Information Links
Related Conferences
Previous Issues Volume 5, Issue 1 - 2020
Inflammatory Bowel Disease with Crohn’s-like Phenotype in Patient with Common Variable Immunodeficiency: A Case Report
Aya Hammami*, Raida Harbi, Amani Hassine, Nour Elleuch, Wafa Dahmani, Wafa Ben Ameur, Hanen Jaziri, Ali Jmaa
University of Sousse, Faculty of medecine of Sousse, Department of Gastroenterology, University Hospital of Sahloul- Sousse, Tunisia
*Corresponding author: Aya Hammami, University of Sousse, Faculty of medecine of Sousse, Department of Gastroenterology, University Hospital of Sahloul- Sousse, Tunisia, Tel: +21654868578; E-mail: [email protected]
Received Date: December 07, 2020 Published Date: December 31, 2020 Copyright: Hammami A, et al. (2020) Citation: Hammami A, et al. (2020). Inflammatory bowel disease with Crohn’s-like phenotype in patient with Common Variable immunodeficiency: A case report. Mathews J Gastroenterol Hepatol. (5)1:14.
ABSTRACT
Background: Common variable immunodeficiency (CVID) is the most prevalent type of primary immunodeficiency syndrome in adults. It is characterized by reduced levels of serum immunoglobulins responsible for recurrent bacterial infections and increased susceptibility to inflammatory affections, particularly autoimmune diseases, such as inflammatory bowel disease.
Case summary: Herein, we present the case of a 32-year-old patient with past medical history of recurrent rhinopulmonary infections, who presented with digestive symptoms consisting of chronic diarrhea, associated to abdominal pain, and fever, being diagnosed of Crohn-like disease associated to CVID. Our patient was treated with adalimumab and achieved favorable clinical response with no adverse reaction or increased susceptibility to infections. He has been in clinical remission for 2 years. The treatment with immunomodulators in this immunodeficiency status is a real therapeutic challenge. To the best of our knowledge, it is the first case reported in Tunisia.
Conclusions: We described this case to share our experience in the management of Crohn’s-like disease associated to common variable immune deficiency and to support that anti-TNF alpha could be a safe and efficient therapeutic option in similar cases.
KEYWORDS: Common Variable immunodeficiency; Inflammatory bowel disease; Crohn’s-like disease; Adalimumab
INTRODUCTION
Common variable immunodeficiency (CVID) is the most frequent symptomatic disorder of the adaptive immune system in adults that affects similarly males and females [1,2]. It is characterized by decreased serum immunoglobulin levels, resulting in an increased susceptibility to recurrent infections mainly sinopulmonary ones, higher incidence of malignant and autoimmune manifestations [3], and inflammatory conditions including gastrointestinal (GI) manifestations. Recently, emerging cases of inflammatory bowel disease (IBD) associated to CVID have been reported in the literature with an incidence of 2–4% [4]. Herein, we report the case of a patient with CVID-associated inflammatory enteropathy treated with Adalimumab with successful outcome.
CASE REPORT
A 32- year-old man, complaining of chronic diarrhea, accompanied by abdominal pain, and fever, was referred to our department. He has a past medical history of recurrent rhinopulmonary infections. His family history was unremarkable.
He was diagnosed, seven months earlier, with ileocolic Crohn’s disease (CD), complicated with severe acute colitis. The diagnosis of CD was confirmed as the other causes of colitis have been ruled out particularly infectious etiologies, since stool cultures and parasitological examination of feces were negative. Results of Clostridium Difficile testing including detection of toxin A/B in stools were also negative and there was no evidence of cytomegalovirus infection on rectal biopsies.
Thus, our patient was treated with intravenous corticosteroids with clinical and endoscopic improvement (Figure 1). Then, he was switched with oral corticosteroids and was started on Azathioprine (AZA) at the dose of 2.5mg/kg/day as maintenance therapy. Two weeks after the initiation of AZA, the patient developed papulopustular eruption on the lower limbs and forearms with no pruritus. As these lesions were compatible with AZA hypersensitivity reaction, the treatment was discontinued.
On admission, he had good general condition. Clinical evaluation was unremarkable, without any abnormal findings or ano-perineal lesions.
Laboratory tests of our patient revealed hemoglobin (Hb) 10 g/dL, white blood cell count 6020 /μL with lymphopenia of 670/mm³, platelet count 396000 /μL.
The C-reactive protein was within normal limits. Total serum proteins was 54 g/L, with albumin fraction of 30g/L. Quantitative analysis of serum immunoglobulins (Ig) revealed undetectable IgM (< 0.06 g/L), whereas IgA was< 0.066 g/L and IgG < 4.2 g/L.
Therefore, the patient was diagnosed with CVID. Flow cytometric analysis on peripheral blood lymphocytes showed 0.5% of lymphocytes B and decrease of T lymphocyte subsets.
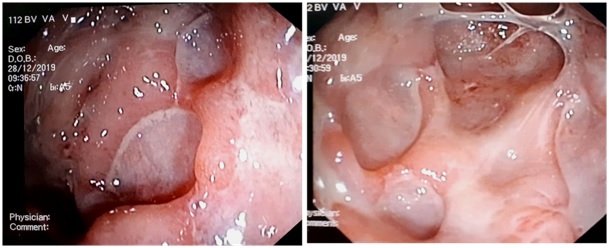
A Colonoscopy with ileoscopy was performed and showed congestive mucosa with multiple various sized longitudinal ulcerations in the terminal ileum, in the ascending colon, the sigmoid and the rectum. Histopathological examination of the samples reported chronic active inflammation in the ileum, leukocyte accumulation and granulation tissue showing architectural distortion of the crypts with no granulomas in the colon.
Upper gastrointestinal endoscopy showed no abnormalities and biopsies performed in the bulb and the duodenum excluded celiac disease.
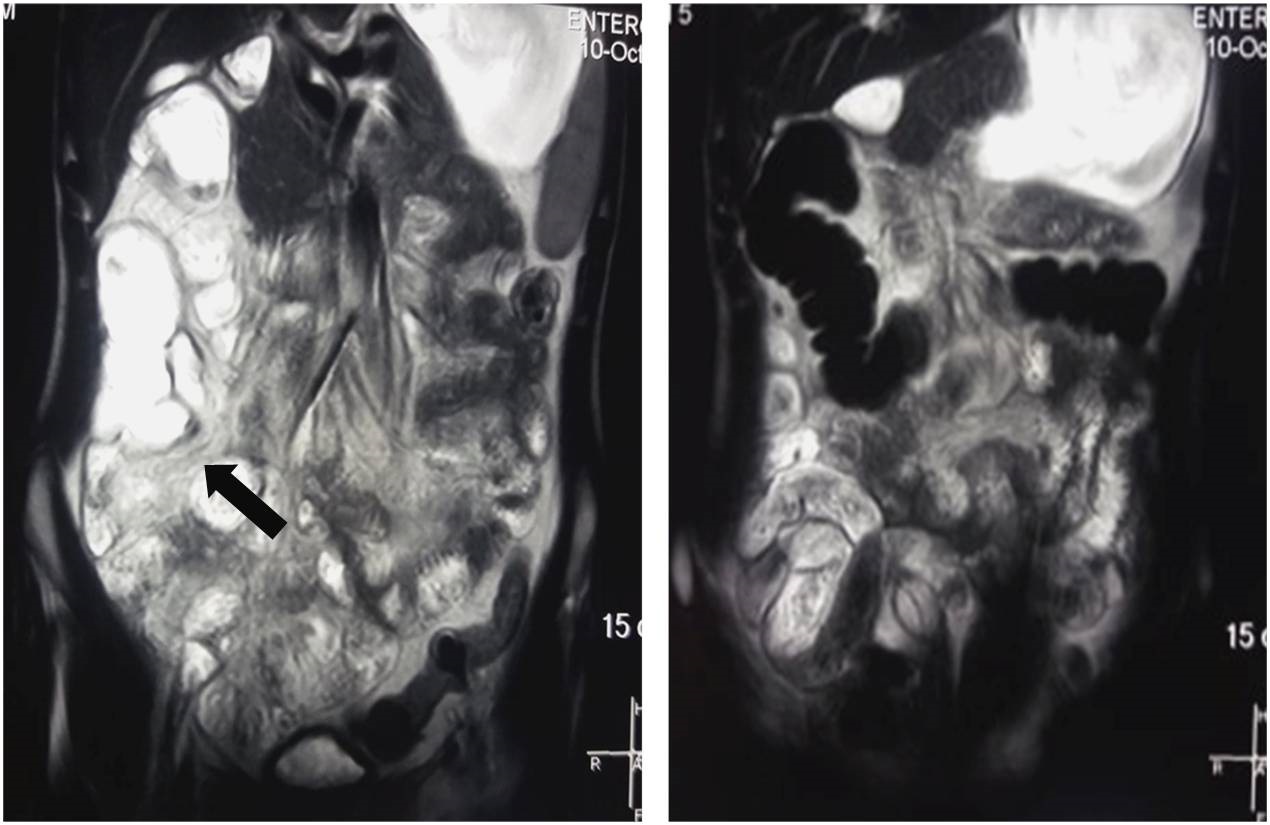
At admission, radiological study by MRI enterography was performed, revealing a diffuse and homogeneous ileal thickening with abnormal high signal and intense contrast-enhancement (Figure 2). These signs were compatible with inflammatory bowel disease at ileal and colon levels.
Figure 1: Colonoscopy images of Crohn’s-like disease demonstrating multiple deep ulcerations in the colon with edematous mucosa and loss of vascular markings.
Figure 2: Magnetic resonance (MR) enterography: Ileum thickening with submucosal enhancement (arraow).
Treatment with anti-TNF was decided. Subcutaneous injections of Adalimumab (ADA) were started with induction dose of 160 mg, 80 mg at week 2, and 40 mg at weeks 4, and then continued at the dose of 40 mg every two weeks.
After 24 weeks of ADA, the patient achieved clinical improvement in his general health and quality of life with mucosal healing at control. No side effects, namely infectious, were noticed during follow-up and the patient remained in clinical remission for two years. The follow-up visits showed a progressive increase in serum immunoglobulins.
DISCUSSION
Common variable immunodeficiency (CVID) is the most frequent primary immunodeficiency (1/25.000-1/50.000) in adults, characterized by antibody deficiency [5]. The diagnosis is based on reduced serum levels of at least two classes of immunoglobulins with lack of production of antibodies in response to infection and vaccines because of failure of B-lymphocytes differentiation from plasma cells. The underlying genetic basis of these disorders has not been well identified [6].
The clinical spectrum is heterogeneous, with specifically an increased susceptibility to infections and association to a high incidence of gastrointestinal manifestations including infectious colitis, lymphocytic colitis, collagenous enteritis, celiac diseases, and IBD-like colitis [7,8].
Actually, a dysfunction of the intestinal immune system that maintains the balance between active immunity and tolerance is thought to play the determining role in the pathogenesis of CD. Crohn’s-like disease is an inflammatory bowel disorder that may affect the gastrointestinal tract in the same way as CD, resulting in mucosal inflammation. Excluding infectious disease is very crucial and can be challenging, in CVID patients, since there is a high prevalence of bacterial infections in these patients that can show symptoms mimicking IBD [8].
It has been shown that a persistent immune activation of the tumor necrosis factor-α in a subgroup of patients with CVID, may be involved in the pathogenesis of the disease [9]. In most cases, intestinal manifestations are diagnosed after the onset of CVID. Less commonly, colitis can occur years before underlying immunodeficiency is discovered [10]. The endoscopic aspects may be similar to classical Crohn’s disease [3]. The distinction of idiopathic CD from CVID-associated CD may be based on histological findings, with absence of plasma cells that are characteristic of the latter disorder [11].
However, there is no clear evidence regarding the appropriate therapeutic options of CVID-associated inflammatory enteropathy as there have been a limited number of reported cases. It has been suggested that patients with Crohn’s-like symptoms in CVID could be treated similarly to the patients with classic IBD, by corticosteroids and immunosuppressive agents. However, the efficacy of these drugs was not well established [12].
The first two cases of CVID-associated inflammatory enteropathy, successfully treated with infliximab, were published in 2006 by Nos et al [13]. These patients did not experience an increased risk of severe infections. Since then, some papers have documented the efficacy of anti-TNF-α agents (infliximab and adalimumab) to achieve long-lasting significant improvement of severe intestinal manifestations in these patients, and without a greater risk of infections [14]. Actually, an increased production of TNF-α has been found in a subgroup of patients with CVID, suggesting that specific inhibition of TNF-α could improve CVID. Despite limited experience with biological therapy in Crohn’s-like disease associated to CVID, emerging case studies have shown significant clinical outcomes and endoscopic improvement in these patients, supporting biologic agents as a first-line treatment [4,15].
In this paper, we have described a patient with clinical and histopathologic signs of Crohn‘s disease-like and CVID. To our knowledge, it is the first case reported in our country. Our patient, treated with ADA, achieved clinical remission and mucosal healing without increased risk of bacterial infections.
This case provided evidence that Adalimumab could be a safe treatment option in such clinical circumstances. Nevertheless, the risk of developing infection is important and so caution is necessary.
STATEMENT OF ETHICS
This study did not require informed consent nor review/approval by the appropriate ethics committee.
DISCLOSURE STATEMENT
None of the authors have any conflicts of interest to declare.
FUNDING SOURCES
There were no funding sources.
AUTHOR CONTRIBUTIONS
Aya Hammami wrote the manuscript; Nour Elleuch, Hanen Jaziri : have been involved in drafting the manuscript or revising it critically for important intellectual content;Wafa Dahmani, Wafa Ben Ameur, Ali Jmaa: have given final approval of the version to be published.
REFERENCES
- Abbott JK, Gelfand EW. (2015). Common Variable Immunodeficiency: Diagnosis, Management, and Treatment. Immunology and allergy clinics of North America. 35(4):637-58.
- Bonilla FA, Barlan I, Chapel H, Costa-Carvalho BT, Cunningham-Rundles C, et al. (2016). International Consensus Document (ICON): Common Variable Immunodeficiency Disorders. The journal of allergy and clinical immunology In practice. 4(1):38-59.
- Bosworth BP, Sanders A, Maltz C. (2006). Common variable immunodeficiency masquerading as Crohn's ileocolitis. Inflammatory bowel diseases. 12(2):151-2.
- Vazquez-Moron JM, Pallares-Manrique H, Martin-Suarez IJ, Benitez-Rodriguez B, Ramos-Lora M. (2013). Crohn's-like disease in a patient with common variable immunodeficiency treated with azathioprine and adalimumab. Revista espanola de enfermedades digestivas : organo oficial de la Sociedad Espanola de Patologia Digestiva. 105(5):299-302.
- Saldana Duenas C, Rubio Iturria S. (2016). Immmunodeficiencies and autoimmune diseases: common variable immunodeficiency and Crohn-like. Revista espanola de enfermedades digestivas : organo oficial de la Sociedad Espanola de Patologia Digestiva. 108(8):520-3.
- Agarwal S, Smereka P, Harpaz N, Cunningham-Rundles C, Mayer L. (2011). Characterization of immunologic defects in patients with common variable immunodeficiency (CVID) with intestinal disease. Inflammatory bowel diseases. 17(1):251-9.
- Sanges M, Spadaro G, Miniero M, Mattera D, Sollazzo R, et al. (2015). Efficacy of subcutaneous immunoglobulins in primary immunodeficiency with Crohn's-like phenotype: report of a case. European review for medical and pharmacological sciences. 19(14):2641-5.
- Akazawa Y, Takeshima F, Yajima H, Imanishi D, Kanda T, et al. (2014). Infliximab therapy for Crohn's-like disease in common variable immunodeficiency complicated by massive intestinal hemorrhage: a case report. BMC research notes. 7:382.
- Aukrust P, Lien E, Kristoffersen AK, Muller F, Haug CJ, et al. (1996). Persistent activation of the tumor necrosis factor system in a subgroup of patients with common variable immunodeficiency--possible immunologic and clinical consequences. Blood. 87(2):674-81.
- Daniels JA, Lederman HM, Maitra A, Montgomery EA. (2007). Gastrointestinal tract pathology in patients with common variable immunodeficiency (CVID): a clinicopathologic study and review. The American journal of surgical pathology. 31(12):1800-12.
- Kalha I, Sellin JH. (2004). Common variable immunodeficiency and the gastrointestinal tract. Current gastroenterology reports. 6(5):377-83.
- Sato T, Kanai T, Watanabe M, Sakuraba A, Okamoto S, et al. (2004). Hyperexpression of inducible costimulator and its contribution on lamina propria T cells in inflammatory bowel disease. Gastroenterology. 126(3):829-39.
- Nos P, Bastida G, Beltran B, Aguas M, Ponce J. (2006). Crohn's disease in common variable immunodeficiency: treatment with antitumor necrosis factor alpha. The American journal of gastroenterology. 101(9):2165-6.
- Arieira C, Dias de Castro F, Moreira MJ, Cotter J. (2018). Common Variable Immunodeficiency-Associated Inflammatory Enteropathy: The New Era of Biological Therapy. GE Port J Gastroenterol. 25(6):322-6.
- Ruiz de Morales JG, Munoz F, Hernando M. (2017). Successful Treatment of Common Variable Immunodeficiency-associated Inflammatory Bowel Disease With Ustekinumab. Journal of Crohn's & colitis. 11(9):1154-5.