa RM, Park J, et al. (2014). Does young age influence the prognosis of colorectal cancer: a population-based analysis. World Journal of Surgical Oncology. 12(1): 370.
- Myers EA, Feingold DL, Forde KA, Arnell T,et al. (2013). Colorectal cancer in patients under 50 years of age: a retrospective analysis of two institutions' experience. World Journal of Gastroenterology: World J Gastroenterol. 19(34): 5651-5657.
- Ahnen DJ, Wade SW, Jones WF, Sifri R, et al. (2014). The increasing incidence of young-onset colorectal cancer: a call to action. Mayo Clinic Procee Mayo Clin Proc. 89(2): 216-224.
- Schellerer VS, Merkel S, Schumann SC, Schlabrakowski A, et al. (2012). Despite aggressive histopathology survival is not impaired in young patients with colorectal cancer : CRC in patients under 50 years of age. Int J Colorectal Dis. 27(1): 71-79.
- https://newsroom.unsw.edu.au/news/health/whats-behind-increase-bowel-cancer-among-younger-australians
- https://www.aihw.gov.au/getmedia/7403256c-c71a-4042-a0d3-bbef1a08ed69/cipa02-11.pdf.aspx?inline=true
- https://www.aihw.gov.au/reports/cancer/cancer-adolescents-young-adults/contents/table-of-contents
- https://www.aihw.gov.au/getmedia/8c9fcf52-0055-41a0-96d9-f81b0feb98cf/aihw-can-123.pdf.aspx?inline=true
- Haggar FA and Boushey RP. (2009). Colorectal cancer epidemiology: incidence, mortality, survival, and risk factors. Clin Colon Rectal Surg. 22(4): 191-197.
- Siegel RL, Miller KD and Jemal A. (2018). Cancer statistics, 2018. CA Cancer J Clin. 68(1): 7-30.
- Malvezzi M, Carioli G, Bertuccio P, Boffetta P, et al. (2018). European cancer mortality predictions for the year 2018 with focus on colorectal cancer. Ann Oncol. 29(4): 1016-1022.
- Feletto E, Yu XQ, Lew JB, St John DJB, et al. (2019). Trends in Colon and Rectal Cancer Incidence in Australia from 1982 to 2014: Analysis of Data on Over 375,000 Cases. Cancer Epidemiol Biomarkers Prev. 28(1): 83-90.
- Wolf AMD, Fontham ETH, Church TR, Flowers CR, et al. (2018). Colorectal cancer screening for average-risk adults: 2018 guideline update from the American Cancer Society. CA Cancer J Clin. 68(4): 250-281.
- https://www.aihw.gov.au/getmedia/efaa639f-d6b8-4e19-8d38-bcba7646360e/14757.pdf.aspx?inline=true
- Cole SR, Tucker GR, Osborne JM, Byrne SE, et al. (2013). Shift to earlier stage at diagnosis as a consequence of the National Bowel Cancer Screening Program. Med J Aust. 198(6): 327-330.
- Weinberg BA and Marshall JL. (2019). Colon Cancer in Young Adults: Trends and Their Implications. Curr Oncol Rep. 21(1): 3.
- Feletto E, Yu XQ, Lew J-B, St John DJB, et al. (2019). Trends in Colon and Rectal Cancer Incidence in Australia from 1982 to 2014: Analysis of Data on Over 375,000 Cases. Cancer Epidemiol Biomarkers Prev. 28(1): 83-90.
- Young JP, Win AK, Rosty C, Flight I, et al. (2015). Rising incidence of early-onset colorectal cancer in Australia over two decades: report and review. J Gastroenterol Hepatol. 30(1): 6-13.
- Bailey CE, Hu CY, You YN, Bednarski BK, et al. (2015). Increasing disparities in the age-related incidences of colon and rectal cancers in the United States, 1975-2010. JAMA Surg. 150(1): 17-22.
- Amini AQ, Samo KA and Memon AS. (2013). Colorectal cancer in younger population: our experience. J Pak Med Assoc. 63(10): 1275-1277.
- Nasira T, Rehan T, and Salman T. (2018). Incidence of Colorectal carcinoma in Young Population of Islamabad. Int J Cancer Res Ther. 4(1): 1-6.
- Gupta S, Bhattacharya D, Acharya A, Majumdar S, et al. (2010). Colorectal carcinoma in young adults: a retrospective study on Indian patients: 2000–2008. Colorectal Dis. 12(10 Online): e182-e189.
- Fazeli MS, Adel MG and Lebaschi AH. (2007). Colorectal carcinoma: a retrospective, descriptive study of age, gender, subsite, stage, and differentiation in Iran from 1995 to 2001 as observed in Tehran University. Dis Colon Rectum. 50(7): 990-995.
- Ibrahim OK, Afolayan AE, Adeniji KA, Buhari OM, et al. (2012). Colorectal carcinoma in children and young adults in Ilorin, Nigeria. West African journal of medicine. 30(3): 202-205.
- Singh Y, Vaidya P, Hemandas AK, Singh KP, et al. (2002). Colorectal carcinoma in Nepalese young adults: presentation and outcome. Gan To Kagaku Ryoho. 29 Suppl 1: 223-229.
- Ahmed G, Basel Ebeid, Aida A and Anthony A. (2014). Colorectal cancer in Egypt is commoner in young people: Is this cause for alarm? Alexandria Journal of Medicine. 50(3): 197-201.
- Chung SJ, Kim YS, Yang SY, Song JH, et al. (2010). Prevalence and risk of colorectal adenoma in asymptomatic Koreans aged 40-49 years undergoing screening colonoscopy. J Gastroenterol Hepatol. 25(3): 519-525.
- Al-Jaberi TM, Yaghan RJ and El-Heis HA. (2003). Colorectal cancer in young patients under 40 years of age. Comparison with old patients in a well defined Jordanian population. Saudi Med J. 24(8): 871-874.
- Zorluoglu A, Yilmazlar T, Ozguc H, Bagcivan E, et al. (2004). Colorectal cancers under 45 years of age. Hepatogastroenterology. 51(55): 118-120.
- De Silva M, Fernando M and Fernando D. (2000). Comparison of some clinical and histological features of colorectal carcinoma occurring in patients below and above 40 years. Ceylon Med J. 45(4): 166-168.
- Chiang JM, Chen MC, Changchien CR, Chen JS, et al. (2003). Favorable influence of age on tumor characteristics of sporadic colorectal adenocarcinoma: patients 30 years of age or younger may be a distinct patient group. Diseases of the Colon and Rectum. 46(7): 904-910.
- Al-Ahwal MS and Abdo Al-Ghamdi A. (2005). Pattern of colorectal cancer at two hospitals in the western region of Saudi Arabia. Saudi J Gastroenterol. 11(3): 164-169.
- Chew MH, Koh PK, Ng KH and Eu KW. (2009). Improved survival in an Asian cohort of young colorectal cancer patients: an analysis of 523 patients from a single institution. Int J Colorectal Dis. 24(9): 1075-1083.
- http://www.cancer.ca/Canadian-Cancer-Statistics-2017-EN
- Crosbie AB, Roche LM, Johnson LM, Pawlish KS, et al. (2018). Trends in colorectal cancer incidence among younger adults-Disparities by age, sex, race, ethnicity, and subsite. Cancer Med. 7(8): 4077-4086.
- https://bowelcancerorguk.s3.amazonaws.com/Test%20images/NeverTooYoungreport2015.pdf
- Ashktorab H, Vilmenay K, Brim H, Laiyemo AO, et al. (2016). Colorectal Cancer in Young African Americans: Is It Time to Revisit Guidelines and Prevention? Dig Dis Sci. 61(10): 3026-3030.
- Shaw C, Blakely T, Sarfati S, Fawcett J, et al. (2007). Trends in colorectal cancer mortality by ethnicity and socio‐economic position in New Zealand, 1981‐99: one country, many stories. Aust N Z J Public Health. 30(1): 64-70.
- Weir K, Supramaniam R, Gibberd A, Dillon A, et al. (2016). Comparing colorectal cancer treatment and survival for Aboriginal and non-Aboriginal people in New South Wales. Med J Aust. 204(4): 156.
- Siegel RL, Jemal A and Ward EM. (2009). Increase in incidence of colorectal cancer among young men and women in the United States. Cancer Epidemiol Biomarkers Prev. 18(6): 1695-1698.
- Siegel RL, Miller KD, Fedewa SA, Ahnen DJ, et al. (2017). Colorectal cancer statistics, 2017. CA Cancer J Clin. 67(3): 177-193.
- Goldvaser H, Purim O, Kundel Y, Shepshelovich D, et al. (2016). Colorectal cancer in young patients: is it a distinct clinical entity? Int J Clin Oncol. 21(4): 684-695.
- Lee SY, Song WH, Oh SC, Min BW, et al. (2018). Anatomical distribution and detection rate of colorectal neoplasms according to age in the colonoscopic screening of a Korean population. Ann Surg Treat Res. 94(1): 36-43.
- Andrea B-H, John DP, Jessica C, Douglas C, et al. (2018). Tumor characteristics and treatment in early-onset colorectal cancer. J Clin Oncol. 36(Suppl 4): 566
- Segev L, Kalady MF and Church JM. (2018). Left-sided dominance of early-onset colorectal cancers: a rationale for screening flexible sigmoidoscopy in the young. Dis Colon Rectum. 61(8): 897-902.
- You YN, Xing Y, Feig BW, Chang GJ, et al. (2012). Young-onset colorectal cancer: is it time to pay attention? Arch Intern Med. 172(3): 287-289.
- Carethers JM. (2011). One colon lumen but two organs. Gastroenterology. 141(2): 411- 412.
- Patel SG and Ahnen DJ. (2012). Familial colon cancer syndromes: an update of a rapidly evolving field. Curr Gastroenterol Rep. 14(5): 428-438.
- Butterworth AS, Higgins JP and Pharoah P. (2006). Relative and absolute risk of colorectal cancer for individuals with a family history: a meta-analysis. Eur J Cancer. 42(2): 216-227.
- St John DJ, McDermott FT, Hopper JL, Debney EA, et al. (1993). Cancer risk in relatives of patients with common colorectal cancer. Ann Intern Med. 118(10): 785-790.
- Salovaara R, Loukola A, Kristo P, Kaariainen H, et al. (2000). Population-based molecular detection of hereditary nonpolyposis colorectal cancer. J Clin Oncol. 18(11): 2193-2200.
- Olsson L and Lindblom A. (2003). Family history of colorectal cancer in a Sweden county. Fam Cancer. 2(2): 87-93.
- Half E, Bercovich D and Rozen P. (2009). Familial adenomatous polyposis. Orphanet J Rare Dis. 4: 22.
- Stoffel EM, Koeppe E, Everett J, Ulintz P, et al. (2018). Germline Genetic Features of Young Individuals with Colorectal Cancer. Gastroenterology. 154(4): 897-905. e1.
- Bonadona V, Bonaiti B, Olschwang S, Grandjouan S, et al. (2011). Cancer risks associated with germline mutations in MLH1, MSH2, and MSH6 genes in Lynch syndrome. JAMA. 305(22): 2304-2310.
- Senter L, Clendenning M, Sotamaa K, Hampel H, et al. (2008). The clinical phenotype of Lynch syndrome due to germ-line PMS2 mutations. Gastroenterology. 135(2): 419-428.
- AK Tiwari, HK Roy and HT Lynch. (2016). Lynch syndrome in the 21st century: clinical perspectives. OJM. 109(3): 151-158.
- Marjolijn JLL, Kuiper RP, Chan TL, Goossens M, et al. (2009). Heritable somatic methylation and inactivation of MSH2 in families with Lynch syndrome due to deletion of the 3′ exons of TACSTD1. Nat Genet. 41: 112-127.
- Niessen RC, Hofstra RM, Westers H, Ligtenberg MJ, et al. (2009). Germline hypermethylation of MLH1 and EPCAM deletions are a frequent cause of Lynch syndrome. Genes Chromosomes Cancer. 48(8): 737-744.
- Kempers MJ, Kuiper RP, Ockeloen CW, Chappuis PO, et al. (2011). Risk of colorectal and endometrial cancers in EPCAM deletion-positive Lynch syndrome: a cohort study. Lancet Oncol. 12(1): 49-55.
- Lynch HT, Lynch PM, Lanspa SJ, Snyder CL, et al. (2009). Review of the Lynch syndrome: history, molecular genetics, screening, differential diagnosis, and medicolegal ramifications. Clin Genet. 76(1):1-18.
- Lynch HT and de la Chapelle A. (2003). Hereditary colorectal cancer. N Engl J Med. 348(10): 919-932.
- Lynch HT, Lynch JF, Lynch PM and Attard T. (2008). Hereditary colorectal cancer syndromes: molecular genetics, genetic counseling, diagnosis and management. Fam Cancer. 7(1): 27-39.
- Stoffel EM and Chittenden A. (2010). Genetic testing for hereditary colorectal cancer: challenges in identifying, counseling, and managing high-risk patients. Gastroenterology. 139(5): 1436-1441 DOI: 10.1053/j.gastro.2010.09.018
- Pearlman R, Frankel WL, Swanson B, Zhao W, et al. (2017). Prevalence and Spectrum of Germline Cancer Susceptibility Gene Mutations among Patients with Early-Onset Colorectal Cancer. JAMA Oncol. 3(4): 464-471.
- Mork ME, You YN, Ying J, Bannon SA, et al. (2015). High Prevalence of Hereditary Cancer Syndromes in Adolescents and Young Adults with Colorectal Cancer. J Clin Oncol. 33(31): 3544-3549.
- Gallinger S, Aronson M, Shayan K, Ratcliffe EM, et al. (2004). Gastrointestinal cancers and neurofibromatosis type 1 features in children with a germline homozygous MLH1 mutation. Gastroenterology. 126(2): 576-85.
- Jackson CC, Holter S, Pollett A, Clendenning M, et al. (2008). Cafe-au-lait macules and pediatric malignancy caused by biallelic mutations in the DNA mismatch repair (MMR) gene PMS2. Pediatr Blood Cancer. 50(6): 1268-1270.
- Trimbath JD, Petersen GM, Erdman SH, Ferre M, et al. (2001). Cafe-au-lait spots and early onset colorectal neoplasia: a variant of HNPCC? Fam Cancer. 1(2): 101-105.
- Wimmer K and Etzler J. (2008). Constitutional mismatch repair-deficiency syndrome: have we so far seen only the tip of an iceberg? Hum Genet. 124(2): 105-122.
- Al-Sukhni W, Aronson M and Gallinger S. (2007). Hereditary colorectal cancer with DNA microsatellite instability. Surg Oncol. 16.
- Chen E, Xu X and Liu T. (2018). Hereditary Nonpolyposis Colorectal Cancer and Cancer Syndromes: Recent Basic and Clinical Discoveries. J Oncol. AID 3979135: 11.
- Bolocan A, Ion D, Stoian RV and Serban MB. (2011). Map syndrome (MYH Associated Polyposis) colorectal cancer, etiopathological connections. J Med Life. 4(1): 109-111.
- Landon M, Ceulemans S, Saraiya DS, Strike B, et al. (2015). Analysis of current testing practices for biallelic MUTYH mutations in MUTYH-associated polyposis. Clin Genet. 87(4): 368-372.
- http://atlasgeneticsoncology.org/Kprones/MAPID10092.html
- Guarinos C, Juarez M, Egoavil C, Rodriguez-Soler M, et al. (2014). Prevalence and characteristics of MUTYH-associated polyposis in patients with multiple adenomatous and serrated polyps. Clin Cancer Res. 20(5): 1158-1168.
- Snover DC. (2010). Serrated polyps of the colon and rectum and serrated polyposis, WHO classification of tumours of the digestive system. 2010: 160-165.
- Boparai KS, Mathus-Vliegen EM, Koornstra JJ, Nagengast FM, et al. (2010). Increased colorectal cancer risk during follow-up in patients with hyperplastic polyposis syndrome: a multicentre cohort study. Gut. 59(8): 1094-1100.
- Lucci-Cordisco E, Risio M, Venesio T and Genuardi M. (2013). The growing complexity of the intestinal polyposis syndromes. Am J Med Genet A. 161A(11): 2777-2787.
- Rosty C, Parry S and Young JP. (2011). Serrated polyposis: an enigmatic model of colorectal cancer predisposition. Patholog Res Int. 1-13.
- Walls HL, Magliano DJ, Stevenson CE, Backholer K, et al. (2012). Projected progression of the prevalence of obesity in Australia. Obesity (Silver Spring). 20(4): 872-878.
- Garnett SP, Baur LA, Jones AM and Hardy LL. (2016). Trends in the Prevalence of Morbid and Severe Obesity in Australian Children Aged 7-15 Years, 1985-2012. PLoS One. 11(5): e0154879.
- https://www.abs.gov.au/ausstats/[email protected]/Lookup/by%20Subject/4364.0.55.001~2017-18~Main%20Features~Overweight%20and%20obesity~90
- https://data.oecd.org/agroutput/meat-consumption.htm
- Nguyen LH, Liu PH, Zheng X, Keum N, et al. (2018). Sedentary Behaviors, TV Viewing Time, and Risk of Young-Onset Colorectal Cancer. JNCI Cancer Spectr. 2(4): pky073.
- de Kort S, Masclee AAM, Sanduleanu S, Weijenberg MP, et al. (2017). Higher risk of colorectal cancer in patients with newly diagnosed diabetes mellitus before the age of colorectal cancer screening initiation. Sci Rep. 7: 46527.
- Triantafillidis JK, Nasioulas G and Kosmidis PA. (2009). Colorectal cancer and inflammatory bowel disease: epidemiology, risk factors, mechanisms of carcinogenesis and prevention strategies. Anticancer Res. 29(7): 2727-2737.
- Safaeian M, Robbins HA, Berndt SI, Lynch CF, et al. (2016). Risk of Colorectal Cancer After Solid Organ Transplantation in the United States. Am J Transplantat. 16(3): 960-967.
- Enblad M, Birgisson H, Ekbom A, Sandin F, et al. (2017). Increased incidence of bowel cancer after non-surgical treatment of appendicitis. Eur J Surg Oncol 43(11): 2067-2075.
- Golshiri P, Rasooli S, Emami M and Najimi A. (2016). Effects of Physical Activity on Risk of Colorectal Cancer: A Case-control Study. Int J Prev Med. 7: 32.
- Liu PH, Wu K, Song M, Zauber AG, et al. (2018). Association of Obesity With Risk of Early-Onset Colorectal Cancer Among Women. JAMA Oncol. 5(1): 37-44.
- Larsson SC and Wolk A. (2006). Meat consumption and risk of colorectal cancer: a meta-analysis of prospective studies. Int J Cancer. 119(11): 2657-2664.
- Terry P, Giovannucci E, Michels KB, Bergkvist L, et al. (2001). Fruit, vegetables, dietary fiber, and risk of colorectal cancer. J Natl Cancer Inst. 93(7): 525-533.
- Garland C, Shekelle RB, Barrett-Connor E, Criqui MH, et al. (1985). Dietary vitamin D and calcium and risk of colorectal cancer: a 19-year prospective study in men. Lancet. 1(8424): 307-309.
- Song M, Nishihara R, Wang M, Chan AT, et al. (2016). Plasma 25-hydroxyvitamin D and colorectal cancer risk according to tumour immunity status. Gut. 65(2): 296-304.
- Cho E, Smith-Warner SA, Spiegelman D, Beeson WL, et al. (2004). Dairy foods, calcium, and colorectal cancer: a pooled analysis of 10 cohort studies. J Natl Cancer Inst. 96(13): 1015-1022.
- Anderson JC, Attam R, Alpern Z, Messina CR, et al. (2003). Prevalence of colorectal neoplasia in smokers. Am J Gastroenterol. 98(12): 2777-2783.
- Lieberman DA, Prindiville S, Weiss DG and Willett W. (2003). Risk factors for advanced colonic neoplasia and hyperplastic polyps in asymptomatic individuals. JAMA. 290(22): 2959-2967.
- de Castilhos ED, Horta BL, Gigante DP, Demarco FF, et al. (2012). Association between obesity and periodontal disease in young adults: a population-based birth cohort. J Clin Periodontol. 39(8): 717-724.
- Vrieze A, Van Nood E, Holleman F, Salojarvi J, et al. (2012). Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 143(4): 913-916. e7.
- Kato I, Vasquez AA, Moyerbrailean G, Land S, et al. (2016). Oral microbiome and history of smoking and colorectal cancer. J Epidemiol Res. 2(2): 92-101.
- Lakkis D, Bissada NF, Saber A, Khaitan L, et al. (2012). Response to periodontal therapy in patients who had weight loss after bariatric surgery and obese counterparts: a pilot study. J Periodontol. 83(6): 684-689.
- Flynn KJ, Ruffin MT, Turgeon DK and Schloss PD. (2018). Spatial variation of the native colon microbiota in healthy adults. Cancer Prev Res. 11(7): 393-402.
- Mima K, Cao Y, Chan AT, Qian ZR, et al. (2016). Fusobacterium nucleatum in Colorectal Carcinoma Tissue According to Tumor Location. Clin Transl Gastroenterol. 7(11): e200.
- Bullman S, Pedamallu CS, Sicinska E, Clancy TE, et al. (2017). Analysis of Fusobacterium persistence and antibiotic response in colorectal cancer. Science. 358(6369): 1443-1448.
- https://bowel-cancer.canceraustralia.gov.au/statistics
- Van Cutsem E, Cervantes A, Nordlinger B and Arnold D. (2014). Metastatic colorectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 25(Suppl 3): iii1-9.
- Quah HM, Joseph R, Schrag D, Shia J, et al. (2007). Young age influences treatment but not outcome of colon cancer. Ann Surg Oncol. 14(10): 2759-2765.
- Blanke CD, Bot BM, Thomas DM, Bleyer A, et al. (2011). Impact of young age on treatment efficacy and safety in advanced colorectal cancer: a pooled analysis of patients from nine first-line phase III chemotherapy trials. J Clin Oncol. 29(20): 2781-2786.
- Patel SG and Ahnen DJ. (2018). Colorectal Cancer in the Young. Curr Gastroenterol Rep. 20(4): 15.
- Inra JA and Syngal S. (2015). Colorectal cancer in young adults. Dig Dis Sci. 60(3): 722-733.
- Chen FW, Sundaram V, Chew TA and Ladabaum U. (2017). Advanced-Stage Colorectal Cancer in Persons Younger Than 50 Years Not Associated With Longer Duration of Symptoms or Time to Diagnosis. Clin Gastroenterol and Hepatol. 15(5): 728-737.e3.
- Rho YS, Gilabert M, Polom K, Aladashvili A, et al. (2017). Comparing Clinical Characteristics and Outcomes of Young-onset and Late-onset Colorectal Cancer: An International Collaborative Study. Clin Colorectal Cancer. 16(4): 334-342.
- Boyce S, Nassar N, Lee CY, Suen MK, et al. (2016). Young-onset colorectal cancer in New South Wales: a population-based study. Med J Aust. 205(10): 465-470.
- Yeo H, Betel D, Abelson JS, Zheng XE, et al. (2017). Early-onset Colorectal Cancer is Distinct From Traditional Colorectal Cancer. Clin Colorectal Cancer. 16(4): 293-299.e6.
- Kneuertz PJ, Chang GJ, Hu CY, Rodriguez-Bigas MA, et al. (2015). Overtreatment of young adults with colon cancer: more intense treatments with unmatched survival gains. JAMA Surg. 150(5): 402-409.
- Indini A, Bisogno G, Cecchetto G, Vitellaro M, et al. (2017). Gastrointestinal tract carcinoma in pediatric and adolescent age: The Italian TREP project experience. Pediatr Blood Cancer. 64(12).
- Kam MH, Eu KW, Barben CP and Seow-Choen F. (2004). Colorectal cancer in the young: a 12-year review of patients 30 years or less. Colorectal Dis. 6(3): 191-194.
- Ferrari A, Rognone A, Casanova M, Zaffignani E, et al. (2008). Colorectal carcinoma in children and adolescents: the experience of the Istituto Nazionale Tumori of Milan, Italy. Pediatr Blood Cancer. 50(3): 588-593.
- Russo AG, Andreano A, Sartore-Bianchi A, Mauri G, et al. (2019). Increased incidence of colon cancer among individuals younger than 50 years: A 17 years analysis from the cancer registry of the municipality of Milan, Italy. Cancer Epidemiology. 60: 134-140.
- Kasi PM, Shahjehan F, Cochuyt JJ, Li Z, et al. (2019). Rising Proportion of Young Individuals With Rectal and Colon Cancer. Clin Colorectal Cancer. 18(1): e87-e95.
- Rodriguez L, Brennan K, Karim S, Nanji S, et al. (2018). Disease characteristics, clinical management, and outcomes of young patients with colon cancer: a population-based study. Clin Colorectal Cancer. 17(4): e651-e61.
- Vatandoust S, Price TJ, Ullah S, Roy AC, et al. (2016). Metastatic Colorectal Cancer in Young Adults: A Study From the South Australian Population-Based Registry. Clin Colorectal Cancer. 15(1): 32-36.
- Willauer AN, Liu Y, Pereira AAL, Lam M, et al. (2019). Clinical and molecular characterization of early-onset colorectal cancer. Cancer. 125(12): 2002-2010.
- Li Q, Cai G, Li D, Wang Y, et al. (2014). Better long-term survival in young patients with non-metastatic colorectal cancer after surgery, an analysis of 69,835 patients in SEER database. PLoS One 9(4): e93756.
- Yantiss RK, Goodarzi M, Zhou XK, Rennert H, et al. (2009). Clinical, pathologic, and molecular features of early-onset colorectal carcinoma. Am J Surg Pathol. 33(4): 572-582.
- Chang DT, Pai RK, Rybicki LA, Dimaio MA, et al. (2012). Clinicopathologic and molecular features of sporadic early-onset colorectal adenocarcinoma: an adenocarcinoma with frequent signet ring cell differentiation, rectal and sigmoid involvement, and adverse morphologic features. Mod Pathol. 25(8): 1128-1139.
- Verhulst J, Ferdinande L, Demetter P and Ceelen W. (2012). Mucinous subtype as prognostic factor in colorectal cancer: a systematic review and meta-analysis. J Clin Pathol. 65(5): 381-388.
- Chew MH, Yeo SA, Ng ZP, Lim KH, et al. (2010). Critical analysis of mucin and signet ring cell as prognostic factors in an Asian population of 2,764 sporadic colorectal cancers. Int J Colorectal Dis. 25(10): 1221-1229.
- Borger ME, Gosens MJ, Jeuken JW, van Kempen LC, et al. (2007). Signet ring cell differentiation in mucinous colorectal carcinoma. J Pathol. 212(3): 278-286.
- Gopalan V, Smith RA, Ho YH and Lam AK. (2011). Signet-ring cell carcinoma of colorectum--current perspectives and molecular biology. Int J Colorectal Dis. 26(2): 127-133.
- Kang H, O'Connell JB, Maggard MA, Sack J, et al. (2005). A 10-year outcomes evaluation of mucinous and signet-ring cell carcinoma of the colon and rectum. Dis Colon Rectum. 48(6): 1161-1168.
- Chen JS, Hsieh PS, Chiang JM, Yeh CY, et al. (2010). Clinical outcome of signet ring cell carcinoma and mucinous adenocarcinoma of the colon. Chang Gung Med J. 33(1): 51-57.
- Jennifer L, Matthew FK and James C. (2015). Young age of onset colorectal cancers. Int J Colorectal Dis. 30(12): 1653-1657.
- Burt R. (2007). Inheritance of Colorectal Cancer. Drug Discov Today Dis Mech. 4(4): 293-300.
- Silla IO, Rueda D, Rodriguez Y, Garcia JL, et al. (2014). Early-onset colorectal cancer: a separate subset of colorectal cancer. World J Gastroenterol. 20(46): 17288-17296.
- Valle L, de Voer RM, Goldberg Y, Sjursen W, et al. (2019). Update on genetic predisposition to colorectal cancer and polyposis. Mol Aspects Med. DOI: 10.1016/j.mam.2019.03.001
- Snover DC. (2011). Update on the serrated pathway to colorectal carcinoma. Human Pathol. 42(1): 1-10.
- Jass JR. (2007). Classification of colorectal cancer based on correlation of clinical, morphological and molecular features. Histopathology. 50(1): 113-130.
- Toyota M, Ahuja N, Ohe-Toyota M, Herman JG, et al. (1999). CpG island methylator phenotype in colorectal cancer. Proc Natl Acad Sci USA. 96(15): 8681-8686.
- Mauri G, Sartore-Bianchi A, Russo AG, Marsoni S, et al. (2019). Early-onset colorectal cancer in young individuals. Mol Oncol. 13(2): 109-131.
- Armaghany T, Wilson JD, Chu Q and Mills G. (2012). Genetic alterations in colorectal cancer. Gastrointest Cancer Res. 5(1): 19-27.
- Cisyk AL, Nugent Z, Wightman RH, Singh H, et al. (2018). Characterizing Microsatellite Instability and Chromosome Instability in Interval Colorectal Cancers. Neoplasia. 20(9): 943-950.
- Lengauer C, Kinzler KW and Vogelstein B. (1998). Genetic instabilities in human cancers. Nature. 396(6712): 643-649.
- Markowitz SD and Bertagnolli MM. (2009). Molecular origins of cancer: Molecular basis of colorectal cancer. N Engl J Med. 361(25): 2449-2460.
- Pino MS and Chung DC. (2010). The chromosomal instability pathway in colon cancer. Gastroenterology. 138(6): 2059-2072.
- Antelo M, Balaguer F, Shia J, Shen Y, et al. (2012). A high degree of LINE-1 hypomethylation is a unique feature of early-onset colorectal cancer. PLoS One. 7(9): e45357.
- Boardman LA, Johnson RA, Petersen GM, Oberg AL, et al. (2007). Higher frequency of diploidy in young-onset microsatellite-stable colorectal cancer. Clin Cancer Res. 13(8): 2323-2328.
- Tan C and Du X. (2012). KRAS mutation testing in metastatic colorectal cancer. World J Gastroenterol. 18(37): 5171-5180.
- Karapetis CS, Khambata-Ford S, Jonker DJ, O'callaghan CJ, et al. (2008). K-ras mutations and benefit from cetuximab in advanced colorectal cancer. N Engl J Med. 359(17): 1757-1765.
- Phipps AI, Limburg PJ, Baron JA, Burnett-Hartman AN, et al. (2015). Association between molecular subtypes of colorectal cancer and patient survival. Gastroenterology. 148(1): 77-87. e2.
- Gryfe R, Kim H, Hsieh ET, Aronson MD, et al. (2000). Tumor microsatellite instability and clinical outcome in young patients with colorectal cancer. N Engl J Med. 342(2): 69-77.
- Goel A, Nagasaka T, Spiegel J, Meyer R, et al. (2010). Low frequency of Lynch syndrome among young patients with non-familial colorectal cancer. Clin Gastroenterol Hepatol. 8(11): 966-971.
- Alsop K, Mead L, Smith LD, Royce SG, et al. (2006). Low somatic K-ras mutation frequency in colorectal cancer diagnosed under the age of 45 years. Eur J Cancer. 42(10): 1357-1361.
- Watson R, Liu TC and Ruzinova MB. (2016.) High frequency of KRAS mutation in early onset colorectal adenocarcinoma: implications for pathogenesis. Hum Pathol. 56: 163-170.
- Silva AC, Vicentini MFB, Mendoza EZ, Fujiki FK, et al. (2019). Young-age onset colorectal cancer in Brazil: Analysis of incidence, clinical features, and outcomes in a tertiary cancer center. Curr Probl Cancer. https://doi.org/10.1016/j.currproblcancer.2019.01.009
- Chouhan H, Ferrandon S, DeVecchio J, Kalady MF, et al. (2019). A Changing Spectrum of Colorectal Cancer Biology with Age: Implications for the Young Patient. Dis Colon Rectum. 62(1): 21-26.
- Magnani G, Furlan D, Sahnane N, Reggiani Bonetti L, et al. (2015). Molecular Features and Methylation Status in Early Onset (</=40 Years) Colorectal Cancer: A Population Based, Case-Control Study. Gastroenterol Res Pract. 2015: 132190
- Antelo M, Balaguer F, Shia J, Shen Y, et al. (2012). A high degree of LINE-1 hypomethylation is a unique feature of early-onset colorectal cancer. PLoS One. 7(9): e45357.
- van Roon EH, van Puijenbroek M, Middeldorp A, van Eijk R, et al. (2010). Early onset MSI-H colon cancer with MLH1 promoter methylation, is there a genetic predisposition? BMC Cancer. 10: 180.
- Giraldez MD, Balaguer F, Bujanda L, Cuatrecasas M, et al. (2010). MSH6 and MUTYH deficiency is a frequent event in early-onset colorectal cancer. Clin Cancer Res. 16(22): 5402-5413.
- Bardhan K and Liu K. (2002). Epigenetics and colorectal cancer pathogenesis. Cancers (Basel). 5(2): 676-713.
- Leslie A, Carey FA, Pratt NR and Steele RJ. (2002). The colorectal adenoma-carcinoma sequence. Br J Surg. 89(7): 845-860.
- Khosraviani K. (1996). Colorectal adenoma-carcinoma sequence. Gut. 39(2): 342.
- Fernandez‐Rozadilla C, Brea‐Fernández A, Bessa X, Álvarez‐Urturi C, et al. (2013). BMPR1A mutations in early‐onset colorectal cancer with mismatch repair proficiency. Clin Genet. 84(1): 94-96.
- Kim Y-J, Jung Y-D, Kim T-O and Kim H-S. (2013). Alu-related transcript of TJP2 gene as a marker for colorectal cancer. Gene. 524(2): 268-274.
- Berg M, Ågesen TH, Thiis-Evensen E, Merok MA, et al. (2010). Distinct high resolution genome profiles of early onset and late onset colorectal cancer integrated with gene expression data identify candidate susceptibility loci. Mol Cancer. 9(1): 100.
- Freyaldenhoven BS, Fried C and Wielckens K. (2002). FOXD4a and FOXD4b, two new winged helix transcription factors, are expressed in human leukemia cell lines. Gene. 294(1-2): 131-140.
- Sugai T and Habano W. (2016). [Pathological Diagnosis and Its Molecular Basis in Colorectal Cancer]. Gan To Kagaku Ryoho. 43(3): 294-299.
- Perea J, Rueda D, Canal A, Rodriguez Y, et al. (2014). Age at onset should be a major criterion for subclassification of colorectal cancer. J Mol Diagn. 6(1): 116-126.
- Tejpar S, Stintzing S, Ciardiello F, Tabernero J, et al. (2016). Prognostic and Predictive Relevance of Primary Tumor Location in Patients With RAS Wild-Type Metastatic Colorectal Cancer: Retrospective Analyses of the CRYSTAL and FIRE-3 Trials. JAMA Oncol. 3(2): 194-201.
- Brule SY, Jonker DJ, Karapetis CS, O'Callaghan CJ, et al. (2015). Location of colon cancer (right-sided versus left-sided) as a prognostic factor and a predictor of benefit from cetuximab in NCIC CO.17. Eur J Cancer. 51(11): 1405-1414.
- Venook AP, Niedzwiecki D, Innocenti F, Fruth B, et al. (2016). Impact of primary (1º) tumor location on overall survival (OS) and progression-free survival (PFS) in patients (pts) with metastatic colorectal cancer (mCRC): Analysis of CALGB/SWOG 80405 (Alliance). J Clin Oncol. 34(Supple 15): 3504.
- Schrag D, Weng S, Brooks G, Meyerhardt JA, et al. (2016). The relationship between primary tumor sidedness and prognosis in colorectal cancer. J Clin Oncol. 34(Supple 15): 3505.
- Hendifar A, Yang D, Lenz F, Lurje G, et al. (2009). Gender disparities in metastatic colorectal cancer survival. Clin Cancer Res. 15(20): 6391-6397.
- Slattery ML, Potter JD, Curtin K, Edwards S, et al. (2001). Estrogens reduce and withdrawal of estrogens increase risk of microsatellite instability-positive colon cancer. Cancer Res. 61(1): 126-130.
- Puccini A, Lenz HJ, Marshall JL, Arguello D, et al. (2018). Impact of Patient Age on Molecular Alterations of Left-Sided Colorectal Tumors. Oncologist. 24(3): 319-326.
- Schmoll HJ, Van Cutsem E, Stein A, Valentini V, et al. (2012). ESMO Consensus Guidelines for management of patients with colon and rectal cancer. a personalized approach to clinical decision making. Ann Oncol. 23(10): 2479-2516.
- https://wiki.cancer.org.au/australia/Guidelines:Colorectal_cancer
- Ballester V, Rashtak S and Boardman L. (2016). Clinical and molecular features of young-onset colorectal cancer. World J Gastroenterol. 22(5): 1736-1744.
- Cavestro GM, Mannucci A, Zuppardo RA, Di Leo M, et al. (2018). Early onset sporadic colorectal cancer: Worrisome trends and oncogenic features. Dig Liver Dis. 50(6): 521-532.
- Cremolini C, Schirripa M, Antoniotti C, Moretto R, et al. (2015). First-line chemotherapy for mCRC-a review and evidence-based algorithm. Nat Rev Clin Oncol. 12(10): 607-619.
- Hu CY, Bailey CE, You YN, Skibber JM, et al. (2015). Time trend analysis of primary tumor resection for stage IV colorectal cancer: less surgery, improved survival. JAMA Surg. 150(3): 245-251.
- Cook AD, Single R and McCahill LE. (2005). Surgical resection of primary tumors in patients who present with stage IV colorectal cancer: an analysis of surveillance, epidemiology, and end results data, 1988 to 2000. Ann Surg Oncol. 12(8): 637-645.
- de Mestier L, Manceau G, Neuzillet C, Bachet JB, et al. (2014). Primary tumor resection in colorectal cancer with unresectable synchronous metastases: A review. World J Gastrointest Oncol. 6(6): 156-169.
- Temple LK, Hsieh L, Wong WD, Saltz L, et al. (2004). Use of surgery among elderly patients with stage IV colorectal cancer. J Clin Oncol. 22(17): 3475-3484.
- Venderbosch S, de Wilt JH, Teerenstra S, Loosveld OJ, et al. (2011). Prognostic value of resection of primary tumor in patients with stage IV colorectal cancer: retrospective analysis of two randomized studies and a review of the literature. Ann Surg Oncol. 18(12): 3252-3260.
- Karoui M, Roudot-Thoraval F, Mesli F, Mitry E, et al. (2011). Primary colectomy in patients with stage IV colon cancer and unresectable distant metastases improves overall survival: results of a multicentric study. Dis Colon Rectum. 54(8): 930-938.
- Ferrand F, Malka D, Bourredjem A, Allonier C, et al. (2013). Impact of primary tumour resection on survival of patients with colorectal cancer and synchronous metastases treated by chemotherapy: results from the multicenter, randomised trial Federation Francophone de Cancerologie Digestive 9601. Eur J Cancer. 49(1): 90-97.
- Stillwell AP, Buettner PG and Ho YH. (2010). Meta-analysis of survival of patients with stage IV colorectal cancer managed with surgical resection versus chemotherapy alone. World J Surg. 34(4): 797-807.
- You YN, Dozois EJ, Boardman LA, Aakre J, et al. (2011). Young-onset rectal cancer: presentation, pattern of care and long-term oncologic outcomes compared to a matched older-onset cohort. Ann Surg Oncol. 18(9): 2469-2476.
- Fossum CC, Alabbad JY, Romak LB, Hallemeier CL, et al. (2017). The role of neoadjuvant radiotherapy for locally-advanced rectal cancer with resectable synchronous metastasis. J Gastrointest Oncol. 8(4): 650-658.