Information Links
Related Conferences
Previous Issues Volume 4, Issue 2 - 2020
An Overview on Cancer-Fighting Phytochemicals from Selected Medicinal Plants in Bengal
Sanghita Das1,2, Anindita Dey2,3*, Sukhen Das1, Papiya Nandy3
1Department of Physics, Jadavpur University, Kolkata-700032, India
2Department of Botany, Asutosh College, Kolkata-700026, India
3Centre for Interdisciplinary Research and Education, 404B, Jodhpur Park, Kolkata-700068, India
*Corresponding author: Anindita Dey, Department of Botany, Asutosh College, Kolkata-700026, India, E-mail: [email protected].
Received Date: November 07, 2020
Publication Date: December 09, 2020
Copyright: Sanghita D. ©2020.
Citation: Sanghita D, et al. (2020). An Overview on Cancer-Fighting Phytochemicals from Selected Medicinal Plants in Bengal. Mathews J Pharm Sci. 4(2):05.
ABSTRACT
Bengal possesses a diverse gene pool of ethno-botanically important plant species for alternative medicinal agents. Herbal remedies, also known as botanical medicine, have been recognized as a promising complementary treatment for cancer. In this article, we have methodically highlighted and summarized most popular and effective Bengal plants which possess phytocomponents with anti-cancer properties. Several in vitro as well as in vivo studies demonstrated the details of plant derived metabolites and their promising efficacy against different cancer cell lines. Therefore, recapitulated data about the bioactivity of these phytochemicals, with special emphasis on Bengal medicinal plants will enrich our knowledge about developing carefully designed standardized drug in controlling the carcinogenic processes traditionally over conventional therapies to prevent this global health crisis.
KEYWORDS: Phytochemicals; Anti-cancer; Medicinal plant; Traditional medicine
INTRODUCTION
Cancer remains to be one of the leading causes of mortality worldwide. Though the modern conventional therapeutic approach has indisputably enhanced survival rate, metastasized cancer remains untreatable. Hence, continued searching for more efficient and effective chemoprevention is clearly desirable to improve the treatment [1]. According to an estimation of World Health Organization, 80 % of rural population rely chiefly on medicinal herbs and traditional medicine as a primary healthcare system [2]. In the Ayurveda, numerous plants with medicinal properties are documented in various texts but it requires newer guidelines of standardization, production and quality control. It has been reported that approximately 50% of all modern pharmaceutic in clinical use are plant derived [3]. Many of these have been recognized to have apoptotic activity in various cancer cells of human origin [1]. Any part of a medicinal plant such as seeds, bark, leaves, roots, flowers, fruits can contain some bioactive substances that can be used for therapeutic or medicinal purposes. Phytochemicals are the secondary metabolites that are taxonomically extremely diverse in nature and an excellent reservoir of potential precursors of new drugs based on their modes of pharmacological action [4,5]. Moreover, these bioactive compounds such as flavonoids, terpenoids and alkaloids have received considerable attention for their anti-mutagenic, anti-malignant, antineoplastic and potential chemo preventive properties through their effects on signal transduction in cell proliferation and angiogenesis [5].
India is a heritage country in term of natural resources and biodiversity. West Bengal (a state occupies only 2.7% of India’s land area) possesses an enormous number of medicinal plants [6]. The tropic of cancer passes almost through the middle of the state. Diverse climatic conditions of West Bengal include a tropical wet-dry climate in the southern part and a humid subtropical climate in the north (http://www.westbengalforest.gov.in). The total forested area of West Bengal is 11879 sq. km. which is 13.38% of the total geographic area of the state (http://www.westbengalforest.gov.in/history.php). At present West Bengal has 23 districts which are distributed in five agro-climatic zones i.e. Darjeeling Himalayan hill region, Tarai – Dooars region, western undulating high land and plateau, north and southern plains of Bengal and Gangetic deltaic regions are favourable to establish the diversity of plants [7]. But unscientific and unorganized harvesting and production of raw materials, lesser concern about quality control, fluctuation in demand and supply, lack of coordination, research and inefficient marketing infrastructure are the main difficulties to promote these therapeutic plants effectively. As per recommendations of the National Medicinal Plant Board (NMPB) as well as West Bengal State Medicinal Plant Board (WBSMPB) some medicinal plant species are recognized for the scientific cultivation in West Bengal. These herbal plants are prioritized because of its vast uses in Indian System of Medicine and Homeopathy (ISM & H). Department of AYUSH (Ayurveda, Yoga, Unani, Siddha and Homeopathy systems) of the Government of India regulates researches on indigenous alternative medicines and their quality control and practices (https://www.ayush.gov.in/). Therefore, scientific cultivation, conservation, suitable maintenance measures regarding harvesting and marketing of medicinal plants may lead to greater success in cancer prevention.
PLANTS WITH ANTICANCER ACTIVITY
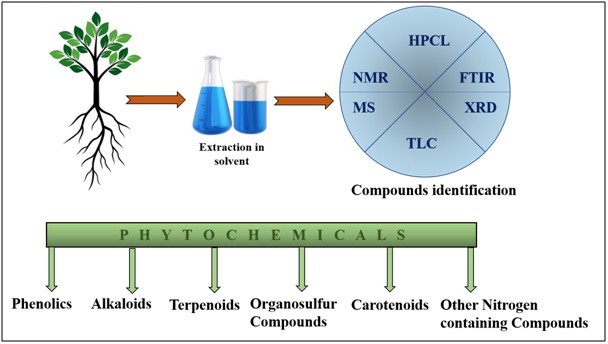
According to previous reports, these medicinal plants contain some important active components i.e. vitamins, carotene, enzymes, minerals, polysaccharides, polyphenols, flavonoids, lignin, xanthones, etc. [Figure 1] which exert potent anticarcinogenic and antimetastatic activities [1]. Plants described in this study are endemic in West Bengal state and also well acknowledged possessing several antioxidants. A significant number of research work has been done about the anticancer efficacy of these plants. Thus, the various combinations of the phytochemicals extracted from these plants may undergo further assessment for their synergistic activity after identification. With the above background, this review article enumerates 20 medicinal plants from West Bengal, according to their suppressive and antiproliferative effect on specific cancer types as well as anti-tumor, antimetastatic and antioxidant properties [Table 1]. We have chosen these plant species based on their availability throughout the state, their popularity among people and last but not the least their significant ability to cure the deadly disease cancer to some extent.
Figure 1: Schematic representation of isolation techniques and types of phytochemicals.
|
Serial No. |
Scientific Name with Family |
Active Components |
Effect in Cancer |
|
1 |
Achyranthes aspera (Amaranthaceae) |
Achyranthine, phenolic compounds |
|
|
2 |
Aerva lanata (Amaranthaceae)
|
Aervitrin, aervolanine, campesterol, kaempferol |
|
|
3 |
Allium sativum (Liliaceae)
|
Allicin, alliin, allixin, Z-again |
|
|
4 |
Alstonia scholaris (Apocynaceae)
|
Echitamine, alstonidine, alstonin |
|
|
5 |
Andrographis paniculate (Acanthaceae)
|
Andrographolide, xanthones, stigmasterols |
|
|
6 |
Artemisia indica (Asteraceae)
|
Ludartin, lupeol |
|
|
7 |
Azadirachta indica (Meliaceae)
|
Nimbolide,azadirachtins, nimocinol, isomeldenin, azadirachtol, |
|
|
8 |
Bauhinia variegata (Fabaceae)
|
Flavonoids, anthraquinones, saponins |
|
|
9 |
Butea monosperma (Fabaceae)
|
Butrin, butein, butin, isobutrin, isocoreopsin |
|
|
10 |
Calotrophis gigantea (Asclepiadaceae)
|
Pregnanes, terols, flavonol glycosides, usharin, gigantin, giganteol, giganteol |
|
|
11 |
Camellia sinensis (Theaceae)
|
Epigallocatechin-3-gallate, epigallocatechin, epicatechin-3-gallate, epicatechin |
|
|
12 |
Cassia fistula (Leguminosae)
|
Rhein, emodine, physion, chrysophanol, Obtusin, chrysoobtusin |
|
|
13 |
Centella asiatica (Apiaceae)
|
Asiatic acid, kaempferol, asiaticosside |
|
|
14 |
Curcuma longa L. (Zingiberaceae)
|
Curcumin, demethoxycurcumin, bisdemethoxycurcumin |
|
|
15 |
Emblica officinalis (Euphorbiaceae)
|
Pyrogallol, gallic acid |
|
|
16 |
Oroxylum indicum (Bignoniaceae)
|
Baicalein, oroxylin A |
|
|
17 |
Semecarpus anacardium (Anacardiaceae)
|
Galluflavanone, phenolic compounds |
|
|
18 |
Syzygium cumini (Myrtaceae)
|
Betulinic acid, Kaempferol 7-O-methylether isoquercitin, quercetin, |
|
|
19 |
Vitex negundo (Verbenaceae)
|
Evn-50 |
|
|
20 |
Withania somnifera (Solanaceae)
|
Withaferin A |
|
Table 1: Name of the selected Bengal plants, active components and their role in anticancer studies.
Achyranthes aspera
Family: Amaranthaceae Parts Used: Leaf, stem, seed
Activity: In traditional medicinal system of Ayurveda A. aspera (local name: Apang) is well known for hepatoprotective, diuretic, immunostimulatory and anti-cancer properties [8,9]. A. aspera, an annual shrub frequently found in tropical and warmer regions in India reportedly contains fatty acids, oleonic acid, triterpenoid based saponins, oleanolic acid, triacontanol, dihydroxy ketones, betaine, achyranthine and various amino acids [10]. The methanol extract of A. aspera shows higher quantity of phenolic compounds compared to aqueous extract [9]. It has been reported that leaf extract is used to treat cancer, particularly breast and cervix cancer [10]. Alkaloid extract of leaf induced apoptosis in breast cancer cell through p53 pathways [11]. Several studies demonstrated that the methanolic extract of leaves contains potent antiproliferative and cytotoxic activity against pancreatic cancer cell lines through the inhibition in the expression of pro metastatic and angiogenic genes [8].
Aerva lanata
Family: Amaranthaceae Parts Used: Aerial parts
Activity: In traditional Ayurvedic medicines, A. lanata (local name: Chaya) was found to be effective against several medical conditions for its antihyperglycemic, hepatoprotective, anti-diabetic, anti-urolithiasis, immunomodulatory properties [12]. A. lanata extracts have significant amount of biologically active secondary metabolites like polyphenols, flavanoid glycosides, aervitrin, aervolanine, aervoside, kaempferol, amyrin betulin, campesterol [12]. Some previous studies proved free radical scavenging activity of ethanol, chloroform and hexane extracts of A. lanata leaves [13]. A. lanata displays strong antiproliferative activity and induced apoptosis of Hep3B (hepatic cancer cells) cell lines [14]. Previous studies showed that p53 mRNA expression was found to decrease Hep3B cells in a dose dependent manner and induced apoptotic activity when treated with petroleum ether extract of A. lanata [14]. The methanolic callus extract of this plant contains potential anticancer property on MCF – 7 cell lines for its significant anti- proliferative activity by induction of the apoptosis in cancer cells. Also the methanol extract of the aerial parts of A. lanata has proven to be a source of potent anticancer and antioxidant compounds when treated on Ehrlich Ascites Carcinoma (EAC) cells in Swiss albino mice by monitoring inhibition of tumor cell growth , measurement of tumor weight and survival time of mice [15].
Allium sativum
Family: Liliaceae Parts Used: Bulb
Activity: A. sativum (local name: Rasun) has been attributed in the Indian medicinal system to possess several medicinal effects. The consumption of garlic offers multiple beneficial properties for its chemo-preventive as well as anti-tumor activity [16]. It has been reported that garlic extract contains some organosulfur phytochemicals like diallylthiosulfinate (allicin), S-allylcysteine sulfoxide (alliin), allixin, adenosine, allyl1,5-hexadienyl trisulphide, allyl methyl trisulphide and eight vital amino acids [17]. There is convincing evidence that the consumption of garlic bulb extract reduces the risk of colorectal, lung and esophageal cancers [16]. Studies showed that garlic can also act against stomach cancer by repressing Helicobacter pylori [18]. Its organosulfur components which have free radical scavenging activity also cause cell cycle arrest [17]. It has been found from studies that garlic in several forms can change carcinogen metabolism, reduce formation of carcinogenic products, induce phase II detoxification enzymes including glutathione transferases, quinone reductase, promote apoptosis in cancer cells and inhibit tumour initiation [16]. Several studies demonstrated that the bioactive phytochemicals of garlic modifying the cytokine pattern which leads to an inhibition of a NFĸB, a prime regulator of pro-inflammatory gene expression [19]. The immune modulatory activity of garlic shifts a proinflammatory and immunosuppressive cellular environment to an enhanced anti-tumor response which helps in tumorsuppression. Garlic contains two very effective trace metals, germanium and selenium, which have potential therapeutic value in cancer treatment [17]. Researchers hypothesized that the phytocomponents present in garlic evokes anti oxidative, immune-modulating and anti-inflammatory responses which suppress a developing malignancy [18]. Z-again, a component derived from garlic reportedly has a range of biological properties like anti proliferative effects on cancer stem cells (CSC) of brain malignancies like Glioblastoma multiforme (GBM) [18].
Alstonia scholaris
Family: Apocynaceae Parts Used: Bark
Activity: A. scholaris (local name: Saptaparni) is a medicinal plant, whose bark have been pharmacologically proven to possess anticancer properties [20]. It is most extensively used in different cultures and civilizations such as India, in herbal formulations for many years [20]. The bark of this species are rich in alkaloids, steroids, triterpenoids, and flavonoids but it is valued for its alkaloids such as echitamine, alstonidine, alstonin, ditain, ditainealstovenine, echicaoutchin, echicerin, echiretin, porphyrine, echaitein, chlorogenine, porphyrosine, and reserpine [21]. The powerful alkaloids of A. scholaris protect cells from the damage by free radicals [21]. From earlier researches it has been revealed that the methanolic extracts of the root bark possess cytotoxic activity against human lung cancer cell lines, adenocarcinoma (MOR-P), and large cell carcinoma (COR-L23). Studies showed that a hydroalcoholic extract of A. scholais also has promising antineoplastic effects [22]. The antineoplastic activity of this bark extract from the same tree in vitro study against HeLa cells (cultured human cervical neoplastic cells) showed that the rate of cell mortality was dependent on the season when the plant bark was harvested and the cytotoxic effects were highest in summer (IC50 of 30 µg/ml) [22]. Echitamine, a bioactive phytochemical of bark extract also has cytotoxicity against HepG2, HL60, HeLa, KB, MCF-7 cells, Vero cells, fibrosarcoma, and Ehrlich ascites carcinoma in vitro [23]. Alstonine, another indole alkaloid present in A scholaris, is reported to possess antineoplastic effect [24]. Multiple reports also demonstrated that the triterpenoid lupeol present in A. scholaris induced cell cycle arrests at G1-S phase and is responsible for increase in the expression of p21 protein in PC-3 cells as well as decrease in cyclin D1, cyclin D2, and cdk2 expressions. It has been reported that bioactive bark components reduce the expression of Ras oncoprotein [22]. Additionally, studies revealed that bark extracts downregulate Bcl2, upregulate Bax, activate caspase-3, and induce poly(ADP) ribose polymerase cleavage, and activate caspase-3, -9, and apaf1 genes in CWR22Rnu1 and PC-3 neoplastic cells which lead to apoptosis [22].
Andrographis paniculate
Family: Acanthaceae Parts Used: Aerial part
Activity: A. paniculate (local name: kalmegh) has been widely recognized as a natural remedy for various physiological disorders. Diversified medicinally active phytochemicals like flavonoids,diterpenoid lactone, xanthones, stigmasterols have been isolated from the extract of A. paniculate [25]. The methanol extract (concentration of 10 µg/mL) of aerial part displays the anti-proliferative activity against HT-29 (colon cancer) cells by 50% but the aqueous extract did not inhibit the proliferation of HT-29 cells [26]. Andrographolide, a diterpenoid, repressed the proliferation of cancer cells promisingly. Previous studies demonstrated that andrographolide exhibited cytotoxic activity against KB (human epidermoid carcinoma) cells and P388 (lymphocytic leukaemia) [26]. Andrographolide 1 (diterpene lactone) of A. paniculate extract also has antitumor activity against breast cancer cell lines and mouse myeloid leukaemia cells [27]. Some recent reports displayed the potential of andrographolide (1) to act as a promising anticancer chemotherapeutic compound as it blocks cell cycle progression by decreasing cyclin-dependant kinase (CDK4) expression [27].
Artemisia indica
Family: Asteraceae Parts Used: Leaves, flowering stems
Activity: Aerial parts of A. indica (local name: Naagdana) has been reported to have anti-parasitic, hepato-protective, anti-helminthic and antiseptic properties [28]. It deserves further research into the chemoprevention and anticancer activity [28]. Among the 43 compounds isolated from extracted essential oils (representing 96.6% of the oil), artemisia ketone (42.1%), germacrene B (8.6%), borneol (6.1%) and cis-chrysanthenyl acetate (4.8%) are some major phytoconstituents exhibiting significant cytotoxic and antioxidant activities [29]. Some biological evaluation demonstrated that the essential oil from A. indica leaves have strong toxic effects on liver cancer cells HepG2 [30]. Essential oil extracted from A. indica exhibited significant reduction of cell viability against the HT-29 cells of colon cancer, THP-1 cells of leukaemia, A-549 cell of lung cancer [30]. A strong inhibitory activity of the ethyl acetate extraction of A. indica (having ludartin and lupeol) was reported against cultured human tumor cell lines MCF-7, BHY, Miapaca-2, Colo-205 and A-549. Researchers also revealed that the anti-proliferative effects of ludartin and lupeol as anticancer agents may be due to the significant DNA damage and loss of mitochondrial membrane potential. However, a favourable interaction between the chemicals may be responsible for the overall antiproliferative action of the extract [28].
Azadirachta indica
Family: Meliaceae Parts Used: Leaf, seed, bark
Activity: A. indica (local name: Neem), a plant containing are markably diverse array of phytochemicals like terpenoids, flavonoids, coumarins, carbohydrates, proteins were found to cure different ailments due to its anti-plasmodial, antioxidant, anti-angiogenic, anti-cancer, anti-bacterial, antiviral, and fungicidal activities [31]. Its varied pharmacological properties attributed to extractions of different parts of these plants containing phytocompounds like azadirachtins, nimocinol, isomeldenin, azadirachtol (a tetranortriterpernoid), 2,3′-dehydrosalanol gedunin, nimbin, nimolicinol, odoratone, azadironolide, isoazadironolide [31]. The chemo-preventive effects of dietary doses of aqueous neem leaf extract are useful for its anticancer activity [32]. Leaf and seed extract have potential antioxidant activity. Previous studies demonstrated that polysaccharides and limonoids present in the neem bark, leaves and seed oil reduced tumors and exhibited efficacy against lymphocytic leukemia [33]. Researchers revealed that nimbolide, a triterpenoid present in the extract, arrested the HT-29 (human colon carcinoma cells) in G2 /M and G0 /G1 stages apparently through upregulation of p21 thereby inhibit tumorigenesis [33]. Nimbolide has also exhibited anticancer activity in numerous cancer types such as lung cancer, osteosarcoma, neuroblastoma, choriocarcinoma, leukemia and melanoma. Also, Azadirachtin and nimbolide of neem suppressed the viability and increase in apoptosis of HeLa cervical cancer cells [34]. The neem extracts also exhibit anti-proliferative effects in both estrogen-dependent as well as independent breast cancer cells and the neem seed oil can inhibit the growth of HeLa cervical cancer cells [34].
Bauhinia variegate
Family: Fabaceae Parts Used: Leaf
Activity: B. variegate (local name: Raktakanchan) is reported to have different phytochemicals, which possess a wide range of activities and give protection against some skin diseases, stomatitis and chronic diseases reported in Indian Ayurvedic medicine [35]. The study revealed the presence of secondary metabolites such as terpenoids, phenolics, flavonoids, anthraquinones, saponins, tannins, and alkaloids in B. variegata leaf extract [35]. Flavonoids extracted from B. variegata stem have been shown to possess cytotoxic activity against Dalton’s ascetic lymphoma, leukemia, and many more cancer cell lines [36]. B. variegata leaf extracts have capability to combat oxidative damage because of its iron binding, radical neutralization ability. It has been reported that extracted flavones are more selective against ovarian cancer cell lines and the presence of flavonoids, anthraquinones, and saponins are responsible for its promising anticancer activity [36]. Ethanol extract of B. variegata showed a significant chemo-preventive and cytotoxic effect against human epithelial larynx cancer (HEp2) and human breast cancer (HBL-100) cell lines [37].
Butea monosperma
Family: Fabaceae Parts Used: Flower
Activity: B. monosperma (local name: Palash) is widely known in the traditional Indian Ayurvedic system for the treatment of a variety of ailments including cancer and liver disorders [38]. The main constituent of the flower is butrin, butein, butin, isobutrin, plastron, and isocoreopsin [39]. The ability of aqueous extract of B. monosperma flowers to impose growth arrest, alter mitogenic signalling and trigger pro-apoptotic death in hepatoma cells associated with its strong chemo-preventive effect in vivo with almost zero cytotoxic effect [38]. Isocoreopsin,a purified flavonoid isolated from flower extract possess significant free radical scavenging activity, showed remarkable efficacy in cell mortality on human colon and liver cancer cell lines (50 μg/ml in HT-29 and 100 μg/ml in HepG2) [40]. Intraperitonial administration of the aqueous extract of flowers of B. monosperma in the X-15-myc onco mice showed antitumorgenic activity [39]. Ethyl acetate, butanol and aqueous solutions derived from total methanol extract of B. monosperma flowers have effective free radical scavenging activities due to the higher phenolic content [39]. Cytotoxic property of B. monosperma floral extracts revealed significant inhibitory effect on HCT-116 cells [41].
Calotrophis gigantea
Family: Asclepiadaceae Parts Used: Leaf, root
Activity: Traditionally extracts and preparations from roots and leaves of C. gigantean (local name: Akanda) are used against abdominal tumours, syphilis, tuberculous, leprosy, skin diseases, piles, wounds, and insect-bites [42]. Several phytochemicals have been isolated from C. gigantea and they include cytotoxic cardenolides, antifeedant nonprotein amino acid, a mixture of tetracyclic triterpene compounds, pregnanes, ursane-type triterpenoids, terols, flavonol glycosides, usharin, gigantin, calcium oxalate, alpha and beta-calotropeol, beta-amyrin., fatty acids (both saturated and unsaturated), giganteol and giganteol [43]. Some experiments explore antitumor activity of methanol extract of C. gigantea root [42]. The plant has been reported to have cytotoxic potentials besides pharmacological properties such as cardiotonic, antimicrobial and many more [44]. Study also revealed that the plant C. gigantea possess anti-oxidant activity and alcoholic extracts of the root and the leaves were found to have anticancer activity against human epidermal carcinoma of the nasopharynx tissue [45].
Camellia sinensis
Family: Theaceae Parts Used: Leaf
Activity: C. sinensis (local name: Cha) is one of the most common drinks consumed worldwide as green tea, a rich source of nutritional flavonoids like epigallocatechin-3-gallate, epigallocatechin, epicatechin-3-gallate and epicatechin [46]. Studies have shown that green tea has a potential to inhibit tumour cell proliferation and induce mortality of leukaemia cells [46]. Some previous research demonstrated that epigallocatechin-3-gallate has free radicals scavenging activity and by arresting cell cycle it suppresses the proliferation of hepatocellular carcinoma cell [47]. Several investigations have suggested that epigallocatechin gallate (EGCG), the major tea polyphenol along with other polyphenols have anti-inflammatory and anti-cancer properties that may help prevent the onset and growth of skin tumors [48]. C. sinensis is a potent anti-carcinogen with no side effects [47]. The antioxidant activity of tea polyphenols is not only due to their ability to scavenge superoxide but also due to increased activity of some detoxifying enzymes such as glutathione peroxidase, glutathione reductase, glutathione-S-transferase, catalase and quinine reductase in small intestine, liver and lungs [49]. Moreover, researchers found that people who drank tea were less susceptible to develop stomach cancer, colorectal cancer, esophageal cancer, pancreatic cancer and lung cancer than those who did not drink green tea [47].
Cassia fistula
Family: Leguminosae Parts Used: Flower, seed, leaf, bark
Activity: C. fistula (local name: Amaltaas), a well-known Indian medicinal plant possesses significant antimicrobial, anti-inflammatory, hepatoprotective, wound healing and hypoglycemic activity [50]. It has been reported that C. fistula leaf extract have a rich amount of anthraquinone glycosides, flavonoids and phenolic compounds [50]. It is also suggested that anthraquinone glycoside (rhein, emodine, physion, chrysophanol, Obtusin, chrysoobtusin etc) have an anticancer activity. C. fistula extracts are high in total phenolics and proanthocyanidin content which are responsible for the synergistic oxidative potency of the extracts [51]. The results of some studies revealed that methanol extract of C. fistula seed has an antitumor activity [52]. Researchers demonstrated the tumour inhibitory activity of methanolic extract of C. fistula seed on the growth of Ehrlich ascites carcinoma [53]. Oral administration of bark extracts in 7, 12-dimethyl benz(a)anthracene (DMBA) induced oral squamous cell carcinoma in hamster showed complete prevention of carcinogenesis due to its remarkable chemopreventive ability [54].
Centella asiatica
Family: Apiaceae Parts Used: leaf
Activity: C. asiatica (local name: Thankuni) is valued in traditional Ayurveda medicine for treating a variety of diseases like skin problems, wound healing, mental fatigue, stomach ulcers, diarrhea, epilepsy, and for revitalizing the nerves and brain cells. The scientific studies have demonstrated a variety of biochemical components i.e. amino acids (mainly Alanine and serine), flavonoids, terpenoids, essential oils, alkaloids present in aerial parts of this herb [55]. The flavonoids of leaf extract include kaempferol, kaempferol-3-o-β-d-glucuronide, quercetin, quercetin-3-o-β-d-glucuronide, castillicetin, castilliferol, apigenin, rutin, luteolin etc and the triterpenes are composed of asiatic acid, madecassic acid, asiaticosside, madecassoside, centelloside, madasiatic acid, brahminoside, brahmoside, brahmic acid, thankiniside, isothankunisode, centic acid, and cenellicacid [55]. C. asiatica methanolic extract showed concentration dependent inhibition of cell proliferation and induction of apoptosis in MCF-7 breast cancer cells [56]. At a concentration above 0.1% of C. asiatica juice, an increased DNA damage and apoptotic cell death was noticed on human HepG2 cell line [57]. Asiatic acid, a phytocompound from C. asiatica showed effective antiproliferative activity on skin and lung cancer cells and also responsible for induction of apoptosis and lowering viability in human melanoma SK-MEL-2 cells [56]. When treated with 40 µg/ml concentration of asiatic acid, a reduction up to 50% in viability in ovarian cancer cells was observed and it also showed cell cycle arrest at the G0/G1 phase followed by increased apoptosis by 7-10 folds [58]. A partially purified fraction of methanol extract of C. asiatica inhibited the tumour growth with no toxic effects on lymphocytes and leaf water extract has a chemo preventive effect on colon tumorigenesis [59].
Curcuma longa L.
Family: Zingiberaceae Parts Used: Root, stem and leaves
Activity: Curcumin, a phenolic compound from the plant Curcuma longa L. (local name: Haldi) has shown a wide-spectrum chemo-preventive, antioxidant and antitumor activities. Curcumin is the natural yellow pigment in turmeric isolated from the rhizome of the plant C. longa [60]. Curcumin was found to inhibit the generation of ROS including superoxide dismutase and hydrogen peroxide in peritoneal macrophages. Curcumin as an anti-inflammatory agent, inhibits the proliferation of several tumour cells [60]. Recently, curcumin has been listed as the third generation of antitumor drug by the US National Cancer Institute (NCI) [61]. Curcumin, demethoxycurcumin and bisdemethoxy curcumin are the most common antitumor constituents in the curcuminoids of turmeric [62]. Curcumin I, curcumin II (monodemethoxycurcumin) and curcumin III (bisdemethoxycurcumin) from C. longa was assayed for their cytotoxicity, antioxidant and anti-inflammatory activities [63]. These compounds showed activity against melanoma, leukemia, colon, CNS, renal, and breast cancer cell lines [63]. Cell viability assays demonstrated the efficacy of rubusoside-solubilized curcumin against human colon, breast, and pancreatic cancer cell lines. Multiple mechanisms of action of curcumin are responsible for various effects on cancer cells including cell cycle arrest at G1/S stage, apoptosis induction which has been observed in different tumor cell lines [64].
Emblica officinalis
Family: Euphorbiaceae Parts Used: Fruit pulp
Activity: E. officinalis (local name: Amlaki) has been used in Asian herbal pharmaceuticals for treatment of various illnesses specially in case of gastrointestinal problems [65]. It has been reported that the medicinal fruit of E. officinalis contains unique biologically active ingredients tannoids and flavanoids, having powerful antioxidant properties and high content of the antioxidant vitamin C, gallic acid [65]. The isolated ingredients from fruit extract have shown their protective effect against lipid peroxidation [66]. From previous studies, it has been proven that fruit extract act as an antimutagen directly as well as against mutagens that need metabolic activation and it also showed anticarcinogenic activity against methylcholanthrene-induced sarcoma formation [67]. The anticancer properties of the bioactive components of fruit extract is exerted through the removal of free radicals and by inhibiting Phase-I enzymes which are required for the activation of carcinogen and activation Phase-II enzymes (antagonist of Phase-I enzyme) [67]. It has been proven that aqueous extracts of E. officinalis cause decrease in the HepG2 cell viability by reducing ROS generation as well as improving reduced intracellular GSH levels. E. officinalis aqueous extracts also induce apoptosis in several cancer cell lines i.e. Hela (cervical), A549 (lung), MDA‑MB‑231 (breast), SK‑OV3 (ovarian) and SW620 (colorectal) [68]. Pyrogallol, a bioactive catechin compound of E. officinalis fruit extracts showed significant anti‑proliferative activity against H520 (lung squamous cell carcinoma) and human lung cancer cell lines‑H441 (lung adenocarcinoma) [69]. Development of pyrogallol based high potency anti‑lung cancer drug needs to be supported by further researches.
Oroxylum indicum
Family: Bignoniaceae Parts Used: bark
Activity: The medicinally active plant O. indicum (local name: Sonapatha) has drawn considerable attention in research because of wide nutritional and medicinal properties to treat biliousness, fevers, intestinal worms, leucoderma, inflammation, diarrhoea, dysentery, diaphoretic, bronchitis pneumonia and respiratory troubles etc [70]. Bioactive phenolic compounds present in O. indicum extract are baicalein, oroxylin A, chrysin and its variety of derivatives. [70]. O. indicum in its methanol and aqueous extracts have previously been reported for its cytotoxicity in MDA-MB-435S and Hep3B cell lines [71]. The bark decoction of O. indicum has also been reported for its use in treating cancer, despite the lack of mechanistic evidence about this therapeutic function [72]. O. indicum bark extracts were furthermore reported to possess anti-proliferative property on human breast cancer cells [73]. The stem bark extract of O. indicum showed effectual cytotoxicity, apoptosis-inducing abilities and distinctive anti-metastatic potentials against estrogen receptor-negative breast cancer [71]. Baicalein, a naturally occurring flavonoid compound isolated from O. indicum possesses effectual anticancer activity when treated against CT-26 colon carcinoma [70].
Semecarpus anacardium
Family: Anacardiaceae Parts Used: Nut
Activity: The fruits of S. anacardium (local name: Bhallatak), a tropical tree growing wild in the Indian subcontinent, are used extensively for the treatment of human cancers in the Ayurvedic medicine [74]. The nut milk extract of this plant exhibits anti-tumor activity by inducing the in vivo antioxidant system or by suppressing hypoxic and angiogenic factors (hypoxia inducible factor-1 alpha), vascular endothelial growth factor, and inducible nitric oxide synthase [74]. The oil extracted from S. anacardium nut is reported to have cytotoxic effects against acute myeloblastic leukemia (HL-60), chronic myelogeincleukemia (K-562), breast adenocarcinoma (MCF-7) and cervical epithelial carcinoma (HeLa) cell lines [75]. Phytochemical analysis of the nut reveals the presence of bioflavonoids (galluflavanone), bhilawanols, phenolic compounds, glycosides and sterols [76]. S. anacardium oil having strong antioxidant capacity showed its anti-tumour activity through a mechanism which does not cause any acute physiological disturbance [77]. Reports have also established that a single injection of nut extract could bring complete inhibition of tumour growth in rats. S. anacardium nut extract may be a potential antineoplastic agent against mammary carcinoma cell [78].
Syzygium cumini
Family: Myrtaceae Parts Used: Fruit, seed
Activity: S. cumini (local name: Kaalojaam), a large evergreen tree native to India has been valued in traditional Ayurveda and Unani medication for its therapeutic potentials [79]. The various parts of the plant (bark, leaf, fruit and seed) is reported to possess antioxidant, anti-inflammatory, anti-microbial, anti-bacterial, anti-HIV, anti-leishmanial, anti-fungal, nitric oxide scavenging, free radical scavenging, anticancer, anti-clastogenic, anti-diarrheal, gastroprotective, anti-ulcerogenic and chemotherapeutic activities [80]. These parts have been extensively investigated for their bioactive phytochemical constituents like maleic acid, oxalic acid, gallic acid, ellagic acid, oleanolic acid, betulinic acid, isoquercitin, quercetin, myricetin, kaempferol, cynidin glycoside, flavonoids, tannins, essential oils and triterpenoids [81]. Some of these components may be collectively responsible for the antineoplastic, radioprotective, chemopreventive properties of such plant extract [81]. Anthocyanins and Betulinic acid, Cyanidin diglycoside, Ferulic acid were reported for their potent anticancer activity. Study showed that ethanol extract of fruit containing Kaempferol 7-O-methylether and Y-Sitosterol is responsible for their antioxidant and anti- leukemia activities [79]. The crude extract of S. cumini fruits inhibited growth and induced apoptosis in human cervical cancer cell lines HeLa and SiHa in a dose and time-dependent manner [82]. S. cumini fruit extract has been further observed to have anti-tumor and anti-oxidative potential against chemically induced stomach carcinogenesis [80]. Some studies also revealed the significant cytotoxic activity of the seed extract of S. cumini on various cancer cell lines (A2780, MCF7, PC-3, H460) [82]. Previous experiments have shown that the standardized fruit extract of this plant possesses antiproliferative as well as pro-apoptotic properties against breast cancer cells.
Vitex negundo
Family: Verbenaceae Parts Used: Leaves, seed
Activity: Traditionally, plant parts of V. negundo (local name: Nishinda) are used for the treatment of skin-ulcers, leukoderma, rheumatoid arthritis, bronchitis, leucoderma, gonorrhea, bronchitis etc. V. negundo also exhibits anti-bacterial, anti-fungal, anti-inflammatory, anti-tumor activity [83]. The phytochemical study of ethanolic extract of leaves of V. nigundo indicated the presence of flavonoids, Alkaloids and terpenoids [84]. The anti-tumour effect shown by the ethanolic extract may be due to antioxidant potential of flavonoids [84]. Evn-50 is a mixture of lignan compounds extracted from V. negundo possesses a broad spectrum of cytotoxic activity for various cancers including hormone dependent and hormone independent cancers ranging from pancreatic cancer, liver cancer, kidney cancer, lung cancer, gastric cancer, and colon cancer [85]. This cytotoxicity of EVn-50 may be due to cell cycle arrest at G2/M phase as observed by flow cytometric study, followed by apoptosis of cancer cells. EVn-50 exerts cytotoxic effect on some hormone related cancers including breast cancer, choriocarcinoma, prostate cancer and ovarian cancer, possibly via apoptosis inducing mechanism and so acknowledged as potential anticancer compound [85].
Withania somnifera
Family: Solanaceae Parts Used: Root, stem and leaves
Activity: In Indian traditional Ayurvedic medicine W. somnifera (local name: Ashwagandha) is well proven as a potential source of various anticancer components due to the presence of several bioactive components acting as free radical scavengers, reducing agents and quenchers of singlet oxygen [86]. Some recent studies using 50% ethanol extract of root, stem and leaves of W. somnifera exhibited in-vitro cytotoxicity against five human cancer cell lines of four different tissues i.e. A-549 (lung), PC-3(prostrate), DU-145 (prostrate), HCT-15 (colon), and IMR-32 (neuroblastoma) [87]. W. somnifera also has anti-inflammatory, anti-tumour and radio-sensitizing actions and analgesic activity [87]. Studies on W. somnifera suggest that it decreases tumour cell proliferation and boosts the efficiency of radiation therapy while potentially mitigating unwanted side effects. Hydro alcoholic (1:1) sample of W. somnifera (leaves) shows efficient cytotoxicity on MCF-7, A549 and PA-1 cancer cell line (breast, lung and ovary respectively) [86]. In a study, W. somnifera was suggested as an alternative long-term therapy to prevent the spread of cancer cells. In this case, the root extracts were tested against vimentin pro-metastatic protein. Thus, different formulations of W. somnifera were used to establish as cell motility inhibitor in case of breast tumours [88]. Withaferin A, an active anticancer agent extracted from the leaf of the plant [89]. Moreover, some experiments proved that the lower concentrations of root extract of W. somnifera can constrain breast cancer metastasis with negligible adverse effects in rat model [88,89]. These findings paved the way for researchers to focus on the bioactivities of this plant and to formulate the composition for medicinal use.
ANTICANCER APPLICATIONS OF PHYTOCHEMICALS
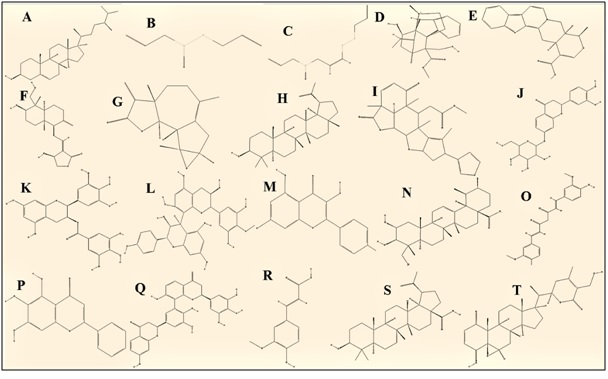
The curative properties of these plants are due to the presence of complex phytochemical constituents of diverse compositions grouped alkaloids, glycosides, corticosteroids, essential oils etc. [Figure 2]. Plant derived medicines are basically multi-compounds extract with complicated compositions in which fractional components possess chemo preventive activity.
Figure 2: Structural representation of phytochemicals having cytotoxic or anticancer properties. The structures and Compound identification number (CID) are taken from PubChem Database of National Centre for Biotechnology Information (NCBI). (A) campesterol, CID: 173183 ; (B) allicin, CID: 65036 (C) Z-ajoene, CID: 9881148 (D) echitamine, CID: 5364099 (E) alstonine CID: 24188474 (F) andrographolide, CID: 5318517; (G) ludartin, CID: 14355826; (H) lupeol, CID: 259846; (I) nimbolide, CID: 100017; (J) isocoreopsin, CID: 12309899; (K) epigallocatechin gallate, CID: 65064; (L) proanthocyanidin, CID: 108065 (M) kaempferol, CID: 5280863 (N) asiatic acid, CID: 119034 (O) curcumin, CID :969516 (P) baicalein, CID : 5281605; (Q) galluflavanone, CID: 101326873 (R) ferulic acid, CID: 445858 (S) betulinic acid, CID: 64971 (T) withaferin A, CID: 265237 (accessed on 19th July 2020).
Several emerging analytical separation methods are reported to have more advantages over conventional procedures to separate and screen the phytochemical constituents of certain medicinal activities. But it is still a challenge to formulate an ideal phytochemical-based medicine containing isolated bioactive anticancer agents. Phytochemical studies revealed that A. aspera, A. scholaris, B. variegate, C. asiatica, V. negundo possess alkaloid compounds with chemo preventive effects. A. scholaris from (Apocynaceae) has been reported to have an alkaloid derivative antineoplastic agent, Alstonine [24]. Manyphenolic compounds are isolated from A. aspera, A. lanata, B. variegate, B. monosperma, C. sinensis, C. longa, O. indicum, S. anacardium. Epigallocatechin gallate, a polyphenol isolated from C. sinensis (Theaceae) leaf reported to have broad chemopreventive efficacy [47]. Proanthocyanidin is another polyphenol (oligomeric flavonoid) identified in C. fistula (Leguminosae) extract has potent anti-carcinogenic activity [51].Curcumin, a natural polyphenol present in C. longa (Zingiberaceae) modulates cell signalling thus interfering cancer cell proliferation and angiogenesis [60]. E. officinalis (Euphorbiaceae) fruit extracts contain a bioactive polyphenol called pyrogallol that exhibited significant anti-proliferative activity [69]. Ferulic acid, a phenolic compound present in S. cumini (Myrtaceae) extract has promising chemo preventive as well as anti-neoplastic activity against various cancer cell lines [80,81].
Campesterol, a phytosterol possessing radical scavenging activity have been isolated from A. lanata (Amaranthaceae) [12,13]. Some organosulfur phytochemicals like allicin from A. sativum (Liliaceae) have promising repressing properties against numerous cancer cell lines [17]. Various bioactive flavonoid compounds are isolated from A. paniculate, A. indica, B. variegate, B. monosperma, C. asiatica, C. gigantea, C. sinensis, C. fistula, S. anacardium and V. negundo. Isocoreopsin, a flavonoid extract of butanol B. monosperma flower showed excellent efficacy against human liver and colon cancer cell lines [40]. Kaempferol, a flavonol present in C. asiatica (Apiaceae) extract was reported to have metastasis and angiogenesis repressing capacity [55]. Baicalein, a flavonoid extracted from O. indicum (Bignoniaceae) was known to suppress invasiveness of colorectal cancer [70]. Galluflavanone, a new biflavonone isolated from S. anacardium nut shells are known to possess cancer preventive potential [76]. Extracts of A. aspera, A. scholaris, A. paniculate, A. indica, B. variegate, C. gigantea, C. asiatica, S. cumini, V. negundo have been reported to contain terpenoid compounds. Andrographolide, a diterpenoid from A. paniculate (Acanthaceae) exhibited cytotoxic activity against human epidermoid carcinoma cells, breast cancer cell lines and lymphocytic leukaemia [26]. Nimbolide, a tetranortriterpernoid isolated from A. indica (Meliaceae) leaf involved in modulating multiple signalling pathways in malignant cells which showed potent chemopreventive activity [33]. Asiatic acid, a pentacyclic triterpenoid extracted from C. asiatica (Apiaceae) possesses excellent anti-proliferative efficacy against various cancer cell lines including human lung cancer [58]. Evn-50, a lignan compounds mixture of V. negundo (Verbenaceae) has excellent broad-spectrum cytotoxic activity specially against SMMC-7721 (liver cancer) and MDA-MB-435 (breast cancer) cell lines [85].
There are some trademarks common in every type of cancer i.e. self-sufficiency in growth signals and uncontrolled cell proliferations, growth inhibitory signal resistance capability, evasion of apoptosis, unrestricted replication capacity, sustained angiogenesis, metastatic activity and invasion. The complicated mechanism of actions requires strong multi-targeted treatment [90]. Bioactive multi-components plant extract involves in modulating various mechanism by interfering cellular transportations, activating pro-drugs to alter metabolites, inhibiting binding to target proteins etc. It has been observed that natural therapeutics exert significant additive or synergistic mode of action at the signalling cascade by which severe toxic side effects associated with conventional cancer therapies can be avoided. In this scenario, we should focus on preclinical studies i.e. quality control, drug designing, delivery strategy, drug safety and therapeutic efficacy as well as clinical studies to overcome the problem of data insufficiency about majority of plant derived drugs. Considering the findings of these ethnopharmacological researches on medicinal plants of Bengal, it is possible to formulate effective anticancer drugs either using single or in combination with other phytochemicals through an extensive scientific analysis.
CONCLUDING REMARKS
Science has long acknowledged the value of natural phytochemical based remedies. These traditional therapeutic-inspired approaches to drug discovery attract considerable attention in cancer therapy due to presence of diverse range of active ingredients. But the effectiveness of any herbal product is dependent upon molecular recognition, rational designing, proper standardization, smart delivery strategy during clinical trials. While some natural formulations have shown to exert promising cytotoxicity against cancer cell lines, many remedies aren't supported by research. Our article highlighted twenty most common Bengal plants having strong anti-cancer properties by promoting anti-tumour or anti carcinogenic activities and boosting up the immunity machineries. Our review helps to make a potent data base on those medicinal plants from different plant families with antiproliferative and anti-carcinogenic effect on some specific cancer types and pave the way for the development and utilization of new Phytotherapeutic agents in medical applications.
DECLARATION OF INTEREST
There are no conflicts to declare.
FUNDING
The research work is not assisted by any kind of funding agencies or research fund.
ACKNOWLEDGEMENTS
The authors would like to thank Biophysics department of Jadavpur University, National Medicinal Plant Board (NMPB), West Bengal State Medicinal Plant Board (WBSMPB) Government of India’s Department of AYUSH. We are thankful to everyone who provided valuable information and guidance during research of this review. We are also grateful to Dr. Ashesh Nandy for careful reading of our article.
REFERENCES
- Pandey G, Sharma M. (2009). Some medicinal plants as natural anticancer agents. Phcog Rev. 3(6):259-263.
- Andualem G, Umar S, Getnet F, Tekewe A, Alemayehu H, et al. (2014). Antimicrobial and Phytochemical Screening of Methanol Extracts of Three Medicinal Plants in Ethiopia. Advances in Biological Research. 8(3):101-106.
- Rosangkima G, Prasad SB. (2004). Antitumour activity of some plants from Meghalaya and Mizoram against murine ascites Dolton's lymphoma. Indian J Exp Biol. 42:981-988.
- Perumal SR, Gopalakrishnakone P. (2010). Therapeutic Potential of Plants as Anti-microbials for Drug Discovery. Advance Access Publication. 7(3):283–294. doi:10.1093/ecam/nen036.
- Hassan W, Nida S, Kazmi Z, Noreen H, Riaz A, et al. (2016). Antimicrobial Activity of Cinnamomum tamala Leaves. J Nutr Disorders Ther. 6:2. http://dx.doi.org/10.4172/2161-0509.1000190
- Sen R. (2009). The Evolution of Industrial Relations in West Bengal, Cornell University ILR School Digital Commons@ILR, 2009 International Labour Organization.
- De B. (1990). West Bengal: A Geographical Introduction, Economic and Political Weekly, May 5-12, 1990. 25:995+997+999-1000.
- Subbarayana PR, Sarkar M, Impellizzeri S, Raymo F, Lokeshwar BL, et al. (2010). Anti-proliferative and anti-cancer properties of Achyranthes aspera: Specific inhibitory activity against pancreatic cancer cells, Journal of Ethnopharmacology. 131;78–82. doi:10.1016/j.jep.2010.06.002.
- Priya CL, Kumar G, Karthik L, Rao KVB. (2012). Phytochemical composition and in vitro antioxidant activity of Achyranthes aspera Linn (Amaranthaceae) leaf extracts. Journal of Agricultural Technology. 8(1):143-156.
- Srivastava PK. (2014). Achyranthes aspera: A potent immunostimulating plant for traditional medicine; IJPSR. 5(5):1601-1611. DOI: 10.13040/IJPSR.0975-8232.5(5).1601-11.
- Adnyana DPA, Meles IDK, Meles W. (2008). Alkaloid Fraction of Jarong (Achyranthes Aspera Linn) Leaf Induced apoptosis breast cancer cell through p53 pathways. The Open Natural Products Journal. 1:44-49.
- Bitasta M, Madan S. (2016). Aervalanata: A blessing of Mother Nature. Journal of Phar macog nosy and Phytoc he mistry. 5(1):92-101.
- Battu GR, Kumar BM. (2012). Invitro antioxidant activity of A. lanata Linn. Int J Pharm Sci. 2:74-8.
- Govindan NK. Antioxidant Aervalanata Extract Supresses Proliferation and Induce Mitochondria Mediated Apoptosis in Human Hepatocelluar carcinoma cell line A. J Exp Integr Med. 6(2). DOI: 10.5455/jeim. 270616.or.153.
- Raihan O, Brishti A, Bahar E, Islam F, Rahman M, et al. (2012). Antioxidant and anticancer effect of methanolic extract of Aervalanata Linn. against Ehrlich Ascites Carcinoma (EAC) in vivo. Orient Pharm Exp Med. 12:219–225. DOI 10.1007/s13596-012-0073-3.
- Thomson M, Ali M. (2003). Garlic [Allium sativum]: A Review of its Potential Use as an Anti-Cancer Agent. Current Cancer Drug Targets. 3:67-81.
- Singh VK, Singh DK. (2008). Pharmacological Effects of Garlic (Allium sativum L.). ARBS Annual Review of Biomedical Sciences. 10:6-26.
- Nouroz F, Mehboob M, Noreen S, Zaidi F, Mobin T. (2015). A Review on Anticancer Activities of Garlic (Allium sativum L.) Middle-East Journal of Scientific Research. 23(6):1145-1151.
- Keiss HP, Dirsch VM, Hartung T, Haffner T, Trueman L, et al. (2003). Garlic (Allium sativum L.) modulates cytokine expression in lipopolysaccharide-activated human blood thereby inhibiting NF-kappa B activity. J Nutr. 133(7):2171-2175.
- Jagetia GC, Baliga MS. (2006). Evaluation of Anticancer Activity of the Alkaloid Fraction of Alstoniascholaris (Sapthaparna) In vitro and In vivo. Phytother Res. 20:103–109. DOI: 10.1002/ptr.1810.
- Jahan S, Chaudhary R, Goyal PK. (2009). Anticancer Activity of an Indian Medicinal Plant, Alstoniascholaris, on Skin Carcinogenesis in Mice. Integrative Cancer Therapies. 8(3):273–279.
- Baliga MS. (2010). Alstoniascholaris Linn R Br in the Treatment and Prevention of Cancer: Past, Present, and Future. Integrative Cancer Therapies. 9(3):261–269.
- Rastogi RM, Mehrotra BN. (1990). Compendium of Indian Medicinal Plants. Lucknow, India: Central Drug Research Institute.
- Beljanski M, Beljanski MS. (1982). Selective inhibition of in vitro synthesis of cancer DNA by alkaloids of beta-carboline class. Exp Cell Biol. 50:79-87.
- Mishra SK, Sangwan NS, Sangwan RS. (2007). Andrographis paniculata (Kalmegh): a review. Pharmacognosy Rev. 1:283-298.
- Okhuarobo A, Falodun JE, Erharuyi O, Imieje V, Falodun A, et al. (2014). Harnessing the medicinal properties of Andrographis paniculata for diseases and beyond: a review of its phytochemistry and pharmacology. Asian Pac J Trop Dis. 4(3):213-222.
- Menon V, Bhat S. (2010). Anticancer Activity of Andrographolide Semisynthetic Derivatives. Natural Product Communications. 5(5):717–720.
- Zeng Y, Jiang J, Lao H, Guo J, Lun Y, et al. (2015). Antitumor and apoptotic activities of the chemical constituents from the ethyl acetate extract of Artemisia indica. Molecular medicine reports. 11:2234-2240.
- Rashid S, Rather MA, Shah WA, Bhat BA. (2013). Chemical composition, antimicrobial, cytotoxic and antioxidant activities of the essential oil of Artemisia indica Willd. Food Chemistry. 138:693–700.
- Bayala B, Bassole HN, Scifo R, Gnoula C, More L, et al. (2014). Anticancer activity of essential oils and their chemical components - a review. Am J Cancer Res. 4(6):591-607.
- Attwood E , Joy C. (2009). Azadirachta indica (neem): a plant of multiple biological and pharmacological activities. Phytochemistry. 8:601–620.
- Kumar GH, Mohan CKV, Rao JA, Nagini S. (2009). Nimbolide a limonoid from Azadirachta indica inhibits proliferation and induces apoptosis of human choriocarcinoma (BeWo) cells. Invest New Drugs. 27:246-52. DOI: 10.1007/s10637- 008-9170-z.
- Paul R, Prasad M, Sah NK. (2011). Anticancer biology of Azadirachta indica L (neem): A mini review. Cancer Biology & Therapy. 12(6):467-476.
- Gupta SC, Prasad S, Tyagi AK, Kunnumakkar AB, Aggarwal BB. (2017). Neem (Azadirachta indica): An indian traditional panacea with modern molecular basis. Phytomedicine. 34:14–20.
- Gunalan G, Krishnamurthy V, Saraswathy A. (2016). GC-MS and HPTLC fingerprinting of Bauhinia variegata leaves for anticancer activity. World Journal of Pharmaceutical Research. 3(9):1313-1336.
- Mishra A, Sharma AK, Kumar S, Saxena AK, Pandey AK. (2013). Bauhinia variegata Leaf Extracts Exhibit Considerable Antibacterial, Antioxidant, and Anticancer Activities. BioMed Research International. Article ID 915436.
- Mali RG, Mahajan SG, Mehta AA. (2007). Plant Review Rakta Kanchan (Bauhinia variegata): Chemistry, Traditional and Medicinal uses- a review. Pharmacognosy Reviews. 1(2).
- Choedon T, Shukla SK, Kumar V. (2010). Chemopreventive and anti-cancer properties of the aqueous extract of flowers of Butea monosperma, Journal of Ethnopharmacology. 129:208–213.
- Sharma AK, Deshwal N. (2011). Butea monosperma A traditional medicinal plant; an overview. International Journal of Pharm Tech Research. 3:867-868.
- Subramaniyan B, Polachi N, Mathan G. (2016). Isocoreopsin: An active constituent of n-butanol extract of Butea monosperma flowers against colorectal cancer (CRC). Journal of Pharmaceutical Analysis. 6:318–325.
- Polachi N, Nagaraja P, Subramaniyan B, Mathan G. (2015). Antiproliferative activity of n-butanol floral extract from Butea monosperma against hct 116 colon cancer cells; drug likeness properties and in silico evaluation of their active compounds toward glycogen synthase kinase-3β/axin and β-catenin/t-cell factor-4 protein complex. Asian J Pharm Clin Res. 8(1):134-141.
- Habib MR, Karim MR. (2011). Evaluation of antitumour activity of Calotropis gigantea L. root bark against Ehrlich ascites carcinoma in Swiss albino mice. Asian Pacific Journal of Tropical Medicine. 786-790.
- Kumar PS, Kalavathy S. (2013). Review on a potential herb Calotropis gigantea (L.) R Br Sch Acad J Pharm. 2(2):135-143.
- Pardesi GS, Chhaya G, Madhav VD, Hamid YH, Babita H, (2008). Preliminary studies on antimitotic and anticancer activity of Calotropis gigantean. Pharmacology online. 1:38-47.
- Ahmed KKM, Rana AC, Dixit VK. (2005). Calotropis Species (Ascelpediaceae) – A Comprehensive Review. Phcog Mag. 2(1):48.
- Parmar N, Rawat M, Vijay KJ. (2012). Camellia Sinensis (Green Tea): A Review, Global Journal of Pharmacology. 6(2):52-59.
- Sharangi AB. (2009). Medicinal and therapeutic potentialities of tea (Camellia sinensis L.) – A review. Food Research International. 42:529–535.
- Katiyar SK, Ahmad N, Mukhtar H, (2000). Green tea and skin. Archives of Dermatology. 136:989–994.
- Frei B, Higdon JV. (2003). Antioxidant activity of tea polyphenols in vivo: Evidence from animal studies. Journal of Nutrition. 133(10):3275S–3284S.
- Singh VK, Khan AM. (1990). Medicinal Plants and Folklores - A Strategy towards Conquest of Human Ailments. 9:67.
- Ramma AL, Bahorun T, Mohammed A, Soobrattee, O, Aruoma I. (2002). Antioxidant Activities of Phenolic, Proanthocyanidin, and Flavonoid Components in Extracts of Cassia fistula. J Agric Food Chem. 50:5042−5047
- Sen AB, Shukia YN. (1968). Chemical examination of Cassia fistula. J Indian Chem Soc. 45:744.
- Gupta M, Mazumder UK, Rath N, Mukhopadhyay DK. (2000). Antitumor activity of methanolic extract of Cassia fistula L. seed against Ehrlich Ascites Carcinoma. Journal of Ethnopharmacology. 72:151–156.
- Md. Danish, Singh P, Mishra G, Srivastava S, Jha KK, et al. (2011). Cassia fistula Linn. (Amulthus)- An Important Medicinal Plant: A Review of Its Traditional Uses. Phytochemistry and Pharmacological Properties. J Nat Prod Plant Resour. 1(1):101-118.
- Zheng CJ, Qin LP. (2007). Chemical components of Centella asiatica and their bioactives. Journal of Chinese Integrative Medicine. 5:348-351.
- Prakash V, Jaiswal N, Srivastava M. (2017). A review on medicinal properties of Centella asiatica. Asian J Pharm Clin Res. 10(10):69-74.
- Hussin F, Eshkoor SA, Rahmat A, Othman F, Akim A. (2014). The Centella asiatica juice effects on DNA damage, apoptosis and gene expression in hepatocellular carcinoma (HCC). BMC Complement Altern Med. 14:32.
- Ren L, Cao QX, Zhai FR, Yang SQ. Zhang HX. (2016). Asiatic acid exerts anticancer potential in human ovarian cancer cells via suppression of PI3K/Akt/mTOR signalling. Pharm Biol. 54(11):2377-82.
- Jamil SS, Nizami Q, Salam M. (2007). Centella asiatica (Linn.) Urban óA review. Natural Product Radiance. 6(2):158-170.
- Dorai T, Cao YC, Dorai B, Buttyan R, Katz AE. (2001). Therapeutic potential of curcumin in human prostate cancer. III. Curcumin inhibits proliferation, induces apoptosis and inhibits angiogenesis of LNCaP.
- Jiang J, Jin X, Zhang H, Su X, QiaoB, et al. (2012). Identification of antitumor constituents in curcuminoids from Curcuma longa L. based on the composition–activity relationship. Journal of Pharmaceutical and Biomedical Analysis. 70;664–670.
- Liang G, Tian JL, Shao LL, Yang SL, Li XK. (2008). Progress in the research on SAR and development of antitumor curcuminoids. Chem Bull. 2:110–111.
- Rarnsewak RS, DeWitt DL, Nair MG. (2000). Cytotoxicity, antioxidant and anti-inflammatory activities of Curcumins I-III from Curcuma longa. Phytomedicine. 7(4):303-308.
- Srivastava RK, Chen Q, Siddiqui I, Sarva K, Shankar L. Linkage of curcumin induced cell cycle arrest and apoptosis by cyclin-dependent kinase inhibitor p21(/WAF1/CIP1). Cell Cycle 2007; 6:2953–61.
- Reddy VD, Padmavathi P, Paramahamsa M, Varadacharyulu NC. (2010). Amelioration of alcohol‑induced oxidative stress by Emblica officinalis (amla) in rats. Indian J Biochem Biophys. 47:20‑5.
- Haque R, Bin-Hafeez B, Ahmad I, Parvez S, Pandey S, et al. (2001). Protective effects of Emblica officinalis Gaertn. in cyclophosphamide-treated mice. Human & Experimental Toxicology. 20:643–650.
- Jose JK, Kuttan G, George J, Kutta R. (1997). Antimutagenic and Anticarcinogenic Activity of Emblicaoficinalis. J Clin Biochem Nutr. 22:171-176.
- Ngamkitidechakul C, Jaijoy K, Hansakul P, Soonthornchareonnon N, Sireeratawong S. (2010). Antitumour effects of Phyllanthus emblica L.: induction of cancer cell apoptosis and inhibition of in vivo tumour promotion and in vitro invasion of human cancer cells. Phytother Res. 24:1405–1413.
- Yang CJ, Wang CS, Hung JY, Huang HW, Chia YC, et al. (2009). Pyrogallol induces G2‑M arrest in human lung cancer cells and inhibits tumor growth in an animal model. Lung Cancer. 66:162‑8.
- Laloua C, Basak A, Mishra P, Mohanta BC, Banik R, et al. (2013). Inhibition of Tumor Cells Proliferation and Migration by the Flavonoid Furin Inhibitor Isolated from Oroxylum indicum. Current Medicinal Chemistry. 20:583-591.
- Kumar DRN, George VC, Suresh PK, Kumar RA. Cytotoxicity, Apoptosis Induction and Anti-Metastatic Potential of Oroxylum indicum in Human Breast Cancer Cells, DOI: http://dx.doi.org/10.7314/APJCP.2012.13.6.2729.
- Mao AA. Oroxylum indicum vent. A potential anticancer medicinal plant. Ind J Trad Know, 2002; 1, 17-21.
- Lambertini E, Piva R, Khan MT. (2004). Effects of extracts from bangladeshi medicinal plants on in vitro proliferation of human breast cancer cell lines and expression of estrogen receptor α gene. Int J Oncol. 24:419-23.
- Premalatha B. (1999). Sachdanandam P. Semecarpus anacardium L. nut extract administration induces the in vivo antioxidant defence system in aflatoxin B1 mediated hepatocellular carcinoma. Journal of Ethnopharmacology. 66:131–139.
- Chakraborthy S, Roy M, Taraphdar AK, Bhattacharya RK. (2004). Cytotoxic effect of root extract of Tiliacoraracemosa and oil of Semecarpus anacardium nut in human tumor cells. Phytotherapy Research. 18:595–600.
- Premalatha B. (2000). Semecarpus anacardium Linn. Nut- A boon in alternative medicine, Indian journal of experimental biology. 38:1177-1182.
- Indap FMA, Ambaye RY, Gokhale SV. (1983). Antitumour and pharmacological effects of the oil from Semecarpus anacardium Linn. Ind J Physio Pharmac.
- Sujatha V, Sachdanandam P. (2002). Recuperative Effect of Semecarpus anacardium Linn. Nut Milk Extract on Carbohydrate Metabolizing Enzymes in Experimental Mammary Carcinoma-bearing Rats Phytother. Res. 16:S14–S18. DOI: 10.1002/ptr.777.
- Choi EJ, Ahn WS. (2008). Kaempferol induced the apoptosis via cell cycle arrest in human breast cancer MDA-MB-453 cells. Nutrition Res Practice. 2(4):322-325.
- Goyal PK, Verma P, Sharma P, Parmar J, Agarwal A. (2010). Evaluation of Anti-Cancer and Anti-Oxidative Potential of SyzygiumCumini Against Benzo[a]pyrene (BaP) Induced Gastric Carcinogenesis in Mice. Asian Pacific J Cancer Prev. 11:753-758.
- Swami SB, Singh N, Thakor J, Patil MM, Haldankar PM. (2012). Jamun (Syzygiumcumini (L.)): A Review of Its Food and Medicinal Uses Food and Nutrition Sciences. 3:1100-1117.
- Yadav SS, Meshram GA, Shinde D, Patil RC, Manohar SM, et al. (2011). Antibacterial and Anticancer Activity of Bioactive Fraction of Syzygiumcumini L. Seeds. Journal of Biosciences. 18(3):118-122.
- Md. Islam S, Most. Akhtar M, Md. Parvez S, Md. Alam J, Alam MF. (2013). Antitumor and antibacterial activity of a crude methanol leaf extract of Vitex negundo. Arch Biol Sci. 65(1):229-238.
- Chitra V, Sharma S, Kayande N. (2009). Evaluation of Anticancer Activity of Vitex negundo in Experimental Animals: An In Vitro & In Vivo Study. Int J Pharm Tech Res. 1(4).
- Xin H, Kong Y, Wang Y, Zhou Y, Zhu Y, et al. (2013). Lignans extracted from Vitex negundo possess cytotoxic activity by G2/M phase cell cycle arrest and apoptosis induction. Phytomedicine. 20:640–647.
- Nema R, Khare S, Jain P, Pradhan A. (2013). Anticancer Activity of Withania Somnifera (Leaves) Flavonoids Compound. Int J Pharm Sci Rev Res. 19(1):103-106.
- Yadav B, Bajaj A, Saxena M, Saxena AK. (2010). In Vitro Anticancer Activity of the Root, Stem and Leaves of Withania Somnifera against Various Human Cancer Cell Lines. Indian J Pharm Sci. 72(5):659–663.
- Yang Z, Garcia A, Xu S. (2013). Withania somnifera root extract inhibits mammary cancer metastasis and epithelial to mesenchymal transition. PLoS One 8: e75069.
- Rai M, Jogee PS, Agarkar G, dos Santos CA. (2016). Anticancer activities of Withania somnifera: Current research, formulations, and future perspectives. Pharm Biol. 54(2):189–197.
- Hanahan D, Weinberg RA. (2000). The hallmarks of cancer. Cell. 100:57-70.