Information Links
Related Conferences
Previous Issues Volume 8, Issue 2 - 2023
The Myriad of Post Herpetic Anterior Uveitis in Herpes Zoster Viral Infection
Rubina Shah*
National Eye Center Lahore, Pakistan
*Corresponding author: Dr. Rubina Shah OD, FAAO, National Eye Center Lahore, Pakistan, Phone: 00923424014487; Email: [email protected].
Received Date: September 01, 2023
Published Date: September 12, 2023
Citation: Shah R, et al. (2023). The Myriad of Post Herpetic Anterior Uveitis in Herpes Zoster Viral Infection. Mathews J Ophthalmol. 8(2):33.
Copyrights: Shah R, et al. © (2023).
ABSTRACT
Herpes viridae is an infective family of virus that is common worldwide. An additional 100 herpes viruses have been categorized, but humans may be affected by only 8 which are Herpes simplex virus (HSV) and varicella-zoster virus (VZV), cytomegalovirus (CMV), Epstein–Barr virus (EBV), human herpes virus [1]. This wide spectrum of viruses can cause various ocular diseases with clinical presentations of keratoconjunctivitis, dendritic epithelial keratitis Blepharitis, geographic or trophic herpetic corneal ulceration, stromal keratitis, and intraocular inflammation. Two major entities may comprise ocular inflammation, anterior uveitis with or second with active corneal involvement as epithelial keratitis or interstitial keratitis and viral retinopathy, which were seen in both immunocompetent and immunocompromised patients. An estimated 90% of intra-ocular inflammation is associated with anterior uveitis and 5 % to 10% of the case of uveitis has chances of herpetic anterior uveitis that is either due to herpes simplex or varicella zoster virus infection [2-4]. It is the common cause of anterior infectious uveitis. This case outlined an overview of post-herpetic anterior uveitis and its occurrence in Herpes Zoster and the effectiveness of medical therapy.
Keywords: HZO (Herpes Zoster Ophthalmicus), OD (Ocular Dexterous), OS (Ocular Sinister), PHN (Post Herpetic Neuralgia), The Herpetic Eye Disease Study (HEDS), Cytomegalovirus (CMV), Epstein–Barr Virus (EBV).
INTRODUCTION
CASE REPORT
A 45-year-old Asian male presented with chief complaints of red eye, lacrimation, persistent intense pain, photophobia, and blurring of vision in the right eye for two days. He denied an ocular history of spectacles. His previous history showed systemic therapy antiviral acyclovir 800mg tablet oral qid for eight days and prednisolone 1% eye drops for Herpes Zoster ophthalmicus prior six months, PHN history of severe pain after three months of infection HZ, and birth history showed measles. He denied any itching and rashes on examination day. Moreover, his cutaneous examination previously showed a scar on the right face due to pustules and vesicles. He denied any insect bites, hearing problems, and shortness of breath. Family ocular history was none. Social history was normal. He had no known drug allergies. He had fatigue, and depression and was not oriented to time, place, and person.
Uncorrected distance visual acuity was counting finger 2Feet OD and 20/40 OS. There was no previous habitual spectacle correction for distance and near. Near acuity showed Nil OD and N12 OS to N5 with +1.50 additions. Manifest refraction revealed no improvement due to incomplete opening of eyes, while the left eye was 20/25 with +1.75DS. Goldman Applanation tonometry measured 17 mmHg OD, and 12 mmHg OS at 10:03 am. The right eye showed blepharospasm with increased sensitivity to light. Pupils were equal, round, and reactive. Slit lamp Biomicroscopy of the right revealed normal adnexa with lid swelling with circumcorneal congestion. And, left eye showed normal lid adnexa, lashes, puncta. The right cornea findings revealed diffused corneal edema inflammation and endothelial decompensation. Epithelial keratitis was absent. Right eye anterior chambers appeared to evidence of cell or flare and Kps, uveitis, and estimation of the chamber angles were 4/4 via the Von Herrick method. The contour of the irides was flat and brown in the left eye whereas sectoral atrophy in the right was noted without posterior synechiae. When pupils were dilated with one drop of 1% Mydriacyl showed a lenticular change posterior subcapsular changes grade III. Marked vitritis was noted in the right eye. A detailed fundus assessment of the left eye revealed normal optic nerve contour with a cup-to-disc ratio of 0.3 in both eyes, but the right fundus couldn’t be revealed due to hazy media and inflammation.
Diagnosed the Disease
The diagnosis was relatively straightforward because of the presence of concomitant skin scar, active interstitial keratitis, and corneal anesthesia. However, in the absence of vague evidence of current or past HSV infection, it requires clinical suspicion and observation. Anterior uveitis secondary to HSV or VZV occurs in association with active or inactive corneal involvement, but anterior uveitis without associated corneal changes also occurs as an isolated entity.
The diagnosed ocular case was anterior herpetic Uveitis with corneal odema, a rare non-ulcerating clinical case associated with Herpes Zoster Varicella HZV Virus. The determined depth of inflammation of stroma and other corneal manifestations required a comprehensive Slit-lamp Biomicroscopy, patient History of HZO, and intraocular pressure guard. For stromal keratitis, which is the most visually significant problem for the cornea, a delicate balance of steroids is sometimes necessary, and even with it, patients can lose vision. Stromal keratitis can also be seen in recurrent cases in 20% of eyes with HZO.
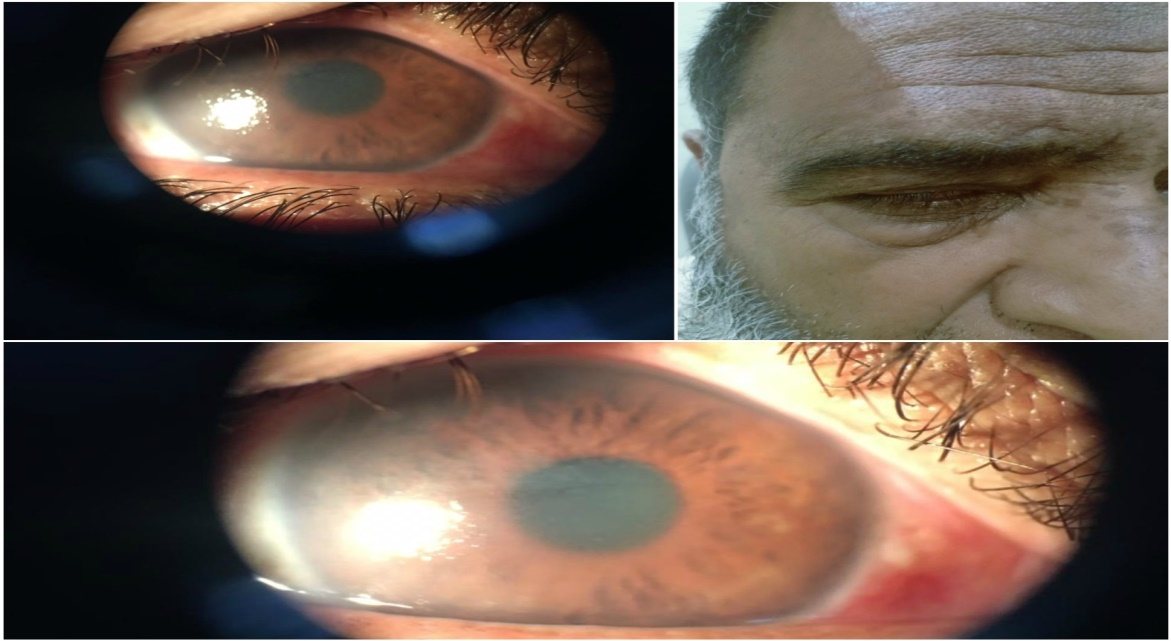
Figure 1. (Photo courtesy Rubina Shah National Eye Centre) NOTE: The Biomicroscopy showed diffuse corneal odema and endothelial decompensation. Cutaneous scarring right side of the face revealed Herpes zoster.
DIFFERENTIAL DIAGNOSIS
An increased awareness of the characteristic clinical features is important for early diagnosis and appropriate treatment.
Fuchs’ uveitis syndrome
Fuchs’ uveitis syndrome [4] can be difficult to discriminate from herpetic anterior uveitis because of the presence of transillumination defects, elevated intraocular pressure, and iris atrophy. However, iris atrophy tends to be concentrated over the iris sphincter and stromal rather than full thickness, the transillumination defects show a radial pattern rather than sectoral, and the cumulative damage to the trabecular meshwork rather than a transient trabeculitis is marked in raised IOP. It is thus unresponsive to topical corticosteroid and antiviral treatment. Additionally, Fuchs’ uveitis syndrome patient is not inclined to form posterior synechiae, whereas posterior synechiae frequently form in patients with herpetic anterior uveitis and were noted in 58% of the patients from the Netherlands. It shows an abnormal uveal pigmentation with chronic low-grade inflammation and ultimately causes iris atrophy and secondary glaucoma. Manifest ocular symptoms have no pain, redness of the external eye, or miosis and may be associated without any additional systemic disease present. Late progression of disease is associated with cataract formation, raised IOP, and, occasionally, vitreous cellular infiltrates. Even though it typically affects unilaterally, 7.8%-10% of affected patients have bilateral conditions [5]. It may be undiagnosed during routine examination or may present with raised IOP [6]. Treatment requires short-term topical corticosteroids for flares. However, might need long-term therapy. Moreover, like other uveitides, to eliminate cells from the anterior chamber a must be prescribed to break down the blood-aqueous barrier and inflammatory activity.
Posner-Schlossman syndrome
Posner-Schlossman syndrome is a glaucomatocyclitic condition of the eye that is self-limited and recurrent episodes of raised intraocular pressure (IOP) and inflammation of the anterior chamber. It is known as secondary inflammatory glaucoma. Posner and Schlossman first diagnosed the features [7]. Glaucomatocyclitic crisis is characterized by a slight decrease in vision, elevated IOP, and open anterior chamber angles, with normal visual fields and optic nerve appearance. In addition, IOP and outflow facility are normal between episodes [8]. It is bilateral and very rarely simultaneous [9,10]. Cytomegalovirus (CMV) infection is supported by literature as the inflammatory precursor to the anterior uveitis and raised IOP [11]. Patients may report blurred vision or halo vision if raised IOP or if there is corneal edema. The history of past attacks of blurred vision lasting several days, monthly, or yearly. The treatment revolves around complete medical care with a history of present illness, any drug allergies, eye examination, a careful explanation of the disorder, and understanding the disease with long-term follow-up [12].
Peripheral corneal endothelitis: The primary focus of inflammation in this condition (peripheral corneal endotheliitis or PCE), initially thought to be autoimmune (Khodadoust et al. 1982), appears to be the corneal endothelium, but a significant anterior uveitis is usual. Patients present with unilateral or bilateral blurring of vision. The central cornea is usually clear, but a variable area of the corneal periphery may be edematous. Essentially this is a white infiltration of deep cornea which may be seen, and the demarcation of the border between apparent normal endothelium may be seen clearly as a distinct line, similar to the Khodadoust line of corneal rejection after corneal transplantation. Generally, large KPs are present at the abnormal area, and gonioscopy may reveal AC anterior chamber angle within the affected loci. Scarce cases are associated with intermediate uveitis as well (Khodadoust et al. 1986). Herpetic inflammation is the common manifestation that causes disciform corneal endotheliitis and other viruses with mumps, varicella-zoster virus (VZV), human herpesvirus 7 (HHV7) and rhabdovirus can also affect the endothelium. Nevertheless, there is the concept of CMV as an etiology of corneal endotheliitis(Chee et al. 2007) and following corneal graft aswell, with linear deposition of KPs and focal endothelial lesions features(Koizumi et al. 2008).
Generally, Fuchs uveitis is associated with HSV and CMV [13, 14-15]. The most helpful diagnosis of herpetic anterior uveitis is sectoral iris atrophy and raised IOP. However, various inflammatory and noninflammatory diseases include unilateral or bilateral iris atrophy that are Fuchs uveitis syndrome, Vogt–Koyanagi–Harada disease, pigment dispersion syndrome, acute angle closure glaucoma, trauma, and Horner’s syndrome. A recent study has shown diffuse depigmentation and bilateral geographical changes in the iris stroma [16]. The differential diagnosis of the acute rise of IOP can also occur in granulomatous anterior uveitis with the systemic association of sarcoidosis, toxoplasmosis, syphilis, leprosy, and also in Posner–Schlossman syndrome [17]. The recurrence is typically noted temporarily with acute onset of Herpetic anterior uveitis disease. Consequently, that challenge the course from chronic entities for example Fuchs uveitis syndrome and Vogt– Koyanagi–Harada disease. The DDx differential diagnosis can also be judged from the distribution of KPs. Herpetic KPs are large, greyish in color, flat, and deposited on the central corneal endothelium. However, Fuchs uveitis syndrome diffuses medium size Kps which is hard to differentiate [18]. While herpetic KPs usually vanish with treatment. The acute episode may present with focal odema that may cause irregular dilation. The clinical presentation of HZV and HSV cannot be judged from the appearance of iris atrophy. However, VZV uveitis may occur in the absence of skin involvement known as (herpes zoster sine herpete) [19]. Currently, a clinical diagnosis of VZV uveitis can be only diagnosed in patients with past attacks of herpes zoster ophthalmicus cutaneous involvement. Van der Lelij et al. [20] termed that aqueous humor analysis revealed HSV in 83% and VZV in 13% with sectoral iris atrophy. However, sectoral iris atrophy can also be seen in recurrent unilateral herpetic anterior uveitis affected by CMV [21]. The IOP elevation is associated with acute trabeculitis and prompt response to anti-inflammatory treatment is noted [22].
TREATMENT
The mainstay therapy of herpetic anterior uveitis is topical corticosteroids and oral antiviral agents. The Herpetic Eye Disease Study [23] demonstrated the number of recurrences of the disease can be hampered by the prophylactic oral acyclovir for 12 months. In a retrospective study, Miserocchi et al. demonstrated the effectiveness of long-term prophylactic acyclovir beyond 12 months [24]. Miserocchi et al. [24] reported the tendency of lifelong treatment with the use of oral acyclovir 800 mg daily for at least 2 years after the onset of herpetic uveitis. It showed 13% of our patients could use oral acyclovir for at least 12 months. The high cost of long-term treatment restricted the use of prophylactic therapy.
The pathogenesis of herpetic iridocyclitis is believed to be involved in active viral replication and the host immune responses. Topical corticosteroids and antiviral agents are commonly used to treat ocular herpetic disease. Topical corticosteroids control iridocyclitis and also acutely decrease intraocular pressure owing to their anti-inflammatory effects on the trabecular meshwork. However, topical and oral antihypertensive agents may be necessary to control the pressure especially initially.
Corticosteroids should be slowly tapered once the inflammation regresses and topical antiviral agents affect herpetic epithelial keratitis, but the much deeper pattern of ocular involvement has not been recognized. It should be used in conjunction with corticosteroid drops to prevent recurrent epithelial keratitis. Yet, another topical anti-herpetic agent of treatment of choice has become gancyclovir gel.
A detail concerning the use of oral acyclovir, demonstrated by the HEDS (herpetic eye disease control group) results suggests a benefit of oral acyclovir in the treatment of herpetic iridocyclitis in patients receiving topical corticosteroids and topical antiviral prophylaxis., Oral acyclovir at a dosage of 400 mg 5 times per day for several weeks is usually used. Intravenous acyclovir (10 mg/kg/d) may be considered in severe anterior uveitis and must be employed in all immunocompromised patients. Alternatively, one may use valacyclovir 1 g 3 times per day or famciclovir 500 mg 3 times daily. Oral acyclovir, 600 to 800 mg/day, given on a long-term basis, can diminish the recurrence of herpetic anterior uveitis.
Ocular Medication
Ocular medication prescribed by the ophthalmologist for endothelial decompensation and uveitis was cyclopen eye drop qid, Eyebradex eye drop eight times a day, synigan eye drop twice a day, and ophthypertonic saline solution 4x days.
The patient had a history of postherpetic neuralgia and was using PHN medication Deltracotril 30 mg analgesics, Tab tramal pain reliever in case of severe pain, Tab tegral( anticonvulsant) 200mg twice a day. The patient had a history of Oral acyclovir 800mg during the attack of HZ herpes Zoster. Ocular Medication used was Tobrex eye qid, Zaviraz ointment tid.
General treatment protocol PHN for the disease turns around by reducing the neuronal membrane and beginning and transmission of the impulse. The tranquilizing effect and anticholinergic effect can be lessened by a group of tricyclic depressants such as amitriptyline(Evanil) may be helpful in severe neuronal pain. Second, the clinical course of antiviral has been very short for antivirals that inhibit complications and recurrences. Such as famciclovir directly acts on Viral DNA synthesis. Third, anticonvulsants can be used for severe muscular spasms and provide restfulness during pain. When compared with placebo treatment certain medications such as gabapentin products (i.e., Neurontin, Gralise, Horizant) describe a statistical progress in pain scores (i.e., a decrease by at least 50% from baseline). Fourth, corticosteroids play a vital role in anti-inflammatory action which modifies the body's immune response to various stimuli as dexamethasone is used for inflammation aswell as prednisone or methylprednisone. Fifth, Studies have shown that some natural chemicals derived from plants of the Solanaceae family may lessen skin and joints insensitive to pain. It is thought to be acting as a chemomediator of pain transmission from the periphery to CNS such as capsaicin. Topical anesthetics such as lidocaine can stabilize neuronal membrane and their neural activity.
FOLLOW UP #1 (week)
On the date of examination, the patient revealed a vision of the affected eye was 20/60 with no improvement with refraction. A slightly regressed odema was noted. Mydriatic pupil and lenticular changes. Slight clearance of vitritis. Fundi were unremarkable. Topical Eyevradex ointment during the night, eyebradex eye drop 5 times a day. Tears plus eye drops four times a day.
FOLLOW UP #2 (2 weeks )
On the date of examination, the patient reveals better odema of the cornea with vision 20/40 in OD. Regressed flare cells. The patient was recovering from the herpetic stage. Corticosteroids were tapered and artificial tears were given. However, variable follow-ups and the nature of the study did not allow calculation and recurrences seemed to be more frequent after discontinuation of treatment. Since we tapered topical steroids very slowly, we did not observe severe bounce attacks.
FOLLOW UP #3( 3 months)
Follow-up examination showed regressed symptoms with visual acuity of 20/30 in the right eye with a manifest refraction of -2.00DS and 20/40 to 20/25 in the left eye +1.75DS. Near acuity showed N10 on the right and N5 on the left with +1,50 addition for near. The patient has advised artificial tears and spectacle correction for distance and near. However, the cataract of grade III PSSC posterior subcapsular cataract was still noted, and educated patients may experience the gradual decline of vision. Further follow-up was required after three month to rule out the recurrence.
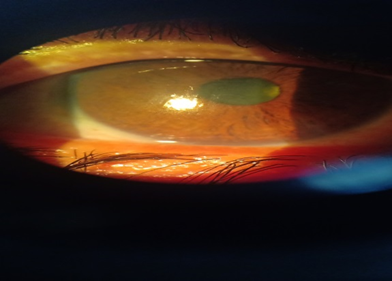
Figure 2: The illustration of the cornea after regressed herpetic keratitis.
DISCUSSION
The major morbidity worldwide is Herpes viruses which have eight distinct viruses with six of them affecting humans. Specifically, herpes simplex virus, herpes zoster virus cytomegalovirus, and HSV2 and VZV3 are the most common ocular pathogens, which have an approximate incidence of 10 to 20/100,000 person-years estimation [25-27].
The case was reviewed and managed for herpetic anterior uveitis associated with a history of post-herpetic neuralgia and herpes zoster infection. Clinical presentation may cover a wide ocular manifestation such as Blepharitis, conjunctivitis, scleritis, keratitis, anterior uveitis, necrotizing retinitis, choroiditis, and optic neuritis, but the manifestation of anterior uveitis was ruled out. The anterior chamber activities of inflammation occur in up to 10% of patients with HSV keratitis [28-30] and 50% of immunocompetent patients with herpes zoster ophthalmicus [30-32]. Although anterior herpetic uveitis diagnosis is usually straightforward due setting of dermatitis or dendritic keratitis, it can be quite difficult in the absence of these signs. Moreover, nonspecific symptoms like patients' complaints of blurred vision, pain, redness, and photophobia are usually seen. The presence of localized corneal scars or edema decreased corneal sensation, geographically or diffusely distributed keratic precipitates, acutely raised intraocular pressure, and iris atrophy, which is frequently localized and tends to produce both pupillary distortion are the clues to diagnose herpetic uveitis.
Additionally, it is almost always unilateral. Perhaps harder to discriminate anterior herpetic uveitis is distinguishing features of HSV from VZV due to anterior chamber inflammation [29,33]. The history of herpes zoster ophthalmicus provides a supportive and striking way to diagnose VZV-induced iritis, but it’s not necessary for cutaneous lesions' presence. The reduction of corneal sensation can be difficult to quantify, but it also suggests the presence of VZV. Similarly, the presence of a pattern of dendritic keratitis may help distinguish HSV from VZV keratitis. The prominence of well-developed terminal end bulbs of HSV may stain with fluorescein, but the clinical features of VZV pseudo dendrites are usually slightly elevated, broader, and with less regular branching, few terminal dilatations or polymorphous that stain centrally with rose Bengal staining or edge pooling with fluorescein. Moreover, sectoral iris atrophy is highly suggestive of VZV-induced anterior uveitis urge some investigations such as serum anti-VZV antibodies have led to suggest iris sectoral atrophy [29,33,34]. It remains likely to measure directly herpes virus deoxyribonucleic acid or antiherpes virus antibodies in the aqueous humor from patients associated with sector iris atrophy. The article by Van der Lelij and colleagues [35] which appeared in the June issue of Ophthalmology addressed this importance where the authors performed anterior chamber paracentesis on a large, seen in unilateral anterior uveitis association with sectoral iris atrophy have no evidence of concurrent epithelial and stromal keratitis that was clinically based cohort of patients from the Netherlands. About 20% of the patients defined by Van der Lelij and associates were initially thought to have Posner-Schlossman syndrome (glaucomatocyclitic crisis). The differential diagnosis of herpetic anterior uveitis [36,31] can also be difficult to distinguish from Fuchs endothelial uveitis because of the presence of transillumination defects, elevated intraocular pressure, and iris atrophy. It is obvious that herpetic anterior uveitis with be treated with antiviral drugs [32,33]. Oral acyclovir, 400 mg twice daily, has been shown to reduce the recurrence rate of HSV [32].
The Herpetic Eye Disease Study (HEDS), a prospective, randomized, double-masked, placebo-controlled multicenter trial supported by the National Eye Institute, established further that the effect of oral acyclovir decreases the recurrence rate of herpetic stromal disease and its beneficial prophylactic effect depends on continued use of suppressive doses of an oral antiviral agent [37]. The HEDS collaborators also examined the effect of oral acyclovir on the duration and number of recurrences of anterior uveitis in patients with prior or concurrent herpetic keratitis [30]. Nevertheless, it failed to demonstrate the statistical significance of the overall effect even notifying the benefit trend of Oral acyclovir therapy. Likewise, various number antiviral agents have recently been available to treat herpetic anterior uveitis. Oral acyclovir has been shown to reach the therapeutic level in both tears and aqueous humor [37] essentially eliminating the requirement of antiviral even in active corneal involvement.
Even though controlled comparisons have yet to be considered in patients with eye disease, the more convenient dosage provided to benefit the patient is valacyclovir, 1 g three times daily, and famciclovir, 500 mg three times daily, appear to be equivalent to oral acyclovir for the treatment. Furthermore, valacyclovir, a benefit have shown a pro-drug of acyclovir that is absorbed much better from the gastrointestinal tract and tends to yield threefold to fourfold higher circulating drug levels as compared to oral acyclovir. Another valacyclovir antiviral agent has been reported to cause thrombocytopenic purpura or hemolytic uremic syndrome in a small number of patients infected by the human immunodeficiency virus. However, it must be probably avoided in this population of patients. Alternatively, topical antiviral agents fail to reach therapeutic levels and absorb in the aqueous humor [38]. The side effects can involve both the corneal and conjunctival epithelium for prolonged dosage, causing 5% to 10% allergic blepharoconjunctivitis or corneal epitheliopathy [39,40]. The single exception is Acyclovir ointment which stabilizes adequate aqueous levels and is well tolerated for prolonged usage. It causes blurred vision but isn’t easily available clinically. However, topical therapy, with ointment does not have any direct effect on viral recurrence in sensory ganglia. For these reasons, practioner prefer oral therapy of acyclovir over ointment after evaluating the condition of the anterior chamber, and is considered to be safest for longer period therapy in HSV and HZV involving uncontrolled eye problems eye. The active disease of the postherpetic uveitic eye also needs corticosteroid therapy and a cycloplegic/mydriatic agent to avoid any posterior synechiae formation reduce pain and provide comfort. A core regimen of treatment might include prednisolone acetate, 1%, along with tropicamide, 1%, each four times daily. Reducing the course of corticosteroids can be challenging and May requires months to achieve the ultimate effect. To control long-term inflammation some patients may require low-dose corticosteroids indefinitely.
Exemplifying the available clinical data, physicians and ophthalmologists should recommend the best treatment systemically and ocularly to prevent any further worsening of the disease. As mentioned for PHAU Post herpetic anterior uveitis current treatment for older age modifies administering the herpes zoster vaccination. Further, clinically a trial systemic medication associated with PHN post-herpetic neuralgia or herpes zoster can also be guarded with the latest treatment such as patching monotherapy before resorting to a systemic therapy must be considered in PHN patients. Alternatively, it may be administered in combination with a tricyclic antidepressant or a gabapentinoid to potentiate analgesic response and reduce the dose requirement of systemic therapies.
CONCLUSION
Postherpetic anterior uveitis is a distinctive condition caused by infection of the zoster virus. Herpes zoster (HZ) infection breaks out when varicella-zoster virus (VZV) reactivates. It remains dormant after the primary attack in the dorsal root of the ganglia. The characteristic raises a painful vesicular rash with unilateral dermatomal distribution. The myriad of infection is approximately 1 million estimated in the USA and one in every three individual get infected from herpes zoster and have worldwide geographic distribution, but annual epidemics are more widespread in temperate climates and over 90 percent of adults in the United States have evidence of VZV infection are at risk for herpes zoster [41]. In Pakistan and certain other Asian countries, the overall prevalence of primary varicella infection is relatively low and late [42,43]. This change in climatic conditions may be responsible for a different seasonal and morphological pattern in herpes zoster. Uveitis is primarily either an ocular condition or related to various systemic disorders. The analyzed frequency of systemic diseases associated with uveitis varies from 18% to 43% in different areas [44,45-49]. It is crucial to manage the ocular involvement and better treatment protocol to manage the patient. The recurrence of the disease can be managed to reduce and prevent vision-threatening conditions such as keratitis, glaucoma, and cataracts in herpetic AU with an early and accurate diagnosis. The developing treatment available for ocular manifestation requires oral antiviral agents and topical steroids which are the current treatment regime for herpetic AU. A prophylactic oral acyclovir is evidenced by a reduction in the number of recurrences of herpetic eye disease. Longer follow-up and long-term treatment can reduce the recurrence of anterior herpetic Uveitis.
ACKNOWLEDGMENTS
None.
DECLARATION OF CONFLICT OF INTERESTS
There is no conflict of interest due to the presence of only one author of the article.
REFERENCES
- Siverio Júnior CD, Imai Y, Cunningham ET Jr. (2002). Diagnosis and management of herpetic anterior uveitis. Int Ophthalmol Clin Winter. 42(1):43-48.
- Cunnigham ET Jr. (2000). Diagnosing and treating herpetic anterior uveitis. Ophthalmology. 107(12):2129-2130.
- Gaynor BD, Margolis TP, Cunnigham ET Jr. (2000). Advances in diagnosis and management of herpetic uveitis. Int Ophthalmol Clin. 40(2):85-109.
- Guney E, Akcay BI, Erdogan G, Unlu C, Akcali G, Bayramlar H. (2012). The etiologic features of anterior uveitis in a Turkish population. Clin Ophthalmol. 6:845-849.
- Jones NP. (1993). Fuchs' heterochromic uveitis: an update. Surv Ophthalmol. 37(4):253-272.
- Norrsell K, Sjodell L. (2008). Fuchs' heterochromic uveitis: a longitudinal clinical study. Acta Ophthalmol. 86(1):58-64.
- Posner A, Schlossman A. (1948). Syndrome of unilateral recurrent attacks of glaucoma with cyclitic symptoms. Arch Ophthalmol. 39(4):517-535.
- Posner A, Schlossman A. (1953). Further observations on the syndrome of glaucomatocyclitic crises. Trans Am Acad Ophthalmol Otolaryngol. 57(4):531-536.
- Levatin P. (1956). Glaucomatocyclitic crisis occurring in both eyes. Am J Ophthalmol. 41(6):1056-1059.
- Dinakaran S, Kayarkar V. (2002). Trabeculectomy in the management of Posner-Schlossman syndrome. Ophthalmic Surg Lasers. 33(4):321-322.
- Chee SP, Jap A. (2008). Presumed fuchs heterochromic iridocyclitis and Posner-Schlossman syndrome: comparison of cytomegalovirus-positive and negative eyes. Am J Ophthalmol. 146(6):883.e1-889.e1.
- Hung PT, Chang JM. (1974). Treatment of glaucomatocyclitic crises. Am J Ophthalmol. 77(2):169-172.
- Chee SP, Bacsal K, Jap A, et al. (2008). Clinical features of cytomegalovirus anterior uveitis in immunocompetent patients. Am J Ophthalmol. 145(5):834-840.
- Barequet IS, Li Q, Wang Y, et al. (2000). Herpes simplex virus DNA identification from aqueous fluid in Fuchs heterochromic iridocyclitis. Am J Ophthalmol. 129(5):672-673.
- de Groot-Mijnes JD, de Visser L, Rothova A, et al. (2006). Rubella virus is associated with Fuchs heterochromic iridocyclitis. Am J Ophthalmol. 141(1):212-214.
- Tugal-Tutkun I, Araz B, Taskapili M, et al. (2009). Bilateral acute depigmentation of the iris: report of 26 new cases and four-year follow-up of two patients. Ophthalmology. 116(8):1552-1557.
- Siverio CD, Imai Y, Cunningham ET. (2002). Diagnosis and management of herpetic anterior uveitis. Int Ophthalmol Clin. 42(1):43-48.
- Tugal-Tutkun I, Guney-Tefekli E, Kamaci-Duman F, Corum I. (2009). A cross-sectional and longitudinal study of Fuchs uveitis syndrome in Turkish patients. Am J Ophthalmol. 148(4):510-515.
- Nakamura M, Tanabe M, Yamada Y, Azumi A. (2000). Zoster sine herpete with bilateral ocular involvement. Am J Ophthalmol. 129(6):809-810.
- Van der Lelij A, Ooijman FM, Kijlstra A, Rothova A. (2000). Anterior uveitis with sectoral iris atrophy in the absence of keratitis. A distinct clinical entity among herpetic eye diseases. Ophthalmology. 107(6):1164-1170.
- Markomichelakis NN, Canakis C, Zafirakis P, et al. (2002). Cytomegalovirus as a cause of anterior uveitis with sectoral iris atrophy. Ophthalmology. 109(5):879-882.
- Green LK, Pavan-Langston D. (2006). Herpes simplex ocular inflammatory disease. Int Ophthalmol Clin. 46(2):27-37.
- Herpetic Eye Disease Study Group. (1998). Acyclovir for the prevention of recurrent herpes simplex virus eye disease. N Engl J Med. 339(5):300-306.
- Miserocchi E, Waheed NK, Dios E et al. (2002). Visual outcome in herpes simplex virus and varicella zoster virus uveitis. A clinical evaluation and comparison. Ophthalmology. 109(8):1532-1537.
- Gaynor BD, Margolis TP, Cunningham ET Jr. (2000). Advances in the diagnosis and management of herpetic uveitis. Int Ophthalmol Clin. 40(2):85-109.
- Liesegang TJ, Melton LJ III, Daly PJ, Ilstrup DM. (1989). Epidemiology of ocular herpes simplex. Incidence in Rochester, Minn, 1950 through 1982. Arch Ophthalmol. 107(8):1155-1159.
- Liesegang TJ. (1989). Epidemiology of ocular herpes simplex. Natural history in Rochester, Minn, 1950 through 1982. Arch Ophthalmol. 107:1160-1165.
- Gaynor BD, Margolis TP, Cunningham ET Jr. (2000). Advances in the diagnosis and management of herpetic uveitis. Int Ophthalmol Clin. 40(2):85-109.
- Liesegang TJ. (1999). Classification of herpes simplex virus keratitis and anterior uveitis. Cornea. 18(2):127-143.
- A controlled trial of oral acyclovir for iridocyclitis caused by herpes simplex virus. The Herpetic Eye Disease Study Group. Arch Ophthalmol. 1996;114(8):1065–1072.
- A controlled trial of oral acyclovir for the prevention of stromal keratitis or iritis in patients with herpes simplex virus epithelial keratitis. The Epithelial Keratitis Trial. The Herpetic Eye Disease Study. Arch Ophthalmol. 1997;115(6):703-712.
- Gaynor BD, Margolis TP, Cunningham ET Jr. (2000). Advances in the diagnosis and management of herpetic uveitis. Int Ophthalmol Clin. 40(2):85-109.
- Liesegang TJ. (1999). Varicella-zoster virus eye disease. Cornea. 18(5):511-531.
- Marsh RJ, Easty DL, Jones BR. (1974). Iritis and iris atrophy in Herpes zoster ophthalmicus. Am J Ophthalmol. 78(2):255-261.
- Van der Lelij A, Ooijman FM, Kijlstra A, Rothova A. (2000). Anterior uveitis with sectoral iris atrophy in the absence of keratitis: a distinct clinical entity among herpetic eye diseases. Ophthalmology. 107(6):1164-1170.
- Jones NP. (1993). Fuchs’ heterochromic uveitis: an update. Surv Ophthalmol. 37(4):253-272.
- Hung SO, Patterson A, Rees PJ. (1984). Pharmacokinetics of oral acyclovir (Zovirax) in the eye. Br J Ophthalmol. 68(3):192-195.
- Wilhelmus KR, Falcon MG, Jones BR. (1981). Herpetic iridocyclitis. Int Ophthalmol. 4(3):143-150.
- A controlled trial of oral acyclovir for iridocyclitis caused by herpes simplex virus. The Herpetic Eye Disease Study Group. Arch Ophthalmol. 1996;114(8):1065-1072.
- A controlled trial of oral acyclovir for the prevention of stromal keratitis or iritis in patients with herpes simplex virus epithelial keratitis. The Epithelial Keratitis Trial. The Herpetic Eye Disease Study Group. Arch Ophthalmol. 1997;115:703-712.
- Akram DS, Qureshi H, Mahmud A, et al. (2000). Seroepidemiology of varicella-zoster in Pakistan. Southeast Asian J Trop Med Public Health. 31(4):646-649.
- Liyanage N, Sirimali F, Malavige G et al. (2007). Seroprevalence of varicella-zoster virus infections in Colombo District, Sri Lanka. Indian J Med Sci. 61(3):128-134.
- Toyama N, Shiraki K. (2009). Epidemiology of herpes zoster and its relationship to varicella in Japan: A 10-year survey of 48,388 herpes zoster cases in Miyazaki prefecture. J Med Virol. 81(12):2053-2058.
- Rothova A, Buitenhuis HJ, Meenken C, et al. (1992). Uveitis and systemic disease. Br J Ophthalmol. 76(3):137-141.
- Smit RL, Baarsma GS, de Vries J. (1993). Classification of 750 consecutive uveitis patients in the Rotterdam Eye Hospital. Int Ophthalmol. 17(2):71-76.
- Ronday MJ, Stilma JS, Barbe RF, et al. (1996). Aetiology of uveitis in Sierra Leone, West Africa. Br J Ophthalmol. 80(11):956-961.
- Rosenbaum JT. (1989). Uveitis: an internist's view. Arch Intern Med. 149(5):1173-1176.
- Chung YM, Yeh TS, Liu JH. (1988). Endogenous uveitis in Chinese—an analysis of 240 cases in a uveitis clinic. Jpn J Ophthalmol. 32(1):64-69.
- Ragozzino MW, Melton LJ III, Kurland LT, Chu CP, Perry HO. (1982). Population-based study of herpes zoster and its sequelae. Medicine (Baltimore). 61(5):310-316.