Previous Issues Volume 1, Issue 1 - 2016
Patterns of Care Among Patients With Genotype 1 Hepatitis C Virus in Europe
Siva Narayanan1
Corresponding Author: Siva Narayanan, SVP and Global Head, Global Evidence, Value and Access, Ipsos Healthcare, 2020 K Street NW, Suite 410, Washington, DC, 20006, USA, Tel: +1-203-297-1208; Email:[email protected] Received Date: 06 May 2016 Accepted Date: 29 Aug 2016 Published Date: 02 Sep 2016 Copyright © 2016 Narayanan S Citation: Narayanan S. (2016). Patterns of Care Among Patients With Genotype 1 Hepatitis C Virus in Europe. Mathews J Gastroenterol Hepatol 1(1): 004. ABSTRACT
To assess clinical characteristics and patterns of care among patients with Genotype 1 (G1) Hepatitis C virus (HCV) in EU, a multi-center retrospective chart-review study of HCV patients was conducted in the EU (France/Germany/Italy/Spain/ UK) in 2014 to collect de-identified data on diagnosis, clinical status, and treatment patterns. Physicians were screened for duration of practice (=3yrs) and patient volume (=15 HCV patients/month) and recruited from a large panel to be geographically representative. Medical charts of the next 10 consecutive HCV patients were abstracted. 2067 eligible G1 HCV patients were included in the final analysis (France: 374/Germany: 506/Italy: 412/Spain: 475/UK: 300). Patient characteristics included (France/Germany/Italy/Spain/UK) G1-subtype A (49%/38%/30%/31%/58%) or B (44%/47%/67%/61%/28%); current fibrosis scores: F0: 9%/15%/5%/7%/18%, F1: 18%/24%/24%/24%/29%, F2: 24%/23%/31%/23%/17%, F3: 20%/14%/17%/19%/10%, F4: 28%/5%/20%/22%/17%; latest mean alanine aminotransferase levels:58/90/95/62/73 IU/ mL; patients with viral load >1Million IU/mL: 23%/12%/33%/27%/14%. Treatment patterns included (France/Germany/Italy/ Spain/UK)- currently treated:23%/28%/20%/17%/20%, treatment naïve/never been treated:38%/40%/41%/41%/52%, not currently treated (previous treatment non-responder, discontinued or relapsed, or therapy complete and awaiting sustained viral response): 21%/20%/29%/30%/22%, not currently treated for other reasons: 19%/12%/11%/13%/6%. Disease burden was high in this cohort of G1 HCV patients and only a small proportion of patients were not treated owing to achieving sustained viral response. KEYWORDS
HCV; Genotype 1; Europe; Burden; Treatments; SVR. INTRODUCTION
Hepatitis C, caused by hepatitis C virus (HCV) is often asymptomatic and is characterized by slow disease progression. An estimated 160 million individuals may be infected with HCV globally and the corresponding estimate for Europe is approximately 5.5 million [1-3]. Globally, genotype 1 (G1) is estimated to account for 46.2% of all HCV cases. G1 is the most predominant genotype in Europe (estimated to be between 59% and 89% depending on the region); subtype 1a is most commonly associated with intravenous drug abuse and subtype 1b is typically observed in patients who have acquired HCV through a blood transfusion. In Europe, approximately 67% of HCV cases are acquired through IV drug use, and approximately 5% through blood and blood products [4-6]. Persistent HCV infection is associated with the development of liver cirrhosis, hepatocellular cancer, liver failure, and death, while HCV is a common cause of death in HIV-positive patients [7, 8]. As incidence of HCV infection decreases in the developed world, deaths from liver disease secondary to HCV infection are expected to increase over the next 20 years [5, 9]. Vaccination against hepatitis C is not yet available; however, current treatment options include antivirals and agents that stop the virus from replicating and may eliminate the infection altogether [5, 10-12]. There is evidence indicating that some patients do not receive treatments to manage their HCV infection. Research highlighting the current patterns of care in the key countries in Europe could help portray the current status of HCV management in that region, especially among the most prevalent HCV patients (i.e., G1 type). MATERIALS AND METHODS
The study was a multi-country, multi-center retrospective medical chart review of adult (>18 years) HCV patients conducted in the big-5 European countries, namely, France, Germany, Italy, Spain & United Kingdom (UK)) in Oct-Dec 2014. Physicians were randomly sampled in each of the countries using online physician panels using geo-dispersion sampling methods (whereby, stakeholders are recruited from a wide selection of clinics/hospitals in a given geography representing the modality of care delivery in HCV arena, with each institution contributing almost equal number of study-eligible patient charts); this sampling methodology ensures physician recruitment from diverse locations (urban, sub-urban and rural centers) and practice settings (hospitals and individual (community/private) clinics), and avoids physician sampling biases occasionally associated with selection/use of only limited set of sites, especially in research related to widely prevalent disease(s), thereby enabling the generalization of study findings in a given geography. Invitations to participate in research were sent to a random set of physicians (hepatologists, gastroenterologists, hepatogastroenterologists, internal medicine and infectious disease specialists) in the existing online physician panels. The physicians representing both hospital-based and private practices in each geography, personally responsible for managing at least 15 HCV patients per month and having >3 years of clinical practice experience were screened for study participation. Each physician reported de-identified anonymous data on 10 next consecutive patients they encountered in their practice within the study recruitment window. HCP patient charts were eligible for the study if the patient was being managed as part of usual care, without any participation in clinical trials. An electronic data collection form was used to collect the following data elements from eligible HCV patient charts: patient demographics, clinical characteristics (incl. classification of liver histology per International Association for Study of the Liver (IASL)), comorbidities, laboratory values (e.g., Viral Load and alanine aminotransferase (ALT)) and HCV treatment patterns/dynamics. Only de-identified anonymous data was collected from the patient charts by the treating physicians. This mode of data collection method met the criteria for local ethics review exemption per the respective physician/site requirements in the respective countries. Study data analysis focused on G1 HCV patients. Descriptive statistics were utilized to analyze the data. RESULTS AND DISCUSSION
Data corresponding to 2067 eligible medical charts of HCV patients with G1 genotype were included in the study analysis; 18% were from France, 24% from Germany, 20% from Italy, 23% from Spain and 15% from the UK. The mean age of patients was 51yrs. The cohort was predominantly male and caucasian; intravenous drug was the main route of infection reported across the studied countries, ranging from 17% (Germany) to 55% (UK). (Table 1). Key comorbidities in the study cohort encompassed cardiovascular disease (12% (range: 3% (UK) – 20% (Germany))), Steatosis (12% (range: 5% (UK) – 17% (Italy))), HIV (11% (range: 4% (Germany) – 18% (France))), depression (11% (range: 5% (Italy) –16% (UK))) and diabetes (11% (range: 8% (UK/Italy) – 13% (Germany))).
Table 1: Patient Demographic and Clinical Characteristics.
| France N=374 | Germany N=506 | Italy N=412 | Spain N=475 | UK N=300 | |
| Age: | |||||
| Mean, yrs | 53 | 49 | 55 | 52 | 45 |
| Missing data, % | 1% | 2% | 0% | 0% | 0% |
| Gender: | |||||
|---|---|---|---|---|---|
| Male, % | 61% | 67% | 61% | 59% | 69% |
| Female, % | 39% | 33% | 39% | 41% | 31% |
| Race: | |||||
| Caucasian, % | 84% | 91% | 95% | 96% | 92% |
| Other, % | 15% | 8% | 5% | 4% | 8% |
| Missing data, % | 1% | 1% | 0% | 0% | 0% |
| Weight: | |||||
| =80 kg, % | 78% | 54% | 79% | 60% | 61% |
| >80 kg, % | 19% | 27% | 17% | 21% | 20% |
| Missing data, % | 3% | 19% | 4% | 19% | 19% |
| Substance abuse: | |||||
| Alcohol, % | 9% | 9% | 3% | 12% | 20% |
| Intravenous drug use, % | 6% | 8% | 1% | 1% | 18% |
| Other, % | 1% | 2% | 1% | 2% | 8% |
| None, % | 70% | 77% | 86% | 78% | 50% |
| Missing data, % | 15% | 5% | 8% | 8% | 4% |
| Route of Infection: | |||||
| Intravenous drug use, % | 37% | 17% | 21% | 27% | 55% |
| Blood transfusion, % | 12% | 14% | 14% | 24% | 5% |
| Homosexual contact, % | 5% | 7% | 7% | 4% | 10% |
| Heterosexual contact, % | 3% | 6% | 15% | 7% | 6% |
| Tattoo/body piercing, % | 4% | 4% | 1% | 1% | 3% |
| Vertical transmission, % | 2% | 2% | 2% | 1% | 1% |
| Other, % | 3% | 3% | 7% | 3% | 0% |
| Missing data, % | 34% | 48% | 32% | 34% | 21% |
| Genotype: | |||||
| Subtype 1a, % | 49% | 38% | 30% | 31% | 58% |
| Subtype 1b, % | 44% | 47% | 67% | 61% | 28% |
| Subtype 1c, % | 0% | 2% | 1% | 0% | 0% |
| Missing data, % | 7% | 12% | 2% | 7% | 14% |
| Alanine aminotransferase (ALT): | |||||
| Mean level, IU/L | 58 | 90 | 95 | 62 | 73 |
| Missing data, % | 4% | 17% | 2% | 2% | 10% |
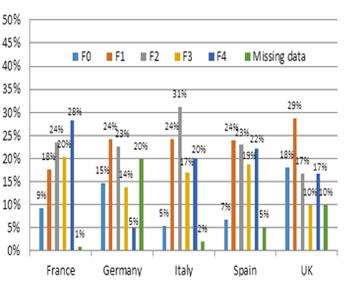
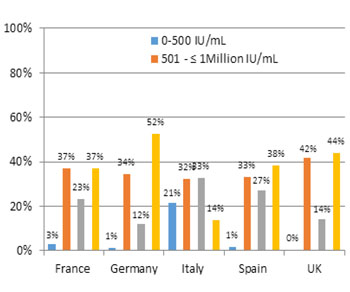
Forty percentage (range: 30% (Italy) – 58% (UK)) of G1 patients in the study had a documented diagnosis of genotype subtype 1a, and 51% (range: 28% (UK) – 67% (Italy)) had subtype 1b and 1% (range: 0% (France/Spain/UK) – 2% (Germany)) had subtype 1c. Mean alanine aminotransferase (ALT) level of patients across the countries was 76 IU/L, and this varied across the countries (Table 1). Distribution of fibrosis scores (per IASL) documented in the patient charts indicated 10% (range: 5% (Italy) – 18% (UK)) had F0 (no liver damage), 24% (range: 18% (France)–29% (UK)) had F1 (minimal liver damage), 24% (range: 17% (UK) – 31% (Italy)) had F2 (moderate liver damage), 16% (range: 10% (UK) – 20% (France)) had F3 (advanced liver damage) and 18% (range: 5% (Germany) – 28% (France)) had F4 (cirrhosis) [Figure 1] Distribution of viral load count (IU/mL) documented in patient charts within the past 3 months varied widely across the studied countries; 5% (range: < 1% (UK) – 21% (Italy)) had an undetectable viral load (< 500 IU/mL); 35% (range: 32% (Italy) – 42% (UK)) had a viral load count of 5001 to < 1 Million IU/mL; 22% (range: 12% (Germany) – 33% (Italy)) had a viral count of >1 Million IU/ mL; 37% however had missing data (range: 14% (Italy) – 52% (Germany)) (Figure 2).
Figure 1: Distribution of Current Fibrosis Score: Percentage of Patients.
Note:Classification of liver histology per International Association for Study of the Liver (IASL): fibrosis score of F0 indicates no liver damage / no fibrosis; F1 indicates minimal liver damage / mild fibrosis; F2 indicates moderate liver damage / moderate fibrosis; F3 indicates advanced liver damage / severe fibrosis; F4 indicates cirrhosis.
Figure 2: Distribution of Viral Load Count: Percentage of Patients.
Forty two percentage of patients (range: 38% (France) – 52% (UK)) had never been treated; 21% (range: 17% (Spain) – 28% (Germany)) were currently treated; 25% (range: 20% (Germany) – 30% (Spain)) were not currently treated (but had prior treatment experience and were either non-responders or discontinued or relapsed or therapy competed and awaiting sustained viral response (SVR)); 13% (range: 6% (UK) – 19% (France)) were not currently treated as they had achieved SVR. The distribution of patient treatment status is depicted in Table 2.
Table 2: Patient's Current Treatment Status.
| France N=374 | Germany N=506 | Italy N=412 | Spain N=475 | UK N=300 | |
| Never been treated, naive | 38% | 40% | 41% | 41% | 52% |
| Currently treated: Previously treatment nave | 14% | 23% | 15% | 13% | 19% |
| Currently treated: Previous treatment non-responder, discontinued / relapsed | 9% | 5% | 5% | 4% | 1% |
| Not currently treated: Previous treatment non-responder/ discontinued/ relapsed | 17% | 14% | 27% | 26% | 19% |
| Not currently treated: Therapy completed and awaiting SVR | 4% | 6% | 2% | 4% | 3% |
| Not currently treated: SVR achieved | 19% | 12% | 11% | 13% | 6% |
Note: SVR is Sustained Viral Response
Telaprevir (TVR) plus peginterferon-a2a (IFN) and ribavirin (RBV) was the most frequently used HCV regimen used among those currently being treated (19%; range: 5% (France) – 43% (UK)), followed by IFN+RBV (19%; range: 1% (France) – 50% (Italy)), sofosbuvir plus daclatasvir (SOF+DCV; 13% (range: 1% (Italy) – 36% (France))), SOF+IFN+RBV (12% (range: 1% (UK/ Italy/Spain) – 34% (Germany))), boceprevir plus IFN+RBV (12% (range: 4% (France) – 21% (Spain))) and SOF + simeprevir + RBV (9% (range: 0% (Italy) – 31% (France))).
This research found that more than half of the HCV patients had moderate/severe liver damage or cirrhosis and only 13% of the patients had achieved SVR (and hence not treated), indicating high disease burden among this cohort of G1 patients in Europe. Compounding this burden is the observation that 42% remained treatment-naïve,and another 25% were not currently treated for other reasons. These observed rates of non-treatment varied by country. Papatheodoridis et al. [2014] had reported the proportion of untreated HCV patients across Europe to be 57% (range: 45-89%; notably, France had a non-treatment rate of 79%), while other studies have reported similar or even higher rate of non-treatment [13-16]. Our study results are directionally consistent with the past reports. Slow progressive nature of HCV infection may imply a potential public health risk of increasing late stage complications in the future unless those infected are successfully treated, as determined by the achievement of a SVR [17-19]. Sub-optimally managed (incl. lack of achievement of SVR) or untreated HCV may result in high morbidity and mortality [7, 8, 12, 19-22]. EASL guidelines recommend the primary goal of HCV therapy to be the cure of infection, associated with resolution of liver disease in patients without cirrhosis [1]. Results from this study denoting the potential suboptimal management of HCV in the studied countries in Europe hence assumes importance. Newer potent direct acting antivirals (DAAs) such as daclatasvir, sofosbuvir and simeprevir are now available to manage HCV aggressively, and several national and international guidelines recommend their use in certain situations [1, 23- 29]. While these therapies are associated with higher SVRs, healthcare systems are concerned about their widespread use on their national/regional budget and thus, the economic sustainability of their policies in HCV arena, owing to the high costs of these drugs [12, 19, 23, 24, 30]. The sporadic use of DAAs observed in this study cohort may have been influenced by these economic and access issues, besides patient characteristics (such as comorbidities, substance abuse, etc.). Further research is warranted to determine the comparative effectiveness of the observed treatments, as a means to discriminate therapeutic choices for optimal patient outcomes and foster best practices amidst a cost-conscious environment. Although physicians were randomly recruited for this study in respective geographies, the findings may represent only the participating physician practices, and may vary from those of non-participating physician practices.
CONCLUSION
In this cohort of G1 HCV patients, treatment rates were low and disease burden was high, and they varied among studied countries in EU; only a small proportion of patients were not treated owing to achieving sustained viral response. Further research is warranted to assess the factors influencing the current patient management strategies to alleviate disease burden in this patient population.
REFERENCES
- European association for the study of the liver (EASL). (2014). EASL Clinical Practice Guidelines: Management of hepatitis C virus infection. J Hepatol. 60(2), 392–420
- Hope VD, Eramova I, Capurro D and Donoghoe MC. (2014). Prevalence and estimation of hepatitis B and C infections in the WHO European Region: a review of data focusing on the countries outside the European Union and the European Free Trade Association. Epidemiol Infect. 142(2), 270-286.
- Blachier M, Leleu H, Peck-Radosavljevic M, Valla DC, et al. (2013). The burden of liver disease in Europe: a review of available epidemiological data. J Hepatol. 58(3), 593-608.
- Zein NN. (2000). Clinical significance of hepatitis C virus genotypes. Clin Microbiol Rev. 13(2), 223-235.
- Messina JP, Humphreys I, Flaxman A, Brown A, et al. (2015). Global distribution and prevalence of hepatitis C virus genotypes. Hepatology. 61(1), 77-87.
- Duffell EF, van de Laar MJ and Amato-Gauci AJ. (2015). Enhanced surveillance of hepatitis C in the EU, 2006 - 2012. J Viral Hepat. 22(7), 590-595.
- Lauer GM and Walker BD. (2001). Hepatitis C virus infection. N Engl J Med. 345(1), 41-52.
- Antiretroviral Therapy Cohort Collaboration (ATCC). (2010). Causes of death in HIV-1-infected patients treated with antiretroviral therapy, 1996-2006: collaborative analysis of 13 HIV cohort studies. Clin Infect Dis. 50(10), 1387-1396.
- Razavi H, ElKhoury AC, Elbasha E, Estes C, et al. (2013). Chronic hepatitis C virus (HCV) disease burden and cost in the United States. Hepatology. 57(6), 2164-2170.
- Mantry PS and Pathak L. (2016). Dasabuvir (ABT333) for the treatment of chronic HCV genotype I: a new face of cure, an expert review. Expert Rev Anti Infect Ther. 14(2), 157-165.
- Ciesek S, Proske V, Otto B, Pischke S, et al. (2016). Efficacy and safety of sofosbuvir/ledipasvir for the treatment of patients with hepatitis C virus re-infection after liver transplantation. Transpl Infect Dis. 18(3), 326-332.
- Maan R and van der Meer AJ. (2016). Recent advances in managing chronic HCV infection: focus on therapy in patients with severe liver disease. 5, 1-14.
- Papatheodoridis GV, Tsochatzis E, Hardtke S and Wedemeyer H. (2014). Barriers to care and treatment for patients with chronic viral hepatitis in Europe: a systematic review. Liver Int. 34(10), 1452-1463.
- Crespo J and Cabezas J. (2015). Barriers to care and treatment for patients with chronic viral hepatitis in Europe: a systematic review. Liver Int. 33, 1775.
- Patruni, Bhanu and Ellen Nolte. (2013). Hepatitis C: A projection of the healthcare and economic burden in the UK. Santa Monica, CA: RAND Corporation.
- Lettmeier B,Mühlberger N, Schwarzer R, Sroczynski G, et al. (2008). Market uptake of new antiviral drugs for the treatment of hepatitis C. J Hepatol. 49(4), 528-536.
- Razavi H, Waked I, Sarrazin C, Myers RP, et al. (2014). The present and future disease burden of hepatitis C virus (HCV) infection with today's treatment paradigm. J Viral Hepat. 21(suppl 1), 34-59.
- McEwan P, Ward T, Bennett H, Kalsekar A, et al. (2015). Estimating the clinical and economic benefit associated with incremental improvements in sustained virologic response in chronic Hepatitis C. PLoS One. 10(1), e0117334.
- Attar BM and Van Thiel DH. (2016). Hepatitis C virus: A time for decisions. Who should be treated and when? World J Gastrointest Pharmacol Ther. 7(1), 33-40.
- Lee MH, Yang HI, Lu SN, Jen CL, et al. (2012). Chronic hepatitis C virus infection increases mortality from hepatic and extrahepatic diseases: a community-based long-term prospective study. J Infect Dis. 206(4), 469-477.
- McCombs J, Matsuda T, Tonnu-Mihara I, Saab S, et al. (2014). The risk of long-term morbidity and mortality in patients with chronic hepatitis C: results from an analysis of data from a Department of Veterans Affairs Clinical Registry. JAMA Intern Med. 174(2), 204-212.
- Smith-Palmer J, Cerri K and Valentine W. (2015). Achieving sustained virologic response in hepatitis C: a systematic review of the clinical, economic and quality of life benefits. BMC Infect Dis. 15, 19.
- Miller MH, Agarwal K, Austin A, Brown A, et al. (2014). Review article: 2014 UK consensus guidelines—hepatitis C management and direct-acting anti-viral therapy. Aliment Pharmacol Ther. 39(12), 1363-1375.
- AASLD-IDSA. Recommendations for testing, managing, and treating hepatitis C.
- Crespo-Casal M. (2015). Treatment guidelines for Hepatitis C in Spain. Rev Esp Sanid Penit. 17(2), 37-47.
- Italian Association for the Study of the Liver (AISF), Coco B, Caraceni P, Aghemo A, et al. (2014). Triple therapy with firstgeneration Protease Inhibitors for patients with genotype 1 chronic hepatitis C: Recommendations of the Italian Association for the Study of the Liver (AISF). Digestive and Liver Disease. 46(1), 18-24.
- Solbach P and Wedemeyer H. (2015). The New Era of Interferon- Free Treatment of Chronic Hepatitis C. Viszeralmedizin. 31(4), 290-296.
- World Health Organization WHO. (2014). Guidelines for the screening, care and treatment of persons with Hepatitis C infection [Internet]. Geneva: WHO.
- Dore GJ. (2012). The changing therapeutic landscape for hepatitis C. Med J Aust. 196(10), 629-632.
- Edwards DJ, Coppens DGM, Prasad TL, Rook LA, et al. (2015). Access to hepatitis C medicines. Bulletin of the World Health Organization. 93(11), 799-805.