Previous Issues Volume 1, Issue 1 - 2016
Gum Arabic Down-Regulate Cholesterol Biosynthesis Enzyme Gene mRNA Expression in Mice Muscle
Abdelkareem A. Ahmed1, Jaafar S. Fedail2, Hassan H. Musa3, Amal Z. Sifaldin4, Taha H. Musa5
1Department of Physiology and Biochemistry, Faculty of Veterinary Science, University of Nyala, Sudan.
2Department of Biology, Faculty of Education, University of Nyala, Sudan.
3Department of Medical Microbiology, Faculty of Medical Laboratory Sciences, University of Khartoum, Sudan.
4Department of Molecular genetics, Institute of Molecular Biology, University of Nyala, Nyala, Sudan.
5Key Laboratory of Environmental Medicine, Ministry of Education, School of Public Health, Southeast University, Nanjing 210009, Jiangsu, China.
Corresponding Author: Abdelkareem Abdallah Ahmed, Department of Physiology and Biochemistry, Faculty of Veterinary Science University of Nyala Nyala, P.O Box: 155 Nyala, Sudan, Tel:+249711833123; E-Mail: [email protected]
Received Date: 17 Jun 2016 Accepted Date: 05 Dec 2016 Published Date: 08 Dec 2016
Copyright © 2016 Ahmed AA
Citation: Ahmed AA, Fedail JS, Musa HH, Sifaldin AZ, et al. (2016). Gum Arabic Down-Regulate Cholesterol Biosynthesis Enzyme Gene Mrna Expression in Mice Muscle. Mathews J Diabetes Obes. 1(1): 003.
ABSTRACT Gum Arabic (GA) is a complex polysaccharide used in the food manufacturing as a thickener and stabilizer. Our previous studies show that GA decreased body weight, visceral adipose tissue and plasma cholesterol level in mice. In addition, GA down regulated peroxisome proliferator-activated receptor gamma and stearoyl-CoA desaturase mRNA expression in mice liver. In the present study, we aimed to reveal the effect of GA on lipid biosynthesis gene mRNA expression in mice. 20 female CD-1 mice at 5 weeks age were divided into two groups, one served as control and the second received 10% of GA in drinking water for 6 weeks. Plasma glucose, total cholesterol, triglyceride, HDL-c and LDL-c were measured. Cholesterol biosynthesis gene mRNA expression of including down-regulated steroid 17-alpha-monooxygenase (CYP17) and 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (HMGR) were measured by Q-PCR. Moreover, glucose 6-phosphatase and fatty acid synthase mRNA was measured. GA significantly (P < 0.05) reduced plasma glucose total cholesterol, HDL-c and LDL-c but not triglyceride when compared to the control group. GA significantly (P< 0.05) down regulated CYP17 and HMGR mRNA expression in mice muscle when compared to the control group. Yet, GA did not affect G6P and FAS mRNA expression. Our data indicate that GA may be of useful in control of obesity.
KEYWORDS
Gum Arabic; Cholesterol Biosynthesis Gene; Mice.
INTRODUCTION The incidence of obesity is increasing in the population worldwide [1, 2]. Obesity is frequently part of the metabolic disease, a situation which includes dyslipidaemia, decreased high density lipoprotein cholesterol and hypertension [3-5]. Metabolic syndrome increases the risk for development of cardiovascular disorders such as heart stroke, coronary artery disease, and chronic renal disease [6-8]. The complications of obesity are not only determined by absolute amount of fat in the body, nevertheless, it depends on distribution of fat [9]. The distribution of adipose tissues in the body was found to be linked with cardio-metabolic risk in many populations [9]. The most important factors that determine deposition of fat in body include de novo fatty acid synthesis, triacylglycerols synthesis, the rate of fatty acids uptake, lipid degradation and synthesis of cholesterol [10-13]. A number of key enzymes are known to play a major role in lipid metabolism in the target tissues. 17-alpha-monooxygenase (CYP17), 3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (HMGR) and fatty acid synthase (FAS) are the key lipids biosynthesis enzymes that play a critical role in lipid biosynthesis [14-16]. HMGR catalyzes conversion of HMG CoA to mevalonate, a precursor of isoprenoid groups that are incorporated into many end-products including cholesterol [17]. HMGR has been well known as the rate-limiting enzyme in cholesterol biosynthesis. Inhibition of HMGR considers the key factor in controlling of hyperlipidemia which could be of clinical importance [18]. Several medicinal plants have been found to regulate HMGR such as Emblica officinalis, arabinoxylan and Quercus mongolica [19-21]. Gum arabic (GA) is an edible, dried sticky exudate from Acacia seyal and Acacia senegal is found to be rich in non-viscous soluble fiber. It has been commonly used in food manufacturing and pharmaceutical field as an emulsifier and preservative. In the Middle East and North Africa, it has been used as an oral hygiene material by various communities for centuries [22]. In our previous studies, we reported that GA suppressed dietinduced obesity by alteration the expression of mRNA levels of genes involved in lipid metabolism in mouse liver [23]. In both human and animal, the majority of studies have examined the anti-obese effects of GA on body mass index and fat deposition [24, 25]. However, the effects of GA on serum lipid profile and its association cholesterol biosynthesis enzyme gene mRNA expression remained unclear. Therefore, in the present study, we used experimental mice to investigate our hypotheses that serum lipid profile could be changed via administrated of GA in mice, and the changes in serum lipid profile may be associated with alterations of cholesterol biosynthesis enzyme gene mRNA expression in the muscle.
MATERIAL AND METHODS
Experimental animals
Twenty female CD-1 mice at 5 weeks age were obtained from the Experimental Animal Center of Nanjing Medical University (Nanjing, China). The mice were housed under controlled lighting (12 h light, 12 h dark), temperature (21 8 C - 22 8 C) and humidity at 65% - 70%. The mice were allowed free access to a commercial pellet diet and drinking water throughout the experiment period. The experimental protocol involving mice was approved in accordance with the guide for the care and use of laboratory animals prepared by the Institutional Animal Care and Use Committee of Nanjing Agricultural University.
Experimental design
After an acclimatization period of a week, mice were randomly divided into two equal groups. The first group continued to receive the same diet without treatment until the end of the study (control group). The second group was given normal food and received 0.5% of gum arabic aqueous solution as drinking water for 7 days, and then 10% solution for further 6 weeks. During the treatment period, the mice were weighed weekly. After 6 weeks, the mice were killed. Serum samples and muscle tissue were collected and stored at -80 ?C.
Plasma lipid profile
Blood glucose, plasma triglycerides, total cholesterol, LDL, VLDL, and HDL were determined using commercially kits (At Nanjing Military Hospital., Nanjing, China).
RNA extraction and Real-time PCR
About 100 mg of muscle was ground in liquid N2 , and a portion of about 50 mg was used for RNA extraction using TRIzol total RNA kit (Invitrogen, Biotechnology Co, Ltd, Carlsbad, CA, USA) according to the manufacturer’s instruction. Two approaches were taken to ensure that all the total RNA preparations are free of genomic DNA contamination. Firstly, total RNAs were treated with 10 U DNase I (Rnase Free, D2215, Takara, Japan) for 30 min at 37°C, and purified according to the manufacturer’s protocol. Secondly, the primers for the reference gene were designed to span an intron, so any genomic DNA contamination can be reported easily with an extra product in the melting curves for real-time PCR. Real-time PCR was performed in Mx3000P (Stratagene, USA) according to the previous publication [23]. Mock RT and No Template Controls were included to monitor the possible contamination of genomic and environmental DNA at both RT and PCR steps. The pooled sample made by mixing equal quantity of RT products (cDNA) from all samples was used for optimizing the PCR condition and tailoring the standard curves for each target gene, and melting curves were performed to insure a single specific PCR product for each gene. The PCR products were sequenced to validate the identity of the amplicons. Primers specific CYP17, HMGR, G6P and FAS (Table 1) were synthesized by Geneary (Shanghai, China). Mice GAPDH were used as a reference gene for normalization purposes. The method of 2−ΔΔCt was used to analyze the real-time PCR data [26]. The mRNA abundances were presented as the fold change relative to the average level of the control group.
| Group | Glucose | Triglyceride | Total cholesterol | HDL | LDL |
| (mmol/L) | (mg/dL) | (mg/dL) | (mg/dL) | (mg/dL) | |
| Control | 8.01±..51a | 49.1±.3.08a | 82.7±.3.4a | 55.82±.2.32a | 75.36±.5.51a |
| Gum | 4.06±.0.70b | 45.4±.2.4a | 63.7±.3.15b | 67.56±.3.14b | 45.61±.3.31b |
Statistical Analysis
Data are expressed as the mean ± SEM and compared by one way analysis of variance and Student’s T test. P < 0.05 as considered significant [27]. All statistical analyses were performed using SPSS 16.0 software (SPSS, Chicago, IL, USA)
RESULTS
Plasma lipid profile and blood glucose
GA treatment significantly (P < 0.05) decreased plasma glucose total cholesterol, HDL-c and LDL-c compared to the control group (Table 2). No changes were observed in triglycerides concentration regarding GA supplementation.
Table 2: Real-Time PCR Primers.
| Target genes | Gene bank number | Product Size | Primer |
| FASN | NM_007988.3 | 129 | F: 5’- GATATTGTCGCTCTGAGGCTGTTG -3’ |
| R: 5’- GGAATGTTACACCTTGCTCCTTGC -3’ | |||
| HMGR | NM_008255.2 | 83 | F: 5’- TGACCTTTCTAGAGCGAGTGCAT -3’ |
| R: 5’- CACGAGCTATATTTTCCCTTACTTCA -3" | |||
| CYP17 | NM_001291508.1 | 2 | F: 5’- CAACTCAGCGGGTGGATACC- 3" |
| R: 5’- GGACCGGGCGTCTATAACAG- 3" | |||
| GAPDH | NM_008084.2 | 141 | F: 5’- ACATGGTCTACATGTTCCAGTA -3’ R: 5’- GGAGTCTACTGGTGTCTTCA- 3’ |
Liver and Muscle 11β-HSD 1 mRNA expression
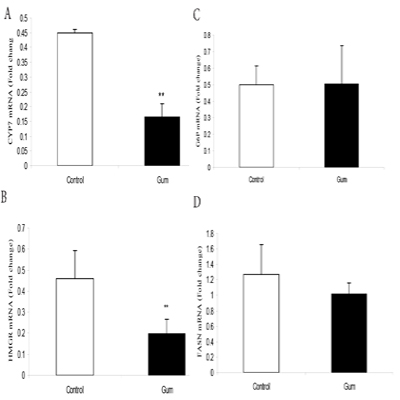
The supplementation of GA significantly (P< 0.05) down regulated CYP17 (Figure. A) and HMGR (Figure. B) mRNA expression in mice muscle compared to the control group. However, supplementation of GA did not change G6P (Figure. C) and FASN (Figure. D) mRNA expression in mice muscle.
DISCUSSION
Obesity is a risk factor for several metabolic diseases such as diabetes, coronary heart disease, heart stroke, nonalcoholic fatty liver disease and many other disorders including cancer [28-30]. In the present study, gum arabic (GA) supplementation significantly reduced blood glucose. Our findings are in line with pervious studies that dietary fibre reduced blood glucose either in normal or fed high fat diet mice [23, 31]. Recent studies reported that GA treatment decreased plasma total cholesterol, triglyceride and low density lipoprotein (LDL) concentrations in human and mice [31, 32]. In agreement with these findings, we reported that treatment of GA decreased plasma total cholesterol, HDL-c and LDL-c. Hyperlipidemia is well known as risk factor for the development of atherosclerosis [33]. Hyperlipidemia can significantly increase the risk of developing cardiovascular disease, including coronary artery disease, diabetes mellitus, hypertension, cerebrovascular disease, and limbs peripheral vascular disease. These conditions can in turn cause heart strokes, heart attacks, and metabolic syndrome [34-40]. The reduction of plasma lipid profile by GA may contribute in the decreasing the metabolic syndrome. Therefore, GA may be promising to be used as Antiobesity choice but requires further studies. Several mechanisms have been projected to disclose the hypocholesterolemic effects of dietary fibre [41]. One of the potential justification is that dietary fibre increases the intestinal contents viscosity, therefore interfering with micelle formation and absorption of nutrient, which, in turn, decreases intestinal lipid absorption [20]. An additional mechanisms proposed that soluble dietary fibers decreases the enterohepatic circulation of bile acids, resultant in increases of bile acid excretion and subsequently decreases plasma cholesterol concentrations [42, 43]. Moreover, the viscosity of fermentable dietary fibers is reported to contribute in lipid lowering in rat [44]. Obesity is frequently associated with type 2 diabetes, hypertension and hypercholesterolemia, which consequently result in coronary heart disease [45]. Based on World Health Organization (WHO) report, risks for type 2 diabetes and hypercholesterolemia are greatly increased in obese patients. In the present study, the reduction of plasma total cholesterol in GA supplemented mice associated with alterations in the expression of cholesterol biosynthesis enzyme gene. Supplementation of GA significantly down-regulated CYP17 and HMGR mRNA expression in mice muscle. Our results are agreed with previous report that supplementation dietary fiber reduced liver HMGR mRNA expression in mice [46, 47]. Inhibition of the key enzyme of cholesterol biosynthesis may prevent hypercholesterolemia in obesity patient and thus it may reduce the obesity complications such as diabetes, coronary artery disease, heart stroke and hypertension [48-50]. Moreover, total ablation of androgen production by potent CYP17 inhibitors may offer effective treatment of prostate cancer patients [51, 52]. However, the supplementation of GA did not change G6P and FASN mRNA levels. Our results are mismatch with previous finding that the consumption of dietary fibre reduced FASN mRNA expression in obese human [53]. The inconsistent of these results may be due to the species differences or experimental design. FAS mRNA levels were unchanged, whereas SREBP-1c increased. The obesity subjects have found to an increased hepatic lipogenesis that may contribute to their excessive fat mass but no evidence for an increased lipogenic capacity of adipose tissue. Therefore, GA may contain principle substance that could contribute in reducing obesity and its complications.
CONCLUSION
The data concludes that gum arabic can reduced blood glucose, plasma total cholesterol associated with down-regulation of cholesterol biosynthesis gene in mice muscle, thus GA is may be of useful in the control of hyperlipidemia in obese patient.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interests.
ACKNOWLEDGEMENTS
The authors are highly grateful to Prof. Shi Fangxiong and Prof. Ruqian Zhao at the Nanjing Agricultural University, China for their support in conducting this research.
REFERENCES
- Nguyen DM and El-Serag HB. (2010). The epidemiology of obesity. Gastroenterol Clin North Am. 39(1), 1-7.
- Karnieli E. (2008). The growing prevalence of obesity worldwide is an increasing concern. Preface. Endocrinology and metabolism clinics of North America. 37(3), xvii-xviii.
- Wahba IM and Mak RH. (2007). Obesity and obesity-initiated metabolic syndrome: mechanistic links to chronic kidney disease. Clin J Am Soc Nephrol. 2(3), 550-562.
- Wang H and Peng D-Q. (2011). New insights into the mechanism of low high-density lipoprotein cholesterol in obesity. Lipids in Health and Disease. 10, 1-10.
- Linhart C, Tukana I, Lin S, Taylor R, et al. (2016). Continued increases in hypertension over three decades in Fiji, and the influence of obesity. Journal of hypertension. 34, 402-409.
- Lu Y, Hajifathalian K, Ezzati M, Woodward M, et al. (2014). Metabolic mediators of the effects of body-mass index, overweight, and obesity on coronary heart disease and stroke: a pooled analysis of 97 prospective cohorts with 1.8 million participants. Lancet. 383, 970-983.
- Talavera-Garcia E, Delgado-Lista J, Garcia-Rios A, Delgado- Casado N, et al. (2016). Correction: Influence of Obesity and Metabolic Disease on Carotid Atherosclerosis in Patients with Coronary Artery Disease (CordioPrev Study). PloS one. 11(6), e0157213.
- Navarro G and Ardiles L. (2015). Association between obesity and chronic renal disease. Revista medica de Chile. 143(1), 77-84.
- Wang T, Ma X, Peng D, Zhang R, et al. (2016). Effects of Obesity Related Genetic Variations on Visceral and Subcutaneous Fat Distribution in a Chinese Population. Scientific reports. 6,20691.
- Ge F, Hu C, Hyodo E, Arai K, et al. (2012). Cardiomyocyte triglyceride accumulation and reduced ventricular function in mice with obesity reflect increased long chain Fatty Acid uptake and de novo Fatty Acid synthesis. Journal of obesity 2012,2012(2012)205648.
- Zhang Y, Liu Y, Wang J, Zhang R, et al. (2010). Medium- and long-chain triacylglycerols reduce body fat and blood triacylglycerols in hypertriacylglycerolemic, overweight but not obese,Chinese individuals. Lipids. 45(6),501-510.
- Khalifeh-Soltani A, McKleroy W, Sakuma S, Cheung YY, et al.(2014). Mfge8 promotes obesity by mediating the uptake of dietary fats and serum fatty acids. Nature medicine. 20(2):175-183.
- Balogh Z, Foris G, Konya G, Paragh G, et al. (2011).Obesity abrogates the concentration-dependent effect of leptin on endogenous cholesterol synthesis in human monocytes. Immunobiology. 216(3), 431-435.
- Liu Y, Yao ZX, and Papadopoulos V. (2005). Cytochrome P450 17a Hydroxylase/17,20 Lyase (CYP17) Function in Cholesterol Biosynthesis: Identification of Squalene Monooxygenase (Epoxidase) Activity Associated with CYP17 in Leydig Cells. Molecular endocrinology. 19, 1918-1931.
- Faulkner RA, Nguyen AD, Jo Y and DeBose-Boyd RA. (2013). Lipid-regulated degradation of HMG-CoA reductase and Insig- 1 through distinct mechanisms in insect cells. Journal of Lipid Research. 54(4), 1011-1022.
- Zhu JJ, Luo J, Sun YT, Shi HB, et al. (2015). Short communication: Effect of inhibition of fatty acid synthase on triglyceride accumulation and effect on lipid metabolism genes in goat mammary epithelial cells. Journal of dairy science. 98(5), 3485-3491.
- DeBose-Boyd RA. (2008). Feedback regulation of cholesterol synthesis: sterol-accelerated ubiquitination and degradation of HMG CoA reductase. Cell research. 18(6), 609-621.
- Bland CM, Bookstaver PB, Lu ZK, Dunn BL, et al. (2014). Musculoskeletal safety outcomes of patients receiving daptomycin with HMG-CoA reductase inhibitors. Antimicrobial agents and chemotherapy. 58(10), 5726-5731.
- Gopa B, Bhatt J and Hemavathi KG. (2012). A comparative clinical study of hypolipidemic efficacy of Amla (Emblica officinalis) with 3-hydroxy-3-methylglutaryl-coenzyme-A reductase inhibitor simvastatin. Indian journal of pharmacology. 44(2), 238-242.
- Lattimer JM and Haub MD. (2010). Effects of dietary fiber and its components on metabolic health. Nutrients. 2(12), 1266-1289.
- Chai YM, Lim BK, Lee JY, Kim MN, et al. (2003). Effects of manufactured soluble dietary fiber from Quercus mongolica on hepatic HMG-CoA reductase and lipoprotein lipase activities in epididymal adipose tissue of rats fed high cholesterol diets. Journal of medicinal food. 6(4), 329-336.
- Tyler VB, Robbers L and Pharmacognosy J. (1977). Philadelphia: Lea & Febiger. 64-68.
- Ahmed AA, Musa HH, Fedail JS, Sifaldin AZ, et al. (2016). Gum arabic suppressed diet-induced obesity by alteration the expression of mRNA levels of genes involved in lipid metabolism in mouse liver. Bioactive Carbohydrates and Dietary Fibre. 7(1), 15-20.
- Babiker R, Merghani TH, Elmusharaf K, Badi RM, et al. (2012). Effects of Gum Arabic ingestion on body mass index and body fat percentage in healthy adult females: two-arm randomized, placebo controlled, double-blind trial. Nutrition journal. 11, 111.
- Ushida K, Hatanaka H, Inoue R, Tsukahara T, et al. (2011). Effect of long term ingestion of gum arabic on the adipose tissues of female mice. Food Hydrocolloids. 25(5), 1344-1349.
- Livak KJ and Schmittgen TD. (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 25(4), 402-408.
- Petaja EM, Sevastianova K, Hakkarainen A, Orho-Melander M, et al. (2013). Adipocyte size is associated with NAFLD independent of obesity, fat distribution, and PNPLA3 genotype. Obesity (Silver Spring). 21(6), 1174-1179.
- Schutter AD, Lavie CJ and Milani RV. (2014). The impact of obesity on risk factors and prevalence and prognosis of coronary heart disease-the obesity paradox. Progress in cardiovascular diseases. 56(4), 401-408.
- Jung UJ and Choi MS. (2014). Obesity and its metabolic complications: the role of adipokines and the relationship between obesity, inflammation, insulin resistance, dyslipidemia and nonalcoholic fatty liver disease. International journal of molecular sciences. 15(4), 6184-6223.
- Pergola GD and Silvestris F. (2013). Obesity as a Major Risk Factor for Cancer. Journal of obesity 2013. 2013, 291546.
- Ahmed AA, Musa HH, Fedail JS, Sifaldin AZ, et al. (2015). Gum arabic decreased visceral adipose tissue associated with downregulation of 11β-hydroxysteroid dehydrogenase type I in liver and muscle of mice. Bioactive Carbohydrates and Dietary Fibre. 6(1), 31-36.
- Mohamed RE, Gadour MO and Adam I. (2015). The lowering effect of Gum Arabic on hyperlipidemia in Sudanese patients. Frontiers in physiology. 6, 160.
- Nimkuntod P and Tongdee P. (2015). Association between Subclinical Atherosclerosis among Hyperlipidemia and Healthy Subjects. Journal of the Medical Association of Thailand. 98(Suppl 4), S51-S57.
- Munshi RP, Joshi SG and Rane BN. (2014). Development of an experimental diet model in rats to study hyperlipidemia and insulin resistance, markers for coronary heart disease. Indian journal of pharmacology. 46(3), 270-276.
- Navar-Boggan AM, Peterson ED, D'Agostino RB, Neely B, et al. (2015). Hyperlipidemia in early adulthood increases longterm risk of coronary heart disease. Circulation. 131(5), 451- 458.
- Bai D, Zhang Y, Shen M, and Sun Y, et al. (2016). Hyperglycemia and hyperlipidemia blunts the Insulin-Inpp5f negative feedback loop in the diabetic heart. Scientific reports. 6, 22068.
- Sugai T, Suzuki Y, Yamazaki M, and Shimoda K, et al. (2016). High Prevalence of Obesity, Hypertension, Hyperlipidemia, and Diabetes Mellitus in Japanese Outpatients with Schizophrenia: A Nationwide Survey. PloS one. 11(11), e0166429.
- Kimura Y, and Uchiyama S. (2001). [Hyperlipidemia and cerebrovascular disease]. Nihon Rinsho. 59 Suppl 2, 685-689.
- Nelson RH. (2013). Hyperlipidemia as a Risk Factor for Cardiovascular Disease. Primary care. 40(1), 195-211.
- Bragg DA, and Walling A. (2015). Metabolic Syndrome: Hyperlipidemia. FP essentials. 435, 17-23.
- Dvir I, Stark AH, Chayoth R, and Madar Z, et al. (2009). Hypocholesterolemic effects of nutraceuticals produced from the red microalga Porphyridium sp in rats. Nutrients. 1(2), 156-167.
- Parnell JA, and Reimer RA. (2010). Effect of prebiotic fibre supplementation on hepatic gene expression and serum lipids: a dose-response study in JCR:LA-cp rats. Br J Nutr. 103(11), 1577-1584.
- Babio N, Balanza R, Basulto J, and Bullo M, et al. (2010). Dietary fibre: influence on body weight, glycemic control and plasma cholesterol profile. Nutr Hosp. 25(3), 327-340.
- Brockman DA, Chen X, and Gallaher DD. (2014). Highviscosity dietary fibers reduce adiposity and decrease hepatic steatosis in rats fed a high-fat diet. J Nutr. 144(9), 1415-1422.
- Aguilar D, and Fernandez ML. (2014). Hypercholesterolemia induces adipose dysfunction in conditions of obesity and nonobesity. Advances in nutrition. 5, 497-502.
- Rideout TC, Harding SV, Jones PJ, and Fan MZ, et al. (2008). Guar gum and similar soluble fibers in the regulation of cholesterol metabolism: current understandings and future research priorities. Vascular health and risk management. 4(5), 1023-1033.
- Kishida T, Nogami H, Ogawa H, and Ebihara K, et al. (2002). The hypocholesterolemic effect of high amylose cornstarch in rats is mediated by an enlarged bile acid pool and increased fecal bile acid excretion, not by cecal fermented products. The Journal of nutrition. 132(9), 2519-2524.
- Asif M. (2014). The prevention and control the type-2 diabetes by changing lifestyle and dietary pattern. Journal of education and health promotion. 3, 1.
- Cole JA, Smith SM, Hart N, and Cupples ME, et al. (2011). Systematic review of the effect of diet and exercise lifestyle interventions in the secondary prevention of coronary heart disease. Cardiology research and practice. 232351.
- Burke V, Hodgson JM, Beilin LJ, and Giangiulioi N, et al. (2001). Dietary protein and soluble fiber reduce ambulatory blood pressure in treated hypertensives. Hypertension. 38(4), 821-826.
- Gianti E and Zauhar RJ. (2012). Modeling androgen receptor flexibility: a binding mode hypothesis of CYP17 inhibitors/ antiandrogens for prostate cancer therapy. Journal of chemical information and modeling. 52(10), 2670-2683.
- Vasaitis TS, Bruno RD, and Njar VC. (2011). CYP17 inhibitors for prostate cancer therapy. The Journal of steroid biochemistry and molecular biology. 125(1-2), 23-31.
- Hudgins LC, Baday A, Hellerstein MK, and Parker TS, et al. (2008). The effect of dietary carbohydrate on genes for fatty acid synthase and inflammatory cytokines in adipose tissues from lean and obese subjects. The Journal of nutritional biochemistry. 19(4), 237-245.