Previous Issues Volume 2, Issue 1 - 2017
Duodenal-Jejunal Bypass Surgery Increased GLP-1 Receptor Expression in Enteric Nervous System of STZ-Induced Diabetic Rats
Zhiqin Gao1, Qingtao Yan1, 2, Xiangpo Pan2, Xiangfeng Meng2, Ruiyan Pan1, Jima Xu1, Xiaoyun Yang1, Wansheng Ji1, 3, Mei-Hua Qu1
1Department of Pharmacology, Key lab of Applied Pharmacology in University of Shandong, Weifang Medical University, Weifang, 261053, China.
2Department of Pediatric Surgery, Weifang People's Hospital, China.
3Affiliated Hospital of Weifang Medical University, China.
Corresponding Author: Mei-Hua Qu, Key Lab of Applied Pharmacology in University of Shandong, Weifang Medical University, Weifang, 261053, China, Tel:086-536-8462466; E-Mail: [email protected]
Received Date: 19 Sep 2016 Accepted Date: 29 Dec 2016 Published Date: 03 Jan 2017
Copyright © 2016 Gao Z
Citation: Qu MH, Gao Z, Yan Q, Pan X, et al. (2016). Duodenal-jejunal bypass surgery increased GLP-1 receptor expression in enteric nervous system of STZ-induced diabetic rats. Mathews J Diabetes Obes 2(1): 004.
ABSTRACT
Aims: Studies have shown that the Duodenal-jejunal bypass (DJB) is highly effective treatment of type 2 diabetes (T2DM) which glucagon like pipetide-1 (GLP-1) plays an important role. This study, we aimed to study DJB surgery effect on the expression of GLP-1R in the enteric nerve system and outset of type 2 diabetic rats. Methods: Type 2 diabetic rats were generated by feeding Wistar rats with high-fat diet and low dose streptozotocin (STZ) injection. DJB and sham operations were performed in T2DM rats. Wistar rats were used as the wild-type control. Fasting blood glucose, insulin and GLP-1 secretion were measured before and eight weeks after surgery.Double-labeling immunofluorescence assay was performed to determine the changesof β cells (insulin) and α cells (glucagon) in pancreatic islets eight weeks after operation. Immunofluorescence and western blotting were performed to determine the expression of GLP-1R in Myenteric plexus (MP) of ENS after operation. Results: DJB decreased the level of fasting blood glucose and the insulin secretion compared to the sham operation. Furthermore, DJB increased the β cell quantities in the pancreas compared to the sham group rats. DJB increased plasma GLP-1 secretionin T2DM rats comparing the sham rats. Immunofluorescence and Western blot results showed that GLP-1R expression in ENS was significantly increased eight weeks after DJB. Conclusion: DJB improved the expression of GLP-1R in the enteric nerve system.And DJB increased GLP-1 secretion and improved the glucose disposal in T2DM and increased the quantity of β cells in the pancreas of T2DM rats.
KEYWORDS
Type 2 diabetes; Duodenal-jejunal bypass; Glucagon-like peptide-1; Glucagon-like peptide-1 receptor; enteric nervous system.
INTRODUCTION
Diabetes is the fifth leading cause of death in the world, and the patients suffer from an increased risk of developing a variety of complications. The number of people with diabetes is likely to increase to approximately 438 million by 2030 (of which 90% is type 2 diabetes mellitus (T2DM)). Conventional therapies of T2DM including lifestyle modifications and ant diabetic drug therapy seem unable to stop the progression of T2DM and cannot cure the disease. In contrast, bariatric surgery has proven an effective ant diabetic treatment modality with reported T2DM remission rates following up to 80% by Roux-en-Y gastric bypass (RYGB) [1-3].In 2007,bariatric surgery was recommended by international diabetes federation (IDF) for T2DM patients with obesity [4]. Currently, the most commonly applied surgical procedure was Roux-en-Y gastric bypass (RYGB) [5]. Although this degree of weight loss has profound metabolic impact, these surgeries seem to have metabolic effects that are independent of weight loss. In support of this is the clinical literature showing rapid resolution of Type 2 diabetes mellitus (T2DM) that occurs before significant weight loss. To gain a complete understanding of the weight loss-independent effects of bariatric surgery, animal models have been developed [6]. In order to eliminate the effect of the restricted stomach volume, Duodenal-jejunal bypass (DJB) surgery was designed to study the weight-loss independent mechanism of bariatric surgery in T2DM [7]. DJB is an experimental metabolic surgery that is similar to RYGB, but without the restriction of the stomach volume [8]. DJB surgery includes duodenal and proximal jejuna exclusion to nutrients, a jejunal Roux-en-Y reconstruction, and an early nutrient delivery to distal small bowel [9]. Until now, the effects of DJB surgery on patients with normal body weight have only been poorly evaluated. Currently, there are two main hypotheses for describing the improvement of glycemia following gastrointestinal surgery: upper intestinal hypothesis and lower intestinal hypothesis [3, 10]. According to the "lower intestinal hypothesis," rapid delivery of nutrients to the lower intestine increases the stimulation of L-cells, resulting in an increase of secretion of hormones that enhance insulin increase and/or insulin action (such as GLP-1) and a subsequent decrease in blood glucose levels.GLP-1 is an incretin hormone secreted from enteroendocrine L cells found in the epithelium of the small and large intestines. L cells have been shown to be present in high numbers along the distal parts of the small intestine and in colon Glucagon-like peptide-1 (GLP-1) receptor (GLP-1R) agonists have emerged as treatment options for type 2 diabetes mellitus (T2DM) [11].GLP-1 and GLP-1 receptor agonists enhance satiety, reduce food intake, and promote weight loss by reducing gastric emptying and suppressing gastric acids secretion in rodents and humans. The enteric nervous system (ENS) has an important role in surgical treatment of T2DM. GLP-1, which is derived from the transcription product of the proglucagon gene, is secreted as a gut hormone by intestinal L-cell inileum [11]. GLP-1R was widely expressed in the submucosal plexus of the entericnervous system (ENS). GLP-1R is also colocalized with cholinergic neurons, nNOS-positive neurons, NPY-positive neurons, somatostatin-positive neurons, and VIP-positive neurons in ENS [12, 13]. GLP-1 could inhibit the electrophysiology of intestinal mucosa plexus and secretion of acetylcholine (Ach). Studies showed that GLP-1 could regulate thelevel of glucose by stimulating the secretion of many hormones in gut [14, 15]. GLP-1also has the ability to stimulate motility. Therefore, ENS has an important role in the surgical treatment of T2DM. In this study, we aimed to investigate GLP-1 secrection and GLP-1, rexpression in ENS of the T2DM rats and the relationship of GLP-1 signaling in the intestine with T2DM treatment by DJB.
MATERIALS & METHODS
Animals and the feeding condition
This study was approved by Weifang Medical University Animal Care and Use Committee. Male Wistar rats (10-12 week's age) were purchased from ShandongLukang Pharmaceutical Group Co, Ltd., China. All rats were kept in a controlled setting at 22°Con a 12:12 h light/dark cycle, fed with standard chow and water adlibitum, except for instances necessitated by preand postoperative care.
STZ-induced T2DM diabetic rat model
A diabetic state was induced by feeding a modified diet containing 41.2% fat for weeks followed by a single intraperitoneal injection of streptozotocin (STZ) (Sigma, USA) at a low dose (35 mg/kg body weight, dissolved in 0.05 M citrate buffer, pH 4.5, immediately before use). One week after injection, fasting blood glucose (FBG) levels were determined from the tail blood using a glucose meter (OneTouch Ultra, USA). Twenty rats with FBG levels above 13.9 mmol/L were randomly divided into DJB surgery and sham groups (n=10 for each group). Ten Wistar rats were used as wild type control.
DJB and Sham Surgeries
DJB and Sham surgeries were performed within one week of T2DM model setting up. Preoperative procedures were performed as described previously [16]. Rats were deprived of food for 16 to 18 h and then anesthetized with ketamine (150 mg/kgbody weight). Prior to the initial incision, morphine (10 mg/kg) was administered subcutaneously. Rats were kept on a water-circulated heating pad throughout the duration of the surgery. The duodenum was separated from the stomach, and bowel continuity was interrupted 10 cm from the ligament of Treitz. The distal limb was connected to the stomach (gastrojejunal anastomosis), and the proximal limb that carries the biliopancreatic juices was reconnected downward to the alimentary limb at a distance of 10 cm from the gastrojejunal anastomosis (Roux-en-Y reconstruction). For the sham operation, transections and reanastomosis of the gastrointestinal tract were performed at sites similar to those of the enterotomies made for DJB, however the physiologic circuit of food was maintained through the gut [16]. Two days after surgery, the rats consumed the modified diet and tap water during the experimental period.
Prior or postoperation observation of blood glucose, insulin and insulin resistance
Fasting blood glucose (FBG) was performed from the tail vein by One Touch glucose meter (Johnson & Johnson Co., USA) after 12 h on fasting. FBG wasdetected one week prior to operation (p.r.), and one, two, four, eight week's post-operation (p.o.). 0.5ml blood sample was collected from the angular vein after 12 h on fasting prior to operation and eight weeks post operation. Blood samples were subsequently centrifuged to separate plasma and stored at−80°C. Insulin level was determined by a rat/mouse ELISA kit (R&D, USA). Anti-rat insulin antibody was added to the 96-well plate (100 μl per well) and incubated at37°C for 60 min. After three times of washing, 100 μl of concentrated enzyme solution was added to each well, and the plate was incubated at 37°C for 30 min. After washing, 100 μl of substrate solution was applied and the plate was incubated at 37°C for 25 min in dark. Finally, the sealer was removed and the stop solution was loaded (100 μl for each well). The absorbance at 450 nm was recorded by the plate reader (ELX808, BioTek, USA) immediately after thorough mixing. RIN-m5F the insulinoma cell line was used as a control. The insulin concentration in the supernatant of RIN-m5F culture medium was examined in a similar manner. The Homeostasis Model Assessment Insulin Resistance (HOMA-IR) index was calculated to estimate insulin resistance according to the following equation: HOMA-IR index = [fasting glucose (mmol/L) × fasting insulin (mIu/L)]/22.5 [17]. Insulin Sensitivity Index (ISI) was calculated according to the following equation: ISI = 1/[fasting glucose (mmol/L) × fasting insulin (mIu/L)].
Immunofluresence examination of the pancreatic islet morphology after surgery
The rats were sacrificed eight weeks after the DJB surgery. Pancreas was removed and then fixed in 4% paraformaldehyde then frozen sections was made for further study. Pancreatic frozen sections were cut at a ten μm thickness. For double-labeled immunofluorescence, pancreatic sections were incubated with mouse anti-insulin antibody (Santa Cruz, USA) and Cy3-conjugated donkey anti-mouse antibody (Jackson immune Research Co., USA); followed by rabbit anti-glucagon (Santa Cruz, USA) and FITC-conjugated donkey-anti-rabbit antibody (Jackson immune Research Co., USA). Insulin and glucagon were visualized as red and green filaments respectively under fluorescence microscopy (Olypus, Japan). To quantify the difference, 10-15 imaging fields (400×) containing islet clusters were randomly selected from pancreas of the 3 experimental groups. The insulin- and glucagon- positive cells in each pancreatic islet were counted, and the percentage of the insulin-positive β cells was calculated, t-test was performed for the groups with ten pancreas islets counted.
GLP-1 secretion after DJB surgery
0.5 ml of blood sample was collected from the angular vein after 12 h of fasting prior to surgery, and subsequently one, two, four, eight week post-surgery. The level of GLP-1(mU/L) was measured by using ELISA kits (R&D, USA) following the protocol of the kit.
GLP-1R expression in enteric nerve system detected by immunofluorescence and western blotting assays
For immune histochemical studies, segments of the ileum were removed and placed in the chilled Krebs’ solution. Small intestinal specimens were opened along the mesenteric border, stretched tautly, and pinned out flat with mucosal side up to Sylgard at the bottom of the dish immediate before fixing Zamboni’s fixative (4%formaldehyde plus 0.2% picric acid in 0.1 M sodium phosphate buffer, pH 7.0) for three hours at room temperature. After fixation, tissues were subsequently washed (3×10 minutes) in phosphate-buffered saline (PBS; 0.9% NaCl in 0.1 M sodiumphosphate buffer, pH 7.0). Wholemounts of the myternic nerve system were microdissected from these segments The myteric plexuses (MPs) were incubated in 10% normal horse serum in PBS for 1 h at room temperature (RT) before exposure to the primary antiserum diluted in hypertonic PBS containing 10% normal horse serum and 0.3% Triton X-100. The preparation was placed in humidified chambers and processed for indirectimmunofluorescence staining by incubation for 18 h at RT. The preparations wereincubated for 24 h with rabbit anti-GLP-1R (1:500 dilution) at RT followed by triplicate 10 min washes with PBS and then incubated in FITC-labeled donkey anti rabbit secondary IgG (Jackson Immuno Research Laboratories, USA) for 1 h at RTand triplicate 10 min washes with PBS. The preparation was placed in the coverslip inVectorshield (Vector Laboratories, USA) and examined by fluorescence microscopy The specificity of the anti-GLP-1R antibodies was controlled by pre-absorbing the antibodies with corresponding blocking peptides provided by the manufacturerswhile samples processed with omission of either the primary or the secondary antibodies were used as negative controls. Protein extracts (100 μg/well) from rat MP preparations were separated on 10%SDS-polyacrylamide gel, transferred to PVDF membrane, blocked with 10% skimmed milk in Tris-buffered saline containing Tween 20 (TBS-T), and then probed withrabbit anti-GLP-1R (1:500 dilution) at 4°C overnight. Horseradish peroxidase-conjugated anti-rabbit IgG were used with a dilution of 1:2000 for 2 h at RT. After washing with TBS-T three times, immune reactive bands were visualized using Pierce ECL Western Blotting Substrate system. β-actin (1:1000 dilution) was used as theloading control. The band intensity was further quantified using BandScan software.
Statistical Analysis
Data were expressed as mean ± SEM, and Graph pad Prism 5.0 software wasapplied to perform statistical analysis. Statistical differences were determined usingone-way ANOVA between different groups, and paired Student's t tests were adoptedto compare the changes in the single pre- and post-operative indicators. A P-value less than 0.05 was considered statistically significant.
RESULTS
DJB surgery improved glucose tolerance and insulin resistance of diabetic rats
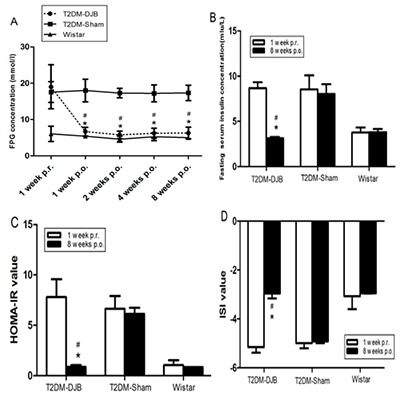
DJB surgery had high efficacy on T2DM rats, evidenced by the increase of glycemic control and insulin resistance of diabetic rats. We measured the levels of fasting blood glucose (FBG) value of each group one week prior to and one, two, four, eight weeks after surgery. As shown in (Figure. 1A), the FBG of the established T2DM group increased significantly compared to the control group, and remained high one week before operation (including T2DM-Sham and T2DM-DJB subgroups). FBG values in the T2DM-DJB group significantly decreased from 19.02 ± 2.15 mmol/L to 6.67 ± 0.69mmol/L 1 week after surgery. T2DM-Sham group remained unchanged (P < 0.05). In T2DM-DJB group, FBG was 5.77 ± 0.60 mmol/L, 6.23 ± 0.79mmol/L, 6.29 ±0.56mmol/L at two, four and eight weeks after operation respectively. While FBG in T2DM-Sham group was 17.03 ± 0.75 mmol/L, 17.20 ± 1.35 mmol/L, 17.33 ± 0.75mmol/L at the correspondingtime points. Analysis of the data from two subgroups showed that FBG in T2DM-DJB is significantly reduced after surgery compared totheT2DM-Sham group. Throughout the whole period of the experiment, FBG in Wistar rats remains largely unchanged. DJB surgery could reduce the fasting insulin level of diabetic rats. Fasting insulin was detected by ELISA assay 1week prior till eight weeks post-surgery The results showed that the fasting insulin also decreased significantly in the DJB surgery group (Figure. 1B) from 8.67 ± 0.38 to 3.14 ± 0.08 (mlu/L). T2DM-Shams surgery group rats did not significantly change eight weeks after surgery. DJB surgery decreased HOMA-IR index of diabetic rats. (Figure. 1C) showed an impaired glucose tolerance that was in accordance with a low insulin action, as demonstrated by 7.8-fold and 6.62-fold increases in HOMA-IR index inT2DM-DJB vs. T2DM-Sham surgery rats or before surgery, respectively. DJB surgery enhanced glucose tolerance and Insulin Sensitivity(ISI) in T2DM-DJB rats. As shown in (Figure. 1C), HOMA-IR was significantly reduced from 7.8 ± 1.02 to 0.87 ± 0.10 eight weeks after DJB surgery. By contrast, T2DM-Sham rats did not show any significant changes compared to those before surgery. DJB surgery increased ISI of diabetic rats. As shown in (Figure 1) D, ISI significantly increased from -5.15 ± 0.13 to -2.96 ± 0.11 eight weeks after DJB surgery. T2DM-Sham and Wistar rats did not show any significant changes within the period of experiments.
Figure 1: Improvement in glucose tolerance and insulin resistance of STZinduceddiabetic rats by DJB. (A) FPG concentrations in all groups of rats (n = 6) weremeasured one week pre-operation (p. r.) and 1, 2, 4, and 8 weeks post-operation (p. o.)by blood glucose meter. (B) Serum insulin levels in all of the groups (n = 6) wereevaluated by ELISA at one week preoperation and eight weeks post-operation. (C)Homeostasis model assessment insulin resistance (HOMA-IR) index was calculated to estimate insulin resistance according to the following equation: HOMA-IR index =[fasting glucose (mmol/L) × fasting insulin (mIu/L)]/22.5 [17]. (D) Insulin sensitivityindex (ISI) was calculated according to the following equation: ISI = 1/ [fasting glucose (mmol/L) × fasting insulin (mIu/L)]. #P < 0.05 compared with T2DM-Shamgroup at the same point; *P < 0.05 compared with pre-operation.
Distribution and percentage of pancreatic α and β cells within the islets
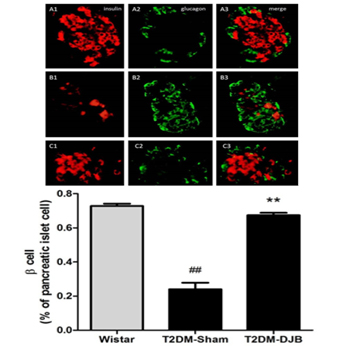
The quantity of β cells (insulin-positive) and α cells (glucagon- positive) in pancreas were measured by double labelling immunofluorescence. Insulin-positive cells and glucagon positive cells are shown in (Figure 2), insulin was labeled with Cy3(red) and glucagon FITC (green). Insulin and glucagon immune reactivity (IR) were detected in the pancreas islets of Wistar rats (A1-3), T2DM-Sham rats (B1-3) and T2DM-DJB rats (C1-3). (Figure 2A) showed the double labelling of insulin and glucagonin pancreas islet of Wistar rats. In normal Wistar rats, insulin was widely distributed inthe pancreas islet, and that the islet appeared round and normal. Glucagon is expressed at the edge of pancreas islet. The quantity of glucagon-positive cells was less than insulin-positive cells. The percentage of insulin positive cell is 72.8±3.1%when counting 1000 cells in 6 rats. (Figure 2B) represents the double labelling of insulin and glucagon in the pancreas islet of T2DM-Sham rats 8 weeks after surgery. The results indicated that insulin positive cells were significantly decreased and glucagon positive cells increased sharply in T2DM sham rats eight weeks after surgery, as compared to, control wistar rats. In the pancreatic islets of T2DM-Sham rats, insulin positive cells decreased to 24.1±8.5%. (Figure 2C) showed double labelling of insulin and glucagon in pancreas islet of T2DM DJB rats eight weeks after operation. The percentage of insulin positive cells increased back to 67.4±3.3% in the pancreas islet after DJB treatment for eight weeks. These results suggested that the number of insulin positive cells gradually increased. On the other side, glucagon positive cells increased significantly in T2DM-Sham rats and decreased after DJB eight weeks after the surgery.
Insulin and glucagon expression in the rat pancreas islet were examined bydouble labelingimmuneflurences. Insulin was labeled with Cy3 (red). Glucagon waslabeled with FITC (green). Insulin and glucagon immune reactivity (IR) wasexpressed in the pancreas islet of Wistar (A1- 3), T2DM-Sham rats (B1-3) and T2DMDJBrats (C1-3). The results showed that DJB surgery could increase the quantityinsulin positive cells. A. Double labeling of insulin and glucagon in pancreas islet ofWistar rats. In the normal Wistar rats, Insulin was expressed widely in the pancreasislet, and the islet showed round and normal. Glucagon expressed in the edge ofpancreas islet. The quantity of glucagon positive cells was less than insulin positivecells. B. Double labeling of insulin and glucagon in pancreas islet of T2DM-Shamrats 8 weeks after operation. Insulin positive cells decreased significantly comparing to Wistar control rats and glucagon positive cells increased sharply instead. C. Doublelabeling of insulin and glucagon in pancreas islet of T2DM DJB rats 8 weeks post operation(p.o). The results showed insulin positive cells recovered but not reaching to thenormal level, and the glucagon positive cells decreased respectively
DJB surgery increased plasma GLP-1 secretion of diabetic rats
GLP-1 secretion was detected by ELISA. As shown in Table.1, the plasma GLP-1 secretion was significantly decreased in the T2DM-Sham and T2DM-DJB rats, as compared to, Wistar rats (10.10 ± 0.82, 9.96 ± 0.36 vs. 17.50 ± 0.46uIU/L). This result indicated that the GLP-1 secretion in T2DM rats significantly decreased in diabetic condition. Two weeks after DJB treatment, GLP-1 secretion significantly increased to 20.19 ±1.67 uIU/L, as compared to, the Sham group at 10.44 ± 0.53 (P < 0.05). GLP-1 was maintained at 21.31 ± 2.42 and 16.76 ±1.50 uIU/L 4, 8 weeks after DJB surgery respectively, while GLP-1 was unchanged in 10.38± 0.62 and 10.53 ±0.59 in T2DM-Sham rats at the corresponding periods. The secretion of GLP-1 in T2DM DJB significantly increased two weeks after DJB treatment compared to the Shame group.
Table1: Fasting plasma GLP-1 concentration (mlu/L).
| T2DM-DJB | T2DM-Sham | Wistar | |
|---|---|---|---|
| 1 week p.r. | 10.10±0.82 | 9.96±0.36 | 17.50±0.46# |
| 2 weeks p.o. | 20.19±1.67#* | 10.44±0.53 | 16.23±0.59# |
| 4 weeks p.o. | 21.31±2.42#* | 10.38±0.62 | 16.98±0.64# |
| 8 weeks p.o. | 16.76±1.50#* | 10.53±0.59 | 18.12±0.42# |
DJB surgery increased plasma GLP-1 secretion of diabetic rats. The plasma GLP-1 level was analyzed by the assay of ELISA 1 week prior(p.r) to and 2, 4, 8 weeks post operation (p.o) (n = 6). *P < 0.05 vs. 1 week prior to DJB surgery; #P < 0.05 compared with T2DM-Sham.
DJB surgery unregulated GLP-1 receptor expression in ENS
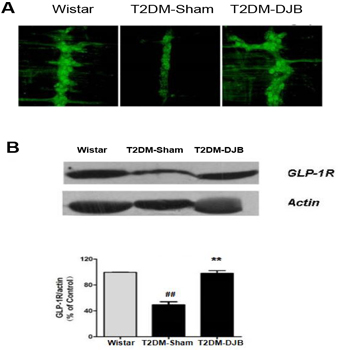
Eight weeks after surgery, GLP-1R expression in the myteric nerve plexes (MP) tissues was detected by immunohistochemistry and Western blotting. Immunofluorescence showed theincrease of GLP-1R in myteric nerve system in rat groups. As shown in Figure 3A, GLP-1R in MP in ENS of the Wistar rats (Figure 4B left) was significantly decreased in T2DM sham group (Figure 3A, middle). Eight weeks after DJB surgery, this reduction was significantly reversed. Figure 3A (right) showed the high density of GLP-1R in MPs of ENS. The expression of GLP-1R in MPs of ENS in three rat groups was also determined by Western blotting. As shown in Figure 3B and 3C, GLP-1R expression in T2DM rats was significantly decreased by 61% in T2DM as compared to Wistar group. DJB treatment resulted in the recovery of GLP-1R expression by 100% as compared to the Wistar control (Figure 3B and 3C right).
Eight weeks after surgery, the GLP-1R expression in the rat myenteric nervous plexuses tissues was examined by immunohistochemistry and Western blotting. A.Fluorescence immune histochemisty for GLP-1R in the myenteric plexuses preparations in Wistar rats (a), T2DM-Sham rats 8 weeks after operation (b) T2DM-DJB 8 weeksafter operation (c) (40×). B. Western blotting analyzed GLP-1R expression in ENS.Molecular weight of GLP-1R and β-actin is 56 and 42 kDa, respectively. C. Therelative optical density (ROD) of immunoblot band is represented as percentagevalues corrected for GLP-1R with ×-actin (n = 3 per group). The bars indicate themeans ± SEM. (**P < 0.01 vs. Wistar group; ## P < 0.01, vs. diabetic group).
DISCUSSION
ENS is composed of nerve cells in gastric wall cell body and its lobes, gastrointestinal submucosa, and links between the longitudinal muscle layers. It iswell known that ENS is independent of the external brain neural networks [5]. Our previous study reported that GLP-1R expressed in ENS and GLP-1 modulates neutrally evoked mucosal chloride secretion in the intestine of Guinea pigc [6]. Here we further investigated the function of GLP-1 and GLP-1R in ENS during the development and treatment of T2DM by DJB [18]. Numerous studies have indicated that DJB surgery prevented a direct contact between the ingested nutrients and the duodenumand proximal jejunum without any gastric restriction or exclusion. Thus DJB has been considered as an alternative surgical therapy for promoting glucose homeostasis without significant weight loss. Some reports have shown a remarkable improvement in glucose homeostasis at the early stage following DJB surgery [19]. This study showed that DJB surgery increased GLP-1 secretion and GLP-1Rexpression in ENS connected with the reduced blood glucose level of the T2DM rats.The role of ENS and the signal of GLP-1 in ENS under DJB surgery remains to be further discovery. Considering the physiological consequences bariatric surgery treating T2DM, resulted in dramatically elevated GLP-1 levels, antagonism of which largely abolishes postsurgical improvements in glucose-stimulated insulin release. GLP-1 mimetics, however, are markedly less effective than surgery in treating type 2 diabetes. Although it was believed that GLP-1 target β-cells through the circulation, but GLP-1 is rapidly cleaved by dipeptidyl peptidase 4(DPP4) when it enters the bloodstream [20]. This led to the idea that receptors localized close to L-cells may act as local sensors of GLP-1 before it is inactivated. And this is the reason we have studied the GLP-1R expression in ENS when DJB was performed in treating T2DM rats. Paul Richardsshowed that GLP-1R was widely expressed in different organs [21-25].In pancreatic islets, GLP-1R expression was largely restricted to the β-and δ-cell populations, only less than 10% a-cells expressed GLP-1R. In the gastrointestinal system (GI), GLP-1R is localized in afferent and entericneuronal cell bodies and nerve fibers within the intestinal mucosa including gastricantrum/pylorus, enteric neurones, vagal and dorsal root ganglia. Patch clamp recording that enteric neurons positive for GLP-1R were activated by GLP-1.Enteric and vagal neurons positive for GLP-1R activated by GLP-1 and may contribute to intestinal and central responses to locally released GLP-1, regulation of intestinal secretomotor activity and appetite, remains to be researched .To the best of our knowledge, this study offers the first direct evidence on the relationship between a change in GLP-1 signaling in ENS induced by DJB surgery and the subsequent improvement in postoperative glucose tolerance.
Compliance with Ethical Standards
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in this study involving animals were approved by the Ethics Committee of Weifang Medical University, Weifang, China.
ACKNOWLEDGEMENTS
This project was supported by grants from National Natural Science Foundation of China (31671208, 81274093, Zhiqin Gao), Shandong Province Natural Science Foundation (ZR2015HL128, Mei-Hua Qu), Health department of shandong province (2014WS0478, Mei-Hua Qu), project of Shandong Province Higher Educational Science and Technology Program ( J14LK15, Xiaoyun Yang).
REFERENCES
- Schauer PR, Burguera B, Ikramuddin S, Dan Cottam, et al. (2003). Effect of laparoscopic Roux-en Y gastric bypass on type 2 diabetes mellitus. Ann Surg. 238, 467-85.
- Cohen R, Pinheiro JS, Correa JL and Schiavon CA. (2006). Laparoscopic Roux-en-Y gastric bypass for BMI _35 kg/m (2): a tailored approach. Surg Obes Relat Dis. 2, 401-404
- Cohen RV, Schiavon CA, Pinheiro JS, Correa JL, et al. (2007). Duodenal-jejuna bypass for the treatment of type 2 diabetes in patients with body mass index of 22-34 kg/m2: a report of 2 cases. Surg Obes Relat Dis. 3,195-197.
- Krein SL, Funnell MM and Piette JD. (2006). Economics of diabetes mellitus. Nurs. Clin. North Am. 41, 499-511.
- Mehaffey JH, Mullen MG, Mehaffey RL, Turrentine FE, et al. (2016). Type 2 diabetes remission following gastric bypass: does diarem stand the test of time?. Surg Endosc.
- Rubino F, Schauer PR, Kaplan LM and David E. (2010). Cummings. Metabolic Surgery to Treat Type 2 Diabetes: Clinical outcomes and mechanisms of action. Annu Rev Med. 61, 393- 411
- Laville M and Disse E. (2009). Bariatric surgery for diabetes treatment: why should we go rapidly to surgery. Diabetes & Metabolism. 35, 562-563.
- Jurowich CF, Rikkala PR, Thalheimer A, Wichelmann C, et al. (2016). Duodenal-Jejunal bypass improves glycemia and decreases SGLT1-mediated glucose absorption in rats with streptozotocin-induced type 2 diabetes. Ann Surg. 258, 89-97.
- Kindel T L, Yoder S M, Seeley R J, D'Alessio DA, et al. (2009). Duodenal-jejunal exclusion improves glucose tolerance in the diabetic, Goto-Kakizaki rat by a GLP-1 receptor-mediated mechanism. J J Gastrointest Surg. 3(10), 1762-1772.
- Rubino F and Marescaux J. (2004). Effect of duodenal-jejunal exclusion in a non-obese animal model of type 2 diabetes: a new perspective for an old disease. Annals of Surgery. 239(1), 1-11.
- Mason EE. (1999). Ileal transposition and enteroglucagon/ GLP-l in obesity surgery. Obes Surg. 9(3), 223-228.
- Lund A, Vilsboll T, Bagger JI, Holst JJ, et al. (2011). The separate and combined impact of the intestinal hormones, GIP, GLP- 1, and GLP-2, on glucagon secretion in type 2 diabetes. Am J Physiol Endocrinol Metab. 300, E1038-1046.
- Calanna S, Christensen M, Holst JJ, Laferrere B, et al. (2013). Knop Secretion of glucagon-like peptide-1 in patients with type 2 diabetes mellitus: systematic review and metaanalyses of clinical studies. Diabetologia. 56(5), 965-972
- Baldassano S, Wang G D, Mule F and Wood JD. (2012). Glucagon-like peptide-1 modulates neurally evoked mucosal chloride secretion in guinea pig small intestine in vitro. Am J Physiol Gastrointest Liver Physiol. 302, G352-G358.
- Drucker D J. (2007). The role of gut hormones in glucose homeostasis. J Clinical Invest. 117, 24-32.
- Sun D, Wang K, Yan Z, Zhang G, et al. (2013). Duodenaljejunal bypass surgery up-regulates the expression of the hepatic insulin signaling proteins and the key regulatory enzymes of intestinal gluconeogenesis in diabetic Goto-Kakizaki rats. Obes Surg. 23(11), 1734-1742.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, et al. (1985). Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasmaglucose and insulin cencentrations in man. Diabetologia. 28(7), 412-419.
- Mei-Hua Qu, JiMa Xu, ChunYan Fang, TingTun Zhao, et al. (2012). The Study of GLP-1R expression in enteric nerve system of type 2 diabetic rat after duodenal-jejunal bypass surgery. Neurogastroenterology and Motility. 2, 132-133.
- . Donglei Z, Liesheng L, Xun J, Chenzhu Z, et al. (2012). Effects and mechanism of duodenal-jejunal bypass and sleeve gastrectomy on GLUT2 and glucokinase in diabetic Goto-Kakizaki rats. Eur J Med Res. 17(1), 15-16.
- Janardhan S and Sastry GN. (2014). Dipeptidyl peptidase IV inhibitors: a new paradigm in type 2 diabetes treatment. Curr Drug Targets. 15(6), 600-21.
- Richards P, Parker HE, Adriaenssens AE, Hodgson JM, et al. Identification and characterization of GLP-1 receptor-expressing cells using a new transgenic mouse model. Diabetes. 63(4), 1224-33.
- Cohen RV, Schiavon CA, Pinheiro JS, Correa JL, et al. (2007). Duodenal-jejuanl bypass for the treatment of type 2 diabetes in patients with the body mass index of 22-34kg/m2: a report of 2 cases. Surg Obes Relat Dis. 3, 195-197.
- Marceau P, Hould FS, Simard S, Lebel S, et al. (1998). Biliopancreatic diversion with duodenal switch. World J Surg. 22, 947-954.
- Toyoda K, Okitsu T, Yamane S, Uonaga T, et al. (2008). GLP- 1 receptor signaling protects pancreatic beta cells in intraportal islet transplant by inhibiting apoptosis. Biochem Biophys Res Commun. 367(4), 793-8.
- Saeidi N, Meoli L, Nestoridi E, Gupta NK, et al. (2013). Reprogramming of intestinal glucose metabolism and glycemic control in rats after gastric bypass. Science. 341(6144), 406-410.