Information Links
Related Conferences
Previous Issues Volume 8, Issue 1 - 2024
The Battle of the Physic and Chemistry against Pathogenic Microorganism
Ferruccio Trifirò*, Paolo Zanirato#
Academy of Sciences of Bologna, via Zamboni 31, Bologna, Italy
*Corresponding author: Dr. Ferruccio Trifirò, Academy of Sciences of Bologna, via Zamboni 31, Bologna, Italy, E-mail: [email protected]
#Co-corresponding author: Dr. Paolo Zanirato, Academy of Sciences of Bologna, via Zamboni 31, Bologna, Italy, E-mail: [email protected]
Received Date: April 24, 2024
Published Date: May 18, 2024
Citation: Trifirò F, et al. (2024). The Battle of the Physic and Chemistry against Pathogenic Microorganism. Mathews J Pharma Sci. 8(1):27.
Copyrights: Trifirò F, et al © (2024).
ABSTRACT
Global analysis of the active ingredients of the substances used such as:
Antiseptics on living tissues (intact, damaged skin and mucous membranes) are: chlorhexidine, the electrolytic chloroxidant, povidone iodine, hydrogen peroxide and ethyl and isopropyl alcohols. The least used ones are iodine tincture, eosin, chloramine T, benzalkonium chloride, triclosan and silver.
Sterilizers, chemical products used to destroy not only all microorganisms, but also the spores present on inanimate surfaces, such as medical devices and surgical instruments with a high and medium level of risk for people, but also hospital and civil environments with a high and medium risk of contagion.
Disinfectants, high level of surfaces, articles and non-critical medical instruments, i.e. those that do not come into contact with patients or with damaged skin or mucous membranes and, in particular, they are also the disinfectants selected against SARS-CoV-2 and intermediate level, used for the elimination of pathogenic microorganisms that contaminate inanimate surfaces, medical and surgical equipment and healthcare, hospital and extra-hospital and low-level environments, which are based on a single active ingredient and in an aqueous solution and are used for disinfection in hospitals in places not occupied by patients and for devices that do not come into contact with patients and in civil environments. Some of these disinfectants may have the same active ingredients as high- and intermediate-level antiseptics and disinfectants, but are used at lower concentrations and for shorter contact times.
Keywords: Antiseptics, Sterilant, Disinfectnt, Chloroxidant, Hydrogen Peroxide Ethyl and Isopropyl Alcohols, Iodine Tincture, Chloramine T
THE ANTISEPTICS
The most used active ingredients of the substances used as antiseptics on living tissues (intact, damaged skin and mucous membranes) are:
- Chlorhexidine,
- Electrolytic chloroxidant,
- Povidone iodine,
- Hydrogen peroxide
- Ethyl and isopropyl alcohols (hydrocarbon C2-C3 alcohol).
- Iodine tincture,
- Eosin,
- Chloramine T,
- Benzalkonium chloride,
- Triclosan and
- Silver.
The active ingredients contained in pharmaceutical and chemical products used to prevent the growth or cause the destruction of pathogenic and non-pathogenic bacteria, viruses, mycobacteria, fungi and spores are differentiated into three classes: antiseptics, disinfectants and sterilisers.
Antiseptics and disinfectants are active against some or all of the first four families of microorganisms, (Viruses, Bacteria, Fungi and Protist) while sterilants destroy all microorganisms, including bacterial spores [1-6].
Antiseptics are used only for skin and living tissues, such as damaged skin, intact skin (before medical and surgical interventions) and mucous membranes of humans and animals (antiseptics for veterinary use), to destroy or avoid the multiplication of almost all microorganisms pathogens except spores. A necessary requirement of the antiseptic is the absence of acute and chronic toxicity and irritant action on the organism on which it is used and to be active even in the presence of organic substances. Antiseptics for damaged skin and mucous membranes are registered as medicinal specialties. All antiseptics for intact skin in the past were considered medical-surgical devices, but since 2016 the Medicines Regulatory Agency (HMRA) has decided that even some products that treat intact skin, such as antiseptics for pre-surgery, must be considered medicinal specialities.
Disinfectants are used on objects, inanimate surfaces, the environment and healthcare equipment, are registered as medical devices and inhibit or kill almost all pathogenic microorganisms, but not spores.
Sterilants are used to disinfect medical and surgical equipment, are registered as medical-surgical devices and kill all forms of microbial life including spores.
In addition, almost all antiseptics have disinfectant capabilities, but at higher concentrations. Some antiseptics and disinfectants also have sterilizing properties (e.g. hydrogen peroxide and glutaraldehyde).
Finally, other products only have a sterilizing action (e.g. ethylene oxide). The products are also classified as bactericidal when they are capable of killing microorganisms or bacteriostatic when they are only able to prevent the development of bacteria and, therefore, inhibit their multiplication.
Antiseptics
The active ingredients of antiseptics are formulated (i.e. they contain other substances in addition to the active ingredients) and have the property of blocking the reproduction or killing of pathogenic microorganisms present on living surfaces to prevent septicemia, which can lead to death [7-15]. Most antiseptics are used in hospitals and those for intact skin may be called biocides. They are used to avoid infections, putrefaction and sepsis in the antisepsis of damaged skin, in particular in the treatment of traumatic and non-traumatic wounds, burns, scalds, abrasions and abrasions; in the antisepsis of intact skin, in particular in the antiseptic and surgical hand washing of healthcare workers; in the pre-operative antisepsis of patients, before intramuscular or intravenous injection therapies, venous and arterial sampling, for the positioning of central or peripheral cannulae; for the antisepsis of mucous membranes (which are a stratified structure that covers the internal surface of the cavities and channels of the organism communicating with the outside), in particular in nasal, vaginal, urethral, oral, respiratory and visual disinfections and auricular, in the preparation of the mucous membranes before surgical interventions and invasive tests, dressing of the umbilical cord, in the insertion of venous and bladder catheters, in the antisepsis of the perineum, in the pre-operative antisepsis of the cesarean section and of the vagina (antisepsis of the new mother).
The most commonly used antiseptics currently are chlorhexidine, electrolytic chloroxidant, povidone iodine, hydrocarbon C2-C3 alcohols and hydrogen peroxide. Other antiseptics, now little used, are iodine tincture, benzalkonium chloride, chloramine T, eosin, boric acid, triclosan and silver. The areas of use of individual antiseptics depend on their chemical nature, but also on their concentration and the solvent used, generally water or alcohols, where the latter enhance the biocidal action. These antiseptics can be liquids, soapy substances and even wipes.
Chlorhexidine
The chlorhexidine as active principle is a biguanide cationica lipophilic, used salified with gluconic acid (for this reason it is called chlorhexidine gluconate) to make it soluble in water or alcohols. It is active against gram positive and gram negative bacteria, on lipophilic viruses, moderately against fungi, and slightly on mycobacteria [7,8,12].
Its antimicrobial activity is due to the ability to disorganize the protein structure of the bacterial cell membrane, as the positive charges of chlorhexidine gluconate react with the negative charges of the bacterial cells, altering them. Chlorhexdine at low concentration (0.5-1%) is bacteriostatic, while at high concentration (>2%) it is bactericidal, and is used in various antiseptic activities with the following compositions: on intact skin at 0.5% in 70% 0.015% chlorhexidine gluconate + 0.15% cetrimide (cetyl-trimethylamonium bromide) in aqueous solution for the following antisepsis: damaged skin, mucous membranes, pre- and post-operative washing, bladder catheterization, external antisepsis in urology, gynecology and obstetrics, before insertion of catheters and for external cleaning of the genitals.
Some products on the market are Neoxina, Hibiscrub, and concentrated Baxidin and concentrated Farvickett. Chlorhexidine gluconate is also used as a disinfectant and sterilizer for medical equipment.
Electrolytic chloroxidizer
The electrolytic chloroxidizer is a 1.1% sodium hypochlorite in denatured water, obtained by partial electrolysis of NaCl. It presents greater purity, stability and histocompatibility (as it operates at neutral pH), compared to sodium hypochlorite obtained by dilution of an industrial hypochlorite (used as a disinfectant) [7,8,12]. This antiseptic works by decomposing into NaCl and oxygen radical which oxidizes the sulphide groups of the enzymatic systems, necessary for the metabolism of bacterial cells, making it effective against gram positive and gram negative bacteria, mycobacteria, viruses and, to a lesser extent, against fungi, and it is also active on spores at high concentrations.
The synthesis of sodium hypochlorite as an electrochemical disinfectant was patented in 1923 by Oronzio De Nora, an electrical engineer, who was awarded an honorary degree in Industrial Chemistry in Bologna in 1989.
Electrolytic chloroxidant is used in the following compositions: with a concentration of 0.05% for disinfection and cleaning of damaged skin and mucous membranes (oral, genital, bladder catheterization, etc.), for disinfection of external genitalia and nipples in breastfeeding; with a concentration of 0.1% in the treatment of all antisepsis of intact skin. As a disinfectant of surfaces, medical devices and water it is used in a concentration of 1.1%, while at concentrations lower than 0.015% it is used for the antiseptic washing of fruit and vegetables. Some products on the market are DECS cute and Amukine Med.
Povidone iodine
Povidone iodine is a soluble complex of iodine with an organic molecule at high molecular weight organic molecule (a neutral polymer of polyvinyl pyrrolidone), which functions as an iodine transporter [7,8,12]. It replaces the old iodine tincture, being less toxic, it is soluble in water, releases iodine gradually and, being a surfactant, penetrates more easily into the cell walls of microorganisms. The antiseptic action is due to the oxidizing action of free iodine which blocks the N-H groups of amino acids, subverting the protein structures, oxidizes the S-H groups of proteins and breaks the S-S disulfide bridges. Kills gram positive and gram negative bacteria, viruses, mycobacteria, and fungi. In 10% solution it contains 1% I2. It is used in all the different antiseptic activities, with the exception of traumatic wounds, with the following compositions: in a concentration of 5-10% in water in the cleaning and disinfection of damaged skin (abrasions, small cuts, superficial wounds and burns), in the antisepsis of the mucous membranes and in many uses on intact skin; in a concentration of 7.5% in water (soap solution) for antiseptic and surgical hand washing; 1% in ethyl and isopropyl alcohol (>60%) for the pre-operative preparation of intact skin; with a concentration of 10% in ethyl or isopropyl alcohol for antisepsis of intact skin, for intramuscular and intravenous injection therapy, for venous sampling and chemistry arterial diseases, preparation for biopsy and exploratory puncture (even in water); 10% in 85% polyethylene glycol for bedsores, small wounds, cuts, abrasions and superficial burns; in a concentration of 5% against fungal or bacterial infections.
Some products on the market are Povidone Scrub, Povidone Iodo Ointment, Betadine and Braunol with different composition. Povidone iodine is also used as a surface disinfectant particularly in the food industry.
Hydrocarbon C2-C3 alcohols
Ethyl alcohol, isopropyl alcohol and propyl alcohol (not in Europe) are used, even in mixtures with each other, with a concentration in water of 60 to 80%. The concentration of ethyl alcohol is generally 70%, while 75% was recommended for coronavirus disinfection, the one used for isopropyl alcohol is 60%, because it is more powerful [8,12]. The antibacterial action of alcohols derives from their ability to denature the proteins of microorganism cells, eliminating gram positive and negative bacteria, mycobacteria, many fungi and inactivating lipophilic viruses. Alcohols alone are used only for antisepsis of intact skin and, above all, for the antiseptic washing of hands at 70-75%, of the oral cavity, lips at 70% and for the treatment of the umbilical stump and skin before injections with a concentration of 80%.
For all other uses for intact skin, mixtures of alcohols with other antiseptics are preferred, such as chlorhexidine, povidone iodine and quaternary ammonium salts, of which they enhance the bactericidal and bacteriostatic action. Alcohols are not used in the antisepsis of damaged skin and mucous membranes due to their irritating and dehydrating action, as well as the possible formation of clots which facilitate the proliferation of germs. Alcohols are used as surface disinfectants.
Some products on the market are: Esosan and Sanigel for hand washing, containing 75% ethyl alcohol, 80% Disinfect Puravir Gel for the skin before injections and STI 70% alcoholic spray solution for disinfection of the oral cavity and of the mouth. The WHO (World Healt Organization) recently provided indications for obtaining a "do-it-yourself" disinfectant liquid, containing alcohol, hydrogen peroxide, glycerin and distilled water.
Hydrogen peroxide
H2O2 is very active only against gram positive bacteria against gram positive and negative bacteria, and less against mycobacterial and fungal viruses and is more used as a surface disinfectant and as a sterilizer than as an antiseptic [7,8,12]. It is used as an antiseptic in a concentration of 3% by weight (10% by volume of oxygen released) in aqueous solution and acts by developing OH- (reaction catalyzed by the Fe2+ and Cu2+ ions present in bacteria), which eliminates microorganisms and produces oxygen bubbles that clean contaminated surfaces.
It is not very active on living tissues, but has an effective mechanical cleansing with the removal of small debris and necrotic tissues in damaged skin: for this reason it is used to disinfect ulcers, dirty wounds, small wounds, abrasions and wounds believed to be infected with tetanus spores. However, its disinfectant action is short, because it is a volatile substance, therefore it is necessary, after its use, to proceed with other antiseptics. It can be used as a mouthwash for the effective cleansing of the oral cavity in a concentration of 1.5%, to lighten the teeth, for patients suffering from gingivitis and cold sores. It is also used as a sterilizer and disinfectant in some medical devices (contact lenses, etc.).
Less used antiseptics
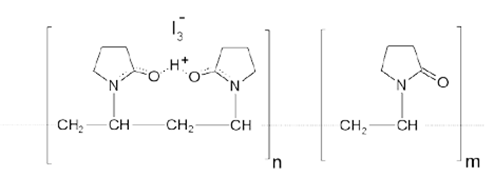
Iodine tincture is a mixture of iodine and potassium iodide in water and is used as a disinfectant of intact skin and oral with composition of 7% I2 and 5% KI in 47% ethanol in water, while it is used with a composition of 2% I2 and 2.4% KI, always in ethanol and water, for the disinfection of the oral cavity [16]. Iodine tincture releases iodine following the reaction: I2 + I- → I-3.
Benzalkonium chloride is a mixture of alkyl-benzyl-dimethylammonium chlorides, with the alkyl group ranging from octyl to octadecyl [17]. It is able to react rapidly with negatively charged surfaces, such as bacterial membranes, intercalating with the membranes themselves, altering their normal structure and leading to their destruction. It is active against gram positive and gram negative bacteria and some fungi. It is used in the following compositions: in a 0.1% concentration in water for the antisepsis of damaged skin (wounds, abrasions, abrasions) and mucous membranes; at 0.1-0.2% in alcohol for intact skin (disinfection and cleansing of patients' hands and skin even before surgery) and for external cleaning and antisepsis in obstetrics and gynaecology; at 0.005% for eye rinses, oral cavity rinses and vaginal irrigations. Some products on the market are Citrosil, Benalcon, LH antiseptic disinfectant and Iridina light
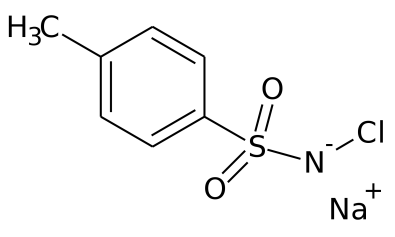
Chloramine T (sodium toluen-p-sulfonchloramide) is the active ingredient of, for example, Euchlorin (which is the name of the antiseptic) and is active against gram positive and negative bacteria and also against some fungi. Its action is predominantly bacteriostatic and, in part, bactericidal, and is due to the oxidizing activity of sodium hypochlorite which is released following the interaction with the sulphide residues of amino acids, altering the functionality of the proteins of pathogenic microorganisms [18]. Chloramine T is used in concentrations from 1 to 2.5% for the following uses: for the disinfection of intact skin before operating procedures, in the dressing of wounds, abrasions, infected sores, abscesses, boils, whitlows, vesicles and skin ulcers and disinfection of mucous membranes.
Eosin (tetrabromofluorescein) is a bacteriostatic and exerts its activity essentially on gram positive and negative bacteria and on fungi and appears to act by interacting with some ion channels of the cell membrane of microorganisms [7]. 2% eosin in aqueous solution is used in the following antiseptic activities: as a healing agent on damaged skin only on small wounds, burns, insect bites, diaper rashes, bedsores and sores; on intact skin on the intergluteal and inguinal area of infants and in the inframammary folds; in the antisepsis of pseudo mucous membranes, such as lips and genitals. Some products on the market are: Novoltre and Eosin NA 2%.
Boric acid (H3BO3) is used in concentrations of 3%, as an antiseptic in the disinfection of irritated or chapped skin, mild sunburn and in the disinfection of minor burns, insect bites and in the treatment of acne [19]. Its function consists in denaturing the intracellular and membrane proteins of pathogenic microorganisms, however it does not possess bactericidal activity, but only bacteriostatic. Boric acid is also used as an eye and ear bath, but diluted. Boric acid is present on the market for example as Boric acid Sella cutaneous solution.
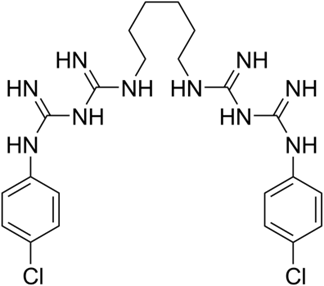
Triclosan is a chlorinated phenol that works by blocking an enzyme necessary for the growth of bacteria [20]. It was used in the following antisepsis: with a concentration of 0.2-2% for intact skin, in particular for antiseptic and surgical hand washing and pre-perative skin disinfection at 1%; with a concentration of 0.1% for the antisepsis of mucous membranes and damaged skin; with a concentration of 0.005% for vaginal irrigations. Triclosan was used for patients who had cases of intolerance towards chlorhexidine and povidone-iodine and was used largely in cosmetics and detergents at 0.3%. Since 2016, the European Commission has banned its use due to its toxicity for humans and the environment, being a POP substance. An example of products on the market that contained it is Citrosil and Irgasan.
Silver, like Ag+, is active against bacteria, fungi and viruses. It is relatively inert, but its interaction with the humidity of the lesions leads to the release of silver ions which bind to the DNA or RNA of the microorganisms, or alter the structure of their walls, blocking their multiplication. In particular, it is the environment of the skin lesion that determines the quantity of active Ag+. Silver is used in the form of protein silver, (bound to organic substances, from 0.5 to 2%) [21] and colloidal silver (silver ions in double-distilled water with Ag particles measuring from 1 to 500 nm , from 5 to 40 ppm) [22], used as antiseptics for burns and wounds, of the oral cavity, nasal, ear, gingival, rectal, vaginal disinfectants and for hands.
The sterilant
Another class of active ingredients are the sterilants products used to destroy not only all microorganisms, but also viruses, fungi and bacterial spores present on inanimate surfaces. Their chemical formulation kill all pathogenic and non-pathogenic microbial forms that contaminate only inanimate surfaces, such as: medical and surgical equipment, injectable drugs, support materials for carrying out aseptic procedures, some foods to be preserved, environments hospital and civil, culture media for bacteriology.
Sterilization determines the denaturation of proteins, nucleic acids and the degradation of the components of the membranes and cell walls of microorganisms. Sterilization is the procedure that gives the highest statistical probability of obtaining sterile material and, in fact, after the use of a sterilant the probability of finding a microorganism inside a sterilized material must be less than 1 in a million [23-27].
Disinfectants, however, not only do not kill spores, but may not kill all the microorganisms present on inanimate surfaces and are divided into high, medium and low level disinfectants, depending on the type of microorganisms destroyed [28]. The medical devices and surgical instruments subject to sterilization procedures, according to the historic Spaulding classification of 1968 [23] are defined as critical, semi-critical and non-critical instruments. Critical instruments (at high risk for patients) are instruments that come into contact with normally sterile tissues or with damaged skin or mucous membranes (which penetrate tissues and the vascular system, for example surgical and dental instruments, vascular catheters, needles, etc.) and must be subjected to a sterilization process before use. Semi-critical (medium risk) instruments are those that come into direct or indirect contact with intact mucous membranes (for example fiber optic endoscopes, vaginal probes, urinary catheters, assisted breathing equipment and laryngoscopes) must be subjected to sterilization, but if this is not possible, they can be subjected to high-level disinfection. Non-critical (low risk) instruments are those that only come into contact with intact skin or that do not come into contact with the patient and are only subjected to medium and low level disinfection.
Environments are also defined as having a high, medium and low risk of causing infections and the high-risk environments that must certainly be subjected to sterilization are clinics and hospitals, food industries and retirement homes for the elderly and scientific investigation environments [28].
Sterilization is carried out using physical, chemical-physical and chemical methods. Physical sterilants are: heat (dry or humid at T>100 °C), radiation (gamma rays, UV, X and microwaves), filtration (for liquid and gaseous substances).
Chemical-physical sterilants are gases or vapors used together with radiation and chemical sterilants are liquid or gas-phase substances.
The active ingredients of chemical and chemical-physical sterilants. All sterilants, with the exception of ethylene oxide, are also used as disinfectants at different levels, at lower concentrations and times of use and, therefore, also used against COVID 19.
Chemical and chemical-physical sterilization is used for instruments or substrates that cannot be brought to high temperatures (in particular plastic products) or which cannot be subjected to radiation; furthermore, chemical sterilization is generally also used to sterilize environments, in this case we often talk about sanitisation. Sterilization is involved in decontamination processes where there is first cleansing followed by sterilization or disinfection.
Chemical sterilization in gas phase
Gas phase sterilization is carried out with gas, such as ethylene oxide or ozone, or with vapors, such as hydrogen peroxide and, to a small extent, peracetic acid on medical devices and surgical equipment within autoclave-like apparatus. Furthermore, H2O2 and O3 are used for the sterilization of closed environments. Gas phase sterilization has the advantage not only of operating at low temperatures, but of acting with the instrumentation inside packages, where the gas migrates through the walls of the breathable packaging.
Sterilization with ethylene oxide
Ethylene oxide (ETO) acts as a sterilizer by alkylation, i.e. by substitution of a hydrogen atom with an alkyl group of the sulfhydryl, amino, carboxyl, phenolic and hydroxyl groups of the spores and vegetative cells, destroying them [29,30]. ETO is always used inside equipment at a 10% concentration in the presence of 90% HCFC (hydrochlorofluorocarbons), or CO2 or N2, to avoid explosions and fires, according to action times of 3 to 6 hours and temperatures between 50 and 60 °C in the presence of humidity (water participates in the alkylation reaction and favors the penetration of ETO through non-polar polymer films). ETO sterilization is used for medical-surgical equipment and pharmaceutical products. The disadvantages of the use of ETO are flammability and explosiveness, the acute toxicity towards the personnel involved (and for these reasons it is a technology used only in hospitals), and also the duration of the sterilization process, especially for the post-treatment of ventilation necessary to eliminate ETO residues.
Sterilization with H2O2 vapors
Sterilization is carried out in the gas phase, but with H2O2 vapours, i.e. with aerosols of 0.3-0.5 μm, starting from a 30-40% solution of H2O, at a temperature between 40-60 °C, for one or two hours: H2O2 vapors form •OH radicals. Which, by depositing on surfaces, destroy microorganisms and spores [31-33]. Sterilization is carried out not only in vacuum autoclaves on medical devices and surgical equipment, but also for the treatment of environments hospital and civil (aircraft and sealed rooms) for H2O2 nebulization. H2O2 vapors cannot be used on materials that can adsorb them (liquids and powders) and cellulose, while the positive aspects of their use are that at the end of the sterilization process only water and oxygen are formed. H2O2 has more limited penetration into packaging than ETO. There is also the sterilant "Silver Clean" [34] on the market, a liquid product based on hydrogen peroxide and silver ions, used for nebulization to sanitize environments where the presence of silver ions has antimicrobial properties synergistic and catalyzing with hydrogen peroxide in the destruction of viruses, bacteria and spores.
Sterilization with ozone
Ozone is used as a sterilizer because it produces atomic oxygen which destroys microorganisms and spores, but, as it can also alter medical and surgical devices, it is more used in closed environments [35]. Ozone, being a toxic and unstable gas, is produced in situ with electrical discharges starting from the air. In Italy it is more used in industrial environments to sterilize water and air and as a disinfectant for surfaces; we essentially talk about environmental sanitization with ozone [36,37], for 30-90 minutes, while abroad the word sterilization with ozone is used more [38].
Sterilization with peracetic acid vapours
Peracetic acid in the vapor phase is used for the treatment of materials that are not compatible with peracetic acid in the liquid phase, such as copper compounds. It also has the advantage of working at a lower temperature (18 and 30°C) compared to ETO (40°C) and H2O2 (50°C), with shorter sterilization times. In Italy it doesn't seem to be used, while it is abroad. For example, in the United States there is REVOX technology [39] for the nebulization of peracetic acid produced in situ with acetic acid and H2O2 in the presence of a catalyst used inside equipment to sterilize medical instruments, pharmaceutical and industrial products.
Chemical-physical sterilization
Chemical-physical sterilization is carried out in closed equipment to sterilize medical devices, surgical instruments and materials usable by patients essentially with H2O2 or even to a lesser extent with ozone, together with radiation or electric fields and/or ultrasound. The advantage is that the materials can be used immediately after sterilization and the sterilization process occurs with shorter times than with the use of gases and vapors alone.
H2O2 vapors with plasma gas
Plasma gas sterilization occurs with H2O2 vapors in the presence of electric or magnetic fields and ionizing radiation (largely UV radiation) or microwaves with the formation of a cloud (which constitutes plasma gas, the fourth sector of matter) of electrons, free radical ions, positive and negative ions which essentially kill microorganisms and spores by oxidation at temperatures between 35-50 °C (in shorter times than with H2O2 vapors) [40,41]. This technology, in addition to having the advantage over ETO of leaving no residues other than oxygen and water at the end of sterilization, operates at low temperature and low humidity and in the absence of pressure, with low contact times and there are no risks for the staff and for the environment. It is not corrosive to materials, in fact it is possible to sterilize metals, plastics and glass, but it is not suitable for porous materials, textiles, cellulose, powders, paper and liquids; It has the drawback of having poorer penetrability into packaging compared to ETO and can be used for small-sized equipment.
UV-ultrasound-ozone sterilizer
The UV-ultrasonic-ozone sterilizer [42] allows you to obtain optimal cleaning results using a triple combined technology to be used in medical and dental practices for the sterilization of the tools used, scissors, nail clippers, stethoscopes, disposable gloves, but also for environments doctors and civilians.
Sterilization in liquid phase
Liquid sterilants are also called cold sterilants, they act by immersing medical devices and surgical instruments in containers that contain them and are used for instruments that cannot withstand high temperatures (e.g. bronchoscopes, hemodialysis endoscopes, surgical instruments, etc.), but which are not altered by liquids. The liquid sterilants used are glutaraldehyde, peracetic acid and hydrogen peroxide (which is used together with peracetic acid) and in the past orthophthaldehyde was also used, which is now considered only a high-level disinfectant, and formaldehyde which has been eliminated because carcinogenic.
Glutaraldehyde
Glutaraldehyde (glutaric aldehyde) is active as a sterilizer due to the presence of the two terminal carbonyl groups capable of alkylating amino, hydroxyl and sulfhydryl groups of the proteins of microorganisms and spores, destroying them [43-44]. Glutaraldehyde is used at 2% in aqueous solution at 25°C for the following activities: as a sterilizer for 10 hours, as a high level disinfectant for 20 minutes and intermediate disinfectant for 10 minutes, as a sterile preservative for medical instruments at 0.4 %. The positive aspects of glutaraldehyde are that it is not corrosive to metals, operates at low temperatures and can be used for plastics, rubbers and instruments with lenses. The negative aspects are its toxicity for the personnel involved and the long sterilization times, also due to the long and careful rinsing times of the sterilized instruments to eliminate residues [45]. Acidic glutaraldehyde is only active in the destruction of bacteria, but not in that of spores, however it is stable over time and can be preserved for a long time; basic glutaraldehyde, on the other hand, is also active in the destruction of spores, but is not stable, in fact, after 15 days it is deactivated by polymerizing (due to the aldehyde groups) and for this reason it must be synthesized in situ. Glutaraldehyde becomes sporicidal, therefore sterilizing, in basic form (pH 7.5-8) and for this reason it must be previously activated by adding sodium bicarbonate or monobasic potassium phosphate, or mixing it with other bactericides, such as mixtures of phenols and phenates.
Sterilization with peracetic acid
Peracetic acid is active as a sterilant because it denatures the proteins and DNA of microorganisms and spores through oxidation. Peracetic acid in concentrations from 0.2 to 0.35% is used in the treatment of medical-surgical devices, especially for endoscopic devices where sterilization is required between one use and another and for those used in the dental field and intended for contact with the mucous membranes of the oral cavity; however, it is not compatible with copper and corresponding alloys (brass and bronze), aluminum and natural rubber [46]. Furthermore, the degradation products are not toxic and dissolve easily by washing the devices with sterile water. Peracetic acid is unstable and decomposes into acetic acid and H2O2, which then degrades into water and oxygen, and for this reason it must be synthesized in situ, generally by mixing sodium percarbonate powder or, to a lesser extent, sodium perborate, which releases H2O2 in aqueous solution, and tetraacetylethylenediamine (TAED) powder which releases acetyl groups in water. To a minimal extent it is also produced in situ from solutions of H2O2 and donors of N- or O-acetyl groups or from acetic acid and hydrogen peroxide in the presence of a catalyst or peracetic acid with H2O2 and acetic acid. Most peracetic acid is sold in powder form when it is to be used with the first reagents indicated in order to allow the composition suitable for the type of application to be prepared in situ; sold in aqueous solution only when used as a sterilant.
The application times are different depending on the composition of the product, which is why only the data for two industrial products will be reported.
[47] is a powder made up of sodium percarbonate and tetraacetylenediamine which, dissolved in water, releases 2% peracetic acid at 35°C (forming 0.26% peracetic acid) and is sterilizing for 10 minutes while at 1% is high level disinfectant for 5 minutes.
HY Care Cetic 2.0 [48] is just a liquid sterilant obtained by adding a solution of H2O2 o a solution of an O-acetyl donor to obtain 0.2% peracetic acid, making it active for 15 minutes.
The disinfectants
High-level disinfectants used for the elimination of pathogenic microorganisms that contaminate inanimate surfaces, medical and surgical equipment and healthcare environments, whether hospital or extra-hospital. Substances used on damaged or intact skin are also commonly called antiseptic [49] and sterlant [50], but these are officially defined as "antiseptics" and treated previously on Chapter 1 & 2.
Disinfectants lead to the elimination of pathogens through physical mechanisms of action, for example, through heat or radiation, or chemical ones, with active ingredients capable of degrading the structure or inhibiting the functionality of the unwanted microorganism.
Disinfection can be classified into three levels: high, medium and low level [51,52].
- High-level disinfection consists in the destruction of all microorganisms, with the exception of spores [53]: Gram positive and Gram negative bacteria, all viruses, fungi and mycobacteria. Disinfectants of this type are used on semi-critical instruments (according to the Spaulding classification) [54,55], i.e. those devices and devices that are used for intact mucous membranes or for non-intact skin, but which do not come into direct contact with internal tissues or with blood.
- Medium-level disinfection consists in the inactivation of bacteria in vegetative form, mycobacteria, fungi and most viruses, with the exclusion of non-lipid ones. Disinfectants of this type are used for non-critical instruments, [56] which do not come into contact with mucosa and/or intact skin.
- Low-level disinfection consists in the inactivation of only vegetative bacteria, some fungi and some viruses (those with pericaspis, commonly called coated), but not tuberculosis mycobacteria (tuberculosis bacteria). Such disinfection is used on non-critical instruments.
In critical and semi-critical hospital environments, in which an immuno-compromised patient is present, even for a short time, or in which high-risk invasive maneuvers are performed, high-level disinfectants are used to ensure the inactivation of pathogenic bacteria in medical devices. The level of effectiveness of disinfectants depends on the chemical nature, the concentration of use, the contact times and the co-presence of organic material, which for some disinfectants leads to a decrease in effectiveness. Furthermore, their use it also depends on their ability not to alter the materials subjected to disinfection and not to cause harm to people, in terms of acute toxicity or generating harmful by-products in the short or long term.
Many high-level disinfectants contain active ingredients completely similar to those of sterilants (e.g. glutaraldehyde, peracetic acid, hydrogen peroxide or ozone) [50], however using reduced contact times and/or lower concentrations. These products also contain some active ingredients that do not have sterilizing properties, such as sodium hypochlorite or ortho-phthalaldehyde and synergistic mixtures of active ingredients of intermediate and low level disinfectants. This note reviews the main active ingredients used in high-level disinfectants on the market [56].
Glutaraldehyde (see Chapt 2)
Glutaraldehyde (glutaricdehyde [43-44] is used at a concentration of 2% (w/w) together with an activator, as a high-level disinfectant for 10 hours or, at lower times, as an intermediate-level disinfectant, immersing medical devices and surgical equipment in its solutions. Glutaraldehyde is toxic to humans and its action is compatible with all materials, even in the presence of blood and proteins; it is used for the disinfection of endoscopes and semi-critical medical devices. Below are two types of high-level disinfectants that contain glutarldehyde.
The main formulation of this type of disinfectant contains a 2% w/w aqueous solution of glutaraldehyde with the addition, before use, of co-formulants, such as non-ionic surfactants, or of pH regulating activators, such as monobasic potassium phosphate (2%), sodium hydroxide or sodium hydrogen carbonate, to bring the pH to 7.5-8 at which glutaraldehyde exhibits its maximum activity. This type of disinfectant is usually used for: high-level disinfection, with a contact time of 10 minutes, at room temperature; for the disinfection of invasive and non-invasive medical devices, in particular those for thermosensitive and non-autoclavable endoscopes (laparoscopes and arthroscopes and other fiber optic instruments used for diagnostic purposes); but it is also used for intermediate-level disinfection of medical-surgical devices, fiber optic equipment used for therapeutic purposes and face masks, breathing tubes and other respiratory therapy equipment. Typically these formulations are also used for the disinfection before sterilization of surgical instruments, diagnostic devices, including thermolabile instruments with cemented optical parts (endoscopes, etc.), dialysis plates and devices, objects made of rubber, glass, plastic and stainless steel, such as endotracheal tubes, probes, fittings, cannulae, and aspirator tubes. At lower concentrations (approx. 0.4% w/w) glutaraldehyde can be used for the aseptic conservation of previously sterilized instruments.
Ortho-phthalaldehyde
Ortho-phthalaldehyde is active only as a high-level disinfectant and is used for surgical equipment and medical devices, as it destroys the proteins of microorganisms, while in the past it was also used as a sterilizing agent. This active ingredient operates at the same neutral pH as glutaraldehyde, but compared to the latter it is less toxic as it is less volatile; furthermore, it does not require activation and operates quickly minor contact, as well as being compatible with all the materials present in the various devices used in hospitals. It should be remembered, however, that due to its potential irritant, sensitizing or allergenic power, it cannot be used to treat instruments that come into contact with patients sensitive to ortho-phthalaldehyde.
Typical formulations containing this aldehyde have a concentration of 0.55% w/w in water and are usually used for 5 minutes of contact, at room temperature for the reprocessing and disinfection of reusable medical instruments, especially fiber equipment optics, used for diagnostic purposes in various healthcare facilities, such as cystoscopes, digestive endoscopy equipment, flexible bronchoscopes or other types of probes.
Peracetic acid
Peroxoacetic (peracetic) acid is normally used as a sterilant, however it can also be used as a high level disinfectant at lower concentrations; in this case, it finds application for surgical equipment and invasive and non-invasive medical devices. Peracetic acid is unstable and must therefore be synthesized in situ, starting from precursors in powder form in order to have the right composition for the different applications. However, it cannot be used on devices based on aluminium, copper and corresponding alloys (brass, bronze, etc.), but it has the advantage of not being toxic to humans at concentrations lower than 2%.
The products on the market are generally made up of a mixture of sodium percarbonate, tetracetylenediamine, citric acid and chelating agents. Once dissolved in water at 35°C, the formulation releases peracetic acid (2 g of powder releases 0.26 g of peracetic acid) which can be used at 2% as a sterilizer with a contact time of 10 minutes, while with dilution at 1%, as a high-level disinfectant with a contact time of 5 minutes for the disinfection of medical-surgical devices, including heat-sensitive endoscopes, in hospitals, medical and especially dental clinics intended for contact with the mucous membranes of the cable oral (such as mirrors, probes, etc.). Such mixtures for in situ preparation of peracetic acid are also an excellent disinfectant for face masks, breathing tubes and other equipment. For respiratory therapy they are usually preferred to other disinfectants described previously. In fact, rinsing with sterile water is sufficient to eliminate the risk of irritation to the respiratory tract due to residues of the active ingredient.
Sodium hypochlorite
Sodium hypochlorite is contained in bleach (bleach used as a laundry detergent) in a concentration of around 5% or is produced in situ by hydrolysis of electrolytic chlorine or by chloroamine (sodium tosylchloroamide) at a concentration of 1% which produces active chlorine (HClO) in water.
HClO decomposes into HCl and atomic O which acts as a disinfectant by oxidizing the sulfhydryl groups of the enzymes that regulate cellular metabolism. Sodium hypochlorite must be used as a disinfectant in the absence of organic substances and the following materials: low-alloy steel, polyurethanes, iron and metals in general. The hypochlorite obtained from bleach is essentially used to disinfect environments and for low-level disinfection while the hypochlorite obtained from the electrochemical chloroxidizer is used for high-level and intermediate-level disinfection due to its higher degree of purity.
Below are some applications of hypochlorite at different concentrations.
As a widely used disinfectant, at a concentration of 2.7% of active chlorine in water, it is used, after dilution, for the following applications:
- At 0.27% for containers for high-risk contaminated fluids with a contact time of 5 minutes;
- At 0.135% for disinfection of operating rooms and their hard surfaces, laboratories and equipment, clinics and emergency rooms.
With a concentration of 1.15% active chlorine it is used diluted to 0.055% for the disinfection of hard surfaces with a contact time of 5 minutes.
With a concentration of 0.55% of active chlorine it is used as a disinfectant for dialysis equipment; with a concentration of 0.1% for medical devices and equipment in the healthcare and laboratory fields, for medical devices for endoscopic use and all those articles of use in plastic, glass, steel, ceramic which are considered medical devices. On the market you can also find wipes soaked in 100 ml of disinfectant which contain 0.1% active chlorine and which are used for the disinfection of non-invasive medical devices, equipment and worktops of medical devices, medical instruments and items not sensitive to chlorine.
Hydrogen peroxide
Hydrogen peroxide is used as an antiseptic in the liquid phase, as a sterilant in the vapor phase and as a disinfectant at high levels and at lower levels, both in the vapor and liquid phases. Furthermore, it finds a wide range of applications, since, being less dangerous for humans and for the environment than other disinfectants, and it is also relatively less aggressive towards materials. Its sterilizing action occurs in 6 hours at a concentration of 6.0-7.5%.
Concentrated solutions of 60% H2O2 are used as a high-level disinfectant at concentrations around 6% of H2O2, and are used: for surfaces and medical devices, equipment in the operating block, laboratories, clinics, particularly for clean rooms, filters, aspirators, small medical devices and surgical devices, stainless steel products, laboratory machines and worktops, vertical and horizontal walls, porous walls, dental practices and industrial plants, machinery in the pharmaceutical sector.
There are also broad-spectrum disinfectant solutions on the market, which, consisting of a mixture of 5% w/w hydrogen peroxide and 9% w/w ethanol, are sprayed on the surfaces or objects to be disinfected.
The biocidal activity of this product is given both by the presence of ethanol, which accelerates contact with the surface of microorganisms, and by the oxidizing properties of hydrogen peroxide. The mixture is suitable for disinfection, which is rapid (in the order of 10 minutes), effective and persistent over time with simultaneous cleaning of surfaces, medical devices, invasive and non-invasive medical devices, non-immersible medical devices, surfaces in general of hospital departments , worktops, operating beds, other medical devices biomedical and dental equipment.
Ozone
Ozone generators are electronic equipment that allow the generation of ozone from atmospheric oxygen through electrical discharges to disinfect private homes, offices, gyms, medical and/or veterinary clinics, healthcare vehicles, etc. in a time of approximately 20 minutes. Ozone, being a toxic and unstable gas, must be produced in situ. Ozone in Italy is most used in industrial environments for the sterilization of water and air, as well as a disinfectant for surfaces. In the latter case we are talking about environmental sanitization with ozone, with the advantage of leaving no residues.
Mixtures of active ingredients
There are now also some types of formulations made up of active ingredients which alone are intermediate or low level disinfectants, but which combined together show a synergistic effect which increases the biocidal property of the individual ingredients, so that they can therefore be proposed as high level disinfectants [57].
An alcoholic solution based on 0.10% quaternary ammonium salt, 0.02% ortho-phenylphenol and isopropanol can be used in the form of a spray for 3-4 seconds, leaving it to act for 10 minutes to achieve adequate levels of disinfection. In this mode it is particularly effective in environments with high attendance and greater risk of infection, such as clinics, waiting rooms, school classrooms, canteens, sports centres, gyms, reception centres, and public toilets and in all those places where the passage of people can compromise the quality of hygiene.
A powder mixture composed of potassium peroxomonosulphate, benzenesulfonic acid and other components in smaller percentages acts as a high-level disinfectant by developing active oxygen in the water. This solution can be used for disinfections in the following sectors: public (gyms, kindergartens, schools, etc.); healthcare (hospital recovery rooms, operating rooms, etc.); industrial (transport, agri-food industries, etc.).
A mixture of powdered potassium peroxomonosulphate and povidone iodine can be actively used to carry out a high-level disinfectant activity on the surfaces of healthcare environments, community and elderly residences, and means of transport.
Capt. 4. Intermediate level of disinfectants
A number of active ingredients of intermediate-level disinfectants (sometimes defined as medium level) [58-59] but inactivate mycobacterium tuberculosis, all vegetative bacterial forms, most viruses (only lipid viruses) and fungi, but do not deactivate bacterial spores and they are used to disinfect medical devices, articles and non-critical surfaces, which carry little risk of transmitting infectious agents, which do not come into contact with patients or with mucous membranes, but only with the intact skin of humans and animals. These devices, items and non-critical surfaces can also be treated with low-level disinfectants in particular situations. Precisely, because theoretically they can be treated with low-level disinfectants, attention must also be paid to non-critical surfaces, such as hospital surfaces and (non-surgical) instruments, civil environments and surfaces contaminated by blood or biological liquids or infected for example by SARS-CoV -2 [60-62], because they could transmit infections. Furthermore, there is material that requires intermediate-level disinfection in gyms, bathrooms, toilet seats and showers, including work surfaces in non-agri-food companies. Finally, these disinfectants are used for the disinfection of sharp (non-surgical) instruments such as scissors, tweezers, knives and blades that require thorough disinfection. These disinfectants are used with the following procedures: by nebulization, which consists of a disinfectant dissolved in water and a nebulization device for spraying into the environment; for the production of aerosols, i.e. drops of 0.5-5 microns; for spraying for wetting floor surfaces and for disinfecting streets with a disinfectant dissolved in water and applied to the surfaces to be treated. Instead, medical instruments and devices, when it is possible, they are also disinfected by immersion in disinfectant liquid. There are three classes of intermediate-level disinfectants: those based on a single active ingredient (which are those mostly present in disinfectant reviews), those with low-level active ingredients dissolved in an alcoholic solution and those based on mixtures of ingredients mostly low-level active ingredients dissolved in aqueous solution, both of these last two classes of disinfectants are essentially present in the documentation of disinfectant manufacturing companies.
Intermediate level disinfectants with a single active ingredient
Intermediate level disinfectants, which contain only one active ingredient, are the following: hydrogen peroxide, peracetic acid, sodium hypochlorite, glutaraldehyde, all also used as high-level disinfectants; ethyl and/or isopropyl alcohol, sodium hypochlorite, electrolytic chloroxidant, phenols and polyphenols, iodophors, all also used as antiseptics.
Solutions of 73 to 75% w/w of ethyl alcohol and isopropyl alcohol, in mixtures or even alone, are used [63] for the following disinfections: hospital ward surfaces, worktops, operating beds, clinical thermometers, facial masks, etc.
An aqueous solution based on H2O2 diluted to concentrations of 0.5% is used [64] for disinfection by wetting surfaces and floors, by nebulizing environments, by immersing objects, in particular also against SARS-CoV-2 (while at a higher concentration it is used as a high level sterilizer and disinfectant).
An aqueous solution containing glutarldehyde at 2% w/w is used [65] for disinfections of medical-surgical devices (face masks, breathing tubes and other respiratory therapy equipment) for 20 minutes (for high-level disinfection for 30 minutes).
A powder of 53% w/w sodium percarbonate and 23% w/w tetraacetylenediamine (TAED) in aqueous solution produces in situ peracetic acid which is the active ingredient (a 2% w/w solution develops 4400 ppm of acid peracetic), and is used [61] as a disinfectant for medical-surgical instruments and medical devices for 10 min. (while always diluted at 2% w/w for 30 min. it is used as a high level disinfectant).
An aqueous solution with 1.15% w/w sodium hypochlorite is used [62], by dilution in water for the following disinfections: of hard surfaces with 0.055% active chlorine for 5 minutes; of objects used in neonatal age at 0.022% active chlorine for 30 min.
An aqueous solution at 2.7% w/w of active chlorine is used [63] for dilution in water for the disinfection and cleansing of: containers of organic fluids and surfaces contaminated by them at 0.27% w/w of chlorine active; of hospital environments and surfaces, food industries, classrooms, toilets and shop windows at 0.135% w/w of active chlorine.
An aqueous solution containing a sodium salt of o-phenylphenol at 2.5% w/w is used [64] as a disinfectant and is used in all sectors of public health, where continuous and/or periodic environmental disinfection is required, diluted from 0.5 to 2% in particular of wall surfaces, toilets and their furnishings (tiles, sinks, tubs, cups), hospitals, clinics and clinics. Intermediate level disinfectants are used against SARS-COV-2 and, in particular, the most used are: sodium hypochlorite with concentrations from 0.1-0.5% w/w, H2O2 at 0.5% w/w, peracetic acid at 2% mp/w, ethyl and isopropyl alcohol at 70-75% w/w.
Intermediate level disinfectants based on alcoholic solutions of other active ingredients
Ethyl and/or isopropyl alcohols, which alone are intermediate level disinfectants, are also used in mixtures with other active ingredients for the same disinfection to make the product, following the presence of synergistic effects between them, a broad-based disinfectant. Spectrum of action, and furthermore alcohol facilitates the conveyance of the other active ingredients towards the specific target, improves their conservation and avoids rinsing at the end of disinfection. All these disinfectants are also defined as alcoholic solutions due to the quantities of alcohol present, and are used as intermediate level disinfectants in the healthcare sector for the treatment of non-critical and even some semi-critical medical devices and biomedical equipment, and of hospital ward surfaces and non-critical biomedical environments. The active ingredients, which are largely low-level disinfectants in aqueous solution if used alone, become intermediate-level disinfectants in alcoholic solution. Disinfection occurs by direct vaporization on the surfaces and then passing a coat and waiting at least 5 minutes before use or by indirect vaporization on a coat which is then passed over the surfaces to be treated. These disinfectants are used for the disinfection of medical devices (such as face masks, electrocardiogram electrodes and bedpans) and in dentistry and for the temporary sterile storage of surgical instruments.
Benzalkonium chloride (alkyldimethylbenzylammonochloride, with alkyls ranging from octyl to dioctyl), chlorhexidine (1,1'-hexamethylenebis[5-(p-chlorophenyl)biguanide]) and quaternary ammonium salts (R4NCl) are the principles most used active ingredients in alcoholic solution.
A solution based on 56% w/w ethyl alcohol, 0.3% w/w benzalkonium chloride is used [65] for the disinfection of equipment and surfaces in civil environments, in particular, company work surfaces agri-food and to disinfect non-surgical instruments.
An alcoholic solution containing benzalkonium chloride 0.040% w/w, didecyldimethylammonium chloride 0.060% w/w, chlorhexidine digluconate (esterified with gluconic acid to make it soluble) 0.050% w/w, ethyl alcohol 31.5% w/w p and isopropyl alcohol at 31.5% w/w is used [66] for the following disinfections: in the healthcare sector for the treatment of: hospital ward surfaces, worktops, operating beds, touch screen monitors, operating lamps, armchairs in dentistry and for dialysis, keyboards and beds.
An alcoholic solution containing 0.5% w/w chlorhexidine digluconate and 70% w/w ethyl alcohol is used [67] in public and private healthcare activities for the following disinfections: for the decontamination of surgical instruments and medical devices in facilities healthcare before sterilization, for the disinfection of medical devices, in dentistry and for the temporary sterile conservation of surgical instruments.
An alcoholic solution containing didecyl-dimethylammonium chloride 0.1 g, o-phenyl-phenol 0.05 g and ethyl alcohol 72 g non-invasive hospital and operating room solutions; for the external disinfection of dialysis equipment and dental practices and the decontamination of medical devices, before their sterilization.
An alcoholic solution of benzalkonium chloride 10 g in 100 g of the mixture/100 g, which contains o-phenyl-phenol and isopropyl alcohol is used for the pre-sterilization of dental and hospital instruments [68].
An alcoholic solution based on benzalkonium chloride 0.3% w/w, chlorhexidine gluconate (19-21%) 0.05% w/w and ethyl alcohol 68% w/w is used [69] for the following disinfections: all the material, equipment and instruments, including sharp ones, which require careful disinfection.
An alcoholic solution containing benzalkonium chloride at 1 g per 100 g of the mixture, o-phenylphenol, isopropyl alcohol and ethyl alcohol is used [70] for the following disinfections: bio-medical equipment, hospital equipment, medical devices and dental and it is used by nebulizing for spray.
Intermediate level disinfectants with mixtures of active ingredients
The presence of multiple active ingredients increases the antimicrobial spectrum of disinfectants and practically the active ingredients which alone are low level disinfectants, become intermediate level, following the synergistic effect of their simultaneous presence.
An aqueous solution of 100 ml containing chlorhexidine digluconate 1.50 g and cetrimide (cetyl-trimethylammonium bromide) 15 g mixture which is also an antiseptic, is used [65] for the following disinfections in the healthcare and laboratory fields: decontamination of contaminated medical devices before of sterilization diluted at 2% w/w for 20 min.; disinfection of surfaces and hospital equipment diluted at 4% w/w for 15 minutes; disinfection of equipment in the operating room (trolleys, basins, beds, furniture, etc.) diluted 4% w/w; for the aseptic conservation of surgical instruments diluted at 4% w/w.
An aqueous emulsion based on p-tert-amylphenol 2.30% w/ An aqueous emulsion based on p-tert-amylphenol 2.30% w/w, o-phenylphenol 11.40% w/w and o-benzyl-p-chlorophenol 8.20% w/w is used [65] diluted at 0.5% w/w for 30 min. for the following disinfections: biomedical equipment and biomedical equipment (operating beds, dental chairs, dialysis chairs, anesthesia equipment (non-critical items), clinical thermometers, ECG monitors, peristaltic pumps, respirators, surgical lamps, etc. and for the decontamination of surgical instruments and medical devices.
An aqueous solution of 100 ml of o-benzyl-p-chlorophenol 5.25 g and o-phenylphenol 1 g is used [65] for the following disinfections: for decontamination of medical devices before cleaning and sterilization with conc. 3% w/w for 10 min.; for the disinfection of medical devices cleaned by immersion with conc. 3.0% w/w, for 5 min.; for the disinfection of suction circuits in dental laboratories dilution from 1.5-3% w/w for 10 min.
An aqueous solution containing didecyl-diethylammomium chloride 0.14% w/w and chlorhexidine gluconate 0.10% w/w is used [71] for the following disinfections: hospital wards surfaces (worktops), biomedical devices and equipment (instrumentation for dialysis, ventilators, counterpulsator.
An aqueous solution containing 1.5% w/w of H2O2 and 0.003% w/w of Ag is used [72] for nebulization for the disinfection of surfaces in the healthcare field, the Ag ions enhance the sanitizing action of the H2O2.
A gas containing hydrocarbons (50% propane and butane), with o-phenylphenol 0.10% w/w and N,N-dodecyl-N-methyl-poly(oxyethyl) ammonium propionate (quaternary ammonium salt) at 0.175 % w/w and isopropyl alcohol at 15-20% w/w is used [73] in the disinfection of ambulances, medical vehicles, public assistance areas, doctors' offices, clinics, dental offices, in the absence of the operator.
All chemical substances used as active ingredients for intermediate level disinfectants are also used as active ingredients for high level sterilants, antiseptics and disinfectants, but at higher concentrations and longer contact times and for low level disinfectants at lower concentrations and shorter contact times and not mixed with different active ingredients. Finally, it is good to remember that intermediate level disinfectants are those used for the prevention and fight against the SARS-CoV-2 coronavirus [60]. Matteo Guidotti, in an interview recommended using 70% ethyl alcohol, or 0.5% hydrogen peroxide, or sodium hypochlorite (common bleach), all substances available at home that they are intermediate level disinfectants [74]
Chemical products on the market of low-level disinfectants [75]
Low-level disinfectants kill most bacteria, some viruses and some fungi (fungi), but are unable to kill more resistant bacteria, such as tubercle bacilli or bacterial spores (Mycobacterium Tuberculosis). In the historic Spaulding classification [76] (year 1968) there are non-critical surfaces, equipment and items that come into contact with only intact skin that need low or intermediate level disinfection, so low level disinfectants can be alternatives to intermediate level disinfectants. In hospitals they should be used to treat instruments, where there is no need for thorough disinfection, in particular for instruments that do not come into contact with the patient or only come into contact with intact skin and not with the mucous membranes. In hospital environments they are present in environments where there are no patients. In the civilian field they are selected in areas with a greater risk of contagion such as hygiene environments and canteens. However, for civil areas at greater risk, intermediate disinfectants are alternatives. While for low risk areas detergents can be chosen as an alternative to low level disinfectants. These disinfectants are used with the following procedures: by nebulization, which consists of a disinfectant dissolved in water and a device; nebulization for spray in the environment; for the production of aerosols, i.e. drops of 0.5-5 microns; for spraying for wetting floor surfaces and for disinfecting streets with a disinfectant dissolved in water and applied to the surfaces to be treated. Instead the tools and medical devices, when possible, are also disinfected by immersion in disinfectant liquid.
Non-critical medical devices are: bedpans, trays, thermometers, parrots, face masks, electrocardiogram electrodes, etc. Non-critical items are: linens, mattresses, environmental surfaces that do not normally come into contact with mucous membranes or damaged skin. Non-critical hospital surfaces are: floors, walls, fixtures in all rooms. Non-critical social environments that may need disinfection may include: schools, boarding schools, hospices; hotels and other community centers; offices, shops, and industries. Below is the chemical nature of some active ingredients of low-level disinfectants marketed for use on medical devices, articles and non-critical social environments.
Low level disinfectants
In all the articles that deal with low-level disinfectants, quaternary ammonium salts are always mentioned, which are sometimes the only ones mentioned, then among the most cited are polyphenols and phenolic derivatives (some formulations), and to a lesser extent, the compounds that produce active chlorine, ethyl or propyl alcohols, chlorhexidine gluconate and glycine derivatives [77-81].
Compounds based on quaternary ammonium salts in aqueous solution
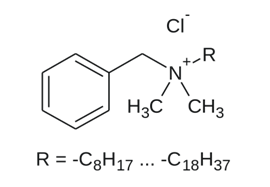
Compounds based on quaternary ammonium salts in aqueous solution, which are called disinfectant-detergents, are also cationic surfactants. These disinfectants should not be mixed with anionic surfactants, and in general with detergents, because they deactivate them and are chosen as low level disinfectants in concentrations between 0.1 and 1% for times of 10 minutes. The quaternary ammonium salts used in greater quantities are the following: Benzalkonium chloride (see adjacent formula) which is an aqueous solution of mixtures of alkyl dimethylbenzylammonium chlorides, with the alkyl group ranging from octyl (C8H17) to dioctyl (C18H37); Cetrimide or Benzoxonium chloride which is didecyldimethylammonium chloride. These disinfectants are ideal for ordinary cleaning of environments on non-critical surfaces, such as floors, furniture and walls.
Polyphenols and phenolic derivatives
These compounds, chosen as low-level disinfectants, are polyphenols and some phenolic derivatives, always used together with detergents (anionic surfactants) at 0.4%. Some more selected phenolic derivatives are the following: alkyl and aryl phenols (for example ortho-phenylphenol, (see formula alongside); polyphenol (o-phenylphenol+orthobenzyl-para-chlorophenol+p-tert-amylphenol); halogenated phenols (para-chloro-metacresol and orthobenzyl-para-chlorophenol). Phenols can be inactivated by organic materials and the disinfectant power is linked to the presence of the free hydroxyl group and is selected in concentrations of 0.4% for 10 minutes. These compounds can also be chosen as intermediate disinfectants at 1% for 30 minutes. These disinfectants are essentially chosen for the decontamination of non-critical metal surgical and medical instruments, for the cleansing of equipment surfaces, furnishings and healthcare devices, for beds and furnishings, thermometers and respirators.
Other low-level disinfectants
Chlorine derivatives that produce >100 <500 ppm (typically 200 ppm) of active chlorine for a contact time of 10 minutes are chosen as low-level disinfectants. A classic example of this family is sodium hypochlorite (bleach), other disinfectants are chloramines (NH2Cl, NCl3, NHCl2) and electrolytic chloroxidant (Amuchina 1.1%).
70% ethyl or isopropyl alcohol (or from 50 to 70%) are also used for 10 minutes, for the disinfection of surfaces (toilets and furnishings).
Chlorhexidine digluconate is also chosen, where Chlorhexidine (a cationic biguanide, see formula alongside) is linked to a mixture of glycols which, while keeping the active ingredient stable, do not negatively interfere with the biocidal activity, enhancing the virucidal one. Furthermore, 1.5% Chlorhexidine + 15% Cetrimide in aqueous solution is used for low-level cleansing and disinfection of metal equipment and furniture surfaces. Finally, glycine derivatives are chosen, which derive from amino acids present in human proteins (Glycine: NH2-CH2-COOH) in which one or two hydrogens of the amino group (NH2+) are replaced by alkyl radicals, these compounds behave such as quaternary ammonium compounds and are commonly used in the food, pharmaceutical and livestock industries, for the disinfection of walls, floors and small objects.
CONCLUSIONS
Low level disinfectants are practically those that have only one active ingredient and are in an aqueous solution, while intermediate level disinfectants are largely in alcoholic solution and/or are present in a mixture with different active ingredients. However, many of the chemicals chosen as active ingredients for low-level disinfectants are also used as active ingredients for high- and intermediate-level sterilants, antiseptics and disinfectants, but at higher concentrations and longer contact times.
REFERENCES
- Katzung BJ, Trevor AJ. Farmacologia Generale e Clinica. XII Edn. 50, 2017, Ed. Piccin.
- http://www.simpios.eu/wp-content/uploads/2017/12/CROSASSO.pdf
- https://innovareacademics.in/journals/index.php/ajpcr/article/view/21218
- https://www.my-personaltrainer.it/benessere/antisettici.html
- https://opbgfad.accmed.org/file.php/1/12FE0262_demo/ata/n/010620_prontuario_antisettici_disinfettanti.pdf
- https://www.aslteramo.it/wp-content/uploads/2018/05/Linee-Guida-sull%C2%B4uso-dei-disinfettanti.pdf
- www.aslcn2.it/media/2013/07/PRONTUARIO-disinfettanti-2015-rev-5.pdf
- https://www.anmdo.org/wp-content/uploads/2016/10/Linee-guida-per-il-corretto-utilizzo-degli-antisettici---disinfettanti.pdf
- www.aslal.it/allegati/Prontuario%202015.pdf
- internetsfn.aslrme.it/cio/pdf/Protocolli/110714%20prontuarioantisettici disinfettanti.pdf
- https://www.galliera.it/files/documenti/cio/pdf/prontuario.pdf
- http://www.ausl.vda.it/elementi/www/strategica/comunicazione/pubblicazioni/disinfettanti_2pagine.6
- https://www.farmaermann.it/farmaci-sop-e-otc/antisettici.html
- https://www.area-c54.it/public/antisettici%20e%20disinfettanti.pdf
- https://silvanomonarca.files.wordpress.com/2012/05/29-disinfezione.pdf
- https://www.torrinomedica.it/schede-farmaci/iodio_afom_7pc 5pc_soluzione_cutanea_alcoolica/
- https://www.codifa.it/farmaci/b/benalcon-benzalconio-cloruro-antisettici
- https://www.codifa.it/farmaci/e/euclorina-cloramina-antisettici
- https://www.torrinomedica.it/schedefarmaci/acido_borico_marco_viti_3pc_soluzione_cutanea/
- https://www.news-medical.net/health/What-is-Triclosan-and-Why-is-it-Banned-(Italian).aspx
- https://farmaci.agenziafarmaco.gov.it/aifa/servlet/PdfDownloadServlet?pdfFileName=footer_002812_031314_FI.pdf&retry=0&sys=m0b1l3
- https://www.scienzaeconoscenza.it/blog/medicina-non_convenzionale/che-cos-e-argento-colloidale-come-si-usa/
- https://m.my-personaltrainer.it/benessere/argento-colloidale.html
- http://www-3.unipv.it/scienzemotorie/wpcontent/uploads/2016/11/6660DISINFAZIONE_STERILIZZAZIONE.pdf
- http://www.demarco.biz/pub/media/productattach/m/a/manualedisterilizzazione_1.pdf
- Rutala WA, Weber DJ. American Journal of Infection, 2013, 41, 52.
- http://www.aiosterile.org/wordpress/documenti/APPROFONDIMENTI/LINEE_GUIDA.pdf
- http://www.simpios.eu/wp-content/uploads/2017/11/CHAPTER-12_pulizia.pdf
- https://www.naturalmania.it/sterilizzato-e-sanificare-gli-ambienti-in-maniera-ecologica/
- http://win.spazioinfo.com/public/STERILIZZAZIONE%20CON%20OSSIDO%20DI%20ETILENE%202007-2008.pdf
- https://www.steris-ast.com/it/services/ethylene-oxide/
- https://www.steris-ast.com/it/services/peroxide-di-hydrogen-vaporizzato/
- https://www.asccanews.it/itemlist/tag/material
- https://www.sysplorer.com/sanificazione-con-il-peroxide-di-hydrogen
- https://www.pserviceweb.com/home/it/disinfection-decontaminazione-e-conservazione/15515-silver-clean-liquido-igienizzante-per-ambienti-a-based-di-peroxide-di-hydrogen .html
- https://mapservice.it/steriizzazione-degli-ambiente-con-macchine-a-ozono/
- www.dday.it/redazione/35522/sanificazione-ambiente-ozono-faccia-coronavirus
- https://www.puntosicuro.it/sicurezza-sul-lavoro-C-1/coronavirus-covid19-C-131/covid-19-considerazioni-tecniche-sulla-sanificazione-con-l-ozono-AR -20224/
- https://tuttnauer.com/blog/low-temperature-sterilization-methods-ozone
- https://www.medivators.com/sites/default/files/minntech/documents/50097-772%20A%20PAA%20Sterilization%20final.pdf
- http://win.spazioinfo.com/public/STERILIZZAZIONE%20CON%20PLASMA%202006-2007.pdf
- https://www.vitrosteril.com/2020/03/sterilizzazione-gas-plasma-nuovo-peroxide-hydrogen/
- http://www.sosozono.it/negozio/sterilizzatore-uv-ultrasuoni/
- https://www.medicalmarket.it/disinf-glutaster-basica-ml-1000.html
- https://www.territo.it/it/catalogo/07-sterilizzanti-a-freddo/glutavir-4x250ml-a800/freddo.aspx
- https://www.farmafox.eu/file/lhglutaral.pdf
- http://www.ausl.vda.it/elementi/www/tecnicoAmministrativa/Formazione/ANMDO/15.Barbaro.pdf
- https://www.ebranditalia.com/it/sterilizzante-disinfettante-peroxill-2000-1000-gr.html
- https://docplayer.it/8291196-Scheda-tecnica-hy-care-2-0-cetic-disposizione-medico-di-classe-ii-b.html/
- Trifirò F. (2020). La Chimica e l’Industria Newsletter. 7(4):14.
- Trifirò F. (2020). La Chimica e l’Industria Newsletter. 7(6):4.
- Song X, Vossebein L, Zille A. (2019). Efficacy of disinfectant-impregnated wipes used for surface disinfection in hospitals: a review. Antimicrob Resist Infect Control. 8:139.
- Rutala WA, Weber DJ. (2013). Disinfection and sterilization: an overview. Am J Infect Control. 41(5 Suppl):S2-S5.
- Gerding DN, Johnson S. (2020). Clostridial infections. In: Goldman L, Schafer AI, (Eds.). Goldman-Cecil Medicine, 26th Edn. Philadelphia, USA, Elsevier Saunders, chap 280.
- Spaulding EH. (1957). Journal of Hospital Research. 9:5.
- Spaulding EH. (1971). The role of chemical disinfection in the prevention of nosocomial infections, in Proc. of the International Conference on Nosocomial Infections, 1970, Chicago, USA, American Hospital Association. pp. 247.
- Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008 (cdc.gov)
- Trifirò F. (2021). La Chimica e l’Industria Newsletter. 8(3):18.
- Trifirò F. (2021). La Chimica e l'Industria Newsletter. 8(3):18.
- Guidelines for the correct use of antiseptics - disinfectants Layout 1 (anmdo.org)
- Decontamination with antiseptics and disinfectants (UNIBa) Slide 1 (uniba.it)
- Sterilizing disinfectant for instruments and equipment 1000 ml, Dimexid 2000 (ebranditalia.com)
- BIOXIR TECHNICAL DATA SHEET
- TECHNICAL DATA SHEET “DECS PURE”
- DECS AMBIENTE PLUS technical sheet
- TECHNICAL DATA SHEET ST DC001: CRESCOM 90
- Antiseptics and Disinfectants-Lombarda H
- BACTISAN Spray 2000 Disinfectant | Online Professional Cleaning
- Gioclorex 0.5 100 (giochemica.com)
- www.ghero.it BENZIMAX NOVATIS
- ST DH063: HYGESAN PROFESSIONAL PLUS TECHNICAL DATA SHEET
- Cerichem Alcovir Technical Data Sheet - Territory
- Lysoform Medical Spray Foam Technical Data Sheet
- https://www.foridra.it/impianti-termici-civili/aria-conditionata/idracleaairsan-ag
- https://www.cfs.it/disinfettante-per-sanificazione-spray-medical-150-ml-mm0007209
- https://www.cnrweb.tv/disinfettante-fai-da-te-contro-il-coronavirus/
- F. Trifirò. (2022). La Chimica e l'Industria Newsletter. 9(1):16.
- https://www.igeamedia.it/blog/sintesi-degli-agenti-disinfettanti-utiliti-con-maggior-frequenza
- Guidelines for the correct use of antiseptics - disinfectants Layout 1 (anmdo.org)
- Decontamination with antiseptics and disinfectants (UNIBa) Slide 1 (uniba.it)
- Correct use of chemical disinfectants and antiseptics (drexpharma.it)
- Choice of disinfectants: from the Spaulding criteria to the legislation on medical devices (nurse24.it)
- Disinfectant Observatory: indications for use and spectrum of action - Techno One srl (techno-one.it)
- Disinfection Univr
- https://www.igeamedia.it/blog/sintesi-degli-agenti-disinfettanti-utiliti-con-maggior-frequenza
- UniBa disinfectants
- Latin ANTISEPTICS and DISINFECTANTS.ppt (live.com)