Previous Issues Volume 2, Issue 1 - 2018
Trifecta Aortic Valve Replacement in Elderly and Polymorbid Patients: Clinical and Echocardiographic Follow-Up
Francesco Venditti1 , Pierluigi Stefàno2 , Ramona Santangeli2 , Claudio Blanzola2 , Maria Riccarda Del Bene1 , Sabina Caciolli2 , Stefano Del Pace2 , Giuseppe Barletta1
1Cardiology Diagnostics, Cardio-Thoracic and Vascular Department, Careggi University Hospital, Florence, Italy. 2Cardiac Surgery, Cardio-Thoracic and Vascular Department, Careggi University Hospital, Florence, Italy. Corresponding Author: Giuseppe Barletta, Cardiologia Diagnostica – AOU Careggi, Largo Brambilla, 3 - 50124 Florence, Italy, Tel: +39 55 410895; Email: [email protected] Received Date: 03 Oct 2018 Accepted Date: 09 Oct 2018 Published Date: 12 Oct 2018 Copyright © 2018 Barletta G Citation: Venditti F, Stefàno P, Santangeli R, Blanzola C, et al. (2018). Trifecta Aortic Valve Replacement in Elderly and Polymorbid Patients: Clinical and Echocardiographic Follow-Up. M J Surg. 2(1): 013.
ABSTRACT Objectives: We sought to evaluate the hemodynamic performance and clinical outcome of the St. Jude Medical Trifecta aortic bioprosthesis in a large single-center series of consecutive polymorbid, moderate-to-high surgical risk elderly patients. Methods: Between January 2014 and January 2015, 250 consecutive patients undergoing aortic valve replacement at Careggi University Hospital, Florence, received a Trifecta aortic bioprosthesis. 202 patients (46.4% females, mean age 79.9±4 years) were enrolled in the present follow up study. Clinical and echocardiographic data were collected preoperatively and at one year after surgery. Results: the most prevalent indication for aortic valve replacement was stenosis (51.5%), followed by mixed pathology (29.7%), regurgitation (14.4%), prosthetic valve degeneration (3.5%) and endocarditis (1%). Preoperative functional class was NYHA III in 67.8% and NYHA IV in 12.4 %; logistic EuroSCORE was moderately high (13.5±6.4). Most patients (73%) underwent concomitant surgical procedures. Global mortality was 9.6% (24 patients), early mortality being 1.6% (4 patients). Adverse events included five thromboembolic events and four prosthetic valve endocarditis; early prosthetic valve degeneration occurred in one. At one-year follow-up, mean gradients ranged from 6.0±2.7 and 12.1±8.6 for the 27 and 19 mm valve, respectively. Indexed effective prosthesis area ranged from 0.73 to 1.16 cm2/m2 for the 19 and 25 mm valve, respectively. Conclusion: St. Jude Medical Trifecta aortic bioprosthesis confirmed excellent hemodynamic profile and achieved good clinical results when offered to elderly patients with high incidence of co-morbidities and moderately high preoperative risk, who underwent concomitant surgical procedures in high percentage of cases.
KEYWORDS Aortic Valve Replacement; Aortic Bioprosthesis; Doppler Echocardiography; Octuagenarians; Polymorbidity
INTRODUCTION The Trifecta aortic valve (St. Jude Medical, Inc., St. Paul, MN, USA) is a third-generation three-leaflet stented bioprosthesis consisting of bovine pericardium tissue leaflets mounted on a titanium stent covered with swine pericardium, and is specifically designed for the supra-annular aortic position [1]. Since its arrival on the market in April 2011, several studies on the hemodynamic performance and durability of this device have yielded promising results, showing overall optimal trans-prosthetic gradients and low percentages of severe patient-prosthesis mismatch, even in subjects with a small aortic annulus in relation to body size [2]. Other studies have demonstrated that its near-physiologic, hemodynamic profile is superior to that of some of its principal competitors [3-8]. However, the studies conducted so far have often been relatively small and have had a limited period of follow-up; moreover, the populations analysed have been quite heterogeneous in terms of age and indication for aortic valve replacement. The present study sought to analyse the hemodynamic performance of the St. Jude Trifecta aortic bioprosthesis in a population of polymorbid, moderate- to high-risk, octogenarian patients, mainly suffering from severe aortic valve stenosis.
MATERIALS AND METHODS Patient selection Between January 2014 and January 2015, 250 consecutive patients underwent aortic valve replacement with the St. Jude Trifecta aortic bioprosthesis at Careggi University Hospital, Florence. Patients submitted to multiple combined surgical procedures were not excluded from the study. Written informed consent was obtained from all patients and institutional review board approval was granted by the local ethics committee. Surgical technique All patients underwent midline sternotomy and cardiopulmonary bypass; myocardial protection was implemented through moderate hypothermia and cardioplegic arrest. After aortic annulus decalcification, the appropriate prosthesis size was determined by the surgeon with the specific manufacturersupplied replica sizer. Prostheses were implanted by means of three linear sutures. Immediately after weaning from cardiopulmonary bypass, correct bioprosthetic positioning and function were checked by trans-oesophageal echocardiography. None of the patients required an aortic root enlargement procedure. Patient follow-up Upon hospital discharge, each patient was scheduled for a one-year follow-up visit, consisting of clinical examination, ECG and a thorough echocardiographic exam. Of the 226 surviving patients, a total of 202 attended the follow-up visit and were enrolled in the present study. Deceased patients’ medical records were obtained from the patients’ relatives or from their general practitioners. Standardized definitions from the Society of Thoracic Surgeons/American Association for Thoracic Surgery, specified in the ‘‘Guidelines for Reporting Morbidity and Mortality and Cardiac Valvular Operations’’ [9], were used to classify adverse events. Early events were defined as those occurring within 30 days of surgery, mediumterm events were defined as those occurring between 30 days and 6 months after surgery, while late events were classified as those occurring between 6 months and one year after prosthesis implantation. Echocardiographic examination Preoperative and one-year postoperative data from bi-dimensional and Doppler transthoracic echocardiographic examinations (EPIQ, X5-1 transducer, Philips Healthcare, Andover, Massachusetts) of all the patients enrolled in the study were acquired in a single core laboratory and stored in an ad hoc database. Standard prosthetic valve measurements were collected according to the American Society of Echocardiography (ASE) guidelines [10]. Each measure was repeated three times and averaged. Peak and mean systolic gradients were obtained automatically through the modified Bernoulli equation, by integrating the continuous-wave Doppler spectral envelope of flow, sampled where the transducer position could yield maximum velocity across the aortic valve or bioprosthesis. Effective orifice area (EOA) was calculated as (CSALVOT x TVILVOT)/TVIAO, where CSALVOT represents the cross-sectional area of the left ventricular outflow tract (LVOT), while TVILVOT and TVIAO represent the time-velocity integrals (TVI) derived from the pulsed-wave Doppler of LVOT and the continuouswave Doppler of the aortic valve/prosthesis, respectively. Finally, mild-to-moderate patient-prosthesis mismatch (PPM) was defined as any value of EOAi (EOA indexed by patient’s body surface area) between 0.85 cm2 /m2 and 0.65 cm2 /m2 , while severe PPM was defined as any EOAi value below 0.65 cm2 /m2 [11-13]. Left ventricular mass (LVM) was indexed by body surface area (BSA), calculated by means of the Dubois and Dubois formula [14]. Statistical analysis of data Statistical analysis was carried out by means of SPSS, release 22.0 for Windows (IBM SPSS, Inc., Chicago, IL, USA). All data are presented as mean ± standard deviation or as percentages (continuous and categorical variables, respectively). The Kolmogorov–Smirnoff test was used to verify the normality of data distribution. Student’s paired t-test was used to compare continuous variables. Dichotomous variables were compared by means of Fisher’s exact test or χ2 , as appropriate. Gender differences were analysed by using two-way analysis of variance. P < 0.05 was considered statistically significant. RESULTS Patients’ and operative data The mean age of the 202 enrolled patients at the moment of implantation was 79.9 ± 4 years (range 65-89 years); 188 (93%) patients were 75 years old or older, and 116 (46.4%) were females. Preoperatively, the majority of patients were in NYHA functional class III or IV; no patient was in NYHA I class. Preoperative risk, as assessed by means of the logistic EuroSCORE, was moderately high (13.5 ± 6.4). Indications for aortic valve replacement were: aortic valve stenosis in 104 (51.5%) patients, aortic valve regurgitation in 29 (14.4%), and mixed valve pathology in 60 (29.7%). Seven (3.5%) patients underwent aortic valve replacement because of previous prosthetic aortic valve degeneration, and 2 (1%) because of aortic prosthetic valve endocarditis. A total of 12 (5.9%) patients had previously undergone aortic valve replacement. Degenerative calcific stenosis of a three-leaflet valve was present in 164 (81.1%) patients, while a rheumatic aetiology was reported in only 2 (1%) cases. Aortic bicuspidy was observed in 2 (1%) patients. Preoperative baseline characteristics are summarized in Table 1.
|
Variables |
All implants (N = 202) |
|
Age, years |
79.98 ± 4.07 |
|
Female sex |
116 (46.4%) |
|
Weight, kg |
70.82 ± 12.61 |
|
Height, cm |
165.2 ± 8.59 |
|
BSA (Dubois), m2 |
1.77 ± 0.18 |
|
BMI, kg/m2 |
25.86 ± 3.86 |
|
eGFR (Cocroft-Gault), mL/min |
59.87 ± 20.37 |
|
eGFR (MDRD), mL/min |
73.15 ± 14.47 |
|
eGFR (CKD-EPI), mL/min |
64.81 ± 12.82 |
|
Serum creatinine, mg/dL |
0.99 ± 0.39 |
|
Clinical History |
|
|
Known CAD |
42 (20.79%) |
|
Hypertension |
154 (76.2%) |
|
Dyslipidaemia |
72 (35.6%) |
|
Diabetes |
40 (19.8%) |
|
Smoking |
71 (35%) |
|
Family history of CAD |
33 (16.3%) |
|
Mean left ventricular ejection fraction |
57.4 ± 10.6 |
|
Low left ventricular ejection fraction (<45%) |
21 (10.4%) |
|
Aortic bicuspidy |
2 (0.9%) |
|
Carotid artery disease |
28 (13.8%) |
|
Aortic valve replacement indication |
|
|
Aortic stenosis (pure) |
104 (51.5 %) |
|
Aortic mixed valve pathology (prevalent stenosis) |
60 (29.7%) |
|
Aortic regurgitation (prevalent regurgitation) |
29 (14.4%) |
|
Previous prosthesis degeneration |
7 (3.5%) |
|
Prosthesis endocarditis |
2 (1% ) |
|
Predominant heart rhythm |
|
|
Sinus rhythm |
162 (80.2%) |
|
Atrial fibrillation |
27 (13.3%) |
|
Pacemaker |
13 (6.4%) |
|
Preoperative NYHA class |
|
|
|
0 |
|
|
40 (19.8%) |
|
|
137 (67.8%) |
|
|
25 (12.4%) |
|
Logistic EuroSCORE |
13.53 ± 6.40 |
Values are expressed as mean ± standard deviation or numbers (%).
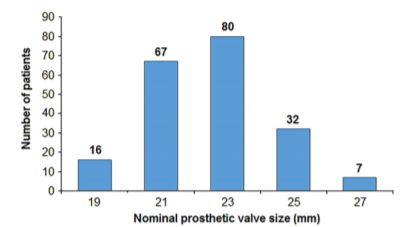
Table 2 shows a list of surgical procedures performed concurrently with aortic valve replacement with the Trifecta bioprosthetic aortic valve in 146 patients (72.3%). Implanted prostheses ranged in size from 19 mm to 27 mm; no patient received a prosthesis with a nominal size of 29 mm. Figure 1 shows the size distribution of the Trifecta aortic valves implanted, on follow-up examination.
Table 2: Surgical procedures concomitant with aortic valve replacement with Trifecta bioprosthetic valve and adverse events.
|
Procedures |
All implants (N = 202) |
|
CABG |
92 (45%) |
|
Mitral valvuloplasty |
21 (10.4%) |
|
Mitral valve replacement |
24 (11.9%) |
|
Tricuspid valvuloplasty |
25 (12.4%) |
|
MAZE |
22 (10.9%) |
|
Morrow’s Myectomy |
5 (2.5%) |
|
Ascending aorta repair/replacement |
16 (7.9%) |
|
Adverse events |
|
|
Endocarditis |
4 (1.98%) |
|
Prosthetic valve detachment (peri-valvular abscess) |
2 (0.99%) |
|
Subacute endocarditis |
2 (0.99%) |
|
Irreversible advanced or complete atrio-ventricular heart block |
15 (7.42%) |
|
Thromboembolic events |
5 (2.47%) |
|
TIA |
4 (1.98%) |
|
Ischaemic stroke |
1 (0.49%) |
|
Haemorrhagic stroke |
0 |
|
Prosthetic valve regurgitation/peri-valvular leaks |
|
|
Absent |
193 (95.5%) |
|
Mild degree |
8 (3.9%) |
|
Moderate degree |
1 (0.5%) |
|
Severe degree |
0 |
|
Re-hospitalization for congestive heart failure |
6 (2.97%) |
|
Mortality |
|
|
Overall mortality |
24 (9.6 %) |
|
Early (within 30 days of implantation) |
4 (1.6 %) |
|
Medium-term (between 30 days and 6 months after AVR) |
14 (6.9 %) |
|
Late (between 6 and 12 months after AVR) |
6 (2.9 %) |
|
Cause of death |
|
|
Acute heart failure |
10 (4.9%) |
|
Ischaemic stroke |
4 (1.6%) |
|
Septic shock |
1 (0.5%) |
|
Neoplasia |
1 (0.5%) |
|
Sudden death |
8 (3.2%) |
Values are expressed as mean ± standard deviation or numbers (%).
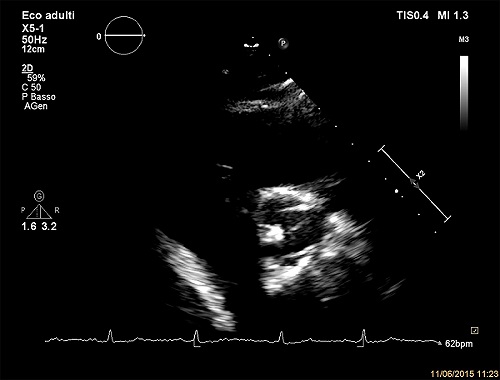
CLINICAL EVENTS A total of 24 (9.6%) patients died. Early mortality occurred in 4 (1.6%) patients. In the immediate postoperative period, 5 (2.47%) patients suffered thromboembolic events: 4 (1%) transient ischemic attacks (TIA) and 1 (0.49%) ischemic stroke; none of these patients had a previous known history of atrial fibrillation. Two (1%) patients underwent postoperative thoracic revision for sternal wound dehiscence. No major bleeding occurred in the postoperative period or during follow-up. There were 4 (1.9%) cases of prosthetic valve endocarditis. Two (1%) patients suffered early prosthetic valve detachment and root abscesses, and needed immediate re-operation. Another 2 (1%) adverse events were incidentally detected during the scheduled one-year follow-up echocardiographic examination; these took the form of subacute endocarditis with sessile vegetation protruding into the prosthesis orifice: after careful re-evaluation by means of trans-esophageal echocardiography, an indication for re-operation was established in both cases. Freedom from prosthetic valve endocarditis was 98% at one year. There were no cases of prosthetic valve thrombosis or significant hemolysis. A de novo, irreversible, advanced or complete atrio-ventricular heart block needing permanent ventricular pacing occurred in 15 (6%) patients. A total of 6 (2.9%) patients underwent re-hospitalization for congestive heart failure during the follow-up period (Table 2).
Clinical and functional evaluation At the one-year follow-up examination, all patients displayed good adherence to optimal medical therapy (Table 3). Arterial blood pressure control was generally satisfactory (143±20/71±10 mmHg). Fifty-four (26.7%) patients were in NYHA functional class I, 117 (57.9%) were in NYHA II and 31 (15.3%) in NYHA III. No patient was in NYHA functional class IV. As already stated, only 6 (2.9%) patients in NYHA III underwent re-hospitalization for an episode of decompensation. The distributions of patients’ pre-operative and follow-up NYHA functional classes are shown in Figure 2.
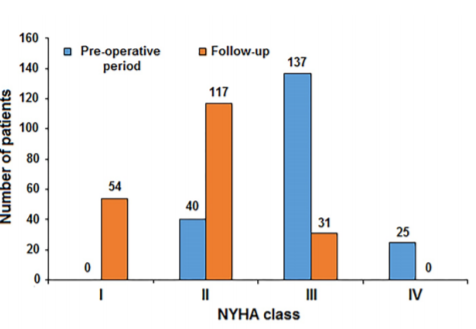
Table 3. Patients’ therapy.
|
Medications |
All implants (N = 202) |
|
Beta-blockers |
141 (69.8%) |
|
ACE-inhibitors |
118 (58.48%) |
|
Sartans |
7 (3.5%) |
|
Diuretics |
183 (90.6%) |
|
Ca-agonists |
12 (5.9%) |
|
Digoxin |
19 (9.4%) |
|
ASA |
143 (70.8% |
|
Statins |
122 (60.4%) |
|
Amiodarone |
66 (32.7%) |
Values are expressed as mean ± standard deviation or numbers (%).
HAEMODYNAMIC RESULTS Haemodynamic results on one-year follow-up examination are reported in Table 4 and in Figure 3. Peak gradients ranged from 8.14 mmHg for the 27 mm valve to 16 mmHg for the 19 mm valve, while mean gradients ranged from 6 mmHg for the 27 mm valve to 12.06 mmHg for the 19 mm valve. Average EOA ranged from 1.18 cm2 for the 19 mm valve to 2.16 cm2 for the 27 mm valve, while EOAi ranged from 0.73 cm2 /m2 for the 19 mm size to 1.16 cm2 /m2 for 25 mm size. Mild-to-moderate PPM was present in 59 (29.2%) patients, while severe PPM occurred in 21 (10.4%) patients. In the early post-operative period and on hospital discharge, no case of moderate or severe prosthetic valve regurgitation/peri-valvular leak was observed. On one-year follow-up examination, 95.5% of patients had no valvular regurgitation or peri-valvular leaks; only 1 (0.5%) case of moderate valve regurgitation was recorded; in this case, early signs of prosthetic aortic valve degeneration were detected (thickening of one of the cusps) (Table 2, Figure 4). On one-year follow-up examination, average LVEF was 58.3% ± 9.1%. A low LVEF< 45% was present in 16 (7.9%) patients. Left ventricular diastolic dysfunction, defined according to the latest definition by the ASE [15], was observed in 57.6% of patients and did not differ significantly between the two genders (females = 63.3%, males = 50.6%, p = ns), though a more severe degree of dysfunction was present in females. Mean left ventricular mass index (LVMi) decreased from 155.41 ± 38.95 g/m2 , pre-operatively, to 114.78 ± 28.83 g/ m2 one-year follow-up examination (paired t-test, p < 0.001), with no differences between genders (p = ns). Complete left ventricular hypertrophy (LVH) regression, which occurred in 57.9% of patients, was also substantially similar in both sexes (p = ns).
Table 4. Haemodynamic characteristics of implanted prosthesis, N =202.
|
Variables |
Nominal size of prosthesis |
||||
|
|
19 |
21 |
23 |
25 |
27 |
|
Number of prosthesis |
16 |
67 |
80 |
32 |
7 |
|
Peak gradient, mmHg |
16.00±7.68 |
13.09±6.17 |
12.54±5.55 |
12.44±4.87 |
8.14±2.04 |
|
Mean gradient, mmHg |
12.06±8.61 |
10.87±6.39 |
8.9±5.11 |
8.22±3.73 |
6.00±2.71 |
|
EOA, cm2 |
1.18±0.38 |
1.59±0.32 |
1.79±0.47 |
2.16±0.59 |
2.16±0.56 |
|
EOAi, cm2/m2 |
0.73±0.23 |
0.94±0.31 |
1.00±0.29 |
1.16±0.37 |
1.09±0.26 |
|
PPM |
|
|
|
|
|
|
> 0.85 cm2/m2 |
2 (0.9%) |
37 (18.3%) |
50 (24.7%) |
27 (13.4%) |
6 (2.9%) |
|
< 0.85 >0.65 cm2/m2 |
8 (3.6%) |
20 (9.9%) |
25 (12.3%) |
5 (2.5%) |
1 (0.5%) |
|
< 0.65 cm2/m2 |
6 (2.9%) |
10 (4.9%) |
5 (2.5%) |
0 |
0 |
Values are expressed as mean ± standard deviation or numbers (%).
.jpg) Figure 3: Top: distribution of average Peak and Mean gradients of implanted Trifecta valves by size, on follow-up examination. Bottom: Distribution of average EOA and EOAi of implanted Trifecta valves by size, on follow-up examination. N = 202.
Figure 3: Top: distribution of average Peak and Mean gradients of implanted Trifecta valves by size, on follow-up examination. Bottom: Distribution of average EOA and EOAi of implanted Trifecta valves by size, on follow-up examination. N = 202.
Figure 4: Short-axis parasternal view of Trifecta valve showing the thickening of cusps.
DISCUSSION The results of the present prospective study confirm the optimal haemodynamic profile of the Trifecta aortic bio-prosthesis, first described in the pilot study by Bavaria and Colleagues [1]. Very low trans-valvular gradients and larger EOAs endow this valve with one of the highest performance levels among currently available pericardial bio-prostheses [3-8]. These features have also been observed in patients with small annuli, relative to body size [2], in whom haemodynamic results have even proved to be comparable to those of stentless bio-prostheses[16]. While several studies have tested the haemodynamic performance of the Trifecta aortic valve and evaluated its durability over variable periods of follow-up [17], very few have reported haemodynamic data collected after one year or longer [1,18,19]; our follow-up data are consistent with their results. The considerable reduction in LVMi and the complete LVH regression attained in more than half of the patient population confirm the excellent haemodynamics of this prosthesis. Our data assume even greater importance if we consider the advanced age, higher number of comorbidities, higher average preoperative risk and elevated number of concomitant procedures of the patients enrolled in the present study. To our knowledge, our population is the oldest one ever to undergo aortic valve replacement with the Trifecta aortic bioprosthesis. Our patients had more comorbidities and relatively higher preoperative risk profiles: nevertheless, mortality was comparable to that expected in a healthy population within the same age-group. Severe PPM, which has been demonstrated to negatively affect patient prognosis [18-21], had a low incidence, occurring in only 21 (10.4%) patients; these were mainly women with little body surface area and small aortic annuli. Diastolic dysfunction, although present in more than half of the population, did not affect the global clinical outcome of patients, probably because of the reported good adherence to optimal medical therapy. Study limitations Our study reflects the annual experience of a single primary center. While the fact that all procedures were carried out by the same surgical team using standard surgical techniques could be an advantage in reducing confounders, the limited size of our population could constitute a limitation to the study. Moreover, although we could document good valve durability on one-year follow-up examination, with only one case of early deterioration, early post-operative and discharge data were not available for comparison, making it impossible to evaluate the temporal trend in the performance of the prosthesis.
CONCLUSION The current study confirmed the already known optimal haemodynamic profile of the Trifecta aortic bio-prosthesis and demonstrated its good durability on one-year follow-up examination in a population of polymorbid, moderate- to high-risk octogenarian patients undergoing AVR and multiple concomitant surgical procedures.
REFERENCES 1. Bavaria JE, Desai ND, Cheung A, Petracek MR, et al. (2014). The St Jude Medical Trifecta aortic pericardial valve: Results from a global, multicenter, prospective clinical study. J Thorac Cardiovasc Surg. 147(2): 590-597. 2. Maruyama M, Daimon M, Kawata T, Kasai T, et al. (2014). Early hemodynamic performance of the trifecta bioprosthetic valve in patients with aortic valve disease. Circ J. 78(6): 1372-1378. 3. Bach DS, Patel HJ, Kolias TJ and Deeb GM. (2016). Randomized comparison of exercise haemodynamics of Freestyle, Magna Ease and Trifecta bioprostheses after aortic valve replacement for severe aortic stenosis. Eur J Cardiothorac Surg. 50(2): 361-367. 4. Fiegl K, Deutsch MA, Rondak IC, Lange R, et al. (2015). Matched Comparison of Two Different Biological Prostheses for Complete Supra-annular Aortic Valve Replacement. Thorac Cardiovasc Surg. 63(6): 459-466. 5. Modi A, Pousios D, Sadeque S, Velissaris T, et al. (2014). Early in-vivo hemodynamic comparison of supra-annular aortic bioprostheses: Trifecta versus Perimount Magna Ease, J Heart Valve Dis. 23(3): 325-332. 6. Minardi G, Pergolini A, Zampi G, Pulignano G, et al. (2013). St. Jude Trifecta versus Carpentier-Edwards Perimount Magna valves for the treatment of aortic stenosis: comparison of early Doppler-echocardiography and hemodynamic performance. Monaldi Arch Chest Dis. 80(3): 126-132. 7. Yadlapati A, Diep J, Barnes M, Grogan T, et al. (2014). Comprehensive hemodynamic comparison and frequency of patient-prosthesis mismatch between the St. Jude Medical Trifecta and Epic Bioprosthetic aortic valves. J Am Soc Echocardiogr. 27(6): 581-589. 8. Ugur M, Suri RM, Daly RC, Dearani JA, et al. (2014). Comparison of early hemodynamic performance of 3 aortic valve bioprostheses. J Thorac Cardiovasc Surg. 148(5): 1940-1946. 9. Edmunds LH Jr, Clark RE, Cohn LH, Grunkemeier GL, et al. (1996). Guidelines for reporting morbidity and mortality after cardiac valvular operations. Ad Hoc Liaison Committee for Standardizing Definitions of Prosthetic Heart Valve Morbidity of The American Association for Thoracic Surgery and The Society of Thoracic Surgeons. J Thorac Cardiovasc Surg. 112(3): 708-711. 10. Zoghbi WA, Chambers JB, Dumesnil JG, Foster E, et al. (2009). Recommendations for evaluation of prosthetic valves with echocardiography and doppler ultrasound: a report From the American Society of Echocardiography’s Guidelines and Standards Committee and the Task Force on Prosthetic Valves, developed in conjunction with the American College of Cardiology Cardiovascular Imaging Committee, Cardiac Imaging Committee of the American Heart Association, the European Association of Echocardiography, a registered branch of the European Society of Cardiology, the Japanese Society of Echocardiography and the Canadian Society of Echocardiography, endorsed by the American College of Cardiology Foundation, American Heart Association, European Association of Echocardiography, a registered branch of the European Society of Cardiology, the Japanese Society of Echocardiography, and Canadian Society of Echocardiography. J Am Soc Echocardiogr, 2009;22:975- 1014. 11. Pibarot P, Dumesnil JG, Lemieux M, Cartier P, et al. (1998). Impact of prosthesis-patient mismatch on hemodynamic and symptomatic status, morbidity and mortality after aortic valve replacement with a bioprosthetic heart valve. J Heart Valve Dis. 7(2):211-218. 12. Pibarot P and Dumesnil JG. (2000).Hemodynamic and clinical impact of prosthesis-patient mismatch in the aortic valve position and its prevention. J Am Coll Cardiol. 36(4):1131-1141. 13. Blais C, Dumesnil JG, Baillot R, Simard S, et al. (2003). Impact of valve prosthesis-patient mismatch on short-term mortality after aortic valve replacement. Circulation. 108(8):983-988. 14. Du Bois D and Du Bois EF. (1989). A formula to estimate the approximate surface area if height and weight be known. 1916 Nutrition. 5(5): 303-311-313. 15. Nagueh SF, Smiseth OA, Appleton CP, Byrd BF 3rd, et al. (2016). Recommendations for the Evaluation of Left Ventricular Diastolic Function by Echocardiography: An Update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging, Eur Heart J Cardiovasc Imaging, 17(12):1321-1360. 16. Tasca G, Martino AS, Giannico F, Riva B, et al. (2015). Hemodynamic comparison between Trifecta and Freestyle valves implanted in small aortic roots. One-year echocardiographic results from a prospective randomized study. J Heart Valve Dis. 24(3):360-367. 17. Phan K, Ha H, Phan S, Misfeld M, et al. (2015). Early hemodynamic performance of the third generation St Jude Trifecta aortic prosthesis: A systematic review and metaanalysis. J Thorac Cardiovasc Surg.149:1567-1575-2. 18. Ugur M, Byrne JG, Bavaria JE, Cheung A, et al. (2014). Suture technique does not affect hemodynamic performance of the small supra-annular Trifecta bioprosthesis. J Thorac Cardiovasc Surg. 148(4):1347-1351. 19. Ruggieri VG, Anselmi A, Chabanne C, Lelong B, et al. (2016). “Three-year haemodynamic performance of the St Jude Trifecta bioprosthesis. Eur J Cardiothorac Surg. 49(3):972-977. 20. Rao V, Jamieson WR, Ivanov J, Armstrong S, et al. (2000). Prosthesis-patient mismatch affects survival after aortic valve replacement. Circulation.102(3): 5-9. 21. Head SJ1, Mokhles MM, Osnabrugge RL, Pibarot P, et al. (2012). The impact of prosthesis-patient mismatch on long-term survival after aortic valve replacement: a systematic review and meta-analysis of 34 observational studies comprising 27 186 patients with 133 141 patientyears. Eur Heart J. 33(12):1518-1529.