Information Links
Related Conferences
Previous Issues Volume 5, Issue 1 - 2021
Fine Needle Aspiration Cytology Findings of Breast Lesions in Female Patients Presenting with Palpable Breast Lumpsat Makerere University College of Health Sciences, Kampala-Uganda
Kinalwa Sulaiman1, Abila Derrick Bary2, and Mwesigwa Boaz3*
1Department of Pathology, School of biomedical sciences, College of Health Sciences, Makerere University, Kampala, Uganda
2Faculty of Biology, Medicine and Health, University of Manchester, United Kingdom
3Cytology Society of Uganda, Kampala, Uganda
*Corresponding Author: Mwesigwa Boaz, Cytology Society of Uganda, Kampala, Uganda, Tel: +256-705-482-996; E-mail:[email protected].
Received Date:January 28, 2021 Published Date:March 20, 2021
Copyright: Sulaiman K, et al. © (2021).
Citation: Sulaiman K, Bary AD, Boaz M. (2021). Fine Needle Aspiration Cytology Findings of Breast Lesions in Female Patients Presenting with Palpable Breast Lumpsat Makerere University College of Health Sciences, Kampala-Uganda. Mathews J Cytol Histol. 5(1):16.
ABSTRACT
Background: Palpable breast lumps are the common manifestation encountered by physicians duringclinical practice.The use of fine-needle aspiration cytology (FNAC)methodhas proven to be quick, simple and cost-effective in theevaluation ofthese lumps for benign, atypical and malignant changes. This study aims to determine the type of breast lesions diagnosed by FNAC and to determine the age-wise distribution for such lesions.
Methodology: A prospective study consisting of 291 femalespresenting with palpable breast lesions was carried out at Makerere College of Health Sciences (MakCHS) pathology laboratory from January 2019 to May 2019. FNAC results were grouped into tier fivereportingcategories as C1, C2, C3, C4 & C5 in accordance to the International Academy of Cytology (IAC).
Results: Out of the 291 cases examined, 14(4.8%) were insufficient (C1), 192(66%) were benign (C2), 9(3.0%) were atypical (C3), 15(5.2%) were suspicious of malignancy (C4) &61(21%) were malignant (C5). In the benign category, lesions of fibroadenoma were the most commonly diagnosed constituting 110/192 (57.3%) whereas under the malignant category lesions of invasive ductal carcinoma were the most commonly diagnosed constituting 27/61(44.3%). The peak agegroup for benign lesions was21-30 years whereas the peak age group for malignant lesions was 41-50 years.
Conclusion: Fine-needle aspiration cytology (FNAC) was found to be an effective diagnostic tool in the categorization of palpable breast lumps into benign, malignant, atypical, suspicious and inadequate categories.
KEYWORDS: Breast cancer; Fine needle aspiration cytology; Palpable breast masses
ABBREVIATIONS: ADH: Atypical ductal hyperplasia; AUS: Atypia of undetermined significance; CBE: Clinical Breast Examination; FA: Fibroadenoma; FCC: Fibrocystic changes; FNAC: Fine Needle Aspiration Cytology; IAC: International Academy of Cytology; IDC: Invasive Ductal Carcinoma; MakCHS: Makerere University College of Health Sciences; SPSS: Statistical Package for Social Sciences.
NTRODUCTION
Breast lump is the localized swelling, protuberance or mass in the breast and is one of the prime indicators of breast disorders. A lump might be a cyst, a benign tumour or a malignancy [1].Symptom misinterpretation, misdiagnosis and late presentation at the primary health care level are reported as one of contributors to the increased progression to breast cancer [2].Globally, over 2 million females arediagnosed with breast canceraccounting foran excess of 626,000 deaths [3]. In 2018, the age-standardized incidence rate and mortality rate for breast cancer in Uganda was 21.3 per 100,000 and 10.3 per 100,000 populations, respectively [4]. These high mortality rates were attributed to a late advanced stage disease diagnosis of women with breast cancer. Up to 89% of the population presenting with a stage of the disease that was too difficult to treat [5]. The recommended approach for the early detection and diagnosis of breast lumps involves the use of triple assessment which includes; clinical breast examination (CBE), imaging (ultrasound or mammography) and fine-needle aspiration cytology [6].Fine-needle aspiration cytology (FNAC) has been in use since the 1930s and is simple, quick and a cost-effective method of diagnosing palpable breast lumps [7]. Whereas this method offers a reliable means of diagnosis during the assessment of breast diseases, its utility in this country is restricted to a few public or private health institutions and limited data is available regarding the prevalence of breast diseases in Uganda. The aim of this study was to document the type of breast diseasesdiagnosed by FNAC at the MakCHS pathology laboratory over a period of 5months (January–May, 2019). Furthermore this study aimed at determining the age-wise distribution of breast diseases at the pathology laboratory over the said period.
MATERIALS AND METHODS
Study Design
The study was a cross-sectional prospective study.
Study Area
This study was conducted at the cytology clinic of the department pathology of Makerere University College of Health Sciences (MakCHS). It is located at Mulago National Referral and Teaching Hospital in Kampala. It is one of fewest public laboratories that offer cytology services.
Study Duration
The study was carried out for five months starting from January 2019 to May 2019.
Study Population
The study population consisted ofall female patients that presented with palpable breast lumps.
Sample Size Estimation
For calculation of sample size, the formula below was used [8];
N= Z2P(1-P)/d2
where Z is the value of 1.96 (z in normal distribution curve),
n is the required sample size,
p is the estimated prevalence, and disprecision error (5%).
A prevalence p of 25.5% [9] was used in the estimation of sample size, n.
Using this equation, a total sample size of 291individuals was computed.
Selection Criteria
Inclusion criteria: All female patients who presented with palpable breast lumps. Patients those who were able togive consent for the study.
Exclusion criteria: The patients with an already known malignancy were excluded. Patients who were severely sick hence couldn’t undergo FNAC procedure.
SAMPLE COLLECTION TECHNIQUE, PROCESSING AND REPORTING
Verbal consent was taken after explanation of the procedure and its benefits to the patient. In the FNAC technique, equipment used involved glass slides, disposable gloves, fixatives (70%, 95% alcohol), swabs, 23 gauge needle and 10 ml syringes.The patient was made to lie supine on an examination couch after exposure of upper half of the body. Gloving of hands was done by the resident pathologist in order to perform a breast examination. This examination involved; an assessment of the size of the breast, overlying skin appearance, quadrants and presence or axillary lymph nodes. Thereafter the skin of the breast over the lump was disinfected with 70% alcohol in a swab.
The lump was immobilised and stabilised between the index finger and the thumb of the left hand. A 23gauge needle attached to a 10 ml syringe was inserted into the nodule. The plunger was retracted to create a vacuum in the needle for suction. Backward and forward movements were used under constant suction with the needle moved at different depths and angles within the confines of the nodule. The process was terminated when fluid appeared in the hub of the needle. The plunger was released to prevent aspiration of the material into the syringe. The needle was removed from the nodule and syringe detached. The syringe was re-attached after withdrawing the plunger and air was used with the needle tip close to the glass slide, the sample was expressed on the slide. At least two passes were made in two different quadrants of the breast lump. With a sterile swab, pressure was applied over the FNA site for about five minutes.
The aspirated material was smeared on a slide labelled with the patient’s laboratory number and another labelled slide was placed on the smear to evenly and thinly spread it between the two slides on pulling them apart, this made two smears per pass and therefore four smears per patient.Two slides were air dried whereas the other two were fixed immediately by immersion into 95% alcohol. The air-dried smears were stained with May-Grunwald-Giemsa (MGG) staining technique and the smears fixed in 95% alcohol were stained with Papanicolaou staining technique. The attending cytotechnologist took part in the assessment of specimen adequacy.
The cytological diagnoses were categorized into five standardized categories in accordance to International academy of cytology reporting criterion for breast cytology; Category I (Insufficient material), Category II (Benign), Category III (Atypical), Category IV (Suspicious, probably in situ or invasive) & Category V (Malignant) [10].The breast samples from FNAC were further sub-classified into definite breast entities.
DATA ENTRY AND ANALYSIS
The data collected was entered using Epidata software and exported to the Statistical Package for Social Sciences(SPSS)version 17.0 for analysis.
RESULTS
Out of 291 female patients presenting with palpable breast lesions at the MakCHS pathology laboratory, the category-wise distribution was: 14 (4.8%) insufficient (C1),192 (66%) benign (C2), 9 (3.0%) atypical (C3), 15 (5.2%) suspicious of malignancy (C4) &61 (21%) malignant (C5). Thus the ratio of benign to malignant lesions was 3:1.In the benign category, fibroadenoma was the most commonly diagnosed lesion constituting110/192 (57.3%) followed by fibrocystic changes constituting of 22/192 (11.5%). In the malignant category, invasive ductal carcinoma was the most commonly diagnosed lesion constituting 27/61 (44.3%) followed byInvasive ductal carcinoma of No Specific type (NST) constituting 14/61 (23.0%).The peak age group for benign lesions was 21-30 years whereas the peak for malignant lesions was 41-50 years.These findings were summarized in table 1, table 2 and table 3 respectively.
|
IAC Classification |
Category |
Frequency |
|
Insufficient |
C1 |
14 (4.8%) |
|
Benign |
C2 |
192 (66%) |
|
Atypical |
C3 |
9 (3.0%) |
|
Suspicious of Malignancy |
C4 |
15 (5.2%) |
|
Malignant |
C5 |
61 (21%) |
|
Total |
|
291 |
Table 1: Category-wise distribution of cases in accordance to International Academy of Cytology.
|
Cytological result |
Frequency (%) |
|
|
|
|
Insufficient C1 |
n=14 |
|
No epithelial cells or histiocytes |
14(100.0) |
|
|
|
|
Benign C2 |
n=192 |
|
Acute mastitis |
7(3.6) |
|
Chronic mastitis |
8(4.2) |
|
Cystic degeneration |
8(4.2) |
|
Duct ectasia |
4(2.1) |
|
Fibroadenoma (FA) |
110(57.3) |
|
Fibroadenoma with chronic mastitis |
1(0.5) |
|
Fibrocystic changes (FCC) |
22(11.5) |
|
Galactocele |
9(4.7) |
|
Lactating adenoma |
1(0.5) |
|
Lipoma |
2(1.0) |
|
Normal breast aspirate (Negative for Malignancy) |
18(9.4) |
|
Tuberculous mastitis |
2(1.0) |
|
|
|
|
Atypical C3 |
n=9 |
|
Atypical ductal cells of undetermined significance (AUS) |
1(11.1) |
|
Atypical ductal hyperplasia (ADH) |
8(88.9) |
|
|
|
|
Suspicious of Malignancy C4 |
n=15 |
|
Low- grade ductal carcinoma in situ (Nuclear grade 1) |
1(6.7) |
|
High-grade ductal carcinoma in situ (Nuclear grade 3) |
14(93.3) |
|
|
|
|
Malignant C5 |
n=61 |
|
Adenocarcinoma of Breast |
10(16.4) |
|
Invasive ductal carcinoma (IDC) |
27(44.3) |
|
Invasive ductal carcinoma of No Specific type (NST) |
14(23.0) |
|
Invasive ductal papilloma |
9(14.8) |
|
Metaplastic carcinoma |
1(1.6) |
Table 2: Type-specific distribution of cases as diagnosed by FNAC.
|
Age-group |
Insufficient(C1) |
Benign(C2) |
Atypical(C3) |
Suspicious(C4) |
Malignant(C5) |
Total |
|
11-20 |
6 |
25 |
- |
- |
- |
31 |
|
21-30 |
5 |
70 |
- |
- |
- |
75 |
|
31-40 |
3 |
52 |
1 |
- |
5 |
61 |
|
41-50 |
- |
20 |
2 |
- |
33 |
55 |
|
51-60 |
- |
15 |
2 |
3 |
10 |
30 |
|
61-70 |
- |
8 |
1 |
6 |
6 |
21 |
|
71-80 |
- |
2 |
3 |
6 |
7 |
18 |
|
Total |
14 |
192 |
9 |
15 |
61 |
291 |
Table 3: Age-wise distribution of cases according to breast categories.
DISCUSSION
Breast lumps are a common clinical presentation in women presenting with breast symptoms. Whether benign or malignant lumps cause anxiety and depression to the patient [11]. Nature of breast lump can be established by a triple test which includes clinical breast examination, imaging and fine-needle aspiration cytology [12]. FNAC is extremely recognized as a reliable procedure for the initial examination of palpable breast masses. It is minimally invasive, cost-effective, safe, simple, rapid and sensitive as compared to biopsy thus reduces the number of open breast biopsies [13].
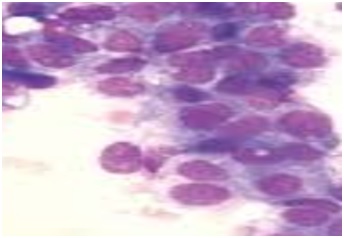
In our study, 4.8% cases had inadequate aspirates and were placed in C1 category which was in concordance with studies done by Sudrat N, et al. [14] whereas Bajwa and Tariq (2010) [15] had a slightly higher rate (13.6%) of C1 cases. An “inadequate” report was issued as due to scanty or a cellular material with in the aspirates, Benign lesionsunder the C2 category were mostcommonly diagnosed with a percentage of 66% (192 cases) was congruent to the study done by Sunita H, et al. [16] andby Ahmed AG, et al. [17]. Fibroadenoma (Figure 1A & Figure 1B) and fibrocystic changes (Figure 2A & Figure 2B) were the most common benign lesion observed in this study similar to that found by Sankaye SB, and Dongre SD (2014) [18]. In this study, atypical lesions in the category C3 comprised of 3% (9 cases) were in agreement to a study done by Sudarat N, et al. [19]. Smears in this category showed ductal epithelial cells benign in nature with overcrowding and mild nuclear enlargement. In the C3 categoryatypical ductal cells of undetermined significance (AUS) was the most diagnosed lesions followed by atypical ductal hyperplasia (ADH). This study showed 5.2% (15cases) in suspicious for malignancy category (C4). Other studies showed variable results in C4 category, namely Bajwa R, et al. [20] in their study found 10.3% in 464 cases and Ibrahim Y, et al.[21] found 13.5% in 66 cases.Smears in this category showed cells. In the category C5, malignant lesions accounted for 21% (61 cases) which was in concordance to the study done by Ibrahim Y, et al. [21]. Invasive ductal carcinoma (figure 3) was the most commonly reported lesionwhich was also found by Bukhari MH, et al.[22].There was a predominance of benign lesions in an age group of 21-40 years and increased malignancy in the age group of 41-50 years. Similar findings were found by Shirish SC, et al.[23] and Almobarak AO, et al. [24].
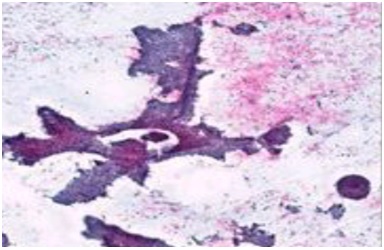
Figure 1A:Fibroadenoma showing papillary-like pattern (Papanicolaou stain, ×50).
Figure 1B:Fibroadenoma showing cohesive groups of ductal epithelial cells Admixed with myoepithelial cells (MGG, ×200).
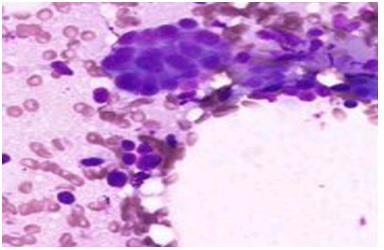
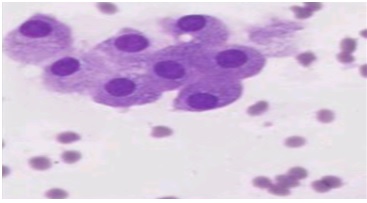
Figure 2A: Fibrocystic change showing apocrine metaplasia (MGG, ×400).
Figure 2B: Fibrocystic changes showing apocrine metaplastic cells in a Honey-comb architecture (Papanicolaou stain, ×200).
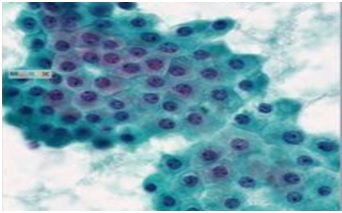
Figure 3: Invasive ductal carcinoma of breast showing coarse chromatin pattern and intranuclear inclusions (MGG, ×200).
LIMITATIONS OF THE STUDY
The occurrence of unsatisfactory (inadequate) smears in 14 cases was a limitation tothis study. This was mainly due the collagenous nature of the lesions and some lesions were less than 1cm in size which caused difficulties in sampling. In future studies, this may be improved through performing FNAC under ultrasound guidance.
CONCLUSION AND RECOMMENDATIONS
This study demonstrated a predominance of benign lesions in young age (reproductive-age) and an increased malignancy in older age (post-menopausal). Fine-needle aspiration cytology (FNAC) was also found to be an effective diagnostic tool in thecategorization ofpalpable breast lumps into benign, malignant, atypical, suspicious and inadequate categories.
DATA AVAILABILITY
Data is available in hard copies and can be accessed on request.
CONFLICT OF INTEREST
Nil
FUNDING
There was no source of funding for this study.
ETHICAL APPROVAL
The permission to carry out this study and disseminate its findings was obtained from the head of department pathology of Makerere University College of Health Sciences.
CONSENT
Informed consent was obtained from the study participants before they were enrolled in the study.
AUTHORS’ CONTRIBUTIONS
Kinalwa Sulaiman designed the study, participated in slide preparation and interpretation collected data, managed and analyzed it. Abila Derrick Bary was responsible for data analysis, manuscript development, and reading of manuscript. Mwesigwa Boaz was responsible for drafting the manuscript. All authors have read and approved the final manuscript.
REFERENCES
- Shah Ta, Shrestha M. (2008). Prevalence of Breast Lump and Risk Factors of Breast Cancer among Reproductive Aged Women of Jabalpur VDC of Sunsari District, Nepal. Journal of Nepal Health Research Council. 2:1.
- Olayide A, SamuelO, Ganiyu R, Julius O, Oladapo K, et al. (2019). Impact of Primary Care Delay on Progression of Breast Cancer in a Black African Population: A Multicentered Survey. Journal of Cancer Epidemiology. 2407138. doi: 10.1155/2019/2407138.
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, et al. (2018). Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin.68:394-424.
- International Agency Research on Cancer, Global cancer observatory: cancer today. 2018. Available from: https://www.iarc.fr/today. [Accessed May 5, 2018].
- Galukande M, Wabinga H, Mirembe F. (2015). Breast cancer survival experiences at a tertiary hospital in sub-Saharan Africa: a cohort study. World J SurgOncol. 13:220.
- Ankit S, Abhinav J, Kalayansunda S. (2019). Epidemiological Study of Breast Lump Cases. International Journal of Contemporary Surgery. 7(1).
- Das DK. (2003). Fine-needle aspiration cytology: its origin, department, and present status with special reference to a developing country, India. DiagnCytopathol. 28(6):345-51.
- Mohamad A, Mohsen V, Mitra R. (2013). Sample size calculation in medical studies. GastroenterolHepatol Bed Bench. 6(1):14-17.
- David E, John A, Olufemi O. (2017). Fine needle aspiration cytology of breast lumps with histopathologic correlation in Owo, Ondo State, Nigeria: a five-year review. Ghana Med J. 51(1):1-5.
- Field A, Raymond W, Rickard M, Arnold L, Brachtel E, et al. (2019). The International Academy of Cytology Yokohama System for Reporting Breast Fine-Aspiration Biopsy Cytopathology. ActaCytol. 63(4):257-273.
- Keyzer-Dekker CMG, van Esch L, de Vries J, Ernst MF, Nieuwenhuijzen GAP, et al. (2012). An abnormal screening mammogram causes more anxiety than a palpable lump in benign breast disease. Breast Cancer Res Treat. 134(1):253-258.
- Ngotho J, Githaiga J, Kaisha W. (2013). Palpable discrete breast masses in young women: two of the components of the modified triple test may be adequate. S Afr J Surg. 51(2):58-60.
- Nguansangiam S, Jesdapatarakul S, Tangjitgamol S. (2009). Accuracy of fine needle aspiration cytology from breast masses in Thailand. Asian Pacific Journal of Cancer Prevention.10(4):623–626.
- Sudarat N, Somneuk J, Siriwan. (2009). Accuracy of fine needle aspiration cytology from breast masses in Thailand. Asian Pac J Cancer Prev. 10:623-6.
- Bajwa R, Tariq Z. (2010). Association of fine needle aspiration cytology with tumor size in palpable breast lesions. Biomedica. 26:124-9.
- Sunita H, Urmila, Sharma DC. (2015). Cytomorpholoogical study of breast lesions with sonomammographic correlation. J Evol Med Dent Sci. 4:137-42.
- Ahmed HG, Ali AS,Almobarak AO. (2009). Utility of fine-needle aspiration as a diagnostic technique in breast lumps. DiagnCytopathol. 37(12):881-4.
- SankayeSB, Dongre SD. (2014). Cytological study of palpable lumps presenting in an indian rural setup. Indian J Med PaediatrOncol.35(2).
- Sudarat N, Somneuk J, Siriwan T. (2009). Accuracy of Fine Needle Aspiration Cytology from Breast Masses in Thailand. Asian Pacific Journal of Cancer Prevention. 10:623-626.
- BajwaR, Zulfiqar T. (2010). Association of Fine Needle Aspiration Cytology with Tumor Size in Palpable breast Lesions. Biomedica. 26:124-129.
- Ibrahim Y, Atanda AT. (2014). Validity of fine needle aspiration cytology of the palpable breast lesions: A teaching hospital experience. Nigerian Journal of Basic and Clinical Sciences. 11(1):36-40.
- Bukhari MH, ArshadM, Jamal S, Niazi S, Bashir S, et al. (2011). Use of fine-needle aspiration in the evaluation of breast lumps. Pathology ResInt. 2011:689521.
- Shirish SC, Kanika G, Arpana AD, Sukanya P, Archana CB, et al. (2014). Pattern of palpable breast lesions on fine needle aspiration: A retrospective analysis of 902 cases. J Midlife Health. 5(4):186-191.
- Almobarak AO, Elhassan TM, Elhoweris MH, Awadalla HM, Elmadhoun WM, et al. (2014). Cytomorpholoogic patterns of breast lesions in Sudanese patients: Lessons learned from fine needle aspiration cytology. Asian Pacific Journal of Cancer Prevention. 15:3411-3413.