Information Links
Related Conferences
Previous Issues Volume 6, Issue 4 - 2023
Tail Parotid Tumor: Warthin’s Tumor: Managed With Surgical Treatment
Pankaj Goyal1,*, Nirupama Kothari2
1Apollo ENT Hospital, Pal Road, Jodhpur, Rajasthan, India
2Sterling Accuris Diagnostics, Saras Dairy Road, Jodhpur, Rajasthan, India
*Corresponding author: Pankaj Goyal, Consultant E.N.T. and Head & Neck surgeon, Apollo E.N.T. Hospital, Jodhpur, Rajasthan, India, ORCID: 0000-0003-4098-7308; E-mail: [email protected].
Received Date: October 10, 2023
Published Date: October 21, 2023
Citation: Goyal P, et al. (2023). Tail Parotid Tumor: Warthin’s Tumor: Managed With Surgical Treatment. Mathews J Surg. 6(4):23.
Copyrights: Goyal P, et al. © (2023).
ABSTRACT
Warthin's tumor, which accounts for around 2-15% of all parotid tumors, is the most common monomorphic adenoma of the main salivary glands. Adenolymphoma, papillary cystadenoma lymphomatosum, and monomorphic adenoma are some of the words used to characterize it. It is a disease of the salivary glands, a benign cystic tumor with an extensive lymphoid stroma and an epithelial component. This tumor, which is found in the parotid gland, is the second most prevalent benign salivary gland tumor. From a clinical perspective, it looks to be a slow-growing tumor that fluctuates frequently due to its cystic form. Complete excision with broad tumor-free margins is considered as the optimum course of treatment. In this case, a 57-year-old male patient had a warthin's tumor that was treated with a parotidectomy and did not recur after six months of follow-up.
Keywords: Warthin’s Tumor, Salivary Gland, Parotid, Surgical Excision, Parotidectomy.
INTRODUCTION
Of all head and neck tumors, salivary gland tumors make up about 3%. The parotid gland is most frequently impacted by about 70–80% of these neoplasms, which are found in the major salivary glands [1-3]. The second most frequent benign salivary gland tumor in the parotid gland, after pleomorphic adenomas, are warthin tumors. They are responsible for 15% of all parotid tumors [4-7]. The first person to identify this lesion as a type of congenital neck cyst was Hildebrand in 1895 [2], but his description was eventually forgotten. The first person to describe it as an unusual entity in English medical literature was the pathologist Aldred Scott Warthin (1866–1931), who did so in 1929 [4].
Adenolymphoma, cystic papillary adenoma, cystadenoma lymphomatosum, monomorphic adenoma, adenolymphoma, or Warthin tumor are some of the names that have been given to this tumor because of its histological appearance with duct-like features and extensive lymphoid stroma [4].
Warthin tumors are typically seen in the inferior part of the parotid gland [8]; less frequently, they can also be found in the submandibular salivary gland or cervical lymph nodes region. They often manifest as a painless, smooth, and soft mass that can be fluctuant on palpation [9] or as an asymptomatic mass that is expanding quite slowly. Males are more likely than females to acquire warthin tumors, with the peak incidence occurring in the fifth and sixth decades. They frequently occur bilaterally or in clusters and are linked to excessive cigarette smoking [10].
According to published research, Warthin tumors can be multicentric, synchronous or metachronous, in 12–20% of instances, and they can also be bilateral in 5%–14% of cases [9,10]. The histogenesis of it is unclear [11]. The Warthin tumor was classified by the World Health Organization (WHO) in 2005 as being made up of glandular and frequently cystic structures; occasionally, these structures have a papillary cystic pattern and are coated with epithelium. The aforementioned epithelium is composed of inner columnar eosinophilic or oncocytic cells that are encircled by smaller basal cells and has a distinctive bilayer arrangement [8].
The lymphoid stroma frequently contains a large number of germinal centers. There may still remain lymphoid tissue in the lymph nodes after some of the neoplastic epithelium there has been partially replaced by it, or there may be an immunological reaction to the neoplastic epithelium [12-15]. Van der Wal, et al. [16] demonstrated that lymph nodes can exist inside healthy salivary gland tissue. At the same time, salivary gland tissue may become entrapped in the structure of the lymph nodes.
Partial, subtotal, or total parotidectomy with facial nerve preservation constitutes the course of treatment [17]. Only 0.1% of cases of malignant transformation have been reported, and they typically develop in the lymphoid tissue tumor's epithelial component [18]. After surgical therapy, the likelihood of recurrence is quite uncommon [19].
CASE REPORT
A 57-year-old obese man with no prior medical history arrived to the Apollo ENT Hospital in Jodhpur with a left-sided infra-auricular swelling that had been present for a year and was gradually getting bigger. There were no signs of the constitution, such as tenderness, fluctuation, colour changes, facial weakness, or other masses. There was no prior radiation exposure, and there was no family history of the same illness.
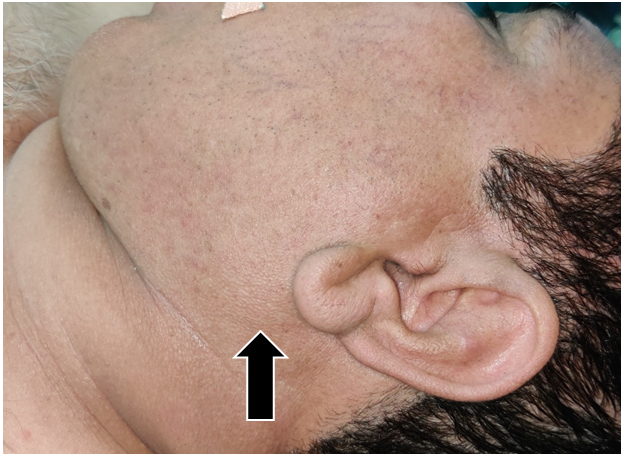
A superficial, painless, soft, and movable mass measuring 2 x 2 cm was discovered during inspection in the left parotid area (especially tail of parotid gland region). (Figure 1) No discoloration or tethering could be found on the skin that covered the mass's surface. In addition, there was no evidence of facial nerve paralysis or cervical lymphadenopathy. There were no sinuses, fistulas or dilated veins over swelling. On palpation, inspectory findings were confirmed. The mass was mobile and non-tender. Skin over swelling was pinchable. There was no palpable lymphadenopathy. There were no noteworthy results from direct flexible laryngoscopy.
Figure 1. Clinical picture showing swelling over left sided parotid-tail region.
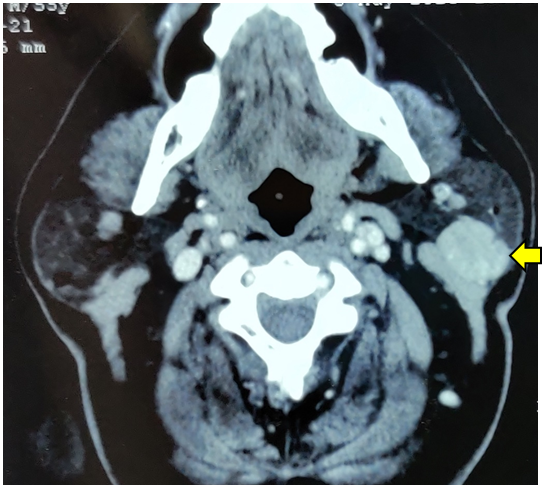
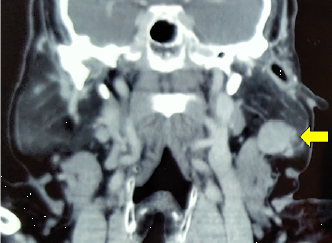
A well-defined oval-shaped soft tissue lesion in left parapharyngeal space measuring 20 x 26.9 x 21 mm and it is involving the superficial lobe of the left parotid gland, without calcification showing heterogenous enhancement on post contrast tomography (CT) scan of the head and neck (Figures 2 A and B). No evidence of extra parotid extension is seen.
Figure 2A. Axial Cut
Figure 2B. Coronal Cut
Figure 2 (A and B). Contrast-enhanced computed tomography (CT) scan (axial and coronal view) of the head and neck revealed enhancement of a well-defined oval-shaped lesion in the left parotid gland's superficial lobe measuring 20 x 26.9 x 21 mm.
Smears that were subjected to fine needle aspiration biopsies revealed cohesive, flat sheets of oncocytes. Oncocytes had rich, granular cytoplasm and spherical, homogeneous nuclei with evenly distributed chromatin. Abundant reactive lymphocytes were observed scattered throughout the background. The features were consistent with a Warthin tumor.
Treatment
After explaining the benefits and possible complications of the surgery, the patient consented to proceed with our method. The patient was treated surgically, with the mass completely removed using a modified Blair's incision for parotidectomy. The surgery went uneventful, and there were no complications afterward.
Surgical technique
The surgery was conducted under general anesthesia by a head and neck team. The patient was resting supine with his neck stretched and his head turned to the contralateral side. The incision was then started at the level of the right tragus in the pre-auricular region, continued along the skin crease, and ended at the ear lobe. It then twisted around the ear lobe and proceeded postero-superiorly along the post-auricular sulcus until it reached the level of the right external auditory canal.
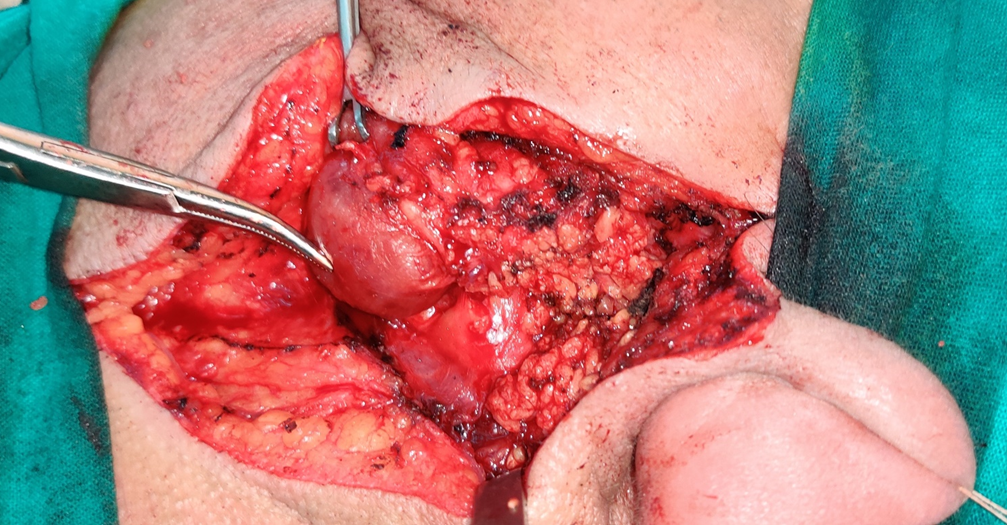
To reveal the parotid gland and anterior border of the right sternocleidomastoid muscle, the skin flap was elevated in three directions: anterior, inferior, and posterior. The parotid fascia was detached from the ear lobe, which was then superiorly retracted. The facial nerve was identified by using the tympanomastoid fissure, retromandibular vein and tragal cartilage as a landmark after parotid gland and right sternocleidomastoid muscle were dissected apart. The relevant branch of the facial nerve was carefully dissected after the flap had sufficiently retracted (Figure 3). The tumor was fully excised, with an adequate resection boundary that included normal parotid tissue (Figure 4). Hemostasis was obtained. The wound was closed in layers.
Figure 3. Intraoperative picture showing mass at left tail parotid region with surrounding parotid gland.
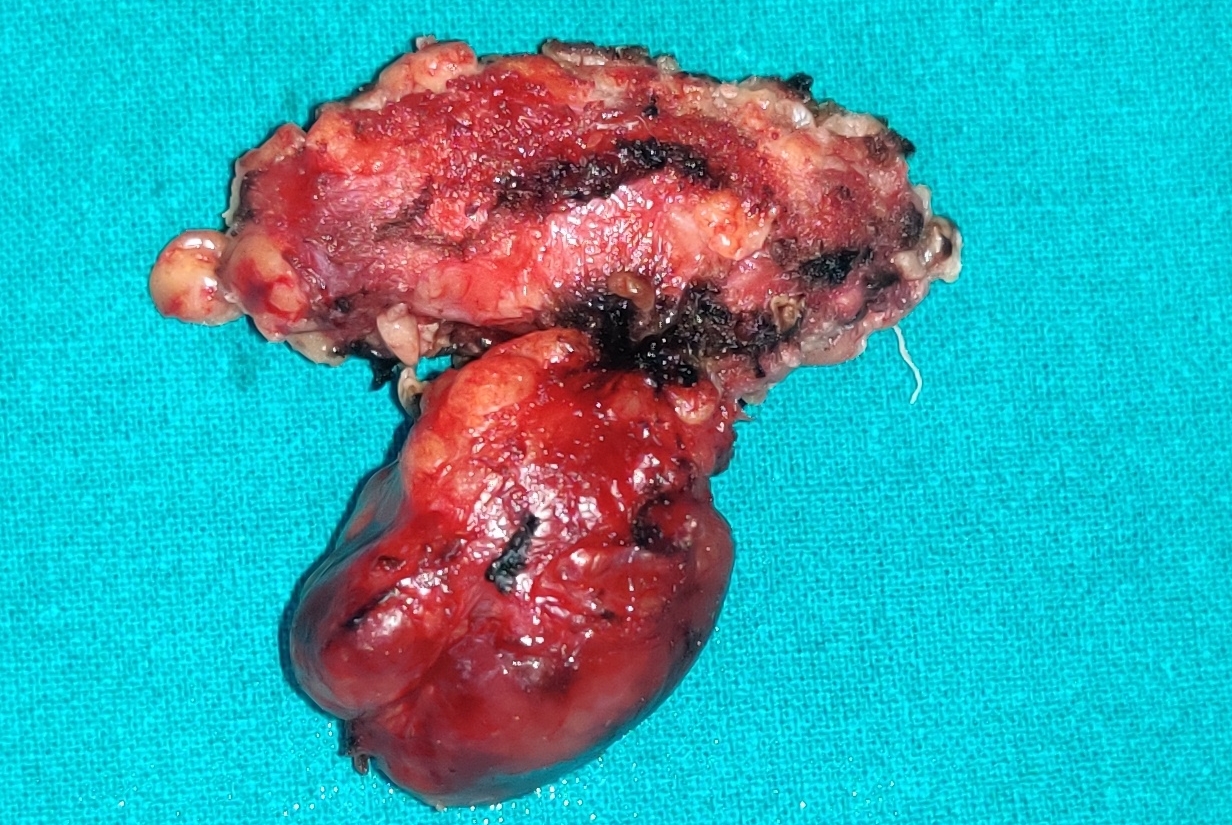
Figure 4. Main surgical specimen.
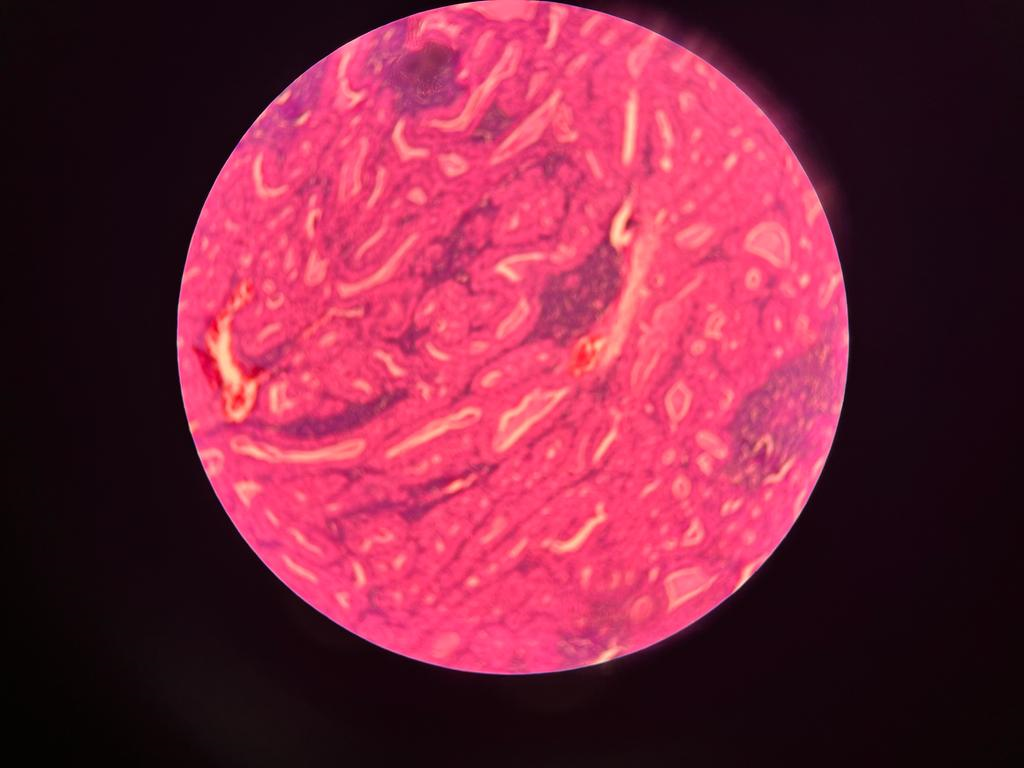
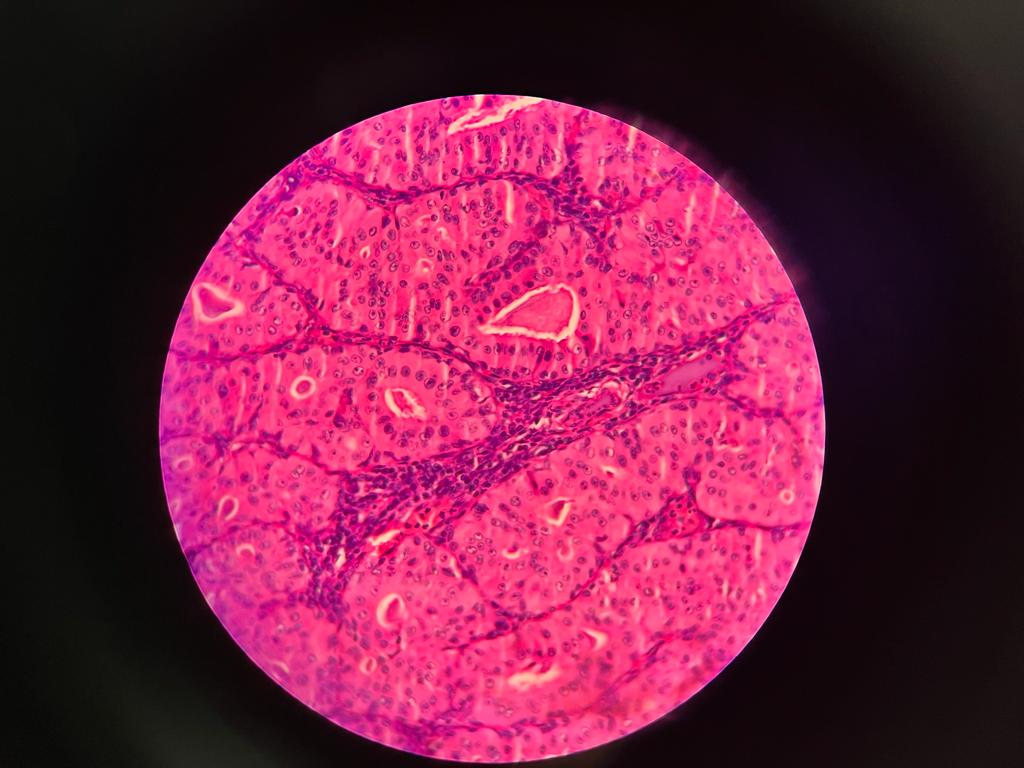
The pre-operative diagnosis of Warthin's tumor was confirmed by pathological investigation, with no evidence of malignancy (Figure 5 A, B, C). The patient has been examined on a regular basis during follow-up, and there have been no recurrences in one year.
Figure 5A
Figure 5B
Figure 5C
Figure 5 (A,B,C). Histopathological slides of Warthin’s tumor showed a well circumscribed tumor composed of papillary structures lined by hyalnized oncocytic epithelium and surrounding dense lymphoid aggregates forming lymphoid follicles few papillae are dilated and filled with eosinophilic material and cholestrol clefts. No atypia seen.
DISCUSSION
Warthin tumor is one of the most common benign salivary gland tumors and it generally involves the parotid gland [20]. It occurs predominantly in the parotid gland and represents approximately 15% of parotid tumours [17,19,21-23]. Fewer than 10% of cases occur outside this gland [19]. Furthermore, bilateralism is described in 5-15% of cases and multifocality in 6-20% [23-26].
The pathophysiology that leads to the development of Warthin tumor is currently unknown. The prevalent theory holds that the lesion develops during embryogenesis, when salivary gland tissue becomes lodged in the parotid lymph nodes.
The lymphoid and epithelial parts of the parotid gland do not compartmentalize inside the gland in the early stages of development. The lymphoid component may still contain epithelial cells, which are the ancestors of the salivary duct-acinar system.
These lymphoid parts will eventually make up the intraglandular lymph nodes. After an unknown tumorigenic event, these epithelial inclusions could result in Warthin's tumor [24]. Due to the presence of numerous lymph nodes in the parotid gland's tail, bilateral or unilateral multiple Warthin's tumors are more prone to form there.
The second theory holds that the Warthin tumor is an adenoma with a lymphocytic infiltrate, suggesting that the presence of tumor epithelial cells triggers either an immunological or a hypersensitive response [27].
The pathophysiology of Warthin tumors may be influenced by certain viruses, however the literature has not yet addressed this issue. One study alone seemed to suggest that the Epstein-Barr virus might be involved, but further studies did not support this theory [28]. Cigarette carcinogens are probably going to be a big risk factor for the development of Warthin tumors. In our case, the patient smoked for a considerable amount of time—more than 28 years—supporting the idea that smoking caused the lesion.
Patients may present with no symptoms or complain of facial pain. Rarely, facial nerve palsy in tumors linked to fibrosis and inflammation might manifest; this characteristic can raise the possibility that the tumor is cancerous. Tinnitus, deafness, and ipsilateral ear pain are rare ear symptoms that can be observed in some people [29]. Our case featured a progressively growing asymptomatic tumor in the parotid gland, similar to prior reports [30].
The tumor size varies from a few millimeters to a few centimeters, with an average of 2 cm to 4 cm in diameter; it is mostly located in the lower pole of the gland, i.e., the jaw angle [31,32]. Our patients presented tumors with dimensions closer to those in the literature.
Fine needle aspiration biopsy (FNAB) and CT scans are necessary for the diagnosis of a Warthin tumor. While FNAB is a quick and easy approach to find a salivary gland tumor, it is not extremely sensitive or specific for detecting Warthin tumors [20]. The FNAB results are still useful in attempting to rule out malignancy, though. Surgery is the most common form of treatment and has a low recurrence rate [33].
Several writers have linked the rate of recurrence to the extent of surgery, which ranges from 0% to 13% [19,25,34]. In our study, we used superficial parotidectomy with facial nerve preservation, and there were no recurrences after a median follow-up of 31 months.
When the circumstances allow, conservative surgery—which does not include the dissection of all facial nerve branches—is being employed increasingly frequently. The use of superficial parotidectomy was associated with a decrease in the incidence of facial nerve dysfunction, according to several research [35,36].
The chosen therapy for Warthin's tumor is either a superficial or total parotidectomy with preservation of the facial nerve, and long-term follow-up has not revealed any instances of tumor recurrence. The two main side effects of this procedure were facial nerve dysfunction and Frey's syndrome.
As a result, if total parotidectomy is an appropriate radical resection of parotid parenchyma that theoretically lowers the risk of recurrence, superficial parotidectomy is also an effective radical procedure with low morbidity in terms of facial nerve dysfunction and Frey's syndrome.
Resections like those of the lower extremity of the superficial pole of the parotid should still be considered since they may be done safely in lower tumors and allow for lower manipulation of the cranial branches of the facial nerve with less morbidity. However, further prospective and randomized researches are required to establish a clearer conclusion.
A thorough examination of patients with multiple Warthin's tumors is therefore advised, and long-term follow-up is required due to the possibility of metachronous salivary and extrasalivary tumors even after long intervals of time [16,37]. This is because Warthin's tumor may be associated with extra-salivary neoplasms.
CONCLUSION
The Warthin tumor, a benign parotid gland tumor, typically affects older smokers who have smoked for a considerable amount of time. Clinicians should consider the Warthin tumor when making a parotid mass differential diagnosis. After diagnosis and treatment, individuals with Warthin tumors should have a sufficient follow-up since metachronous tumors or synchronous ones that are still undiscovered might form even after a lengthy period of time.
COMPLIANCE WITH ETHICAL STANDARDS
The procedure performed in this case report was in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
FUNDING
This study is not funded by any resources.
CONFLICT OF INTEREST
The author (s) declares no potential conflicts of interest with respect to the research, authorship, and/or publication of this paper.
ETHICAL APPROVAL
The study was published with the written consent of the patient.
REFERENCES
- Ellis GL, Auclair PL, Gnepp DR. (1991). Surgical pathology of the salivary glands. Philadelphia, USA: W.B. Saunders. p. 165.
- Loyola AM, de Araujo VC, de Souza SO, et al. (1995). Minor salivary gland tumors. A retrospective study of 164 cases in a Brazilian population. Eur J Cancer B Oral Oncol. 31B:197-201.
- Hilton JM, Phillips JS, Hellquist HB, et al. (2008). Multifocal multi-site Warthin tumour. Eur Arch Otorhinolaryngol. 265:1573-1575.
- Lee DH, Yoon TM, Lee JK, Lim SC. (2019). Surgical treatment strategy in Warthin tumor of the parotid gland. Braz J Otorhinolaryngol. 85(5):546-550.
- Diaz-Segarra N, Young LK, Levin K, Rafferty W, Brody J, Koshkareva Y. (2018). Warthin tumor of the oropharyngeal minor salivary gland. SAGE Open Med Case Rep. 6:2050313X18818712.
- Teymoortash A, Schrader C, Shimoda H, Kato S, Werner JA. (2007). Evidence of lymphangiogenesis in Warthin's tumor of the parotid gland. Oral Oncol. 43(6):614-618.
- Espinoza S, Felter A, Malinvaud D, Badoual C, Chatellier G, Siauve N, et al. (2016). Warthin’s tumor of parotid gland: surgery or follow-up? Diagnostic value of a decisional algorithm with functional MRI. Diagn Interv Imaging. 97(1):37-43.
- Schwalje AT, Uzelac A, Ryan WR. (2015). Growth rate characteristics of Warthin’s tumours of the parotid gland. Int J Oral Maxillofac Surg. 44(12):1474-1479.
- Patel DK, Morton RP. (2016). Demographics of benign parotid tumours: Warthin’s tumour versus other benign salivary tumours. Acta Otolaryngol. 136(1):83-86.
- Lee DH, Yoon TM, Lee JK, Lim SC. (2014). Surgical treatment outcomes of patients with bilateral Warthin tumors in the parotid gland. Indian J Otolaryngol Head Neck Surg. 66(Suupl 1):303-306.
- Peter Klussmann J, Wittekindt C, Florian Preuss S, Al Attab A, Schroeder U, Guntinas-Lichius O. (2006). High risk for bilateral Warthin tumor in heavy smokers–review of 185 cases. Acta Otolaryngol. 126(11):1213-1217.
- Cope W, Naugler C, Taylor SM, Trites J, Hart RD, Bullock MJ. (2014). The association of Warthin tumor with salivary ductal inclusions in intra and periparotid lymph nodes. Head Neck Pathol. 8(1):73-76.
- Teymoortash A, Bohne F, Jonsdottir T, Hoch S, Eivazi B, Roessler M, et al. (2013). Human papilloma virus (HPV) is not implicated in the etiology of Warthin’s tumor of the parotid gland. Acta Otolaryngol. 133(9):972-976.
- Orabona GD, Abbate V, Piombino P, Romano A, Schonauer F, Iaconetta G, et al. (2015). Warthin’s tumour: aetiopathogenesis dilemma, ten years of our experience. J Craniomaxillofac Surg. 43(4):427-431.
- Joo YH, Kim JP, Park JJ, Woo SH. (2014). Two-phase helical computed tomography study of salivary gland Warthin tumors: a radiologic findings and surgical applications. Clin Exp Otorhinolaryngol. 7(3):216-221.
- van der Wal JE, Davids JJ, van der Waal I. (1993). Extraparotid Warthin’s tumours–report of 10 cases. Br J Oral Maxillofac Surg. 31(1):43-44.
- Bussu F, Parrilla C, Rizzo D, Almadori G, Paludetti G, Galli J. (2011). Clinical approach and treatment of benign and malignant parotid masses, personal experience. Acta Otorhinolaryngol Ital. 31(3):135-143.
- Faur A, Lazăr E, Cornianu M, Dema A, Vidita CG, Găluşcan A. (2009). Warthin tumor: a curious entity–case reports and review of literature. Rom J Morphol Embryol. 50(2):269-273.
- Yoo GH, Eisele DW, Askin FB, Driben JS, Johns ME. (1994). Warthin’s tumor: a 40-year experience at The Johns Hopkins Hospital. Laryngoscope. 104(7):799-803.
- Singh Nanda KD, Mehta A, Nanda J. (2012). Fine-needle aspiration cytology: a reliable tool in the diagnosis of salivary gland lesions. J Oral Pathol Med. 41(1):106-112.
- Thangarajah T, Reddy VM, Castellanos-Arango F, Panarese A. (2009). Current controversies in the management of Warthin tumour. Postgrad Med J. 85(999):3-8.
- Piccioni LO, Fabiano B, Gemma M, Sarandria D, Bussi M. (2011). Fine-needle aspiration cytology in the diagnosis of parotid lesions. Acta Otorhinolaryngol Ital. 31(1):1-4.
- Faur A, Lazăr E, Cornianu M, Dema A, Vidita CG, Găluşcan A. (2009). Warthin tumor: a curious entity--case reports and review of literature. Rom J Morphol Embryol. 50(2):269-273.
- Maiorano E, Lo Muzio L, Favia G, Piattelli A. (2002). Warthin's tumour: a study of 78 cases with emphasis on bilaterality, multifocality and association with other malignancies. Oral Oncol. 38(1):35-40.
- Guntinas-Lichius O, Gabriel B, Klussmann JP. (2006). Risk of facial palsy and severe Frey's syndrome after conservative parotidectomy for benign disease: analysis of 610 operations. Acta Otolaryngol. 126(10):1104-1109.
- Teymoortash A, Krasnewicz Y, Werner JA. (2006). Clinical features of cystadenolymphoma (Warthin's tumor) of the parotid gland: a retrospective comparative study of 96 cases. Oral Oncol. 42(6):569-573.
- Zeebregts CJ, Mastboom WJ, van Noort G, van Det RJ. (2003). Synchronous tumours of the unilateral parotid gland: rare or undetected? J Craniomaxillofac Surg. 31(1):62-66.
- Attie JN, Sciubba JJ. (1981). Tumors of major and minor salivary glands: clinical and pathologic features. Curr Probl Surg. 18(2):65-155.
- Iwai T, Baba J, Murata S, Mitsudo K, Maegawa J, Nagahama K, et al. (2012). Warthin tumor arising from the minor salivary gland. J Craniofac Surg. 23(5):e374-e376.
- Liang CH, DI WY, Ren JP, Zhou FM, Hu Y, Mao HJ, et al. (2014). Imaging, clinical and pathological features of salivary gland adenolymphoma. Eur Rev Med Pharmacol Sci. 18(23):3638-3644.
- Wang D, Li Y, He H, Liu L, Wu L, He Z. (2007). Intraoral minor salivary gland tumors in a Chinese population: a retrospective study on 737 cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 104(1):94-100.
- Lukšić I, Virag M, Manojlović S, Macan D. (2012). Salivary gland tumours: 25 years of experience from a single institution in Croatia. J Craniomaxillofac Surg. 40(3):e75-e81.
- Baj A, Beltramini GA, Demarchi M, Combi VA, Giannì AB. (2011). Bilateral SMAS rhytidectomy in parotid recurrent pleomorphic adenoma. Acta Otorhinolaryngol Ital. 31(4):256-258.
- García-Purriños FJ. (2011). Trece años de experiencia en parotidectomía parcial superficial como tratamiento de neoformaciones benignas parotideas [Thirteen years' experience with superficial partial parotidectomy as treatment for benign parotid tumours]. Acta Otorrinolaringol Esp. 62(1):10-13.
- Stathopoulos P, Igoumenakis D, Smith WP. (2018). Partial superficial, superficial, and total parotidectomy in the management of benign parotid gland tumors: a 10-year prospective study of 205 patients. J Oral Maxillofac Surg. 76(2):455-459.
- Sood S, McGurk M, Vaz F. (2016). Management of salivary gland tumours: United Kingdom national multidisciplinary guidelines. J Laryngol Otol. 130(S2):S142-S149.
- Scasso CA, Papini M, Eligi C, Ghelardi F, Cagno MC, Bruschini P. (1998). An unusual neck mass: the Warthin tumor. Acta Otorhinolaryngol Belg. 52(1):55-57.
.png)