Information Links
Related Conferences
Previous Issues Volume 7, Issue 1 - 2023
Proximate Composition and Some Mineral Contents on Flesh of Tetradon Fahaka and Synodotis Schell Fish from River Nile
Haram Hassan Abbas Bakhiet*, Osman MS, Kbashi ER, Noor SA
Department of Fisheries and Wildlife Science College of Animal Production Science and Technology, Sudan University of Science and Technology, Khartoum North, Sudan
*Corresponding author: Haram Hassan Abbas Bakhiet, Department of Fisheries and Wildlife Science College of Animal Production Science and Technology, Sudan University of Science and Technology, Khartoum North, Sudan: Email: [email protected], [email protected]
Received Date: January 9, 2023
Publication Date: January 30, 2023
Citation: Bakhiet HHA, et al. (2023). Proximate Composition and Some Mineral Contents on Flesh of Tetradon Fahaka and Synodotis Schell Fish from River Nile. Mathews J Vet Sci. 7(1):17.
Copyright: Bakhiet HHA, et al. © (2023)
ABSTRACT
Proximate composition (moisture, protein, fat, ash, fiber) and some minerals (iodine, zinc, and iron, copper) content in the flesh of two fish species, Tetradon fahaka and Synodotis schell were investigated. Statistical analysis using ANOVA showed significant differences in the values of the moisture and fat in the two fishes studied (P<0.05). In terms of percentages, moisture content was highest in Synodotis schell (72.1±0.3 %) and lowest in Tetradon fahaka (70.6±0.7 %). Fat content was found to be highest in Tetradon fahaka (2.1±0.8 %) and lowest in Synodotis schell (1.7±0.6 %), there were no significant differences in protein, ash and fiber content while minerals showed significant differences in the content of Iodine, Zinc and iron there was no significant differences in copper content. Iodine and iron were highest in Synodotis schell (0. 24 mg/kg) and (0.19mg/kg) respectively. While Zinc was highest in Tetradon fahaka (0. 3 ppm).
Keywords: Tetradon fahaka, Synodotis schell, Protein, Zinc
INTRODUCTION
Fish is stable diet, particularly in developing nations due to its high protein content and nutritional value of unsaturated fatty matter. It might only be available and affordable source of animal protein for slum housing in urban semi-urban areas. Fish is also widely accepted due to its high palatability, low cholesterol and tender flesh. However the feeding habit, sex, species, seasonal variation and other factors have a significant impact on the nutrient composition of specific fish species (Kefas, M., etal, 2014) [1].
Fishes are good sources of animal protein and other essential nutrients for body and health maintenance. Small indigenous fish species are a rich supply of minerals like calcium and phosphorus in addition to being a source of protein. (Amin et al., 2013) [2].
Essential nutrients are those chemical elements that are necessary for regular upkeep of humans. These elements (Zn, Fe, Cu, I) participate in several biochemical reactions; zinc is mostly found as a cofactor in enzyme reactions, iron forms part of the hemoglobin molecule which transport oxygen around the body. When the body is not adequately supplied with these nutrients primarily through nutrition an individual may develop diseases caused by mineral deficiency such as anemia, stunted growth and genetic problems. (Mogobe, O, et al, 2015) [3]. The human body requires nutrients to function properly and to stay healthy; these nutrients are obtained from foods. Water, protein, carbohydrates, fats, vitamins and minerals are some of the food nutrients. Fishes are known to provide protein, fat and vitamins which are of great benefit to human health. Fish protein is of high quality because it contains almost ideal proportion of essential amino acids. Fish protein is easily digestible, because it contains less collagen fiber than meat protein; allowing the body to make the most of its protein. Fish fats contain unsaturated fatty acids which are beneficial by lowering blood triglycerides. (Victoria, 2015) [4]. Proximate composition refers to the percentage composition of basic constituents within a fish body such as water, protein, lipids, carbohydrate and minerals. Traditionally, the chemical composition of the fish has been used to predict its nutritional value. (Moghaddam et al., 2007; Aberoumand, 2011) [5,6]. It varies greatly between species and also greatly influenced by the feeding habit, sex and seasonal variations (Islam et al., 2005) [7]. The study’s objectives are to determine the proximate composition; (moisture, protein, fat, ash, fiber) and some mineral contents (iodine, iron, zinc, and copper) in tetradon fahaka and synodontis schell fish.
MATERIALS AND METHODS
Samples Collection and Preparation
Samples were collected from fishing landing site in Khartoum between February and March 2022. Two fish species (Synodontis schell) and (Tetradon fahaka) in different weight and length they were obtained fresh and transported to the laboratory and frozen (-180) until examination, which was, done within 24 hours. Sampling of flesh were taken from back area near the dorsal fin, Analysis of moisture, protein, fat, ash, fiber using standard methods according to the Association of Official Analytical Chemists AOAC 2005 (Ndome and Ogar, 2010) [8]. And minerals (iodine, iron, zinc and copper) were analyzed according to the Atomic Absorption Spectrometer (AAS). The data obtained were analyzed using ANOVA tests to determine the variability in the estimates of studied parameters.
RESULTS
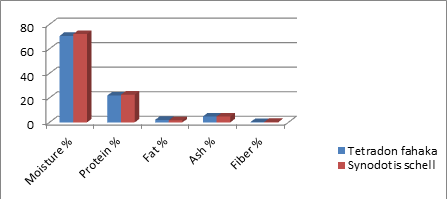
In table 1, the study explicates that there is significant difference in moisture and fat content Which is 70.6±0.7 and 72.1±0.3 for moisture in Tetradon fahaka and Synodotis schell respectively.
Table 1: Explain proximate composition of Tetradon fahaka and Synodotis schell mean ±SD.
|
Fish species |
Moisture% |
protein% |
Fat % |
Ash% |
Fiber% |
|
Tetradon fahaka |
70.6±0.7 |
22±0.3 |
2.1±0.8 |
4.9±0.7 |
0.42±0.1 |
|
Synodotis schell |
72.1±0.3 |
22.7±0.6 |
1.7±0.6 |
4.8±0.6 |
0.44±0.9 |
|
Sig |
* |
NS |
* |
NS |
NS |
Sig: Significant difference, *: high significant difference, NS: Not significant
Figure 1: proximate composition of the fish Tetradon fahaka and Synodotis schell
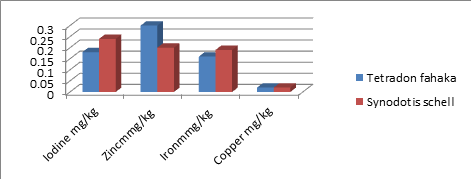
In table 2, there is significant difference in iodine, zinc and iron between the two studied fish. on the other hand, there is no significant difference in copper content.
Table 2: Explain some mineral content (mg/kg) (Mean) in Tetradon fahaka and Synodotis schell
|
Fish species |
Iodine |
Zinc |
Iron |
Copper |
|
Tetradon fahaka |
0. 18 |
0. 3 |
0.16 |
0.02 |
|
Synodotis schell |
0. 24 |
0. 2 |
0.19 |
0.02 |
|
Sig |
* |
* |
* |
NS |
Sig: Significant difference, *: high significant difference, NS: Not significant
Figure 2: Some mineral content in fish Tetradon fahaka and Synodotis schell.
DISCUSSION
The nutritional profile is formed when the proximate components of the fish flesh combine to form a first indication of the fish’s commercial standards that are required for good regulation (Marichamy et al., 2012) [9]. The study of moisture, protein, fat and ash contents of fish beneficial to the consumers, producers and scientists from a variety of perspectives. Besides recognizing the nutritional value of fish, thus a study also assists in comprehending its greater processing and preservation (Mridha et al., 2005) [10].
The results of the proximate composition expressed in g/100g edible portion, are presented in table 1 and figure 1, the moisture contents of the fish samples were 70.6±0.7% and 72.1±0.3 % for Tetradon fahaka and Synodotis schell respectively.
The values obtained in this study are higher than the values reported by (Boran and Karacam, 2011) [12] for shad (57 to 68%) and in the same range for horse mackerel (65 - 75%) from the Black Sea of Turkey. The protein contents of the fishes in this study are within the range of 22.00 to 22. 07 (g/100 g) (Table. 1 and Figure 2). These values indicate that they are high protein sources for consumers. These values are greater than values of other fishes. Palani et al. (2014) who’s found that the protein content of fatty fishes that landed in the Thoothukudi Coast of India was 14% [13]. The high tissue protein content in the present study fish may result from the equally high protein content of their diets. Fish fat contains Omega-3 fatty acids which assist to lower the probability of evolving cardio-vascular illnesses in humans. Additionally, fish fat includes considerable amount of vitamins A and E and is one of the few natural sources of vitamin D. Fishes have fat content less than 5% are considered lean fish (Stansby, 1982; Ackman, 1989) [14]. In the present study the fish species had fat contents that lower than 5%, so they can be considered to be lean. The ash values found in this study are higher than the finding made by (Alfred, Ochkiya and Ndioma (1998) [15] who found (3.38) % ash content for C. macrolepis. Ash is known to be a significant nutrients source in fishes. The identified Ash content range stated that the investigated species are excellent minerals source (Hanna, 1985) [16]. Zinc serves a variety of functions in the body; it is a part of many metallo-enzymes, essential for gene expression and cellular metabolism. The FAO reported ranges of 0.23 - 2.1 mg/100 g and it’s higher than the range for this research (0. 2 - 0.3mg/100 g) and Tao et al. (2012) [17] also obtained the same range of this study (0.64 - 0.81 mg/100 g) in cultured fish in China; French market fish gave a range of 0.13 - 2.5 mg/100 g (Guerin et al., 2011) [18]; some fish from Turkey gave a range of 0.57 - 1.3 mg/100 g (Alas et al., 2014 ) [19] B. Poechii record the highest Zinc content of 8.47 mg/100 g followed by B. Lateris (6.48 mg/100 g). These results are in agreement with results obtained by Kawarazuka and Bene (2011) [20], in studies carried out in Bangladesh and Cambodia which showed higher zinc concentration ranges for small Bangladesh fishes (1.1 - 4.0mg/100g). Iron is necessary for a variety of physiological roles in the body, and very beneficial for which is the transport of oxygen throughout the body. Iron deficiency result in anaemia, (Latham, 1997) [21]. The Fe concentration in Chanoga fish was 1.65 - 6.4 mg/100 g which is higher than the study finding. On the other side the results agree to some extent to the Study by Gokoglu et al. (2004) on fish from a French market recorded much lower concentration levels of iron (0.13 - 1.9 mg/100 g) [22]. According to the literature consulted for this paper, there is a significant regional variance in the amount of iron found in fish, Fish is typically the highest source of iodine in human diets, seaweed and iodized salt. Iodine concentration in various fish species has been the subject of numerous studies from various nations. There was significant variance in the iodine concentration of different fish species around the Norwegian coast, both between individuals of the same species and between fish species (Julshamn, 2001) [23]. Pakistan has also examined the iodine level in several freshwater and saltwater fish species and they found that freshwater fish had an iodine concentration that was 5 to 10 times higher than that of seawater. (Azmat, 2008) [24]. The accumulation of iodine in fish from various aquatic resources varies from species to species and also depends on the nature of the species, environmental factors or it may alter periodically, according to various data provided by researchers Larsen, 1997) [25]. In the present study, Cu values are in agreement with literature values and have not posed a threat to the health of humans.
REFERENCES
- Kefas M, Michael KG, Abubakar KA, Edward A, Wahide JA. (2014). Proximate and mineral contents of flesh and body parts of Oreochromis niloticus And Synodontis clarias in Mubi, Nigeria. Global J Biol Agri Health Sci. 3(3):116-121.
- Amin SMN, Mohd Azim MK, Fatinah SNJ, Arshad A, Rahman MA, Jalal KCA. (2013). Population parameters of Rastrelliger kanagurta (Cuvier, 1816) in the Marudu Bay, Sabah, Malaysia. Ir J Fish Sci. 13(2):262-275.
- Mogobe O, Mosepele K, Masamba WRL. (2015). Essential mineral content of common fish species in Chanoga, Okavango Delta, Botswana. Afs J Food Sci. 9(9):480-486.
- Akpambang VOE. (2015). Proximate composition of some tropical fish species. Pelagia Res Library, 6(4): 125-129.
- Moghaddam HN, Mesgaran MD, Najafabadi HJ, Najafabadi RJ. (2007). Determination of chemical composition, mineral contents and protein quality of Iranian Kilka fish meal. Int J Poultry Sci. 6:354-361.
- Aberoumand A. (2011). Proximate composition of less known some processed and fresh fish species for determination of the nutritive values in Iran. J Agri Technol. 8(3):917–922.
- Islam MD, Tanaka M. (2005). Impact of pollution on coastal and marine ecosystems including coastal and marine fisheries and approach for management: A review and synthesis. Marine Pollution Bull. 48:624-649.
- Ndome O. Oriakpono O, Ogar A. (2010), Proximate Composition and Nutritional Values Of Some Commonly Consumed fishes from the cross river estuary. Tropical Freshwater Biol. 19(1):11-18.
- Marichamy G, Haq MB, Vignesh R, Sedhuraman V, Nazar AR. (2012). Assessment of proximate and mineral composition of twenty Edible fishes of parangipettai coastal waters. Int J Pharma Bio Sci. 3(2):54-64.
- Mridha AR, Lipi SY, Narejo NT, Uddin MS, Kabir MS, Karim M. (2005). Determination of biochemical composition of Cirrhinus reba (Hamilton, 1822) from Jessore, Bangladesh. J Sci Technol Univ Peshwar. 29(1):1–5.
- Boran G, Karacam H. (2011). Seasonal Changes in Proximate Composition of Some Fish Species from the Black Sea. Tur J Fish Aqua Sci. 11:01.
- Palani K M, Ruba A A, S. R., Shanmugam S A, ( 2014). Proximate and Major Mineral Composition of 23 Medium Sized Marine Fin Fishes Landed in the Thoothukudi Coast of India. J Nutr Food Sci. 4:1.
- Stansby ME. (1982). Properties of fish oils and their application to handling of fish and to nutritional and industrial use. In: Martin RE, Flick GJ, Hebard CE, Ward DR. (Eds) Chemistry and Biochemistry of Marine Food Products:75-92.
- Ackman RG. (1989). Nutritional composition of fats in sea foods. Prog. Food Nutr Sci. 13:161-241.
- Alfred-Ockiya JF, Ndioma FO. (1998). Biochemical Composition of Selected fin fishes. Alodl J Pure Appl Sci. 4:199-202.
- Hanna GMR. (1985). Proximate composition of certain red sea fishes. J. Marine Fisheries Rev. 46(3):71-75.
- Tao NP, Wang LY, Gong X, Liu Y. (2012). Comparison of nutritional composition of farmed pufferfish muscles among Fugu obsurus, Fugu flavidus and Fugu rubripes. J Food Compost Anal. 28:40-45.
- Guerin T, Chekri R, Vastel C, Sirot V, Volatier JL, Leblanc JC, Noel L (2011). Determination of 20 trace elements in fish and other seafood from the French market. Food Chem. 127:934-942.
- Alas A, Ozcan MM, Harmankaya M. (2014). Mineral contents of head, caudal, central fleshy part, and spinal columns of some fishes. Environ. Monit Assess. 186:889-894.
- Kawarazuka N, Bene C. (2011). The potential role of small fish species in improving micronutrient deficiencies in developing countries:Building evidence. Public Health Nutr. 14:1927-1938.
- Latham MC. (1997). Human Nutrition in developing world. Rome: FAO Food and Nutrition Series No:29.
- Gokoglu N, Yerlikaya P, Cengiz E. (2004). Effects of cooking methods on the proximate composition and mineral contents of rainbow trout (Oncorhynchus mykiss). Food Chem. 84:19.
- Julshamn K, Dahl L, Eckhoff K. (2001). Determination of iodine in seafood by inductively coupled plasma/mass spectrometry. J AOAC Int. 84:1976-1983.
- Azmat R, Talat R, Syed-Junaid M. (2008). Distribution of iodine in marine and freshwater fishes from Sindh Regions of Pakistan. J Appl Sci 8:1790-1792.
- Larsen EH, Ludwigsen MB. (1997). Determination of iodine in food-related certified reference materials using wet ashing and detection by inductively coupled plasma mass spectrometry. J Anal Atom Spectrum 12:435-439.