Previous Issues Volume 1, Issue 1 - 2018
Evolution of the Septoplasty: Maximizing Functional and Aesthetic Outcomes in Nasal Surgery
Sean W Delaney11,2
1Facial Plastic Surgery Associates, 6655 Travis Street, Suite 900, Houston, Texas.
2Division of Facial Plastic Surgery, Department of Otorhinolaryngology-Head and Neck Surgery, McGovern Medical School, University of Texas Health Science Center in Houston.
Corresponding Author: Sean W Delaney, Facial Plastic Surgery Associates, 6655 Travis Street, Suite 900, Houston, Texas, Tel: +1 (713) 526-5665; E-Mail: [email protected]
Received Date: 07 Jan 2018 Accepted Date: 01 Feb 2018 Published Date: 02 Feb 2018
Copyright © 2018 Delaney SW
Citation: Delaney SW. (2018). Evolution of the Septoplasty: Maximizing Functional and Aesthetic Outcomes in Nasal Surgery. M J Otol. 1(1): 004.
ABSTRACT
Correction of a severely deviated nasal septum remains one of the most challenging operations for the facial plastic surgeon. As an understanding of nasal anatomy and physiology advanced over time, so have septoplasty techniques, which have evolved in sophistication and capability to correct increasingly complex nasoseptal deformities. Consequentially, the term “septoplasty” has become fraught with confusion, even among physicians, as it broadly encompasses several different surgical techniques that all attempt to correct septal deviations. Accurate pre-operative assessment of septal deformities and selection of the most appropriate surgical procedure can help maximize functional and aesthetic outcomes. In this article, we will review the evolution of septal surgery and the differences between the various surgical septoplasty techniques.
KEYWORDS
Septorhinoplasty; Cartilage; Rhinoplasty; Breathing; Nasal Appearance.
ABBREVIATIONS
LLC: Lower Lateral Cartilages; ULC: Upper Lateral Cartilages; INV: Internal Nasal Valve
INTRODUCTION
Septal deviation is one of the most common causes of nasal obstruction [1-3]. In a cadaveric study of 2000 subjects, over 75% were found to have some degree of septal deviation [4]. Approximately 33% of patients who visit an otolaryngologist report nasal obstruction, with up a quarter of these patients seeking surgical intervention [5]. Septoplasty is the third most commonly performed otolaryngologic surgery in the United States, preceded only by ear tube placement and adenotonsillectomy[6]. In the United States, 340,000 septoplasties were performed in 2006 alone [7].
The term “septoplasty” is fraught with confusion, even among physicians, as it broadly describes any technique that attempts to correct a deviated septum, and these techniques may vary considerably in complexity and effectiveness. This confusion may contribute to the selection of inappropriate surgical techniques. Despite being one of the most commonly performed surgeries, septal deviations often persist following primary septoplasty, with persistent anterior septal deviation noted in 51-72% of revision septoplasties and patient dissatisfaction rates upwards of 35% [6-8].
In this article, we will review the evolution of the nasal septal surgery and examine the differences between the described septoplasty techniques.
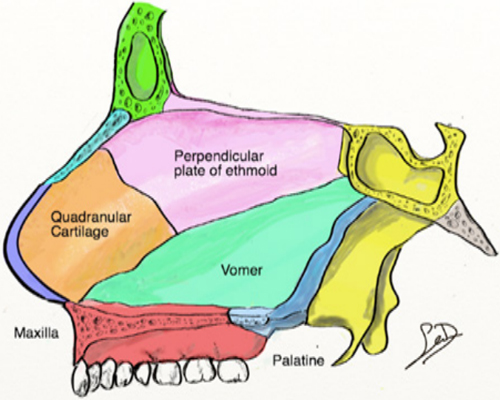
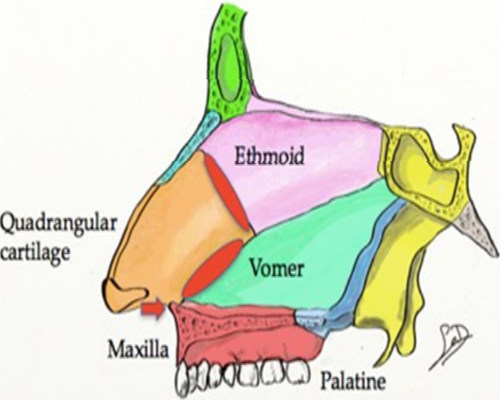
SEPTAL ANATOMY The nasal septum is a midline structure, composed of membranous, cartilaginous and bony components, which divides the nasal cavity into halves and provides structural support to the external nose. The membranous septum, comprised of fibrofatty tissue, is positioned between the medial crura of the lower lateral cartilages (LLC) anteriorly and the cartilaginous septum posteriorly. The cartilaginous septum articulates with the membranous septum anteriorly, nasal bones superiorly, maxillary crest inferiorly and bony septum posteriorly. The cartilaginous septum thins caudally, where it becomes susceptible to traumatic fractures and dislocations [9]. The perpendicular plate of the ethmoid, vomer, maxilla and palatine bones form the bony septum. (Figure 1).
Figure 1: The nasal septum is comprised of three parts: membranous septum, cartilaginous septum, and bony septum.
The septum develops as a downward extension of the frontal prominence [10] that fuses with the primary and secondary palatal shelves. The septum is completely cartilaginous initially. Portions of the septum ossify over time, beginning with the vomer and concluding with the perpendicular plate at puberty. For this reason, children with nasoseptal trauma rarely present with observable fractures, but rather sustain microfractures that alter cartilaginous growth centers and manifest as nasoseptal deviations later in life [11, 12].
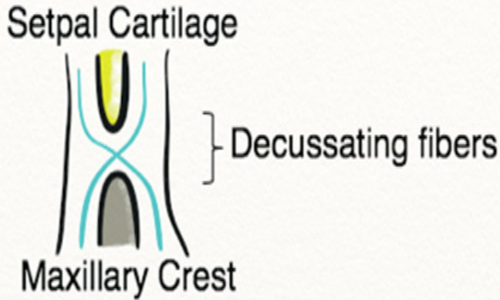
Mucoperichondrium and mucoperiosteum invest the septum bilaterally with fibers that thicken and decussate at the posterior septal angle [2, 13]. (Figure 2).
Figure 2: Investing fibers of the nasal septal mucosa thicken and decussate at the level of the posterior septal angle and maxillary crest.
The septal mucosa is lined with pseudostratified columnar respiratory epithelium inferiorly and olfactory either superiorly.
The narrowest region of the nasal passage termed the internal nasal valve (INV), which contributes the greatest to airway resistance. In the coronal plane, the INV is formed by the borders of the caudal upper lateral cartilage (ULC), head of the inferior turbinate, and septum. Ideally, the angle between the dorsal septum and caudal ULC should be 10° or greater [9]. A straight septum permits laminar airflow during respiration and efficient warming and humidification of inspired air. Therefore, septal deviations that impact the INV may significantly increase airflow resistance, [14] or cause turbulent airflow that alters normal nasal physiology [15]. Inferior turbinate hypertrophy, craniofacial disproportions can also affect the INV but are beyond the scope of this review.
Lastly, the septum serves as a major tip-support mechanism of the external nose through its stabilization of the ULC and LLC [13, 16]. The ULCs attach to the dorsal septum adherently cephalically and loosely caudally. This asymmetric attachment permits dynamic modulation of the internal valve angle at the caudal border of the ULC through dilator naris muscle contraction, widening the INV angle during periods of increased oxygen demand. Intercural ligaments secure the medial crura of the LLCs to the caudal septum and disruption of these attachments may result in decreased tip support and deprojection.
Etiology of Septal Deviation Most individuals do not recall a definitive inciting event for their septal deviation. Occasionally, patients may report a history of nasal trauma. Septal deviation without an attributable traumatic etiology likely develops through the accrual of microfractures early in life that results in asymmetric septal growth that only becomes apparent after growth spurts experienced during puberty [17]. Alternatively, a high-arching palate can also affect septal development [18].
Impact of Septal Deviation Nasal obstruction is the most common symptom associated with septal deviation. Anterior septal deviations may drastically affect nasal airflow dynamics directly and indirectly. Both dorsal and caudal deviations contribute to nasal obstruction through the reduction of the INV cross-sectional area and narrowing of the nasal vestibule [13]. These areas of narrowing behave as Starling resistors during an increased respiratory effort that collapse as the negative pressure generated by airflow through a narrow region, because of Bernoulli’s principle, overcomes the nasal cartilaginous support and thereby further decreases airflow. Posterior septal deviations are better tolerated due to the increased cross-sectional area in this region, [19] but can still affect breathing when severely deviated.
Septal deviations can also cause nasal crusting, epistaxis, recurrent rhinosinusitis, [20] decreased olfaction, [21] obstructive sleep apnea [22] and the appearance of a twisted nose. Headaches associated with septal contact with lateral nasal wall have been described, although the exact mechanism remains unclear [3, 13].
Individuals with septal deviations are more susceptible to rhinosinusitis from both direct obstruction of the ostiomeatal complex and from secondary obstruction due to compensatory mucosal thickening and impaired mucociliary clearance [23-26]. In a meta-analysis of 1621 patients, Orlandi [19] found a robust correlation between septal deviation greater than 10° from midline and rhinosinusitis (p = 0.0004).
Nasal appearance and symmetry are also intimately related to the nasal septum. Dorsal septal deviations create dorsal irregularities that disturb the brow-tip aesthetic line. Caudal septal deviations contribute towards the appearance of a twisted nose through the loss of nasal tip support and tip projection, columellar irregularities [13] and nostril asymmetry.
Evaluating the Deviated Septum The twisted nose can involve the asymmetry of the nasal bones, ULC and LLC, and septum. A comprehensive evaluation entails an examination of each of these components individually and concerning one another. Palpation is essential to determine size, firmness, and elasticity of these structures. [16] The internal and external nasal valves are examined at rest and dynamically. Asking the patient to breathe through one nostril while covering the other can provide information regarding which side has more obstruction. The location and severity of all septal deviations and spurs should be noted. If the patient has had previous nasal surgery, gentle palpation of the septum with a wet cotton-tip applicator can provide an idea of how much septum remains. In certain instances, rigid endoscopy may be useful to evaluate the intranasal anatomy more thoroughly.
Although numerous septal deviation classification schema has been proposed, [27-33] none are widely employed by clinicians and is probably due to the substantial challenge of describing and categorizing the complex three-dimensional nature of septal deviations. Septal deviations can exhibit C-shaped or S-shaped deformities, spurs, regions of thickening, dorsal or caudal deflections, excess cartilage length, and maxillary crest deflection or widening. Additionally, the relation of the septum to the nasal bones, nasal valves, inferior turbinates, maxillary crest, and the nasal soft tissue envelope deserves careful consideration as well. Given the significant individual anatomical differences, there is no single classification system that is robust enough to capture the many factors to be considered in septal surgery at this time.
Acoustic rhinometry, rhinomanometry, peak inspiratory flow, and spectral sound analysis provide objective measurements of septal deviation, [16, 34] but lack the precision of direct visualization in identifying the exact location and severity of septal deviation. Interestingly, subjective ratings of nasal obstruction do not always correlate with objective or physical findings of septal deviation severity. [35, 36] Consequently, assessment of nasal obstruction severity and surgical effectiveness relies heavily upon subjective patient reporting and the patient and surgeon’s satisfaction with nasal appearance.
Sykes et al. [37] proposed the “Four R’s” of septoplasty: Resection, Reposition, Reconstruction and Replacement when considering how to correct a deviated septum. (Table 1)
Table 1: The Four R’s of Septoplasty.
| Technique | Uses |
|---|---|
| Resection | Small deviations, Duplication, Redundancy |
| Reposition | Septal displacement, redistributing structural support |
| Reconstruction | Large deficiency |
| Replacement | Structural support with lack of native cartilage |
Adapted from Sykes JM, Kim JE, Shaye D, Boccieri A. The Importance of the Nasal Septum in the Deviated Nose. Facial Plast Surg, 2011: 27(5):413-421.
Through accurate pre-operative identification of an individual’s nasoseptal deformity, the surgeon can best select the most appropriate surgical technique to maximize the functional and aesthetic outcomes of nasal surgery.
Surgical Management of the Deviated Septum Indications for Septoplasty Septal deviation alone is not a reason for surgical correction. Indications for septoplasty include persistent nasal obstruction refractory to medical management, poor cosmesis from a twisted nose, recurrent epistaxis or sinusitis, chronic headaches from septal contact with the lateral nasal wall, intolerance to continuous positive airway pressure appliance, or access during endoscopic sinus or anterior skull base procedures.
Historical Perspective The earliest written treatise describing the correction of a deviated septum dates back to the Egyptian Edwin Smith Surgical Papyrus (c. 3000 B.C.), which prescribed closed reduction with splinting [38]. In the intervening millennia, advancements in septal deviation treatment stagnated, as clinicians focused primarily on reconstructing nasal defects using pedicled flaps. Quelmaltz proposed daily digital pressure to correct a deviated septum in the 18th century [39]. In 1875 Adams proposed closed reduction and splinting for treatment of a deviated septum [40]. Although what Adams described was not a novel concept and had previously been prescribed by the ancient Egyptians, his publication ushered in a new era of growing interest for septal deviation treatment, as reflected by the multitude of septal surgical techniques that ensued shortly after.
While the physiological role of the nasal septum remained poorly understood during the 19th century, surgeons at the time forayed into septal surgery enthusiastically, excising obstructing segments of the septum in an attempt to relieve nasal obstruction. Many surgeons, such as Krieg (1890s), performed mastectomies, removing entire segments of septal cartilage and bone along with its mucosa, in essence exchanging septal deviations for perforations [39]. These patients enjoyed a short-lived improvement in nasal obstruction, only to suffer nasal whistling, crusting, epistaxis, worsening nasal obstruction from turbulent airflow, and saddle nose deformity, which was attributed to cicatricial forces of healing by the surgeons of the period.
Upon review of his surgical outcomes, Krieg concluded that mucosa preservation and removal of only deviated septal cartilage could decrease complications [39]. Recognizing the need for mucosa preservation, surgeons resected the deviated septum along with the mucosa on the concave side only, preserving the mucosa on the convex side. Similarly, Bosworth sawed-off deviated septal spurs, sacrificing the ipsilateral mucosa while maintaining the contralateral mucosa [38]. Asch (1899) proposed a full cruciate incision through the cartilaginous septum to “destroy the resiliency” of the cartilage while preserving mucosa [41]. Despite these changes in technique, perforation still occurred frequently [42]. In 1882 Ingalls, who some call the father of septoplasty, described the targeted resection of a deviated septum with the preservation of bilateral mucosa [39].
Up to this point, there was little emphasis on the septum’s role in providing structural support for the external nose. Experts at the time considered the septal mucosa to be the only physiologically relevant structure within the nose [42]. Patients with overly aggressive septal cartilage resection eventually developed nasal tip ptosis, columellar retraction, and saddle nose deformity.
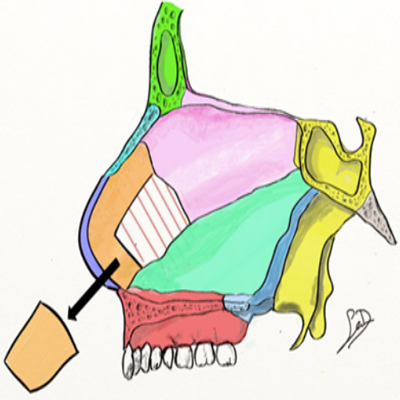
Contemporary Septoplasty Techniques Submucous resection At the turn of the 20th century, Freer [43] and Killian [44] ushered in the era of contemporary septoplasty with their recognition of the importance of the structural support that the septal L-strut provides for the external nose. They proposed submucous resection of the deviated septum while maintaining a dorsal and caudal strut of the cartilage of sufficient size to support the nose. (Figure 3)
Figure 3: Submucous resection of the deviated septal cartilage with the preservation of an L-strut to support the external nose. Segments of the perpendicular plate of the ethmoid and vomer may also be resected as needed to correct posterior septal deviations
The tenets of (1) mucosa preservation and (2) L-strut conservation formed the cornerstone of modern septoplasty.
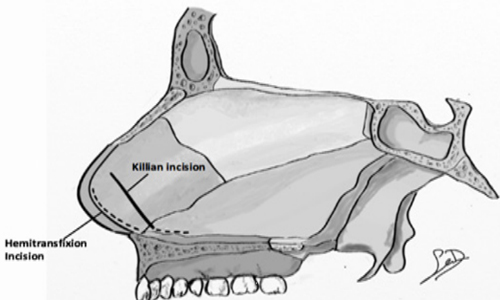
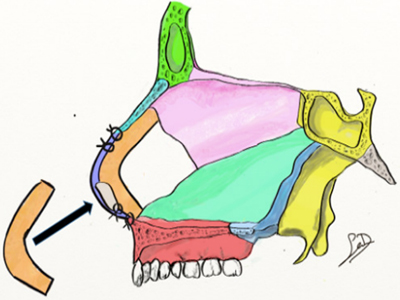
With this technique, the septum is approached endonasal through one of two mucosal incisions: a Killian incision placed behind the septal mucosquamous junction or a hemitransfixion incision that parallels the border of the caudal septum. (Figure 4)
Figure 4: In the endonasal approach, the nasal septum can be accessed via a Killian incision or hemitransfixion incision
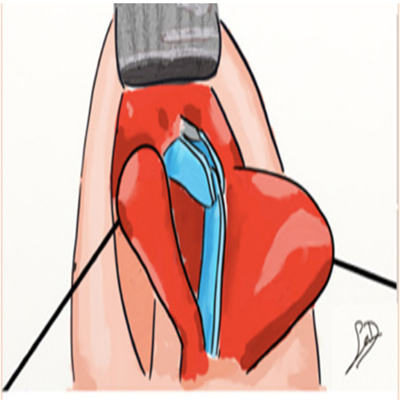
A mucosal flap is elevated broadly, with care to incorporate the perichondrium, which provides the biomechanical strength of the septal lining [45]. Next, a contralateral septal flap is elevated through a transcartilaginous incision in the septal cartilage. With both mucosal flaps elevated and protected, septal cartilage and bone posterior to the L-strut is resected. (Figure 5).
Figure 5: Meticulous bilateral septal mucosal flap elevation and preservation permits access for removal of the deviated septum and decreases the risk of post-operative septal perforation.
The excised cartilage can then be weakened and straightened, before being replaced between the septal flaps to decrease the chance of septal perforation formation. With the introduction of the submucous resection, septal perforations and saddle nose deformities decreased considerably. To this day, this technique is widely practiced and can be effective for mild and posterior septal deviations.
Swinging Door technique for Caudal Septal Deviation
In 1929 Metzenbaum observed that caudal septal deviations were often the result of excess septal length, which causes the septum to deflect off the maxillary crest to one side. To correct the deviated caudal septum without destabilizing the external nose, Metzenbaum proposed the “Swinging Door” technique [46]. Through a hemitransfixion incision, bilateral mucosal flaps are elevated, and the cartilaginous septum is disarticulated from the bony septum, maxillary crest, and nasal spine, leaving only its superior attachment, from which the cartilaginous septum now “swings.” [3] (Figure 6)
Figure 6: Swinging Door Technique. The cartilaginous septum is disarticulated from the nasal spine, maxillary crest groove, and bony septum, leaving only its superior attachments, freeing up the cartilaginous septum to be repositioned and secured in the midline.
Once the excess septal length has been trimmed, the caudal septum is secured to the nasal spine in the midline. [47]. (Figure 7)
Figure 7: Swinging Door Technique. (A) Excess septum above the maxillary crest is resected to permit repositioning of the septum in the midline, (B) where the caudal septum is be secured to the anterior nasal spine. (C) Alternatively, the excess septal length can be swung over to the contralateral side of the maxillary crest, which serves as a “doorstop” to straighten the septum.
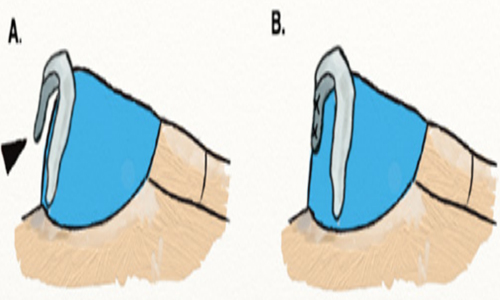
Around the same time, Peer proposed a modification to Metzenbaum’s technique, where a deviated or fractured caudal septum is excised and replaced with a straighter piece of harvested septal cartilage [48]. Pastorek recommended flipping the excess septum to the contralateral side of the nasal spine, acting as a“doorstop” to straighten the septum [49]. The caudal septum can be further stabilized by its insertion into a small pocket created between the medial crura in a “tongue-in-groove” fashion and secured with septocolumellar sutures, as described by Kridel (1999) [50]. (Figure 8)
Figure 8: The caudal septum can be stabilized in the midline by its insertion into a small pocket created between the medial crura (arrowhead) in a “tongue-in-groove” fashion.
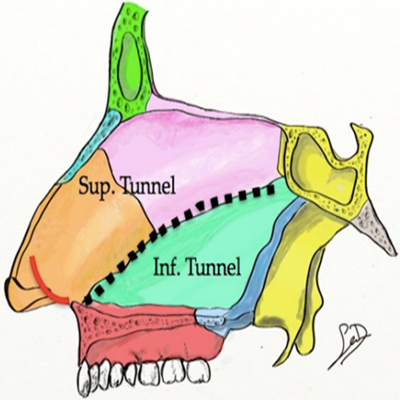
Maxilla Premaxilla Approach Cottle advocated for a more conservative “maxilla-premaxilla” septoplasty approach in 1947 that removed less septal cartilage and bone, intending to reduce post-operative septal perforation and saddle [51]. For this approach, the septum is accessed endonasal through a hemitransfixion incision. After ipsilateral elevation of the septal mucosa, a retrograde premaxillary dissection through the same incision provides exposure of the inferior septum and maxillary crest along the nasal floor. Elevation of the mucosa above and lateral to the maxillary crest provides the broad exposure necessary to safely elevate the mucosa off the maxillary crest, where the decussating investing fibers of the mucoperiosteum are particularly adherent, thereby decreasing the risk of inadvertent mucosal perforation. The elevation of the contralateral flap begins at the caudal septum. However, instead of elevating the entire contralateral flap, only an inferior tunnel of mucosal is elevated to permit resection of deviated septum while minimizing septal blood supply disruption by not elevating a superior mucosal flap [51]. (Figure 9)
Figure 9: In Cottle’sMaxilla Premaxilla approach, only an inferior tunnel of mucoperichondrium is elevated on one side of the septum to minimize blood supply disruption of the septum and decrease the risk of septal perforation.
Through this exposure, the caudal septal deviation may be addressed by the “Swinging Door” technique.
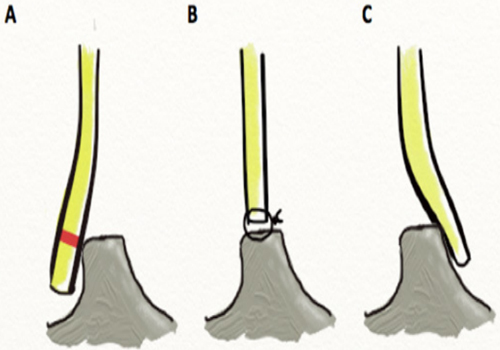
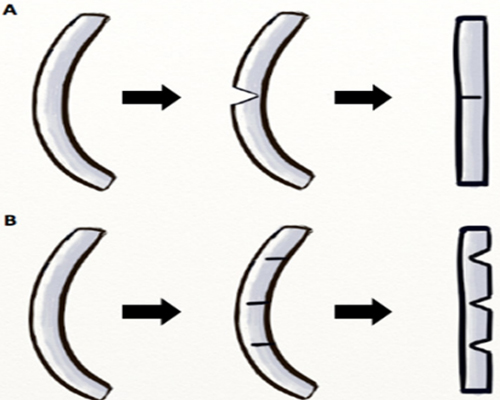
In-situ cartilage modifications To further straighten deviated septal cartilage, several adjunctive techniques that modify cartilage resiliency and strength have been described. Wedge resection and scoring may weaken septal cartilage to coax the cartilage into the desired configuration. Wedge resections placed on the longer convex side of the cartilage in the axis of deviation and bolstered with sutures straighten the septum through a reconciliation of length discrepancies [52]. Scoring the concave side of the deviated cartilage weakens the cartilage, permitting the shorter concave surface to unfurl and straighten. (Figure 10)
Figure 10: Cartilage modification techniques: (A) wedge resection on the convex side, (B) scoring on the concave side.
A mattress suture, similar to the Mustarde stitch used in otoplasty, or asymmetric clocking stitch placed on the convex side of a septal deviation can counteract intrinsic cartilage curvature during the healing phase. Lastly, batten grafting with a thin piece of cartilage or ethmoid bone on the concave surface of a deviate caudal septum may provide the necessary support to straighten the septum.
Open Septorhinoplasty While caudal septal deviations can often be corrected via an endonasal technique, dorsal septal deflections that contribute to the appearance of a twisted nose and require complex septal reconstruction are best addressed through an open approach [47]. The detachment of the ULCs from the dorsal septum permits easier mucoperichondrial flap elevation, unparalleled exposure of the septum, [3] and release of all extrinsic forces that can distort the septum, thereby providing a more thorough assessment of intrinsic septal deformities. Spreader grafts can now be placed to improve breathing and straighten the dorsal septum [53]. Boccieri and Pascali (2003) [54] described a modified spreader graft technique in which a unilateral spreader graft is secured as a “crossbar” on the concave side of the dorsal septum with two intra-cartilaginous notches made perpendicular to the axis of the dorsal septum. It is important to re-attach the ULC to the septum to prevent the collapse of the middle third of the nose, exhibiting as an inverted-V deformity. With the nasal framework exposed through the open approach, additional grafts and sutures may be precisely placed to execute functional and aesthetic improvements.
Extracorporeal Septoplasty King and Ashley introduced extracorporeal septoplasty in 1952 [55] to correct the markedly deviated septum. Both open [56] and closed [3] approaches for extracorporeal septoplasty have been described, but an open approach is generally preferred for its superior exposure. The procedure involves bilateral mucoperichondrial elevation and en bloc removal of the cartilaginous septum in its entirety [3]. Deviated portions of the bony septum are removed as needed. The septal cartilage is then reconfigured ex vivo to fashion a straighter L-strut, or“new septum,” to be reimplanted. (Figure 11)
Figure 11: Extracorporeal septoplasty. The new septum is fixated to the upper lateral cartilages and keystone region dorsally and the lateral cartilages and nasal spine caudally.
The new septum can be crafted out of a single piece of cartilage or, when a single piece of the cartilage of sufficient size is not available, from several smaller pieces of cartilage sutured together. Toriumi (2013) [57] described the use of bilateral extended spreader grafts that articulate with a caudal septal replacement graft to recreate the L-strut. (Figure 12)
Figure 12: Toriumi described the creation of a new L-strut using bilateral extended spreader grafts that articulate with a caudal septal replacement graf.
In the case of a previously operated nose, there may not be sufficient residual septal cartilage to create a straight new septum and non-septal sources of cartilage may be necessary. Although easy to harvest, the curvature and thinness of auricular cartilage make it a poor option for septal reconstruction. Costal cartilage, on the other hand, can be molded into long, straight grafts, and is well-suited for the load-bearing demands of a new septum.
To adequately support the external nose, the new septum must be securely fixated dorsally to the ULCs and keystone region and caudally to the LLCs and nasal spine [58]. If the nasal spine is over-large, deviated, or asymmetric, it may be burred down to create a midline groove for the new septum [16]. Failure to properly fixate the new septum may results in dorsal irregularities, dorsal saddling [59] and septal hypermobility with septal deviation recurrence [53].
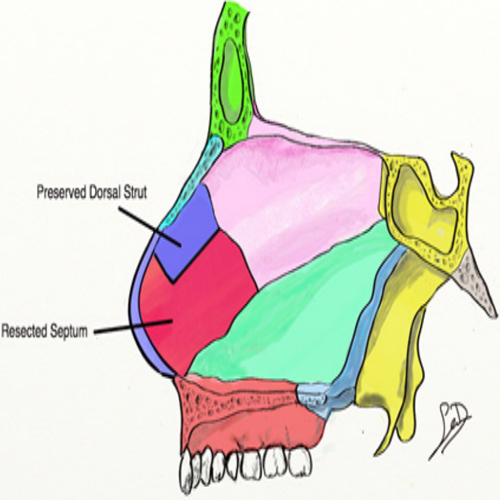
Despite meticulous fixation techniques, however, dorsal irregularities can still be encountered. Gubisch (2008) suggested the placement of dorsal only grafts to camouflage these irregularities preemptively [56]. Most described a modification, termed “anterior septal reconstruction,” where the dorsal strut of cartilage is left in-situ during extracorporeal septoplasty to avoid the risk of subsequent external dorsal irregularities [59]. (Figure 13)
Figure 13: Anterior Septal Reconstruction. A dorsal strut of the cartilaginous septum is left in situ to prevent dorsal irregularities that can be seen with extracorporeal septoplasty with total septal reconstruction.
While unable to treat dorsal septal deviations, anterior septal reconstruction is an effective alternative to extracorporeal septoplasty for correcting severe caudal septal deviations.
DISCUSSION
Septoplasty can be one of the most challenging and yet rewarding operations due to the complicated three-dimensional relationship between the nasal bones, ULCs and LLCs, and septum. Because no two patients’ septal deformities are alike, the septoplasty technique selected for each should be tailored to their particular anatomical needs. Accurate appreciation of each patient’s nasoseptal deformity may help the surgeon determine whether “Resection, Reposition, Reconstruction or Replacement” techniques will be necessary [37].
Since the time of Freer and Killian, there has been considerable apprehension when addressing deviations involving the L-strut, as most surgeons learn early on in their training that an insufficient L-strut may result in tip deprojection and the dreaded saddle nose deformity. Despite being one of the most commonly performed surgeries, septal deviations often persist following primary septoplasty. Septal deviations within the L-strut responsible for nasal obstruction or nasal asymmetry should not be ignored, however, for fear of destabilizing the external nose. Nasal surgeons should be able to accurately identify anterior septal deviations and become comfortable with septoplasty techniques to correct deviations in this region.
In the majority of septal deviations, an endonasal approach can be used. For posterior septal deviations, submucous resection with preservation of an L-strut and septal mucosa is a simple and effective procedure. The maxilla-premaxilla approach combined with the “Swinging Door” technique can correct mild to moderate anterior septal deviations. Adjunctive cartilage modification techniques with suturing, scoring, wedge excision or septal buttressing can further improve outcomes. For the twisted nose with the bony pyramid and ULC asymmetry, an open approach provides the exposure and access necessary to release extrinsic distorting forces on the septum to fully appreciate intrinsic septal deformities. Weaknesses or insufficiencies identified through this approach can be precisely corrected with grafts and sutures to maintain the septum in a midline position and support the external nose.
In the case of the severely deviated L-strut, extracorporeal septoplasty may be necessary, where the entire septal cartilage is removed and replaced by a straighter new septum created ex vivo. When septal cartilage is insufficient, particularly in revision surgeries, autologous or irradiated costal cartilage [60] provides an ample supply of sturdy load-bearing cartilage.
CONCLUSION
As an understanding of nasal septal anatomy and physiology advanced over time, so have septoplasty techniques, which evolved in sophistication and capability to correct increasingly complex nasoseptal deformities. Consequentially, the term “septoplasty” has become fraught with confusion, even among physicians, as it broadly encompasses several different surgical techniques that all aim to correct septal deviations. Whether correcting a mild septal deviation or a severely twisted nose, accurate assessment of the location, extent, and severity of septal deviation can help with the selection of the most appropriate surgical technique to maximize functional and aesthetic outcomes.
REFERENCES
- Simon P and Sidle D. (2012). Augmenting the nasal airway: Beyond septoplasty. Am J Rhinol Allergy. 26(4): 326- 331.
- Gillman GS, Egloff AM and Rivera-Serrano CM. (2014). Revision Septoplasty: A Prospective Disease-Specific Outcome Study. Laryngoscope. 124(6): 1290-1295.
- Fettman N, Sanford T and Sindwani R. (2009). Surgical Management of the Deviated Septum: Techniques in Septoplasty. Otolaryngol Clin N Am. 42(2): 241-252.
- Lang J. (1989). Clinical anatomy of the nose, nasal cavity and paranasal sinuses. New York: Thieme.
- Bateman ND and Woolford TJ. (2003). Informed consent for septal surgery: the evidence base. J Laryngol Otol. 117(3): 186-189.
- Manoukian PD, Wyatt JR, Leopold DA, and Bass EB. (1997). Recent trends in the utilization of procedures in otolaryngology-head and neck surgery. Laryngoscope. 107(4): 472- 477.
- Bhattacharyya NN. (2010). Ambulatory sinus and nasal surgery in the United States: demographics and perioperative outcomes. The Laryngoscope 120(3): 635-638.
- Becker SS, Dobratz EJ, Stowell N, Barker D, et al. (2008). Revision septoplasty: a review of sources of persistent nasal obstruction. Am J Rhinol. 22(4): 440-444.
- Loyo M and Wang TD. (2015). Management of the Deviated Nasal Dorsum. Facial Plast Surg. 31(3): 216-227.
- Sadler TW. (2000). In Lagman's medical embryology. Baltimore: Lippincott Williams & Wilkins.
- Beck JC and Sie KCY. (1999). The growth and development of the nasal airway. Facial Plast Surg Clin North Am. 7: 257-262.
- Vasuki AK, Fenn TK, Devi MN, Hebzibah TD, et al. (2016). Fate and Development of Human Vomeronasal Organ - A Microscopic Fetal Study J Clin Diagn Res. 10(3): AC08-11
- Haack J and Papel ID. (2009). Caudal Septal Deviation. Otolaryngol Clin N Am. 42(3): 427-436
- Cole P, Chaban R, Naito K and Oprysk D. (1998). The obstructive nasal septum: effect of simulated deviations on nasal airflow resistance. Arch Otolaryngol Head. 114(4): 410-412.
- Cummings CW, Fredrickson JM, Harker LA, Krause CJ, et al. (1998). Rhinoplasty in Otolaryngology, Head & Neck Surgery. St Louis, Missouri: Mosby-Yearbook. p. 481-495.
- Palmaa P and Khodaei I. (2014). Surgical management of the distorted caudal septum. Curr Opin Otolaryngol Head Neck Surg. 22 (1): 52-57.
- Holt GR. (1999). Biomechanics of nasal septal trauma. Otolaryngol Clin North Am. 32(4): 615-619.
- Monini S, Malagola C, Villa MP, Caterina Tripodi, et al. (2009). Rapid maxillary expansion for the treatment of nasal obstruction in children younger than 12 years. Arch Otolaryngol Head Neck Surg. 135(1): 22-27.
- Howard BK and Rohrich RJ. (2002). Understanding the nasal airway: Principles and practice. Plast Reconstr Surg. 109: 1128-1146.
- Orlandi RR. (2010). A Systematic Analysis of Septal Deviation Associated With Rhinosinusitis. The Laryngoscope. 120(8): 1687-1695.
- Altundag A, Salihoglu M, Tekeli H, Saglam M, et al. (2014). Lateralized Differences in Olfactory Function and Olfactory Bulb Volume Relate to Nasal Septum Deviation. The Journal of Craniofacial Surgery. 25(2): 359-362.
- Kushida CA, Guilleminault C, Clerk AA, and Dement WC. (1997). Nasal Obstruction and Obstructive Sleep Apnea: A Review. Allergy and Asthma Proc. 18(2): 69-71.
- Setlur J and Goyal P. (2011). Relationship between septal body size and septal body. American Journal of Rhinology & Allergy. 25(6): 397-400.
- Mahler E and Reuven S. (1985). The Role of Turbinectomy in Rhinoplasty. Aesth. Plast. Surg. 9: 277-279.
- Jang YJ, Myong NH, Park KH, Koo TW, et al. (2002). Mucociliary transport and histologic characteristics of the mucosa of the deviated nasal septum. Arch Otolaryngol Head Neck Surg. 128: 421-24.
- Kamani T, Yilmaz T, SurucuS, Bajin MD, et al. (2014). Histopathological changes in nasal mucosa with nasal septum deviation. Eur Arch Otorhinolaryngol. 271(11): 2969-2974.
- Lee JW and Baker S. (2013). Correction of Caudal Septal Deviation and Deformity Using Nasal Septal Bone Grafts... JAMA Fac Plast Sur. 15(2): 96-100.
- Lin JK, Wheatley FC, Handwerker J, Harris NJ, et al. (2014). Analyzing nasal septal deviations to develop a new classification system: a computed tomography study using MATLAB and OsiriX. JAMA Facial Plastic Surgery. 16(3):183-187.
- Mladina R. (1987). The role of maxillar morphology in the development of pathological septal deformities. Rhinology 25(3): 199-205.
- Bauman I and Bauman H. (2007). A new classification of septal deviations. Rhinology. 45(3): 220-223.
- Guyuron B, Uzzo CD and Scull H. (1999). A practical classification of septonasal deviation and an effective guide to septal surgery. Plastic and Reconstructive Surgery. 104(7): 2202-2209.
- Rohrich RJ, Gunter JP, Deuber MA, and Adams Jr WP. (2002). The deviated nose: optimizing results using a simplified classification and algorithmic approach. Plastic and Reconstructive Surgery. 110(6): 1509-1523.
- Codari M, Zago M, Guidugli GA, Pucciarelli V, et al. (2016). The nasal septum deviation index (NSDI) based on CBCT data. Dentomaxillofac Radiol. 45(2): 20150327.
- Aziz T, Biron VL, Ansari K and Flores-Mir C. (2014). Measurement tools for the diagnosis of nasal septal deviation: a systematic review. Journal of Otolaryngology-Head and Neck Surgery. 43: 11.
- Stewart MG, Smith T, Weaver E, Witsell DL, et al. (2004). Outcomes after nasal septoplasty: results from the Nasal septoplasty effectiveness (NOSE) study. Otolaryngol Head Neck Surgy. 130(3): 283-290.
- Verhoeven S and Schmelzer B. (2016). The type and severity of septal deviation are not related to the degree of subjective nasal obstruction. Rhinology. 54(4): 355-360.
- Sykes JM, Kim JE, Shaye D and Boccieri A. (2011). The Importance of the Nasal Septum in the Deviated Nose. Facial Plast Surg. 27(5): 413-421.
- Santoni-Rugiu P and Sykes PJ. (2007). A History of Plastic Surgery. New York: Springer Science & Business Media
- Hinderer KH. (1971). History of septoplasty. Fundamentals of anatomy and surgery of the nose. Birmingham, Alabama: Aesculapius Publishing Co.
- Adams W. (1875). The treatment of the broken nose by forcible straightening and mechanical apparatus. Br Med J. 2(770): 421-422.
- Asch M. (1899). Treatment of nasal stenosis due to deflective septum with and without thickening of the convex side. Laryngoscope. 6: 340.
- Bailey B. (1997). Nasal septal surgery 1896-1899: transition and controversy. Laryngoscope. 107(1): 10-16.
- Freer OT. (1902). The correction of deflections of the nasal septum with a minimum of traumatism. JAMA. 38: 636-642.
- Killian G. (1904). Die submucosa"se fensterresektion der nasenscheidewand. Archiv Fur Laryngologie und Rhinologie. 16: 362-387.
- Kim DW, Egan KK, O'Grady K and Toriumi DM. (2005). Biomechanical strength of human nasal septal lining: comparison of the constituent layers. Laryngoscope. 115(8): 1451-1453.
- Metzenbaum M. (1929). Replacement of the lower end of the dislocated septal cartilage versus submucous resection of the dislocated end of the septal cartilage. Arch Otolaryngol. 9: 282-292.
- Constantine FC, Ahmad J, Geissler J, and Rohrich RJ. (2014). Simplifying the Management of Caudal Septal Deviation in Rhinoplasty. Plast Reconstr Surg. 134(3): 379-388.
- Peer LA. (1929). An operation to repair lateral displacement of the lower border of the septal cartilage. Arch Otolaryngol. 9: 282-296.
- Pastorek NJ. (2000). Treating the caudal septal deflection. Arch Facial Plast Surg. 2: 217-220.
- Kridel RW, Scott BA, and Foda HM. (1999). The tongue in groove technique in septorhinoplasty: a 10-year experience. Arch Facial Plast Surg. 1(4): 246-256.
- Cottle MH, Loring RM, Fischer GG, and Gaynon IE. (1958). The 'maxilla-premaxilla' approach to extensive nasal septum surgery. Arch Otolaryngol Head Neck Surg. 68(3): 301-313.
- Shin JH, Lee MH, Kim YH, Kim SW, et al. (2011). Wedge resection and modified mattress suture for correcting anterior septal deviation: how we do it. Clinical Otolaryngology. 36: 491-513.
- Boccieri A. (2013).The crooked nose. ACTA otorhinolaryngological ita ca. 33(3): 163-168.
- Boccieri A and Pascali M. (2003).Septal Crossbar Graft for the Correction of the Crooked Nose. Plast Recon Surg. 111(2): 629-638.
- King ED and Ashley FL. (1952). The correction of the internally and externally deviated nose. Plast Reconstr Surg. 95: 672-676.
- Gubisch W and Sinha V. (2008). Extracorporeal septoplasty-how we do it at Marien hospital Stuttgart Germany. Indian J. Otolaryngol. Head Neck Surg. 60(1): 16-19.
- Toriumi DM. (2013). Subtotal Septal Reconstruction: An Update. Facial Plast Surg. 29(6): 492-501.
- Gubisch W. (2005). Extracorporeal septoplasty for the markedly deviated septum. Arch Facial Plast Surg. 7(4): 218-226.
- Most SP. (2006). Anterior septal reconstruction: outcomes after a modified extracorporeal septoplasty technique. Arch Facial Plast Surg. 8(3): 202-207.
- Kridel RW, Ashoori F, Liu ES, and Hart CG. (2009). Longterm use and follow-up of irradiated homologous costal cartilage grafts in the nose. Arch Facial Plast Surg. 11(6): 378-394.