Information Links
Related Conferences
Previous Issues Volume 7, Issue 3 - 2023
Exocrine Pancreatic Insufficiency in a Dog: Case Report
Fábia Luvisetto do Nascimento1, André Rinaldi Fukushima2,3,4,*, Juliana Weckx Peña Muñoz1, Vinícius dos Santos Fernandes Conceição1, Bruno Nascimento Lopes2, Steffany Teles de Novais2, Esther Lopes Ricci2,3,5, Maria Aparecida Nicoletti6, Simone Rodrigues Ambrósio7
1Nosso Vet Veterinary Hospital, São Paulo, Brazil
2School of Veterinary Medicine and Animal Science, University of São Paulo, São Paulo, Brazil
3Faculdade de Ciencias da Saúde IGESP, São Paulo, Brazil
4University Center of the Americas–FAM, São Paulo, Brazil
5Mackezie Presbyterian University, São Paulo, Brazil
6Faculty of Pharmaceutical Sciences, University of São Paulo, São Paulo, Brazil
7São Judas Tadeu University, São Paulo, Brazil
*Corresponding author: André Rinaldi Fukushima, School of Veterinary Medicine and Animal Science, University of São Paulo, São Paulo, Brazil, Tel: (11) 98133-7311, ORCID: 0000-0001-6026-3054; E-mail: [email protected].
Received Date: July 14, 2023
Published Date: July 27, 2023
Citation: do Nascimento FL, et al. (2023). Exocrine Pancreatic Insufficiency in a Dog: Case Report. Mathews J Vet Sci. 7(3):24.
Copyrights: do Nascimento FL, et al. © (2023).
ABSTRACT
Exocrine pancreatic insufficiency is a gastrointestinal illness marked by insufficient production of digestive enzymes by pancreatic acini, resulting in signs of poor digestion, malabsorption and malnutrition. It is considered less common in small animals' clinical routine, being more diagnosed in female dogs of the German Shepard, Collie and Cavalier breeds due to genetic predisposition. Therefore, it is reported in this work a case of exocrine pancreatic insufficiency in a male French bulldog. The animal was treated in a private clinic in São Paulo-SP with the main complaint of weight loss, appetite disorders (polyphagia, coprophagy, and abnormal appetite), steatorrhea and flatulence. The serum trypsin-like immunoreactivity test in conjunction with ventral ultrasound has confirmed the diagnosis. Treatment based on enzyme replacement with pancreatin, antibiotic therapy, gastric protection, and vitamin supplementation and diet changes proved to be totally effective after thirty days, because the animal showed significant weight gain, normorexy, normal defecation and improvement of other clinical signs.
Keywords: Canine, Pancreas, EPI, TLI, Pancreatin.
ABBREVIATIONS
IF: Intrinsic Factor; EPI: Exocrine Pancreatic Insufficiency; TLI: Trypsin-Like Immunoreactivity; APA: Acinar Pancreatic Atrophy.
INTRODUCTION
The pancreas is an organ located in the right epigastric region, consisting of a left lobe near the greater curvature of the stomach and a right lobe positioned next to the duodenum. It is considered a mixed gland with both endocrine and exocrine functions. The endocrine function regulates metabolism through the production of hormones such as insulin, glucagon, and somatostatin, while the exocrine function secretes pancreatic juice composed of enzymes, bicarbonate, water, and intrinsic factor (IF), responsible for the digestion and absorption of nutrients and cobalamin. Pancreatic juice is produced in the pancreatic acini and secreted by the main pancreatic duct [1,2].
Exocrine pancreatic insufficiency (EPI) is characterized by inadequate production of digestive enzymes by the pancreatic acini, resulting in poor digestion, malabsorption, and malnutrition [3,4]. Acinar pancreatic atrophy (APA) is considered the main cause of EPI in dogs, but end-stage chronic pancreatitis, racial predisposition mainly in German Shepherds, Collies, and Cavaliers [5], and rarely neoplasms are also observed. It is believed that the disease manifests only when 90% of enzyme production and acinar tissue are compromised. The most observed clinical signs are polyphagia, parorexia, coprophagia, soft stools, steatorrhea, increased fecal volume, borborygmus, flatulence, weight loss, seborrhea, and muscle atrophy [1,2,4].
Together with amylase and lipase, trypsin is one of the enzymes produced by the exocrine pancreas and present in pancreatic juice. It is converted into trypsinogen in the intestine and is of great importance in protein digestion [2,6]. Animals with EPI do not produce sufficient trypsin, and as a result, diagnosis is made through the measurement of serum trypsin-like immunoreactivity (TLI), a test with high specificity and sensitivity. For dogs, the reference value of TLI is between 5.0 and 32.0 ng/mL, and values below 2.5 ng/mL confirm the disease [7,2-4]. It is important to note that amylase and lipase measurements are not used because these enzymes are also produced in other organs unlike trypsin, and typically, hematological, and biochemical test results are within normal limits [2,4,6]. Abdominal ultrasound should also be performed as it allows visualization of changes in size, echotexture, echogenicity, and the presence of nodules or acoustic shadowing due to pancreatic fibrosis [8].
The treatment for EPI is mainly based on enzyme supplementation with pancreatin for an indefinite period, which is crushed pig pancreas provided in powder or capsule form along with food [5-7]. Fresh pancreas can also be used, but it is not recommended due to the possibility of gastrointestinal infection [2,6]. Gastric pH decreases the enzymatic activity of lipase and trypsin. Therefore, the administration of proton pump inhibitors such as omeprazole, one to two times a day for continuous use, is necessary [2,7]. The use of antibiotics such as metronidazole aims to prevent intestinal bacterial overgrowth caused by the loss of antimicrobial factors in pancreatic juice or the increased availability of undigested substrates in the intestine [2,5]. Decreased or absent intrinsic factor causes hypocobalaminemia, so vitamin supplementation should also be part of the treatment [1-3]. The dietary recommendation for dogs with EPI consists of diets based on hydrolyzed protein with high digestibility, low fiber content, and a higher quantity of B-complex vitamins [1,2]. Low-fat content was previously indicated, but it has been found to directly interfere with the animal's weight gain and is therefore only used as a last resort to control steatorrhea [5].
The prognosis varies according to the etiology and overall condition of the animal [8], but the majority of dogs show a good response in the first weeks of treatment, with weight gain and control of other clinical manifestations, resulting in a favorable prognosis [2,7]. The objective of this study is to report a case of exocrine pancreatic insufficiency in a French Bulldog, highlighting the importance of definitive diagnosis and proper treatment for the clinical improvement of the patient.
CASE REPORT
A 3-year-old intact male French Bulldog dog was presented in July 2020 at a private veterinary clinic located in São Paulo, Brazil, with the main complaint of lethargy, progressive weight loss, polyphagia, coprophagia, parorexia, soft stools without foul odor and yellowish coloration, and flatulence. The owner reported that the clinical signs began approximately 2 years ago, coinciding with the transition from puppy to adult food, with alternating periods of soft stools and diarrhea. The patient had been examined by several colleagues who were unable to determine the cause of the clinical manifestations. Additionally, the owner reported that the dog's vaccinations and deworming were up to date and performed ethically. The dog lived in an apartment and had access to the street only for occasional walks. Renal and hepatic biochemical tests were within the reference values for the canine species, and a negative fecal examination for parasites was obtained.
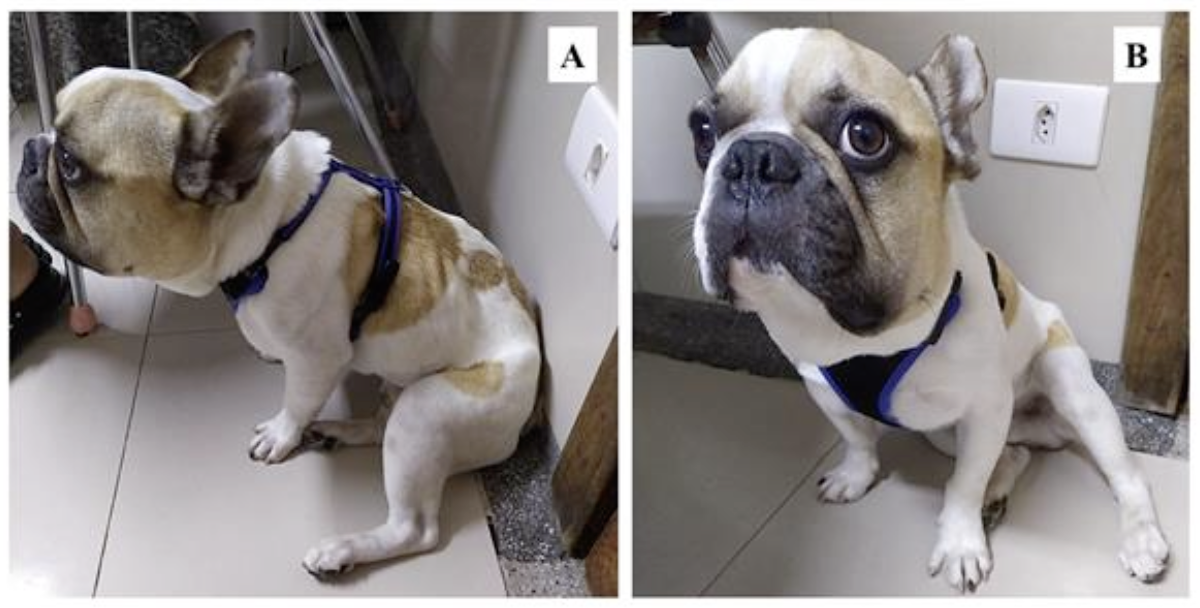
On physical examination, the dog was alert, with a body condition score ranging from thin to cachectic (Figure 1), muscle atrophy, dull and brittle hair, mild dehydration, enlarged submandibular lymph nodes, abdominal tenderness upon palpation in the epimesogastric region, borborygmus, and other vital parameters such as mucosal coloration, capillary refill time, temperature, heart rate, respiratory rate, and cardiopulmonary auscultation were within normal limits."
Figure 1. Photographs of the patient during the initial clinical examination. A) Lateral view of the animal showing a body condition score ranging from thin to cachectic, characterized by the visibility of ribs and muscle atrophy. B) Frontal view of the animal demonstrating lethargy and disproportion between the size of the head and body.
Complete blood count, blood glucose, serum TLI measurement, and abdominal ultrasound were requested. The alterations found included mild leukocytosis with neutrophilia, extremely decreased serum TLI level (Table 1), hepatomegaly, a large amount of gas, and the presence of low cellularity free fluid in the gastric cavity. Evolutionary and accelerated intestinal peristalsis indicated poor absorption, while the pancreas appeared slightly enlarged with increased echogenicity, corresponding to pancreatopathy. Considering the patient's history and clinical findings, these alterations are compatible with EPI.
Table 1. Serum TLI concentration in a 3-year-old intact male Bulldog France's dog diagnosed with EPI.
|
Unit |
Result |
Reference |
|
|
TLI |
ng/mL |
< 1 |
5.0 – 32.0 |
A therapeutic approach was initiated, consisting of pancreatic enzyme replacement therapy with powdered pancreatin at a dose of one teaspoon per meal, equivalent to 5 g/10 kg continuously, omeprazole at a dose of 1 mg/kg once daily continuously, metronidazole at a dose of 15 mg/kg twice daily for 15 days, probiotic at a dose of 2 g/day for 7 days, vitamin supplementation with weekly intramuscular injections of cobalamin at a dose of 0.02 mg/kg, and oral administration of a B-complex polyvitamin once daily continuously. Additionally, the diet was changed to a commercial therapeutic protein-based ration with increased digestibility, B-complex vitamins, and reduced fiber and fat content, until further recommendations.
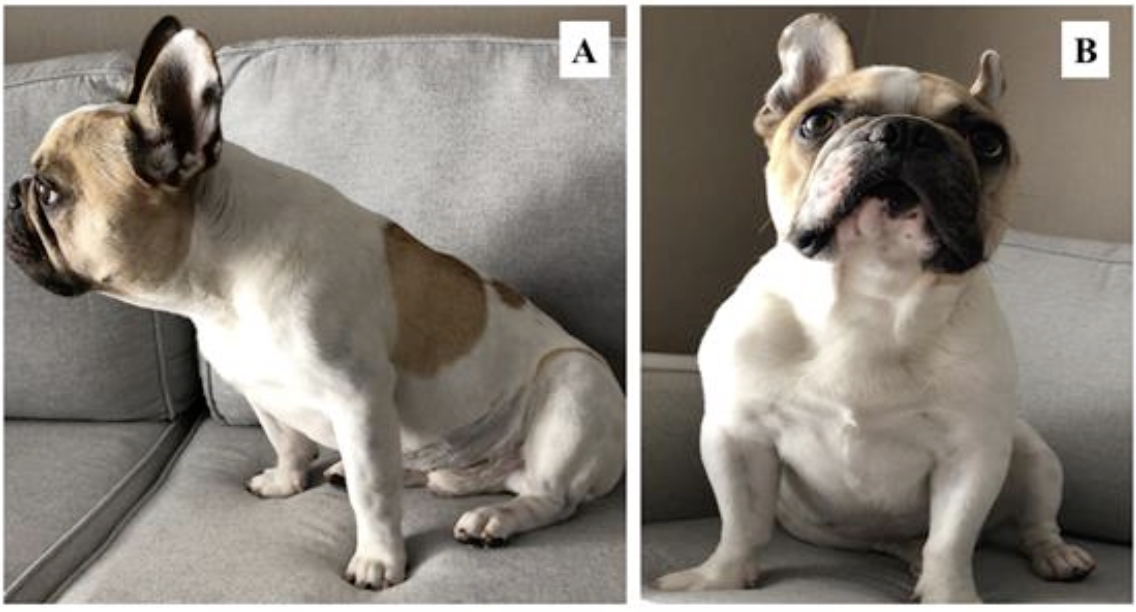
After 30 days of treatment, the animal achieved the ideal body condition score (Figure 2), showing weight gain, normal appetite, and normal bowel movements with reduced borborygmus and flatulence. Moreover, it demonstrated increased muscle mass and significant improvement in coat and behavior, as it resumed playing and interacting with its owners.
Figure 2. Photographs of the patient with 30 days of treatment. A) Lateral view of the animal characterizing the ideal body condition score, without the appearance of ribs. B) Frontal view of the animal, demonstrating muscle mass gain and no evidence of apathy.
DISCUSSION
[3,4,9] report that IPE is more common in dogs of the German Shepherd, Collie and Cavalier breeds, corroborating [5] who states that the prevalence in other breeds such as Boxer, Golden Retriever, Labrador, and Rottweiler is much lower. Matilda KS, et al. [7] and [6] describe cases of PEI in patients without defined breed, but no reports were found in dogs of the French Bulldog breed as in the case in question. The patient described is a male dog. Previously it was defined that males and females were equally affected [10], but studies conducted in the last decade report that are much more affected, regardless of breed [4,5]. Regarding age, clinical signs of PEI are usually seen in young dogs between 1 and 5 years of age [10]. In addition, a recent study defined a mean age of 3.8 years [3], totally close to the reported age.
A study conducted by Raiha and [10] and cited by [11] evaluated 109 dogs diagnosed with PEI, and the main clinical signs described were: yellowish stools (99%), fecal volume increase (95%), weight loss and cachexia (90%), flatulence (88%), coprophagia (61%) and skin changes (14%). The study conducted with 299 dogs by [3] defined yellowish stools (96%), weight loss (84%), coprophagia (18%), and skin changes (6%). These data show that EPI continues to show the same clinical signs in the last 30 years of research [1,2,4], and can consider them as classics.
Although the literature describes that laboratory tests such as blood count, renal and hepatic serum biochemistry are within the reference values in the IPE [7], the patient had mild leukocytosis due to neutrophilia. Matilda KS, et al. [7] and [6] state that for the diagnosis of PEI other causes of malabsorption such as inflammatory bowel disease, diabetes mellitus or parasitosis should be ruled out, justifying the performance of abdominal ultrasound, blood glucose measurement and coproparasitological examination, respectively. With the case reported, it is possible to affirm that the measurement of TLI is really the test of choice for the diagnosis of IPE [2-4,7], since its result dispensed with the need to perform other tests such as the fecal proteolytic activity test [1,6] and enabled the correct treatment.
German AJ [5], describes that pancreatin administered with enteric coatings such as tablets or capsules is more favorable in the treatment of IPE, however the powder administration showed excellent results, and may be justified by the concomitant use of omeprazole [2]. Hypocobalaminemia is highly related to negative prognosis and shorter survival [1-3], but serum vitamin B12 dosage is not accessible to all tutors. Thus, it was decided to perform vitamin supplementation that has shown success to date. Currently there are several rations marketed as adjuvants to the treatments of gastrointestinal diseases, and these are considered good options in cases of IPE [1,2]. Therefore, the chosen diet has all the nutritional needs of the affected patient.
As previously mentioned, the survival of dogs that do not respond to treatment in the first few weeks is low [2,7]. Considering that the patient responded positively and showed significant improvement in the first 30 days, it can be believed that his prognosis is favorable.
CONCLUSION
PEI is not among the main diseases treated in the medical clinic of small animals, but it should be considered in patients who have clinical manifestations of malabsorption and poor digestion. There are predisposed breeds, but the case reported demonstrates that other breeds such as Bulldogs can also be affected. The definitive diagnosis by means of TLI dosage proved to be totally effective, providing the appropriate treatment that ensured the clinical improvement of the patient. It is an incurable disease and because of this it needs the awareness and commitment of the tutor, but it has a favorable prognosis and good survival when treated and monitored properly.
REFERENCES
- Lucca RPV. (2017). Insuficiência pancreática exócrina em cães-relato de caso. Arq. Ciênc. Vet Zool UNIPAR, Umuarama. 20(2):83-86.
- Nelson RW, COUTO CG. (2015). O pâncreas endócrino. In: Medicina interna de pequenos animais. 5th edn. Brazil, Rio de Janeiro: Elsevier. pp. 1767-1830.
- Soetart N, Rochel D, Drut A, Jaillardon L. (2019). Serum cobalamin and folate as prognostic factors in canine exocrine pancreatic insufficiency: An observational cohort study of 299 dogs. Vet J. 243:15-20.
- Westermarck E, Wiberg M. (2012). Exocrine Pancreatic Insufficiency in the Dog: Historical Background, Diagnosis, and Treatment. Top Companion Anim Med. 27(2):96-103.
- German AJ. (2012). Exocrine Pancreatic Insufficiency in the Dog: Breed Associations, Nutritional Considerations, and Long-term Outcome. Top Companion Anim Med. 27(3):104-108.
- Santana AD, Lima VFS, Saints MS, Climacus MSS. (2014). Insuficiência pancreática exócrina em um cão sem raça definida: relato de caso. Enciclopédia Biosfera, Goiânia. 10(18):2046-2053.
- Matilda KS, Gali NM, Romon FG, Machado LHA, Lourenco MLG. (2011). Importância da imunorreatividade tríptica sérica (STI) no diagnóstico definitivo da insuficiência pancreática exócrina: relato de caso. Revista de Educação Continuada em Medicina Veterinária e Zootecnia, São Paulo. 9(2):38-40.
- Conceição NF. (2013). Insuficiência pancreática exócrina em cães: métodos de diagnóstico e alternativas terapêuticas - revisão de literatura. 2011. 78 f. Trabalho de Conclusão de Curso (Graduação)-Faculdade de Agronomia e Medicina Veterinária, Universidade de Brasília, Brasília.
- Silva RD. (2015). Doenças do pâncreas exócrino. In: Jericho MM, Kogika MM, Grandson JPA. Tratado de medicina interna de pequenos animais. 1st edn. Rio de Janeiro: Roca. pp. 3177-3186.
- Westermarck E, Saari SA, Wiberg ME. (2010). Heritability of exocrine pancreatic insufficiency in German Shepherd dogs. J Vet Intern Med. 24(2):450-452.
- Simpson KW. (2014). Função da nutrição na patogênese e manejo das alterações do pâncreas exócrino. Enciclopédia da nutrição clínica canina. Available at: https://vetacademy.royalcanin.es/enciclopedia-de-la-nutricion-canina/.