Information Links
Related Conferences
Current Issue Volume 10, Issue 2 - 2026
Seroprevalence and Risk Factors of Ovine Pasteurellosis in Asagirt District, Ethiopia
Habtamu Yeneneh Alemu*
Livestock and Fishery Resource Development, Animal Health Team, Amanuel, Ethiopia
*Corresponding Author: Habtamu Yeneneh Alemu, Livestock and Fishery Resource Development, Animal Health Team, Amanuel, Ethiopia, Phone: +251-920249926, E-mail: [email protected]
Received Date: December 30, 2025
Published Date: March 09, 2026
Citation: Alemu HY. (2026). Seroprevalence and Risk Factors of Ovine Pasteurellosis in Asagirt District, Ethiopia. Mathews J Vet Sci. 10(2):104.
Copyrights: Alemu HY. © (2026).
ABSTRACT
A cross-sectional study was conducted from January 2022 to July 2022 in Asagirt district in North Shoa Zone, Amhara National Regional State of Ethiopia with the objectives of estimating the prevalence of ovine pasteurellosis, identify the associated risk factors, and to determine the dominant serotypes of pasteurella species. A total of 141 sheep sera was collected to identify the prevailing serotypes and parallelly a questionnaire was administering to the sheep owners to collect information on presumed potential risk factors. The serum samples were examined for six serotypes of species of M. haemolytica, B. trehalose and P. multocida using indirect haemagglutination test at National Veterinary Institute, Bishoftu, Ethiopia. An overall seroprevalence of 67.4% was found in this study. Among the serotypes detected M. haemolytica serotype A2 (43.3%) was the most dominant serotype in the study district followed by serotype A7 (41.8 %). The prevalence of the other serotypes T15, PA, T4 and A1 were 26.2%, 24.8%, 21.7% and 7.8% respectively. Among the risk factors flock size, sex, age, lactation and pregnancy status have statistically significant association with seropositivity of ovine pasteurellosis whereas contact with other flocks, different management practice and exposure to other disease were not have significant association in this study. Generally, ovine pasteurellosis is highly prevalent in the study district. Serotype A1, A2, A7, PA, T4 and T15 are the dominant serotypes circulating in the area. Hence, regular vaccination with a polyvalent vaccine incorporated the dominant serotypes identified should be considered.
Keywords: Asagirt District, Ovine Pasteurellosis, Prevalence, Risk Factor, Serotype.
INTRODUCTION
In Ethiopia, the agricultural sector is a cornerstone for the economic and social life. The sector employs 80-85 per cent of the population and smallholder farmers dominate the agricultural sector. The highlands hold about 95 percent of the cropped area and support two-thirds of the livestock population [1]. The livestock sector is a large contributor to the Ethiopian economy as well as a mainstay in the livelihoods of many Ethiopians. But livestock production and markets vary substantially across space in Ethiopia due to a variety of reasons including topographical variations, market access, water availability, and population characteristics [2].
Ethiopia is believed to be the first in livestock comparing to other livestock population in Africa and tenth in the world and containing about 60.39 million cattle, 31.30 million sheep, 32.74 million goats, 2.01 million horse, 8.85 million donkeys, 0.46 million mules and 1.42 million camels [3]. With this large population livestock sector play a significant role and contributing 15-17% of the total GDP and 35-49% of agricultural GDP in the country by providing export commodities like live animals, hides and skins to earn foreign exchanges. In addition to this, livestock sub sector is sources of food, immediate cash income and promote saving [4].
Despite the large number of livestock in Ethiopia the sector is characterized by low productivity, hence, income derived from this sector is not at the expected level in the development of the country’s economy. The low productivity is attributed to the low genetic potential, poor nutrition and reproductive performance, inadequate management, high disease incidence and parasite burden [5]. One of the diseases that reduce small ruminant productivity and cause substantial losses through high morbidity and mortality not only in Ethiopia but also throughout the world is pasteurellosis [6]. Pneumonic pasteurellosis is one of the most economically important infectious diseases of ruminants with a wide prevalence throughout the country with severe fibrinous or fibrinopurulent bronchopneumonia, fibrinous pleurisy and septicaemia. Infected animals may die within a few days of the onset of clinical signs, but those which survive the acute attack may become chronically infected. M. haemolytica, P. multocida and B. trehalosi are the most important members of the family Pasteurellaceae that pose serious hazards in livestock industry and incriminated in many outbreaks [7].
The disease in sheep is characterized by an acute infection with high fever, coughing, dyspnea, muco-purulent nasal discharge, anorexia and depression that commonly develops when the immune system of the animal is compromised by stress factors [8]. Predisposing factors of pneumonic pasteurollosis like infectious agents that have been incriminated epidemiologically and management and environment factors like transportation, overcrowding (large flock size), climate changes, bad housing condition are important on occurrence and spread of the disease from infected animal to healthy animals by direct contact, by inhalation and consumption of contaminated feed [9].
In Ethiopia, different studies indicated that pasteurellosis is a major threat to sheep production and some works have been conducted on ovine pasteurellosis seroprevalence and detection of most prevalent serotypes, for example, in Tigray regional state at Tanqua-Abergelle district [10], in SNNPR in Wolaita Zone [11], and in Oromiya region, East Shewa Zone at Lume district [9].
M.haemolytica, B. trehalosi and P.multocida have different serotypes. In Ethiopia, however, detailed information is lacking and routine serotyping is not practiced due to the time and expense involved, and the lack of commercially available antisera, so that the serotypes circulating and causing outbreak remain unknown [10]. Furthermore, the prevalence of ovine pasteurellosis is not known in the study district. Therefore, the main objectives of this study were to estimate the prevalence of ovine pasteurellosis, determine the dominant serotypes of the species of M. haemolytica, B. trehalosi and P. multocida, and identify associated risk factors that enable the disease to proliferate in Asagirt district.
MATERIALS AND METHODS
Study area description
The study was conducted in Asagirt district in North Shoa Zone, Amhara National Regional State of Ethiopia. The district is 125.5 kilometers(Km) far from Addis Ababa, the capital city of Ethiopia. It has 15 kebeles (the lowest administrative unit) and located at the eastern edge of the Ethiopian highlands [12]. Asagirt district is situated at about 1500-3200 meters above sea level, and falls in to three agro-ecological zones, Dega (2300-3200 m.a.s.l), Woina Dega (1500-2300 m.a.s.l.) and Kolla (bellow 1500 m.as.l). In the district, the temperature ranges between 9.33-19.17°C with the mean annual temperature of 14.25°C. Rainfall ranges between 925-1816 mm (Ethiopian Metrological Institute).
Study design and Study population
A cross sectional study design was used to estimate the seroprevalence of ovine pasteurellosis, determine the dominant serotypes circulating in Asagirt district and identify the potential risk factors of the disease. The study was carried out from January 2022- July 2022. Apparently healthy local breed sheep of both sex, age of above six months and all management systems. The study animals were in different age group, body conditions and management. The management system practiced in that area was more of semi intensive even extensive and intensive management system also practiced. Sheep which managed under extensive and semi intensive have common area of grazing and watering.
Sample Size and Sampling Methods
The study district was selected purposively due to its large population of sheep with communal grazing and watering practice and no history of vaccination for ovine pasteurellosis. The sample size for the study was determined using the formula given by [13]. To calculate the total sample size, 95% confidence Interval, 5% desired level of precision; and 10% expected prevalence of ovine pasteurellosis were used.
N=1.962Pexp (1-Pexp)
d2
Where N= required sample size, P exp = expected prevalence, d = desired absolute precision, 1.962 = z- value for 95% confidence interval. Hence, by using this formula, the sample size required for the study was 138.
Sample collection and laboratory processing
Blood samples for serum extraction were collected directly from jugular vein using sterile needles and plain vacutainer tubes. From each sheep 5 up to 10 ml of blood was collected aseptically. The blood was allowing to clot without shaking for 2 h at room temperature, and slanted at 45 degrees overnight at 4 °C. The serum will then separate from the clot by centrifugation at 3000 rpm for 10 minutes and transferred to cryovials, label and kept frozen (-20°C) until laboratory analysis. The collected sera were transported on ice to the National Veterinary Institute (NVI), Bishoftu for serological identification using IHAT. Each serum samples were serotyped using the reference serotypes available in NVI using IHAT to detect the presence of antibodies against serotypes of Mannheimia haemolytica, B. trehalosi and P. multocida in the serum sample.
The IHAT antibody titer of all the samples were recorded and Positive results were taken if the level of antibody titer was greater than 1:40 after the following procdure:- 100 of 1/10μl diluted serum in PBS dispenced into first row of microplat in dubilicates and charge all remaining wells of the micrplate with 50μl of 1x PBS. A serial double fold dilution (1:10, 1:20, 1:40, etc.) was made by transferring 50μl of diluted serum from wells of the first row to the next row wells in parallel thorough mixing and discard 50μl diluted sera from the last row after thorough mixing. After this 50 μl SRBC added to into all wells of micro plate and cover plates with micro plat sealer to prevent evaporation and incubated at 37°C in moist chamber for 60 minutes with constant agitation. Negative and positive controls were included in separate microplates. Wells with > 50% agglutination of red blood cells indicates a positive reaction whereas small button of deposited RBCs is a negative reaction.
Questionnaire survey
A structured questionnaire was used to collect information on multifactorial predisposing factors or risk factors of pneumonic pasteurellosis (management system, feeding and watering practice, coinfection and contact with each other’s or other flock, age, vaccination history, use of medications, flock size, sex, if female: its parity, pregnancy and lactation status). Farmers were asked about general farming systems including the number of families, types and number of domestic animals and farming constraints before enter to the actual questions.
Data management and analysis
Data obtained from questionnaire survey as well as sero surveillance were coded and entered in to Microsoft Excel spread sheets in order to make ready for descriptive and statistical analysis. The statistical analysis was done by using SPSS version 20. Descriptive method was used to summarize and display the data in tables, and to calculate the prevalence of ovine pasteurellosis. Chi-square (x2) test was employed to assess the presence of association between the dependent variable seropositivity for Pasteurella species and independent variables (age, sex, flock size, pregenancy status, and health status, lactation, contact, management system).
RESULTS
Pasteurellosis is among the complex respiratory diseases that develop when the immune system of the animal is compromised by different effects [14]. The effects of different environmental stressors are believed to be important components of risk factors for pasteurellosis in many domestic ruminants. The explosive multiplication of M. haemolytica and B. trehalosi and also P. multocida due to suppression of the immune system in sheep by stressors is probably most economically important in inducing acute infectious disease that causes widespread financial losses because of death, reduced live weight, delayed marketing and treatment costs [15].
The questionnaire survey result indicated that semi intensive, extensive system and rarely intensive management systems are practiced in the study districts with a level of 45.3%, 42.5% and 12% respectively. Due to the extensive and semi intensive production system practiced in the area 87.2% of the flock have contact with other flocks. Majority of the farmers (84.4%) have less than ten flock size. Farmers in the study area keep more females (78.7%) than males (21.2%) because they maintain females for breeding purposes whereas they sell males for cash income.
Infected sheep by Pneumonic pasteurellosis develop high fever with clinical evidence of severe respiratory involvement manifested by dyspnea, froth at the mouth, cough and mucopurulent nasal discharges. Young animals are more susceptible than adults and they develop more severe infection in which sudden death may occur with or without any previous warning clinical signs [7].
Septicemia typically occurred in lambs which caused by Bibersteinia trehalosi clinical signs included sudden death, dullness, pyrexia, recumbency, frothy discharge of the mouth, dyspnea, and prostration leading to death within 6-8 hours.
From a total of 141 serum samples collected and tested, 95 samples were found positive for ovine pasteurellosis and this gives an overall prevalence of 67.4% (95/141) (Figure 1).
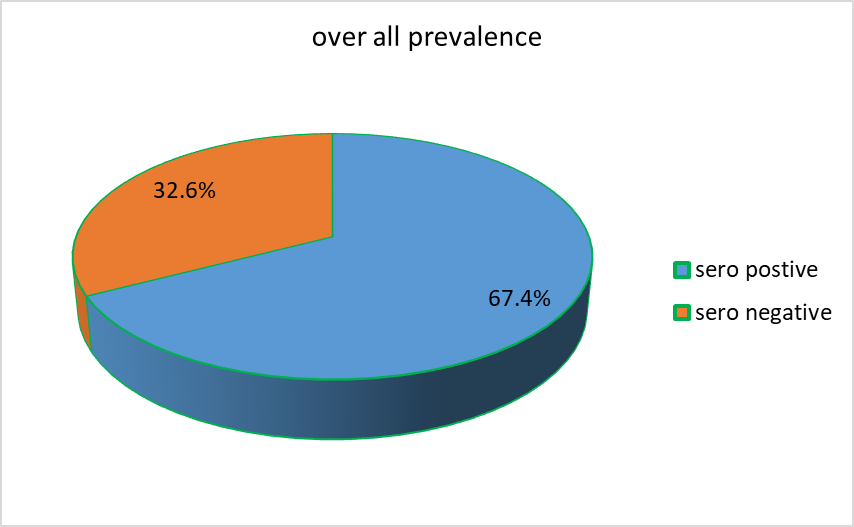
Figure 1. Over all sero prevalence of ovine pasteurellosis.
Among the presumptive risk factors assessed, flock size, sex, age, lactation and pregnancy status have statistically significant association with seropositivity for ovine pasteurellosis. Whereas contact with other flocks, presence of other infection and management system have no statistical significant association with ovine pasteurellosis (Table 1).
Table 1. Chi-square analysis of risk factors association with ovine pasteurellosis
|
Risk Factor |
Level |
No. examined |
No. positive |
Percentage |
χ2-value |
P-value |
|
Flock size |
<10 10-15 >15 |
119 13 9 |
85 8 2 |
71.4 61.5 22.2 |
9.439 |
0.009 |
|
|
||||||
|
Sex |
Female Male |
111 30 |
81 14 |
73.0 46.7 |
7.435 |
0.006 |
|
Age |
<1year 1-2year >2year |
20 27 94 |
8 17 70 |
40.0 63.0 74.5 |
9.209 |
0.010 |
|
Lactation status |
Yes No |
46 65 |
35 46 |
76.1 70.8 |
7.491 |
0.024 |
|
Pregnancy status |
Yes No |
35 76 |
25 56 |
71.4 73.7 |
7.491 |
0.024 |
|
Management system |
Intensive Semi intensive Extensive |
17 64 60 |
12 43 40 |
70.6 67.2 66.7 |
0.95
|
0.954 |
|
Contact |
Yes No |
123 18 |
82 13 |
66.7 72.2 |
0.220 |
0.639 |
|
Other infection |
Yes No |
50 91 |
36 59 |
72.0 64.8 |
0.754 |
0.385 |
Serotyping results
The serotyping test was conducted against to six serotypes under three pasteurella species: M. haemolytica (A1, A2, and A7), P. multocida A and B. trehalose (T4 and T15). The frequency of the serotypes was 7.8%, 43.3%, 41.8%, 24.8%, 21.7% and 26.2% for A1, A2, A7, PA, T4 and T15 respectively (Table 2).
Table 2. The frequency of Pasturella serotypes in sampled sheep sera
|
|
Serotypes |
|||||
|
A1 |
A2 |
A7 |
PA |
T4 |
T15 |
|
|
Frequency |
11 |
61 |
59 |
35 |
31 |
37 |
|
Prevalence (%) |
7.8 |
43.3 |
41.8 |
24.8 |
21 |
26.2 |
When we see at species level Mannheimia haemolytica is most prevalent than B. trehalose and P. multocida (Figure2).
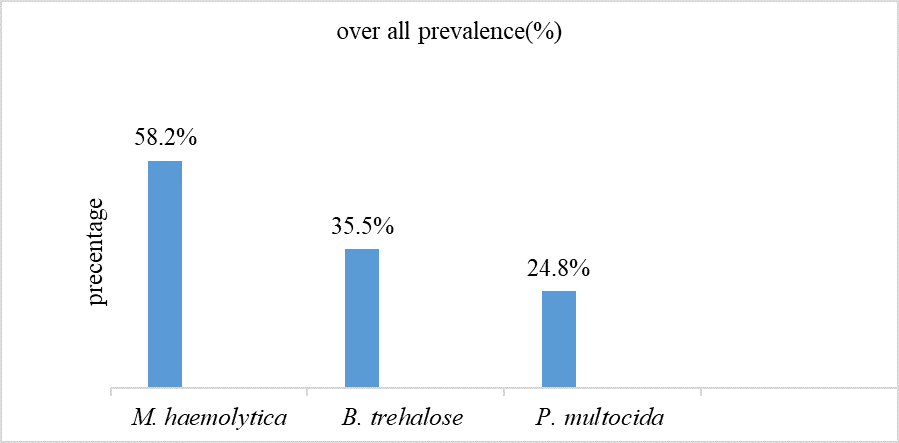
Pasteurella species
Figure 2. Over all prevalence of M. haemolytica, B. trehalose and P. multocida detected from sheep sera tested.
DISCUSSION
In this study, an overall seroprevalence of 67.4% was recorded which coincides with what [16] reported (62.7%) in North showa. However, the current result is higher than [17], who reported 27.6% in metema district, northwest Ethiopia in indigenous Gumuz sheep. On the other hand, it is less than that of 83% which reported in wollo [16]. In this study, species of M. haemolytica B. trehalosi, and P.multocida) were detected in frequency of 58.1%, 35.4%, and 24.8% respectively. These are greater than what [9] reported for M. haemolytica (11.2%), B.trehalose (7.6%) and P.multocida (2.1%) from the nasal swab and 11.7% prevalence for M. haemolytica, 10.3% for trehalose and 2.1% for P.multocida from lung lesions in Lume districts, East Shoa Zone of Oromia region.
In present study, serotype A1, A2, A7, PA, T4 and T15 were detected in the frequency of 7.8%, 43.3%, 41.8%, 24.8%, 21.7% and 26.2% respectively. M. haemolytica A2 was identified in higher frequency than others. The 43.3% prevalence reported for serotype M. haemolytica A2 in the current study is comparable to the 42.6% prevalence reported by [9], but much less than from 81.8% reported by [11] and higher than the 36% prevalence reported by [18]. M. haemolytica A7 (41.8%), the second dominant serotype identified in this study is much less than the 76.8% prevalence reported by [11]. Serotype A1 (7.8%) was the least prevalent serotype in the current study and is lower than the 37.1% [14] and 23.3% [9] reported somewhere else in Ethiopia. These prevalence difference might be due to the difference in type of test employed, sample size and the study area.
Among the factors considered in this study, flock size, sex, age, lactation and pregnancy status was found to have statistically significant association with ovine pasteurellosis. There was statistically significance difference between different age groups. This indicates that difference in age can affect seropositivity, as the age of the animal increases the seropositivity also increases. This finding (the effect of age on seropositivity) coincide with what [19] reported in Fogera woreda, north-west Ethiopia. Sex also found to be a factor for seropositivity of ovine pasteurellosis. This indicates that the distribution of positive sera was dependent of the sex of the sheep. This finding coincide with what [19] reported in Fogera woreda, north-west Ethiopia. But, contrary to this, [9] and [20] reported that sex of sheep does not affect sheep seropositivity for pasteurelosis [21-24].
CONCLUSION AND RECOMMENDATIONS
Pasteurellosis is a complex disease that develops when the immune system of the animal is compromised by stress factors. Ovine pasteurellosis is one of the disease which is caused by Mannhaemia haemolytica, Pastuerella multocida and Bibersteinia trehalosi that result in high rates of morbidity and mortality with widespread financial losses because of reduced live weight, delayed marketing, treatment costs and unthriftness among survivors. The overall prevalence of ovine pasteurellosis (67.4%) found in this study is high. Six serotypes, which are in the species of Mannhaemia haemolytica, Bibersteinia trehalosi, and Pastuerella multocida were detected in high frequency. Serotype A2 and A7 are the dominant serotypes circulating in the study area. Flock size, sex, age, lactation and pregnancy status have statistically significant association with the occurrence of ovine pasteurellosis. Based on the above conclusion, routine epidemiological study in different parts of the country should be practiced to identify prevalence of ovine pasteurelosis and dominant serotypes. Development of polyvalent vaccine that contain locally identified serotypes should be considered. Furthermore, reducing the spread of the disease by avoiding different associated risk factors are mandatory.
ACKNOWLEDGMENT
First of all, I would like to offer glorious gratitude to almighty of God for helping me in everything of my life. Next thanks to all sheep owners and veterinarians participated during data collection
COMPETING INTERESTS
The author declares that he has no competing interests.
REFERENCES
- Sileshi Z, Tsegahun A, Yami A, Tegegne A. (2001). Status of livestock research and development in the highlands of Ethiopia. CGIAR Research centers, Ethiopia. Available at: https://hdl.handle.net/10568/51093
- Tilahun H, Schmidt E. (2012). Spatial analysis of livestock production patterns in Ethiopia. International Food Policy Research Institute, Ethiopia. Available at: https://hdl.handle.net/10568154021
- CSA. (2018). Agricultural sample survey, 2017/18 (2010 E.C.), volume II: report on livestock and livestock characteristics (private peasant holdings). Statistical bulletin 587. Central statistical agency (CSA), Federal Democratic Republic of Ethiopia, Addis Ababa.
- Getu A. (2015). Review on Challenges and Opportunities of Sheep Production in Ethiopia. African J Basic & Appl Sci. 7(4):200-205
- Moges N, Bogale B. (2012). Assessment of major animal production and health problems of livestock development in lay-Armacheho District, Northwestern Ethiopia. Am-Euras J Sci Res. 7(3):136-141.
- Haji S. (2015). Pasteurollosis in sheep and its drug susceptibility pattern in Mojo district, East Shoa Zone. Addis Ababa University. CGIAR research centers. Available at: https://hdl.handle/10568/135988
- Mohamed R, Abdelsalam E. (2008). A review on pneumonic pasteurellosis (respiratory mannheimiosis) with emphasis on pathogenesis, virulence mechanisms and predisposing factors. Bulgarian Journal of Veterinary Medicine. 11(3):139-160.
- Legesse A, Abayneh T, Mamo G, Gelaye E, Tesfaw L, Yami M, et al. (2018). Molecular characterization of Mannheimia haemolytica isolates associated with pneumonic cases of sheep in selected areas of Central Ethiopia. BMC Microbiol. 18(1):205.
- Jarso D. (2016). Epidemiology of Ovine Pasteurellosis in Lume District, East Shewa Zone of Oromiya Region, Ethiopia. J Vet Sci Res. 1(3):000115.
- Berhe K, Weldeselassie G, Bettridge J, Christley RM, Abdi RD. (2017). Small ruminant pasteurellosis in Tigray region, Ethiopia: marked serotype diversity may affect vaccine efficacy. Epidemiol Infect. 145(7):1326-1338.
- Yonas Y, Goa A. Butako B. (2017). Study on the Epidemiology and Strain Identification of Small Ruminant Pneumonic Pasteurellosis for Vaccine Production in Selected Districts of Wolaita Zone, SNNPR. Int J Curr Res Biol Med. 2(3):8-13.
- Negash WD, Tsehay CT, Yazachew L, Asmamaw DB, Desta DZ, Atnafu A. (2022). Health system responsiveness and associated factors among outpatients in primary health care facilities in Ethiopia. BMC Health Serv Res. 22(1):249.
- Thrusfield M. (2007). Describing disease occurrence. Veterinary Epidemiology. Blackwell Publishing, Oxford, England. pp. 46-74.
- Mengstie F. (2015). Serotyping and identification of predisposing factors for the occurrence and distribution of ovine pasteurellosis in Bonga sheep breed. AsianJ Med Biol Res. 1(2):175-181.
- WIrtu A, Kumsa B, Zerabruk E, Albene Y, Tadesse F, et al. (2022). Bacteriological and Molecular Identification of Mannheimia haemolytica, Pasteurella multocida and Bibersteinia trehalosi from Cattle and Sheep from Selected Areas of Ethiopia. J Vet Med Res. 9(3):1234.
- Tadesse B, Alamirew K, Ketema A, Kiflie W, Endashaw M. (2017). Ruminant Pneumonic Pasteurellosis: Review on Epidemiology, Pathogenesis and Virulence Mechanism. Academic Journal of Animal Diseases. 6(2):30-39.
- Nigusu K, Fentie T. (2012). Prevalence and causes of selected respiratory infections in indigenous Gumuz sheep in Metema district, Northwest Ethiopia. Int J Sci. 5(1):14-20.
- Tarikua S. (2004). Epidemiologic and Serologic Investigation of Multifactorial Respiratory Disease of Sheep in the Central Highland of Ethiopia. Intern J Appl Res Vet Med. 2(4):274-278.
- Engdaw TA, Alemneh AT. (2015). Pasteurellosis in Small Ruminants: biochemical Isolation, Characterization and Prevalence Determinationin Relation to Associated Risk Factors in Fogera Woreda, North-West Ethiopia. Advances in Biological Research. 9:330-337.
- Marru HD, Anijajo TT, Hassen AA. (2013). A study on ovine pneumonic pasteurellosis: isolation and identification of Pasteurellae and their antibiogram susceptibility pattern in Haramaya District, Eastern Hararghe, Ethiopia. BMC Vet Res. 9:239.
- Ahmed M. (2003). Colonisation of the ovine respiratory tract by Pasteurella (Mannheimia) haemolytica: a thesis presented in partial fulfilment of the requirements for the degree of Master of Veterinary Studies at Massey University. New Zealand: Massey Research. Available at: https://mro.massey.ac.nz
- Brogden KA, Lehmkuhl HD, Cutlip RC. (1998). Pasteurella haemolytica complicated respiratory infections in sheep and goats. Vet Res. 29(3-4):233-254.
- Hakim A, Bakry M, Nagwa S, Mona S. (2014). Role of molecular techniques in characterization of bacteria causing pneumonia in small ruminants. Life Sci J. 11(6):147-153.
- Kumar J, Dixit SK, Kumar R. (2015). Rapid detection of Mannheimia haemolytica in lung tissues of sheep and from bacterial culture. Vet World. 8(9):1073-1077.