Information Links
Related Conferences
Previous Issues Volume 6, Issue 1 - 2021
Caecal Metastasis: An Exceptional Manifestation Mode Revealing Small Cell Lung Carcinoma: Clinical Case and Review of the Literature
W Ben Makhlouf1, Z Dahmani2, M Hamdani1, J Elghoul3, K Bel Hadj Ali1, A Khabir1*
1Pathology department, Chu Habib Bourguiba of Medenine, Tunisia
2Gastroenterology and Hepatologydepartment, Chu Habib Bourguiba of Medenine, Tunisia
3Department of pneumology, Chu Habib Bourguiba of Medenine, Tunisia
*Corresponding author: Dr. Abdelmajid Khabir, Faculty of Medicine, University of Sfax, Tunisia. Tel : +21698656812, E-mail: [email protected]
Received Date: 13-09-2021
Published Date: October 28, 2021
Copyright: Khabir A, et al. © (2021). This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Citation: Khabir A, et al. (2021). Caecal Metastasis: An Exceptional Manifestation Mode Revealing Small Cell Lung Carcinoma: Clinical Case and Review of the Literature. Mathews J Gastroenterol Hepatol. 6(1):15.
ABSTRACT
Lung cancer is the leading cause of death worldwide. Almost 50% of patients present with distant metastasis at the moment of diagnosis. The most common metastasis sites are lymph nodes, liver, adrenal glands, bones, and brain. However, gastrointestinal tract (GIT) metastasis from primary lung carcinoma is a rare phenomenon and it is considered a late stage of the disease, generally detected in patients with a documented previous history of a primary lung malignancy. By contrast, the finding of a lung cancer initially manifesting with GI-tract involvement is extremely rare and it is usually reported in the literature in isolated case reports. This case study involves a 70 year-old-man, initially presenting with digestive symptoms related to caecal metastasis from primary lung carcinoma.
KEYWORDS: lung cancer, small cell carcinoma, caecal metastasis
INTRODUCTION
Gastrointestinal tract (GIT) metastases from lung cancer are extremely rare. They are usually asymptomatic and often discovered at a late stage of the disease or at autopsy studies. We, herein, report a case of a patient with caecal metastasis from small cell lung carcinoma presenting initially with abdominal symptoms. Histopathological examination and immunohistochemical staining of the caecal biopsies confirmed the diagnosis of metastatic neuroendocrine small cell lung carcinoma.
CASE REPORT
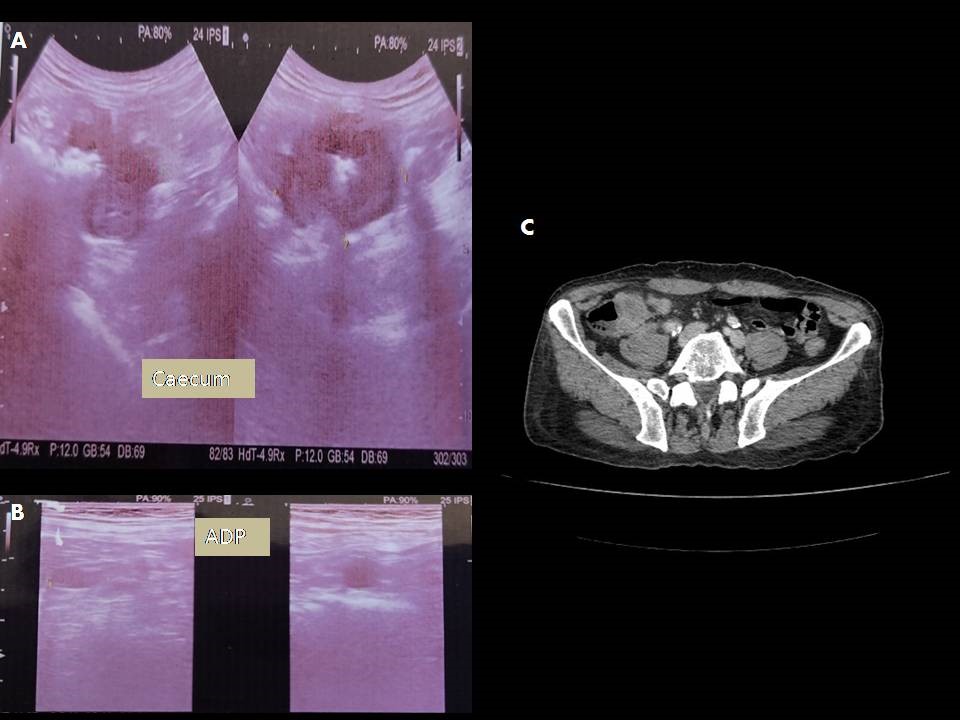
A 70-year-old male patient with no pathological history, a former cigarette smoker (40 Pack-year) who quitted smoking 2 years earlier, consulted our emergency department for cramp-type left flank pain associated with constipation and vomiting progressing for 2 months. The patient also reported asthenia, anorexia, and weight loss. The examination showed generalized abdominal defense. Abdominal ultrasound showed a solid caecal mass with irregular contour, hypoechoic internal echotexture, measuring 6x5x4 cm with multiple enlarged lymph nodes, suggesting a secondary origin. Abdominal CT scan was performed and showed a large caecal mass with exophytic development and heterogeneous enhancement measuring about 45 x 46 x 50 mm in contact with the last ileal loop that appeared to be invaded and it was associated with multiple locoregional necrotic lymphadenopathy in continuity with the tumor and in para aortic region (Figure 1).
Diagnosis based on the clinical and radiological findings was in favor of colonic tumor. A colonoscopy under general anesthesia was performed and showed an obstructing circumferential and ulcerative tumor of the caecum. Biopsies were taken.
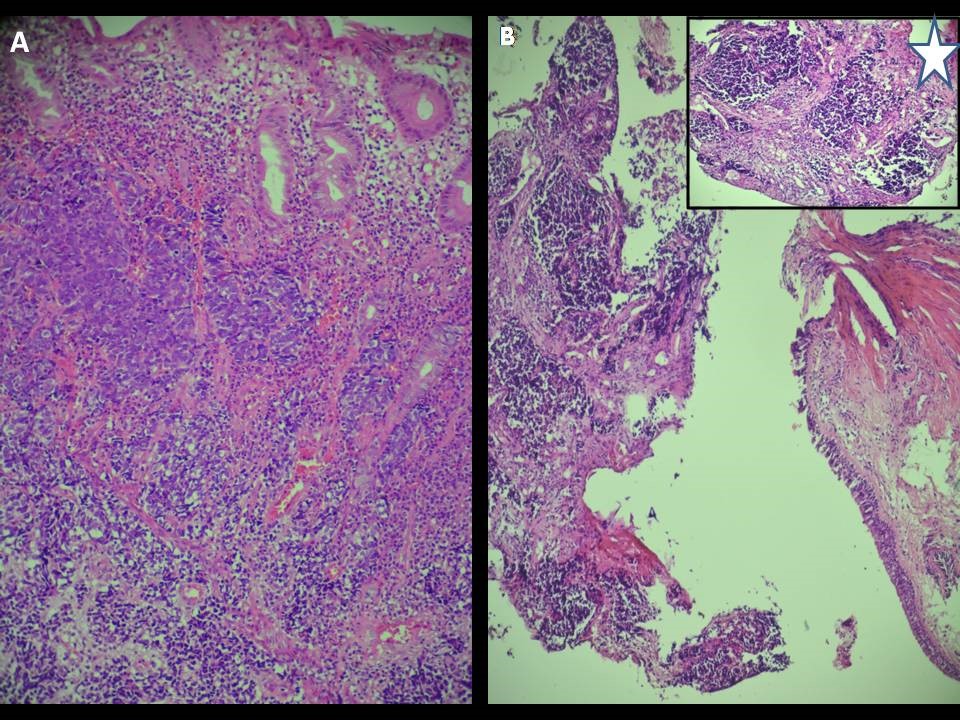
Pathological examination revealed a largely ulcerated colonic mucosa with carcinomatous proliferation, arranged in small and sometimes medium cell clusters with reduced cytoplasm. The nuclei were monomorphic with a salt and pepper appearance. These tumor cells were in some places crushed and poorly delimited. Mitoses were numerous. The stroma was reduced, and having neuroendocrine type (Figure 2).
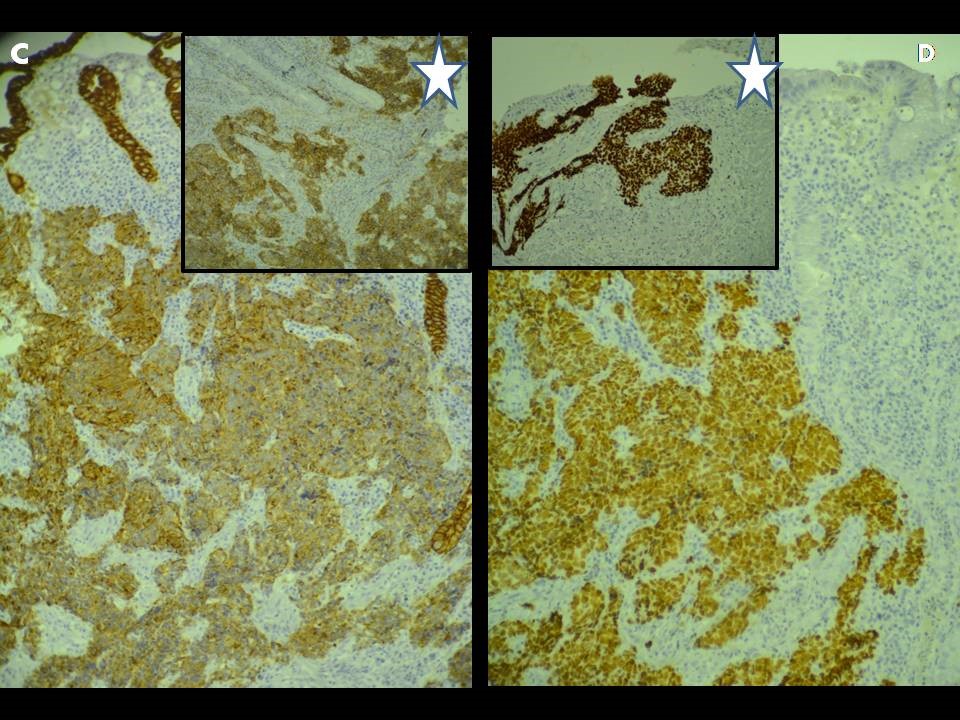
Immunohistochemical studies revealed that the tumor cells were positive for cyto-keratin, weakly positive for synaptophysin, and strongly positive for CD56 and TTF1 (Figure3). Although the latter can be seen in primary neuroendocrine carcinoma of the digestive tract, a search for a primary pulmonary origin should be performed.
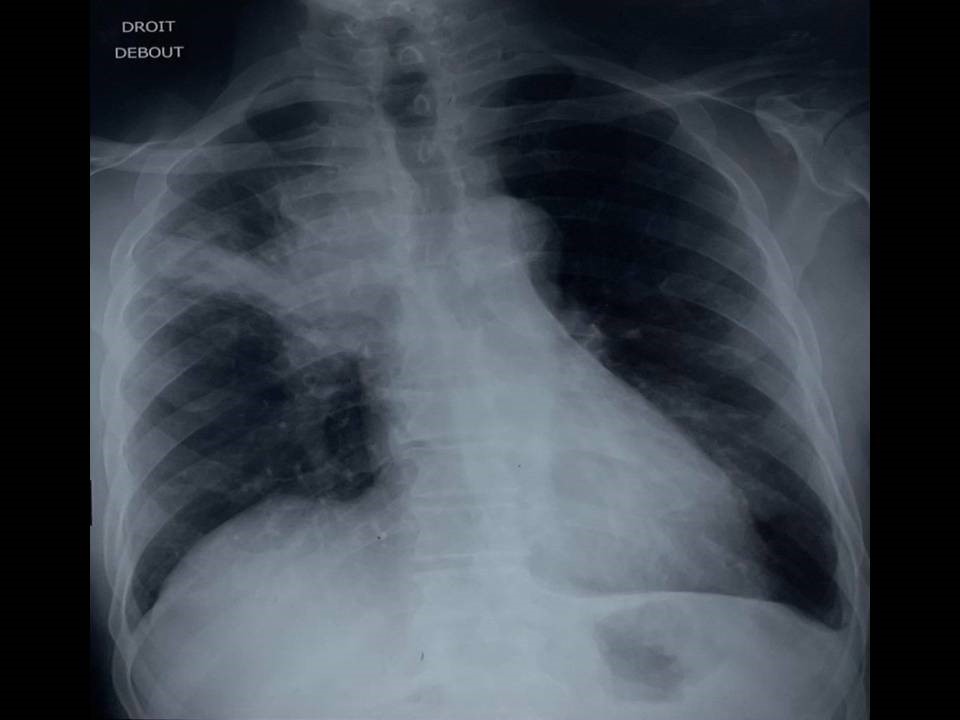
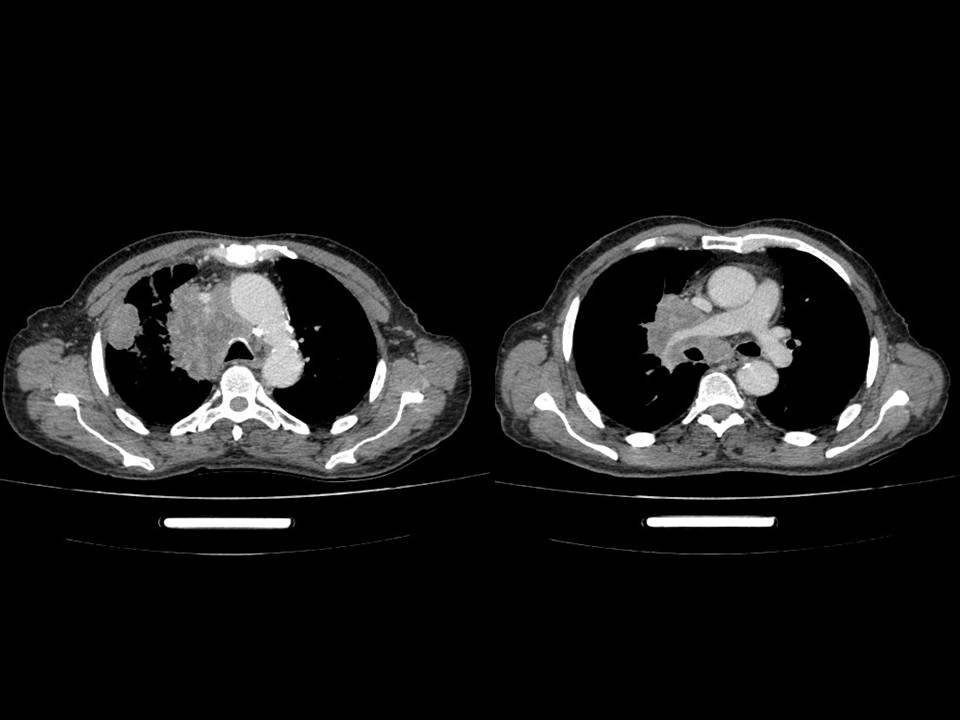
Chest x-ray showed a para-hilar opacity with poorly demarcated external limits in the upper right pulmonary lobe (Figure 4). Thoracic CT scan was therefore performed and it showed a voluminous right mediastino-apical mass with poorly limited contours in close contact with the brachio-cephalic arterial trunk, the trachea, the right main bronchus, and the superior vena cava, which invades it, sheathing the right pulmonary artery. It was associated with a subcarinal lymphadenopathy of 16 x25 mm, locoregional lymph nodes and an average lobar sub-pleural tissue mass of 24 x 30 mm in contact with the parietal suggesting secondary origin (Figure5).
Bronchialfibroscopy revealed complete and impassable obstruction of the right upper lobe bronchus. Pathological and immunohistochemical examination of the lung biopsies showed the same histological appearance of the digestive biopsies described above. Diagnosis of bronchial neuroendocrine small cell carcinoma with caecal metastasis was therefore made.
Figure 1: abdominal ultrasound: [A] solid caecal mass with irregular contour with multiple enlarged lymph nodes [B]. [C] abdominal CT scan: large caecal mass with exophytic development and heterogeneous enhancement.
Figure 2: [A] undifferentiated colic infiltrating carcinoma. Tumor cells are small with scant cytoplasm and are arranged in lobular groups [HE x 200]. [B] Tumor cells infiltrate the bronchial mucosa [HE x 200].
Figure 3: Immunohistochimical staining for keratine, CD56 and TTF1: [A]: Strong cytoplasmic staining for keratine, [A*]: Strong cytoplasmic staining for CD56. [B]: Strong nuclear staining for TTF1 in colic tumor cells. [B*]: Strong nuclear staining for TTF1 in bronchial tumor cells [x 200]
Figure 4: Chest x-ray: para-hilar opacity with poorly demarcated external limits in the upper right pulmonary lobe.
Figure 5: Thoracic CT scan: voluminous right mediastino-apical mass with poorly limited contours with lobar sub-pleural tissue mass.
DISCUSSION
GIT metastasis from primary lung carcinoma are rare with an incidence ranging from 4.7 to 14% in autopsy studies [1,2]. This incidence is much lower when patients are alive ranging from 0,3 to 1,8% [3,4] because of the asymptomatic course of the disease most often [5].
In a large case series involving over 11-year period of assessment by Mac Neill, et al., out of 6,006 patients hospitalized in an institution for bronchial cancer, 46 of the patients were found to have metastasis in the digestive tract at autopsy. All these patients presented at least one other metastatic site, with a mean of 4.8 locations. Only six patients presented this digestive metastatic evolution during their lifetime, which was a mode of discovering the disease for only one patient among them [6].
In a decreasing order of frequency, digestive metastasis sites are esophagus , small intestine, stomach, colon and the anus [7]. The circumstances in which a metastatic digestive lesion of a pulmonary tumor is discovered are quite stereotypical. Regarding patients whose primary lung cancer is already known, the main clinical form is acute perforation. Sometimes, it is revealed by other complications, such as dysphagia in case of esophageal localization, hematemesis in case of gastric or duodenal localization, acute peritonitis, obstruction, acute or chronic GI bleed in case of localization in the small intestine, GI bleed, and microcytic anemia in case of colonic localization. The Discovery of a pulmonary lesion by an inaugural digestive symptomatology is much rarer. The existence of a table involving simple acute abdominal pain without perforation, as found in our case, remains exceptional [7,8].
No histological type seems more conducive to the occurrence of digestive metastasis, however, different autopsy studies have shown that squamous cell carcinoma is the most common with a rate of 33%, followed by large cell (20 to 29%), and small cell lung cancer [13 to 23%) [5,8]. Yet, other more recent papers have observed that poorly differentiated pulmonary adenocarcinomas and large cell undifferentiated carcinomas seem to have a particular predilection for the GI tract [9].
The pathophysiological mechanism of intestinal metastasis is unknown. The lymphatic pathway seems unlikely because the abdominal lymphatic system is a one-way that drains from the abdomen to the chest via the thoracic duct. The hematogenous pathway seems to be the most likely. Indeed, 10% of patients have asymptomatic intracardiac dissemination, detectable only on systematic cardiac ultrasound [7]. The predominant mode of dissemination depends, in part, on the histological type of the tumor. For instance, small cell carcinoma of the lung shows a very high incidence of vascular invasion and is among the most aggressive tumors [5].
Gastrointestinal endoscopy is an accurate method of identifying patients with gastric, duodenal, or colonic metastatic tumors. The endoscopic appearance is extremely variable, possibly appearing as a diffuse involvement of the intestinal mucosa, multiple nodules with either mucosa erosion or ulceration, or even as a single “volcano-like” lesion mimicking a primary GI tumor [10,11]; which was the case of our patient presenting with a stenosing ulcerative budding endoscopic appearance typical of a primary colonic tumor. However, the endoscopic appearance can in no case confirm the primary or metastatic nature of the tumor, which is the same for radiological explorations. Histological examination is the only way for identifying metastatic tumors to the GI tract[2]. Hence, the finding of an undifferentiated tumor with unusual morphology in the GI tract and the absence of dysplasia of the surface mucosa may always give rise to the suspicion of a metastatic malignancy, thus leading the pathologists to employ a small panel of specific markers of pulmonary or GI primary tumor (TTF-1, CDX2, Keratin 7, and Keratin 20) to determine the origin tumor site [2].
Several treatment modalities are considered, including surgery, endoscopic resection, and chemo-radiotherapy. Due to the occurrence of perforation or acute complication such as gastrointestinal bleeding, as a revealing symptom of the disease, therapeutic management has often involved an emergency surgery [8]. Endoscopic resection has been reported in several cases with metastatic tumor of size less than 1 cm. Ito, et al., demonstrated that chemo-radiotherapy with four cycles of cisplatin and etoposide, followed by abdominal irradiation at a dose of 30 Gy to patients with small cell lung cancer with GI metastasis have reasonably good partial response. Zhou, et al., highlighted the potential benefit of tyrosine kinase inhibitors [TKI] in primary lung cancer with driver gene mutations in gastrointestinal metastases [4]. In addition, targeted vascular therapies have gained acceptance in recent years. Patients positive for vascular endothelial growth factor may benefit more from treatment with bevacizumab [12]. Nevertheless, prognosis of GIT metastases from primary lung carcinoma is very poor [3]. The survival limit is from some weeks to a couple of months in the great majority of cases [2]. The median survival is 4.75 months. Overall survival during the first year is 20% and it is nil at 2 years [13].
CONCLUSION
Gastrointestinal metastasis from lung carcinoma are rare and the caecal localization is not common. They represent a sign of late-stage disease and are exceptionally revealing the primary tumor. Their prognosis is poor with an average ranging from some weeks to few months between diagnosis and death. For better results new advances in diagnosis and treatment are required.
REFERENCES
- Pararas N, Kirkilessis G, Pikoulis A, Syrigos K, et al. A Rare Case of a Metastatic Lung Squamous Cell Carcinoma to the Large Bowel and the Liver. Cureus. 13(3):e13867. [Link].
- Rossi G, Marchioni A, Romagnani E, Bertolini F, et al. (2007). Primary Lung Cancer Presenting with Gastrointestinal Tract Involvement: Clinicopathologic and Immunohistochemical Features in a Series of 18 Consecutive Cases. J Thorac Oncol. 2(2):115‑120. [Link]
- Nemoto M, Prasoon P, Ichikawa H, Hanyu T, et al. (2020). Primary lung squamous cell carcinoma and its association with gastric metastasis: A case report and literature review. Thoracic Cancer. 11(6):1708-1711. [Link]
- Kosasih S, Muhammad Nawawi KN, Wong Z, Chia Hsin DC, et al. (2019). Upper Gastrointestinal Bleed due to Duodenal Metastases of Lung Adenocarcinoma: Report of Two Cases and Review of Literature. Case Rep Med. 2019:3437056. [Link].
- Antler AS, Ough Y, Pitchumoni CS, Davidian M, et al. (1982). Gastrointestinal metastases from malignant tumors of the lung. Cancer 49(1):170-172. [Link]
- McNeill PM, Wagman LD, and Neifeld JP. (1987). Small bowel metastases from primary carcinoma of the lung. Cancer. 59(8):1486‑1489. [Link]
- Locher C, Grivaux M, Jeandel R, Blanchon, et al. (2008). Métastases intestinales de carcinome broncho-pulmonaire. EM-Consulte. 23(2):273-276. [Link]
- Metges JP, Labat JP, Giroux MA, Simon H, et al. (2001). Métastases gastroduodénales : un mode de révélation exceptionnel du cancer du poumon.Étude de deux cas et revue de la littérature. Rev Médecine Interne. 22(5): 465‑468. [Link]
- Agaimy A, Daum O, Michal M, Schmidt MW, et al. (2021). Undifferentiated large cell/rhabdoid carcinoma presenting in the intestines of patients with concurrent or recent non-small cell lung cancer [NSCLC): clinicopathologic and molecular analysis of 14 cases indicates an unusual pattern of dedifferentiated metastases. Virchows Archiv. 479:157-167. [Link]
- Hsu CC, Chen JJ, and Changchien CS. (1996). Endoscopic features of metastatic tumors in the upper gastrointestinal tract. Endoscopy. 28(2):249‑253. [Link]
- Lee KA, Lee SK, Seo DW, and Kim MH. (2001). Duodenal metastasis from lung cancer presenting as obstructive jaundice. Gastrointest Endosc. 54(2):228. [Link]
- Li X, Li S, Ma Z, Zhao S, et al. (2018). Multiple gastrointestinal metastases of squamous-cell lung cancer: A case report. Medicine [Baltimore). 97(24):e11027. [Link]
- de O Campoli PM, Ejima FH, Cardoso DMM, da Silva OQ, et al. (2006). Metastatic cancer to the stomach. Gastric Cancer. 9(1):19‑25. [Link]