Information Links
Related Conferences
Previous Issues Volume 4, Issue 1 - 2020
Immunohistochemical studies of the Upregulation of BCL-2 and Downregulation of Bax in the Hippocampus of STZ-induced Type 1 Diabetic Rats Co-administered Melatonin and Magnesium
Elvis T. Godam1*, Wilson O. Hamman2, Sunday Oladele3, Modupeola O. Samaila4, Sunday A. Musa2
1Department of Human Anatomy, Faculty of Basic Medical Sciences, College of Medical Sciences, Rivers State University, Port Harcourt, Nigeria 2Department of Human Anatomy, Faculty of Basic Medical Sciences, Ahmadu Bello University Zaria 3Department of veterinary Pathology, Faculty of Veterinary Medicine Ahmadu Bello University, Zaria 4Department of Pathology, Faculty of Basic Clinical Sciences, Ahmadu Bello University Zaria
Corresponding Author Elvis Tams Godam, Department of Human Anatomy, Faculty of Basic Medical Sciences, College of Medical Sciences, Rivers State University, Port Harcourt, Nigeria, E-mail: [email protected]; Tel: +234-806-333-9602. Received Date: June 25, 2020 Published Date: August 20, 2020 Copyright: Godam ET, et al. ©2020. Citation: Godam ET. (2020). Immunohistochemical studies of the Upregulation of BCL-2 and Downregulation of Bax in the Hippocampus of STZ-induced Type 1 Diabetic Rats Co-administered Melatonin and Magnesium. Mathews J Cytol Histol. (4)1:14.
ABSTRACT The aim of this study was to evaluate the effects of co-administration of melatonin and magnesium on BCL-2 and Bax expression in the Hippocampus of streptozotocin-induced Type 1 diabetic Wistar rats. To achieve this aim sixty-four Wistar rats were used in the study. Streptozotocin (STZ) was used to induce chemical type 1 diabetes mellitus (T1DM) after two weeks acclimatization period in fifty-eight Wistar rats. Fifty-three rats were diabetic and fourty-eight diabetic rats were randomly distributed in eight groups and six normal normoglycaemic rats were used as control. The animals were assigned into nine groups as follows, Normal control group (NC), Diabetic control (DC) group, Melatonin Low dose group of 10 mg/kgb (MLD), Magnesium low dose group of 240 mg/kgbw (MgLD), Melatonin and magnesium combined low dose group of 10mg/kgbw + 240mg/kgbw (MMgLD), Melatonin high dose group of 20mg/kgbw (MHD), Magnesium high dose group of 480mg/kgbw (MgHD), Melatonin and magnesium high dose combined group of 20mg/kgbw + 480mg/kgbw (MMgHD) and Insulin at 500mg/kgbw group (IN). Melatonin and insulin were administered through intraperitoneal injections (IP) while magnesium was by oral administration. The control groups were given placebo and all group treatment was for twenty-one days. At the end of the study, the animals were aestheticized and euthanized to harvest the brain organ. The harvested organs were weighed and their relative weight recorded. They were fixed in neutral buffered formaldehyde (NBF). The Brain tissues were evaluated using BCL-2 mouse polyclonal antibodies to evaluate the level of neuroprotection of hippocampal neurocytes by melatonin and magnesium co-administration. The immunohistochemical test showed that melatonin and magnesium co-administration at low doses increased neuroprotection of hippocampal neurocytes by upregulation of BCL-2 protein and down regulation of Bax proteins.
Keywords: BCL-2; Bax; Diabetes; Neuroprotection; Melatonin; Magnesium
INTRODUCTION Reports published by the World Health Organization (WHO) states that diabetes mellitus (DM) is a common disease with a global prevalence estimated to be 9% among adults aged 18+ years and above [1]. Population-based studies of neuropathy in persons with diabetes indicate that neuropathy is a common complication of insulin-dependent diabetes mellitus (IDDM) and non-insulin- dependent diabetes mellitus (NIDDM), with 60%-70% of patients affected with the disease [2]. Subclinical neuropathy is much more common than clinical neuropathy. Distal symmetrical polyneuropathy is the most common type of neuropathy, followed by carpal tunnel syndrome, mono neuropathies, and autonomic neuropathy [1]. Behavioral and cognitive changes associated with type 1 diabetes mellitus (T1DM) have gained worldwide attention. Concerns about the deleterious effects of T1DM on the central nervous system have grown with the increasing incidence of T1DM in children [3]. Many studies have clearly shown cognitive and behavioral changes in type 1 diabetic rats and humans, which are evident in elevated levels of anxiety, depression, and slowing of mental speed and flexibility [4 6]. Earlier published Reports on diabetes-induced behavioral and cognitive changes are related to several factors and that diabetic complications and reduced central serotonin (5-hydroxytryptamine, 5-HT) synthesis and metabolism are thought to underlie behavioral and cognitive dysfunctions in patients with T1DM [7,8]. It has become evident that insulin and C-peptide deficiencies, including perturbations of their signaling cascades, leads to cerebral dysmetabolism and interference with the regulation of neurotropic factors and their receptors. Ultimately, this cascade of events leads to neuronal loss, causing profound deficits in behavioral and cognitive functions [9]. However, the specific mechanisms underlying these changes and whether they relate to the duration of diabetes are unknown. Although the magnitude of most of these cognitive decrements is relatively modest, even moderate behavioural and cognitive changes can potentially hamper the day-to-day activities of a diabetic child and an adult. These cognitive decrements may present problems in more demanding situations, and critically, can have a negative impact on the quality of life [9].
Melatonin, Magnesium and Diabetes Mellitus Melatonin is a circulating neuro-hormone secreted predominantly at night. It is important in conveying the daily cycle of light and darkness to the body, thus regulating circadian rhythms. In addition to its' regulatory role, melatonin has antioxidative capacity, immunomodulatory potency, and also appears to be protective against certain types of cancers [10]. Type 1 diabetes mellitus is a T cell-mediated autoimmune disease characterized by excess inflammation, independent of adiposity and glycemic control. Melatonin has also been demonstrated to have hypoglycemic effect, pancreatic Beta cells regeneration and up regulation of intracellular antioxidant (GPx, SOD and CAT) and reduction in intracellular free radicals generated Malondialdehyde (MDA) in Streptozotocin (STZ) induced diabetes in wistar rats [11]. Magnesium (Mg) is an electrolyte of chief physiological importance in the body, being the most abundant divalent intracellular cation in the cells, the second most abundant cellular ion next to potassium and the fourth cation in general in the human body [12]. Type 2 diabetes mellitus (T2DM) is often accompanied by alteration of Mg status. An increased prevalence of Mg deficits has been identified in diabetes mellitus patients, especially in those with poorly controlled glycemic profiles, with longer duration of the disease and with the presence of micro and macrovascular chronic complication [13]. Magnesium deficiency may also be a factor implicated in diabetes mellitus complications. It was found that there is a relationship between ionic changes and echocardiographic indices alterations. Further observation is a significant association of reduced cellular Mg with cardiac hypertrophy in diabetes mellitus patients [14,15]. The occurrence of type 1 diabetes mellitus is increasing at 3-5% per year worldwide [16] and this increase cannot be accounted for by known genetic factors. It is well known that both oxidative stress, inflammatory activity and neurodegeneration with cognitive decrement play crucial roles in the pathogenesis of T1DM. Little information is available on the different patterns of type 1 diabetes progression following diagnosis. The research aims to study the upregulation of BCL-2 and down regulation of Bax using immunohistochemical stains (antibody) in the Hippocampus of STZ-induced type 1 Diabetic rat’s administered melatonin and magnesium.
MATERIALS AND METHODS Materials The following materials were used in the study, Plastic Cages, Spectrophotometer auto analyzer, blood sample containers, organ sample containers, Centrifuge, Temperature controlled refrigerator, Microwave oven, water bath, humidity chamber, Leica Auto processor, Leica Auto Stainer, Leica DM750, Camera ICC50 E, AmScope D200 digital camera, MRC spectrophotometer. Bioactive Compounds And Drugs Used In The Study Were Melatonin M5250-1G (Sigma Aldrich USA), Magnesium (Randox, USA) Streptozotocin SP0130 (Sigma Aldrich, USA), BCL-2 Antibody (Bioss, USA) and Bax Antibody (Bioss USA) were used in the study. Source of Animals and Management Sixty-four Male wistar rats weighing of 120–150g, were purchased from the Faculty of Pharmaceutical Sciences Animal House of the Ahmadu Bello University, Zaria and selected for the study. The rats were maintained on a day and night cycle at room temperatures and have free access to food (Standard feeds, standard rat pellets) and water. All experiments were performed between 08:00 and 12 hours daily.
METHODOLOGY Induction of Type 1 Diabetes Mellitus Type 1 diabetes mellitus (T1DM) was induced after two weeks acclimatization period; baseline blood glucose was performed for all test animals. This was done to ensure that the animals were all normoglycaemic to remove. Fifty-Eight male wistar rats were randomly selected and given a single dose of intra peritoneal injection of streptozotocin, (STZ) (Sigma, Aldrich, USA), at 55mg/kg body weight in citrate buffer (0.1M, pH 4.5). The solution (STZ in citrate buffer) was used within 5 minutes to induce chemical diabetes in the wistar rats after an overnight fast for twelve hours. Hyper Glycaemia Screening and Confirmation of T1DM Four days after streptozotocin was used to induce diabetes mellitus, blood was collected from the tail vein following an overnight fast [17 20]. Fasting blood sugar (FBS) was measured with a standard glucometer (Optimum, Germany). The day that hyperglycaemia above 200mg/dl (11 mmol/l) was confirmed was considered to be diabetic day one. Rats with fasting blood glucose levels lower than 200 mg/dL (11mmol/L) were excluded from the study. Anti-Bcl-2 Polyclonal Antibody Anti-BCL-2 polyclonal antibody (Bioss antibodies-bs-4563R) was applied for the study of anti-apoptotic activity in neuronal cells for neuroprotection by melatonin and magnesium in diabetic treated animals. METHODS The brain tissue section was fixed in neutral buffered formaldehyde and embedded in paraffin wax. Antigen retrieval was by citrate buffer (0.1M, pH 6.0) and was placed in boiling bath for 40 minutes. Endogenous peroxidase was blocked by 3% hydrogen peroxide for seven minutes. It was further incubated with unconjugated polyclonal Bcl-2 antibody (bs-4563R), Bioss Antibodies Massachusetts USA, in 1: 400 dilutions for 20 minutes. It was later labeled with Mouse/Rabbit Horseradish enzyme (DB Biotech, Slovakia) for 30 minutes. DAB was for 10 minutes (C-0010) and later stained with haematoxylin for nuclear contrast. Anti Bax Polyclonal Antibody Anti Bax polyclonal antibody (bs-0127R) was used to study the level of apoptosis in diabetic control, melatonin, magnesium and insulin treated groups, antibody from Bioss antibodies (Massachusetts USA) in the cerebral cortex of all animals. Method The brain tissue was fixed in normal buffered formaldehyde and embedded in paraffin wax: antigen retrieval was by citrate buffer (0.1M, pH 6.0) and was placed in boiling water bath for 90 minutes. Endogenous peroxidase was blocked by 3% hydrogen peroxide for 30 minutes and later blocked using buffer (normal goat serum, C-0005) AT 37°C for 20 minutes.
It was further incubated with Rabbits/Mouse Anti-Bax Polyclonal Antibody, unconjugated with (bs-0127R) from Bioss Antibodies Massachusetts USA at 1: 400 dilutions for 20 minutes. It was later conjugated to the secondary antibody (SP-0023) and DAB staining for 10 minutes (C-0010) and later stained with hematoxylin for nuclear contrast [21].
|
Groups |
Treatment |
|
NC |
Normal control + Normal saline (orally) |
|
DC |
Diabetic control + Normal saline (orally) |
|
M |
Diabetic + Melatonin (10mg/kgbw), (IP) |
|
Mg |
Diabetic + Magnesium (240mg/kgbw), (Orally) |
|
MMgLD |
Diabetic + Melatonin (10mg/kgbw) (IP) + Magnesium (240mg/kgbw), (orally) |
|
MHD |
Diabetic + Melatonin(20mg/kgbw) (IP) |
|
MgHD |
Diabetic + Magnesium (480mg/kgbw) (orally)) |
|
MMgHD |
Diabetic + Melatonin(20mg/kgbw) (IP) + Magnesium (480mg/kgbw), (orally) |
|
IN |
Diabetic + Insulin (500mg/kgbw), (IP) |
Table 1: Experimental groupings, treatment, route of administration and durations. Wistar rats were randomly divided into the following groups of six rats in each as shown in the table. NC (control), DC (Diabetic control), MLD (Melatonin low dose), MgLD (Magnesium low dose), MMgLD (Melatonin and magnesium low dose), MgHD (Magnesium high dose), MHD (Melatonin high dose), MgHD (Magnesium high dose), MMgHD (Melatonin and Magnesium high dose), IN (Insulin). n = 6 rats per group, melatonin route of administration = intraperitoneal, Magnesium = oral, Insulin = intraperitoneal, Treatment duration = once daily for 21 days.
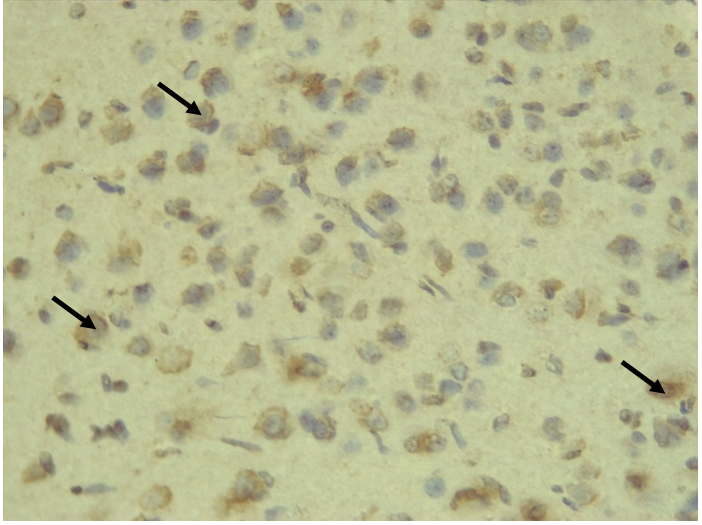
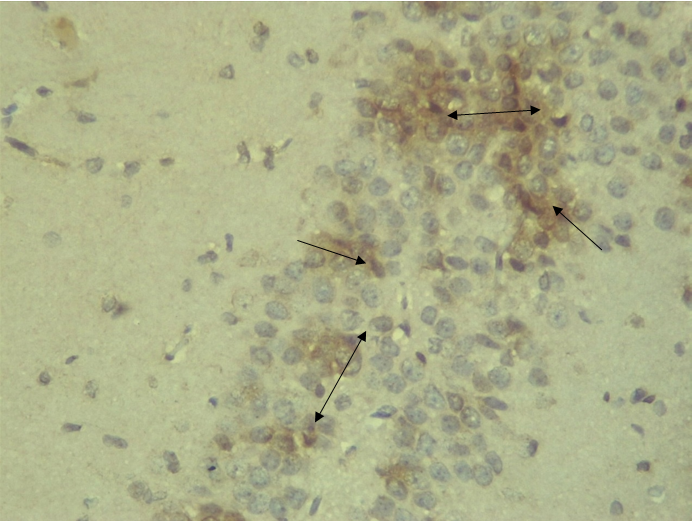
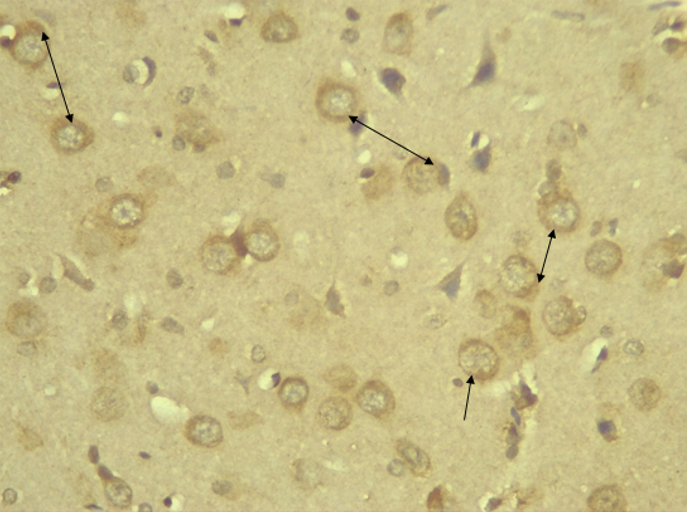
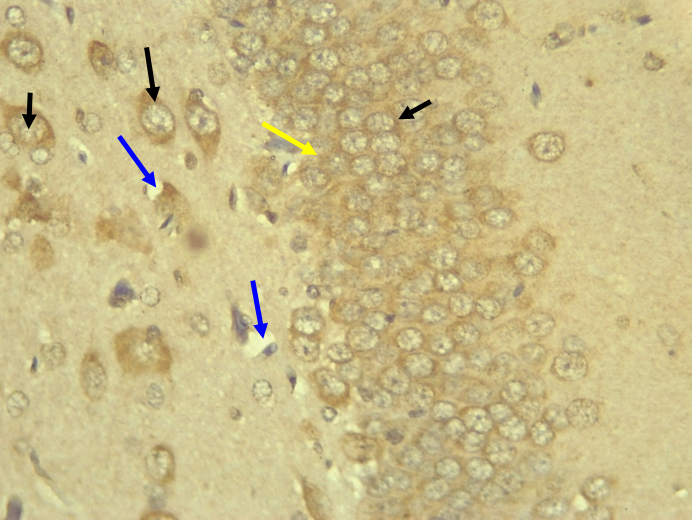
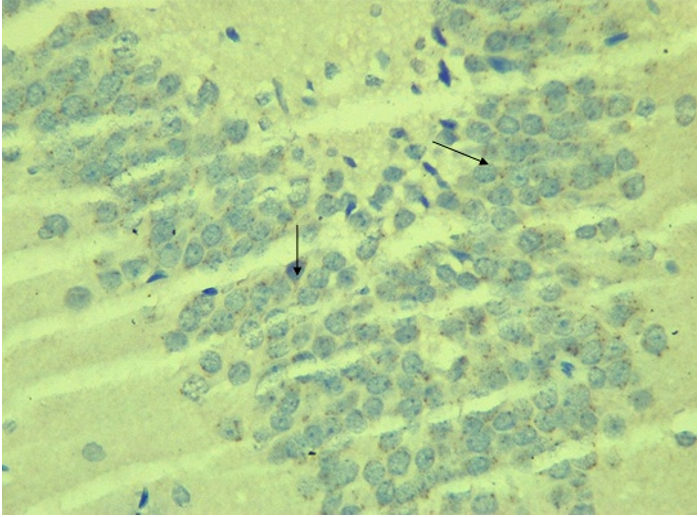
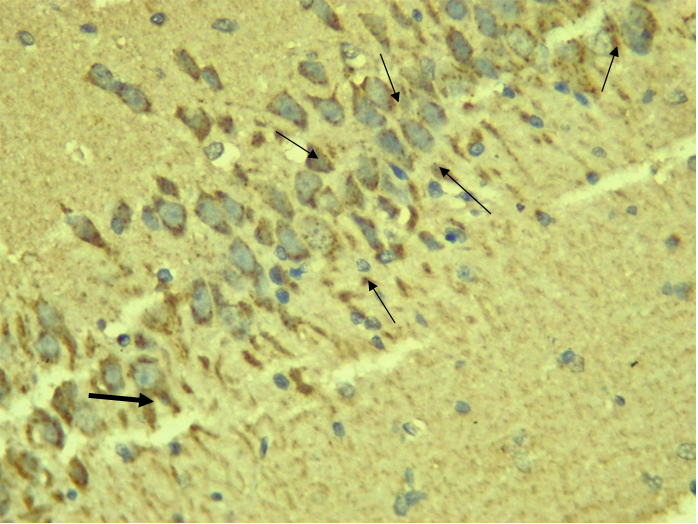
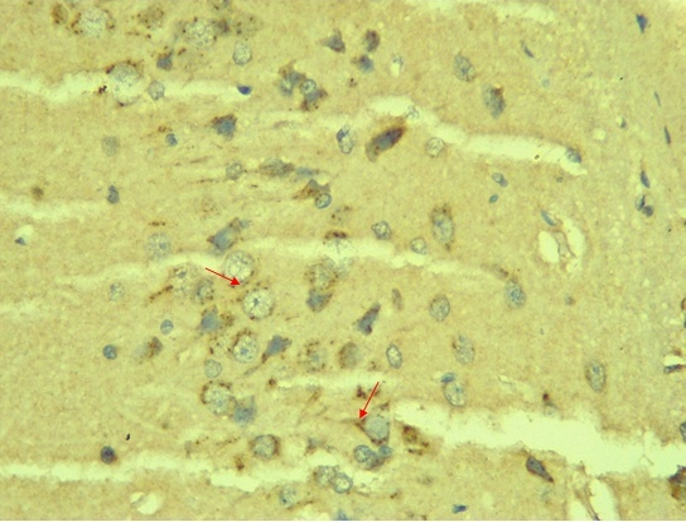
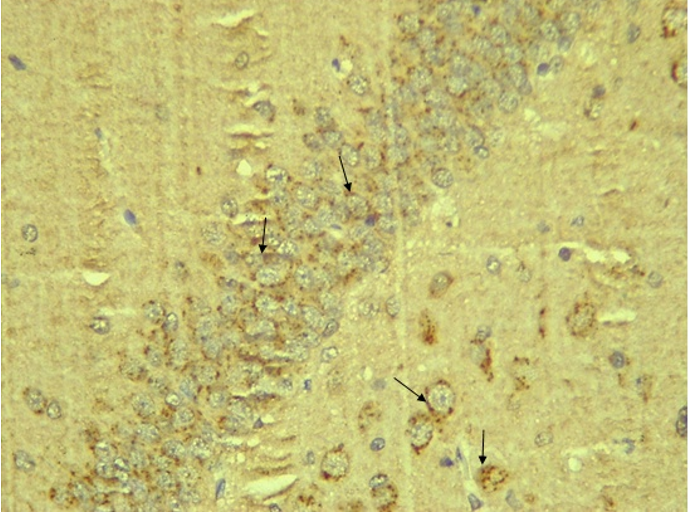
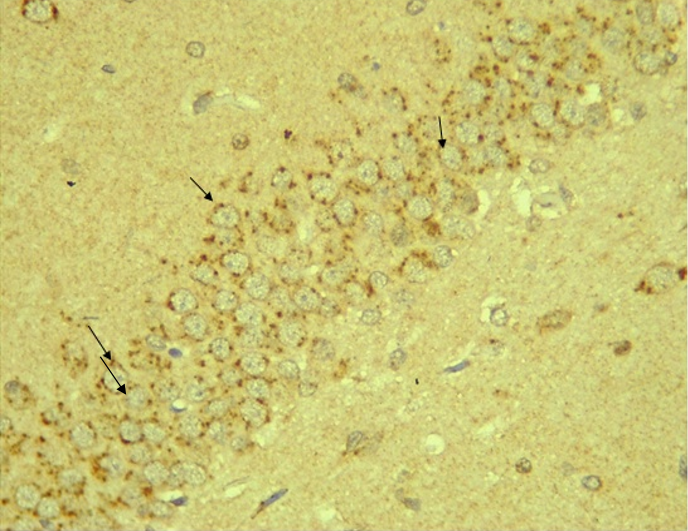
RESULTS Neuroprotection using BCL-2 Polyclonal Antibody (IHC) In the figure 1A, photomicrograph section of normal control rats (NC Group) showing normal BCL-2 expression in the neuronal cells and tissues of the hippocampus. While in figure 1B photomicrograph section of Diabetic control rats (DC group) with no BCL-2 expression in the neuronal cells and tissues of the hippocampus. There is pyknosis and increase in pericellular spaces in the neuronal cells signifying cell death. Microglial cells are enlarged. In the figure 1C, photomicrograph of a section of the brain tissue of STZ induced diabetic rats treated with melatonin at 10 mg/kgbw (MLD Group), showing BCL-2 positive expression in the hippocampus pyramidal and neuronal cells in area CA3 reversing apoptosis. In figure 1D, photomicrograph of a section of brain tissue of STZ induced diabetes treated with magnesium at 240 mg/kgbw (MgLD group), showing no BCL-2 expression in the hippocampus pyramidal and neuronal cells in the molecular layer, hence apoptosis as shown in cellular pyknosis and enlargement of microglial cells. In figure 1E, photomicrograph of a section of brain tissue of STZ induced diabetes rats (MMgLD group) treated with melatonin (10 mg/kgbw) and magnesium treated (240 mg/kgbw) showing BCL-2 positive expression in the hippocampus pyramidal layer in area CA1 and hence reversing apoptosis. In figure 1F, photomicrograph of a section of brain tissue of STZ induced Diabetic rats treated with melatonin (20 mg/kgbw) (MHD Group), showing +2 BCL-2 expression in the pyramidal layer, molecular layer and pleomorphic layer of the CA3 areas of the hippocampus. There is decrease in pyknosis and pericellular spaces in the neuronal cells signifying less apoptosis and neuroprotection (red arrows) and pyramidal cell degeneration. In figure 1F, Photomicrograph of a section of brain tissue (molecular layer of the hippocampus) showing mild membrane and cytoplasmic BCL-2 expression in MgHD group treated with (480 mg/kgbw) in STZ induced diabetic wistar rats. In figure 1G, photomicrograph of a section of brain tissue of STZ induced diabetes rats (molecular layer of the hippocampus) showing mild membrane and cytoplasmic BCL-2 expression in MMgHD treated with melatonin (20 mg/kgbw) and magnesium (480 mg/kgbw) in STZ induced diabetic wistar rats. In figure 1H, photomicrograph of a section of brain tissue (molecular layer of the hippocampus) showing mild membrane and cytoplasmic BCL-2 expression with pyknosis and more glial cells expansion in IN Group treated with Insulin (500mg/kgbw) in STZ induced diabetic wistar rats.
.PNG)
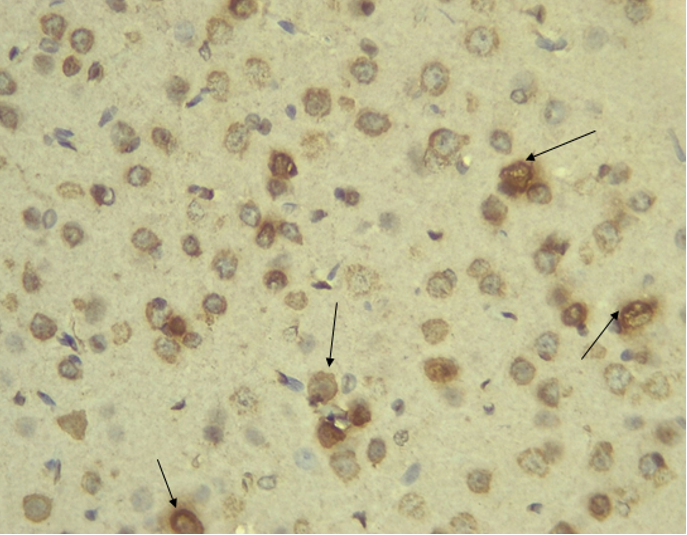
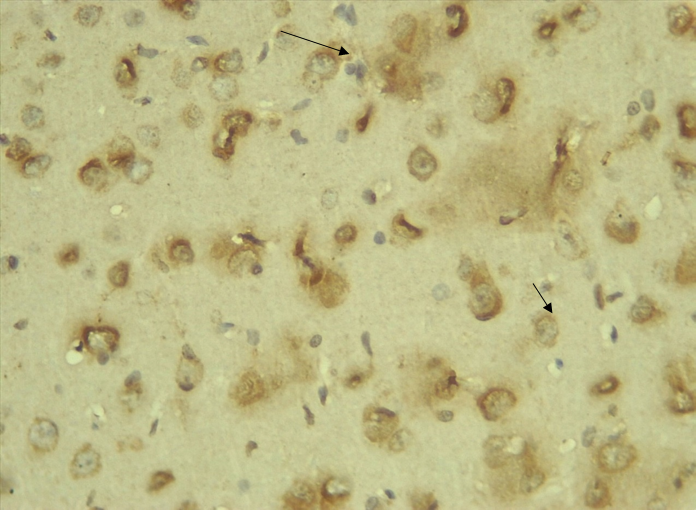
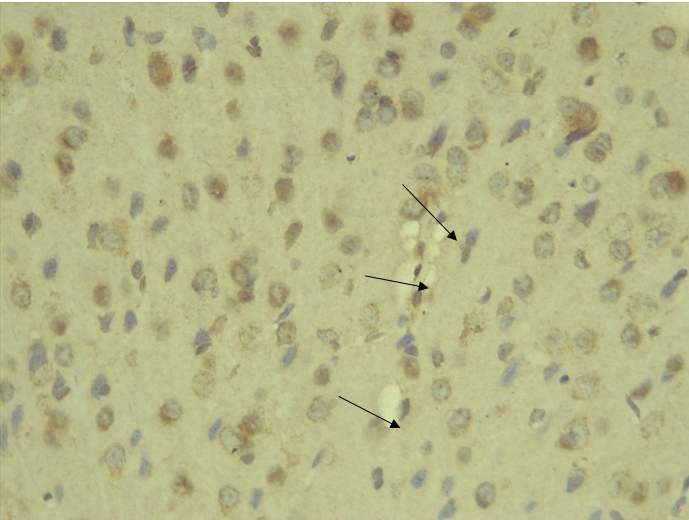
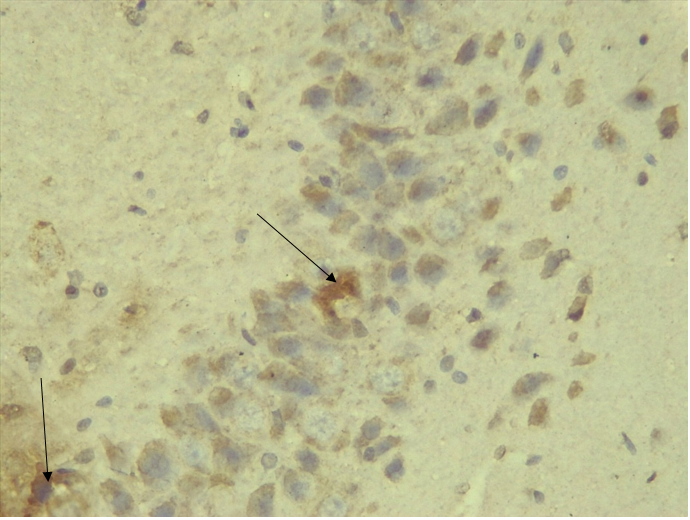
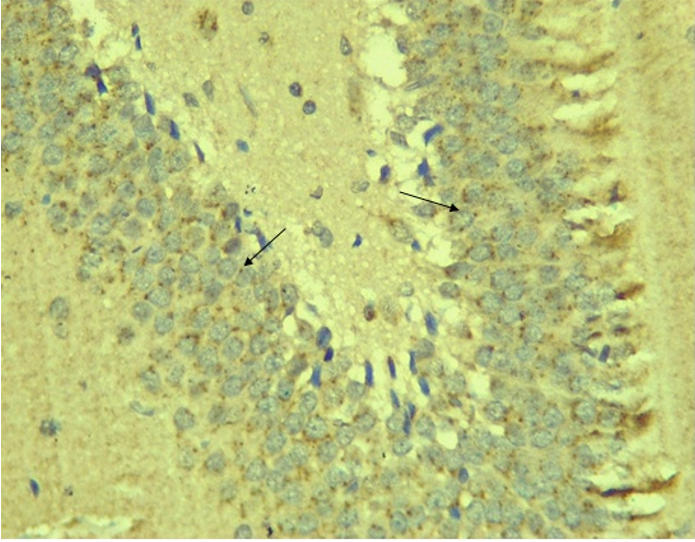
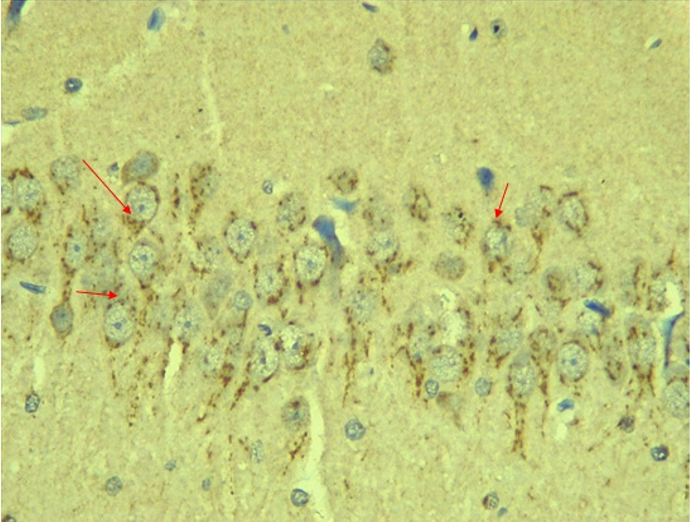
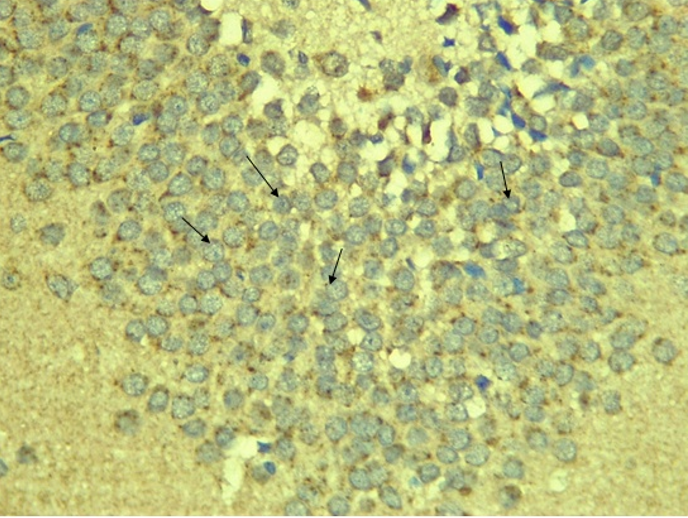
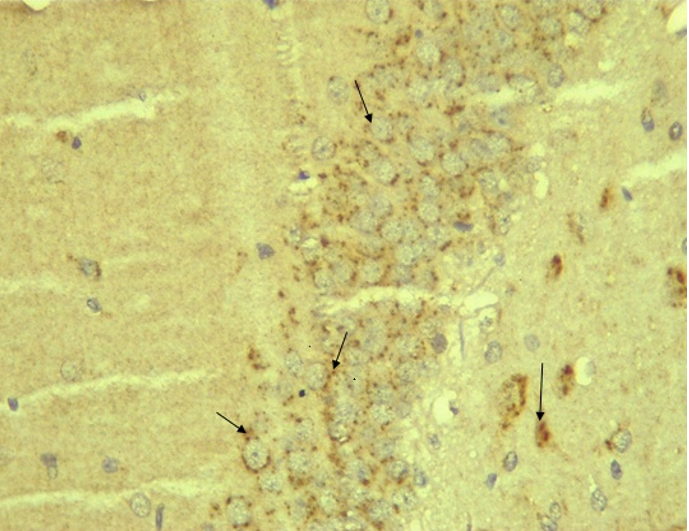
Immunohistochemical Diabetic Neuropathy Studies of Apoptosis in Hippocampal Neurocytes Using Bax In figure 2A, photomicrograph section of the dentate gyrus of the hippocampus in Normal Control Group (NC Group) administered oral normal saline showing negative Bax expression in the neuronal cells (black arrows). This signifies no apoptotic expression in normal control brain tissues. Mild but not significant Bax expression was obtained in figure 2C photomicrograph section (MLD Group) of the brain tissues of STZ induced diabetic wistar rats treated with melatonin (10 mg/kgbw) showing no significant nuclear membrane positive Bax expression (black arrows) in the Dentate Gyrus of the hippocampus, figure 2E, Photomicrograph section of Brain tissue from STZ induced diabetic wistar rats (MMgLD Group) co-administered melatonin (10 mg/kgbw) and magnesium treated (240 mg/kgbw) showing no significant Bax nuclear membrane expression (red arrows) in CA1 area of the Hippocampus and in figure 2F photomicrograph section from brain tissues of STZ induced diabetic wistar rats (MHD Group) treated with melatonin (20 mg/kgbw) showing no significant nuclear membrane positive Bax expression (black arrows) in the Dentate Gyrus of the hippocampus. Figure 2B, photomicrograph section of diabetic control group (DC Group) of STZ induced diabetic rats showing very significant (p ˂ 0.05) Bax positive nuclear membrane expressions (black arrows) in the hippocampus CA1 area and in the cerebral cortex of wistar rats. Similar results were obtained from (MgLD Group) magnesium treated STZ induced diabetic rats at 240 mg/kgbw (Figure 2D) and magnesium treated STZ induced diabetes rats at 480 mg/kgbw, MgHD Group figure 2G and in figure 2H (MMgHD group) the combined dose of melatonin and magnesium at 20 mg/kgbw and 480 mg/kgbw respectively to STZ induced diabetic rats. Figure 2I (IN Group) insulin treated at 500mg/kgbw also showed mild decrease in Bax cytoplasmic expression in neurocytes of STZ induced diabetic rat but was not significant.
DISCUSSION The hippocampus is one of the rare areas of the brain that exhibit neurogenesis. The new neurons created by the hippocampus are important for memory and learning and required brain-derived neurotropic factors (BNDF). BCL-2 is an antiapoptotic protein that suppresses apoptosis. It is localized mainly in the mitochondrial membrane and plays an important role in protecting tissues from apoptotic cell death [22,23]. In the cerebral cortex and especially the hippocampus, the neurocytes in the control group showed positive membranous and cytoplasmic BCL-2 expression as opposed to the negative neurocytes BCL-2 expression in the diabetic control untreated group. The treated melatonin at low and high doses shows neuroprotection in positive BCL-2 expression. Positive membranous and cytoplasmic BCL-2 expression was also observed in the melatonin and magnesium combined group. Mild insignificant BCL-2 expression was observed in magnesium treatment group at low and high doses and when combined with melatonin at high dose. Insulin treated group showed mild neuroprotection in BCL-2 expression. These present observations are consistent with Zang YC, et al. [24] who reported that “hyperglycaemia could induce oxidative stress in the hippocampus, and works by Contreras-Alcantara S, et al. [25] also showed that hyperglycaemia also could results to apoptosis of hippocampal synapses and neuronal loss as observed in the diabetic control group where the above was observed. Apoptosis is a genetically controlled process that occurs both in normal and pathological conditions, [26]. Tang D, et al. [27], showed that there are two apoptotic pathways, which are intrinsic and extrinsic pathways. The extrinsic pathway involves cell surface receptors, while the intrinsic pathway utilizing mitochondria [27]. Apoptosis plays a major role in the development of the central nervous system. Several physiological death signals as well as pathological intracellular damage signals triggers the genetically programmed pathway of apoptosis [28]. Some genes regulating the pathway of apoptosis such as bax and bcl-x, promotes apoptosis, whereas others such as bcl-2 and ced-9 regulates antiapoptotic pathways. It is the balance of these genes in individual neurons that ultimately results in either cell survival or apoptosis [29]. Thus, in this study, we were able to correlates bax positive cytoplasmic and membranous expressions in the diabetic control group where there was significantly high bax expression in the neurocytes of the hippocampus and cerebral cortex. This was due to significantly high hyperglycemic index which negatively affects the intracellular antioxidants (SOD and CAT), increased neurobehavioral and memory deficits and a significantly increase in apoptosis shown as significantly high positive bax expression in diabetic control group hippocampus and cortical neurocytes. The melatonin treatment at low and high doses and when combined with magnesium showed significant negative bax expression in the neurocytes of the hippocampus and cerebral cortical cells similar to the normal control group. Melatonin regulates glucose homeostasis according to reports by Owino S, et al. [30] and that it inhibits apoptosis mediators MHS Pathways and promotes pro-survival BCL-2 Protein expression that down regulates Bax activities. These results are consistent with meta-analysis reviews on subtle cognitive deficits in pediatrics by Gudieri P, et al. [31] and in adults by Brands AM, et al. [32] population with type 1 diabetes. Basic intelligence, psychomotor processing speed, mental flexibility and attention are specific skills noted to be permanently reduced in children with type 1 diabetes [33]. Further reports collaborated the debilitating effects of diabetes stating that children with type 1 diabetes before the onset of five years of age have permanent neurocognitive impairments with age matched peers and siblings [34]. Hence, supporting our result neurocognitive deficits and memory is associated with type 1 diabetes and that melatonin and magnesium attenuates these deficits and cause neurogenesis which further enhanced memory and behavior in STZ induced type 1 diabetic rats.
CONCLUSION Melatonin and magnesium co-administration to streptozotocin induced type 1 diabetic wistar rats upregulates BCL-2 proteins expression showing evidence of neuroprotection of the hippocampal neurocytes and the down regulation of Bax protein expression decreasing neurocytes apoptosis. Thus, our research affirmed the antioxidant nature of the two bioactive agents (melatonin and magnesium) synergistically working to attenuate the neuropathological changes associated with diabetes mellitus as evidenced in this experimental model study in wistar rats.
REFERENCES 1. Bytzer P, Talley NJ, Leemon M, Young LJ, Jones MP, et al. (2001). Prevalence of Gastrointestinal Symptoms associated with Diabetes Mellitus: a population-based survey of 15,000 adults. Archives of Internal Medicine. 161:1989-1996. 2. Ricci JA, Siddique R, Stewart WF, Sandler RS, Sloan S, et al. (2000). Upper gastrointestinal symptoms in a U.S. national sample of adults with diabetes. Scandinavian Journal of Gastroenterology. 35:152-159. 3. Adeghate E, Schattner P, Dunn E. (2006). An update on the Etiology and Epidemiology of Diabetes Mellitus. Annual New York Academic Science. 1084:1–29. 4. Ramanathan M, Jaiswal AK, Bhattacharya SK. (1998). Differential effects of diazepam on anxiety in streptozotocin induced diabetic and non-diabetic rats. Psychopharmacology Journal of Berlin. 135(4):361–367. 5. Kuhad A, Chopra K. (2007). Curcumin attenuate diabetic encephalopathy in rats. Behavioral and biochemical evidences. European Journal of Pharmacology. 576(1–3):34–42. 6. Alvarez EO, Beauquis J, Revsin Y, Banzan AM, Roig P, et al. (2009). Cognitive Dysfunction and Hippocampal Changes in Experimental Type-1 Diabetes. Behavioral Brain Research. 198(1):224–230. 7. Ryan CM, Williams TM. (1993) Effects of insulin-dependent diabetes on learning and memory efficiency in adults. Journal of Clinical Experimental Neuropsychoology. 15(5):685–700. 8. Thorre K, Chaoulof F, Sarre S, Meeusen R, Ebinger G, et al. (1997). Differential effects of restraint stress on hippocampal 5-HT metabolism and extracellular levels of 5-HT in streptozotocin-diabetic rats. Brain Research Journal. 772(1–2):209–216. 9. Sima AA, Zhang W, Muzik O, Kreipke CW, Rafols JA, et al. (2009). Sequential abnormalities in type 1 diabetic encephalopathy and the effects of C-Peptide. Revised Diabetic Studies. 6(3):211–222. 10. Zawilska JB, Skene DJ, Arendt J. (2009). Physiology and pharmacology of melatonin in relation to Biological rythms. Pharmacology and Reproduction. 61:383-386. 11. Godam ET, Samaila MOA, Ibegbu AO, Hamman WO. (2014). Effects of Melatonin and Azadiracta indica administration on serum antioxidant parameters in streptozotocin induced diabetic Wistar rats. Annals of Biological Sciences Journal. 2(3):56-62. 12. Barbagallo M, Dominguez LJ. (2006). Magnesium metabolism in Type-2 Diabetes Mellitus, Metabolic Syndrome and Insulin Resistance. Archive of Biochemistry and Biophysics Journal. 458:40-47. 13. Ramadass S, Basu S, Srinivasan AR. (2015). Serum magnesium levels as an indicator of status of Diabetes Mellitus type 2. Diabetes Metabolism Syndrome. 9:42-45. 14. Barbagallo M, Di Bella G, Brucato V, D'Angelo D, Damiani P. (2014) Serum ionized magnesium in diabetic older persons. Metabolism. 63:502-509. 15. Barbagallo M, Gupta RK, Resnick LM. (1996). Cellular ions in NIDDM: relation of calcium to hyperglycemia and cardiac mass. Diabetes Care. 19:1393-1398. 16. Barbagallo M, Di Bella G, Brucato V, D'Angelo D, Damiani P. (2014). Serum ionized, magnesium in diabetic older persons. Metabolism. 63:502-509. 17. Meiri N, Chelardini C, Tesco G, Galeotti, N, Dahl D, et al. (1997). Reversible antisense inhibition of Shaker-like Kv1.1 potassium channel expression impairs associative memory in mouse and rat. Proc National Academic Science Journal. 94(9):4430–4434. 18. Walther T, Balschun D, Voigt JP, Fink H, Zuschratter W, et al. (1998). Sustained long term potentiation and anxiety in mice lacking the Mas protooncogene. Journal of Biology and Chemistry. 273(19):11867–11873. 19. Kulkarni SK. (1999). Hand Book of experimental pharmacology. Vallabh Prakash Delhi (IN), 3rd ed. 85-89. 20. Godam ET, Samaila MOA, Ibegbu AO, Hamman WO, et al. (2014). Histological effects of Melatonin andAzadirachtaIndicaadministration on the pancreatic tissue in Streptozotocin-Induced Diabetic Wistar Rats. Annals of Biological Sciences Journal. 2(2):27-35. 21. Hegazy R, Hegazy A. (2015) Hegazy’ Simplified Method of Tissue Processing (Consuming Less Time and Chemicals). Annals of International Medical and Dental Research. 1(2):57-6. 22. Yaun J, Yankner BA. (2000). Apoptosis in the nervous systems. Nature Journal. 407:802-809. 23. Chen C, Liu TS, Zhao SC, Yang WZ, Chen ZP, et al. (2018). BCL-2 family protects mitochondrial membrane. Journal of experimental and therapeutic medicine. 15:4587-4593. 24. Zhang YC, Pileggi A, Agarwal A, Molano RD, Powers M, et al. (2003). Adeno-associated virus-mediated IL-10 gene therapy inhibits diabetes recurrence in syngeneic islet cell transplantation of NOD mice. Diabetes Journal. 52:708–716. 25. Contreras-Alcantara S, Baba K, Tosini G. (2010). Removal of melatonin receptor type1 induces insulin resistance in the mouse. Obesity (Silver Spring). 18:1861-1863. 26. Yang Y, Chen S, Pan H. (2017). Safety and efficiency of SGLT2 inhibitor combining with insulin in subjects with diabetes: systematic review and meta-analysis of randomized controlled trials. Medicine (Baltimore). 96:e6944. 27. Tang D, Kang R, Berghe VT, Vandenabeele P, Kroemer G. (2019). The molecular mechanism of cell death. Journal of Cell Research. 29:347-364. 28. Gross A, McDonnell JM, Korsmeyer SJ. (1999). The BCL-2 family members and the mitochondria in apoptosis. Journal of Gene Development. 13(15):1899-1911. 29. Antonsson B, Conti F, Clavatta A. (1997). Inhibition of Bax channel-forming activity by Bcl-2. Science. 277(5324):370-372. 30. Owino S, Contreras-Alcantara S, Baba TG. (2016). Melatonin signaling controls the daily rythms in blood glucose levels independent of peripheral clocks. PLOS ONE. 11(1):eo148214. doi:L10.137/journal.pone.0148214. 31. Gaudieri P, Chen R, Greer T, Homes C. (2008). Cognitive functions in children with type 1 diabetes. Journals of Diabrtic Care. 31:1892-1897. 32. Brands AM, Biessels GJ, De Haan EH, Kappelle LJ, Kessels RP. (2005). The effects of type 1 diabetes on cognitive performance: A meta-analysis. Diabetes Care Journal. 28(3):726 –735. 33. Ryan C.M, Becker DJ. (1999). Hypoglycaemia in Children with type 1 diabetes mellitus: risk factors, cognitive function, and management. Journal of Endocrinology and metabolism clinics of North America. 28(4):883-900. 34. Hyllienmark L, Maltez J, Dandenell A. (2005). EEG abnormalities with and without relation to severe hypoglycaemia in adolescents with type 1 diabetes. Diabetologia. 48:412-419.