Previous Issues Volume 1, Issue 1 - 2017
A New Mechanism of Cartilage Growth in mammals “Involvement of CD117 Positive Undifferentiated Cells in Interstitial Growth”
Soha Abdel-lateif Soliman1*, Hanan Hassan Abd-Elhafeez2, Enas Abdel-Hafez2
1Department of Histology, Faculty of Veterinary Medicine, South Valley University, Qena 83523, Egypt. 2Department of Anatomy and Histology, Faculty of Veterinary. Medicine, Assuit University, 71526, Egypt. Corresponding Author: Soha Abdel-lateif Soliman, Department of Histology, Faculty of Veterinary Medicine, South Valley University, Qena 83523, Egypt, Tel: +201006500848; E-Mail: [email protected]
Received Date: 03 Apr 2017 Accepted Date: 08 Apr 2017 Published Date: 12 Apr 2017
Copyright © 2017 Soha Soliman A
Citation: Soha Soliman A, Hanan Hassan A and Enas A. (2017). A New Mechanism of Cartilage Growth in mammals “Involvement of CD117 Positive Undifferentiated Cells in Interstitial Growth”. Mathews J Cytol Histol. 1(1): 001.
ABSTRACT
Two types of cartilages growth are commonly described; the appositional growth and interstitial growth. The manner of production of new cartilage matrix is different in both types. Appositional type relies on the addition of peripheral matrix upon activation of the perichondrial stem cells and interstitial growth depends on the secretion of new cartilage matrix by chondrocytes progeny. The current study described a different type of cartilage growth in different skeletal elements of camel embryos. Camel embryos were collected, fixed, dehydrated, and embedded in paraffin. Paraffin sections of the whole embryos were stained with H&E, Crossman trichrome and Mallory trichrome. Immunohistochemical staining for CD117 and type II collagen were used in embryonic cartilage. Other samples were processed to be examined by scanning electron microscopy. An Early Embryonic skeleton represented by cartilaginous elements. Undifferentiated cells continuing with the perichondrium penetrated the growing cartilage of ribs, pelvic bone, scapula, and sternum. The cells were CD117 positive and morphologically resembled the undifferentiated mesenchymal cells; they appeared spindle or flattened in shape with an oval nucleus and were connected by cytoplasmic processes. The orientation of the invaded cells could be as low or high cellular densities or as individual cells. The invaded mesenchymal cells transformed to chondrocytes and produce new cartilage matrix. They were immune-stained for Type II collagen The cellular penetration aimed to supply the developing cartilage by undifferentiated cells destined to become chondrocytes and involved in the interstitial growth of the fetal cartilage.
KEYWORDS
Cartilage Growth; Mesenchymal cells; Differentiation; Quail; Embryos
CASE SERIES
Cartilages growth occurs by secretion of new cartilage matrix. Appositional growth depends on activation of the perichondrial cells to secrete additional cartilage matrix encompassing the growing cartilage. Chondrocytes propagation and subsequent interstitial secretion of new cartilage matrix by chondrocyte progenies result in interstitial growth. Appositional growth commits growth of the cartilage width while the increase in cartilage length relies on interstitial growth [1]. An Unusual mode of cartilage growth has been described in the cartilage of quail embryos and the air breathing organ of catfish in which the undifferentiated mesenchymal cells participate in the interstitial type of cartilage growth. The mesenchymal cells derived from the perichondrium invade the interior of the growing cartilage and transformed chondrocytes to secrete new cartilage matrix. In femur and tibia of quail embryos, the mesenchymal invasion is limited to the central hypertrophic zone to provide the cartilage templates by a large population of chondrogenic potential cells [2]. In air-breathing organ of catfish, mesenchymal invasion occurs in multiple sites and involved in growth, renewal, and replacement of the existing cartilage [3]. The current study aimed to investigate a mesenchymal-dependent mode of cartilage growth in the skeleton of camel embryos. Description of a pattern of mesenchymal cells in the cartilage templates. We used immunohistochemical stainings to identify the localization of undifferentiated cells during growth of camel skeleton and type of collagen secreted by the differentiating cells.
MATERIALS AND METHOD
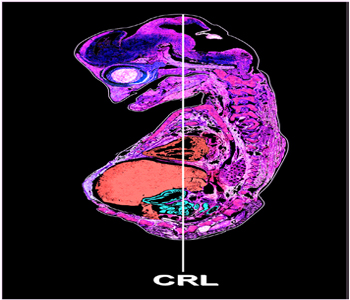
Preparation of Paraffin Embedding Samples The study was carried out on 18 camel embryos. The embryos were obtained form Al-Basatein slaughter house in Cairo. Stages of embryos were evaluated by measuring CRL (Crown-rump length) (Figure 1). The study was conducted using apparently healthy camel embryos measured 5, 6, 6.5, 8, 10, 11, 12 CRL. All embryos were immediately fixed in 10% buffered formalin and Bouin’s solutions. Fixed samples were dehydrated in ascending grades of alcohols at 70%, 80% for (3 hours) at each concentration, 90 % (2hours) and 100% (1 hour). Dehydrated samples were then impregnated and embedded in paraffin wax. Paraffin serial sections of 3-5µm were taken using a Richert Leica RM 2125 Microtome, Germany and mounted on glass slides. Sections were kept in an incubator at 40°C and stained with H&E [4] and Crossman’s trichrome (1937) and Mallory trichrome [5]. Chemical used in the preparation of paraffin sections and staining are summarized in (Table 1 Part I). Stained sections were examined using DMLS light microscope (Leica, Germany) outfitted with MC120 HD camera (Leica, Germany).
Figure 1: Illustration of camel embryo showed CRL measuring.
Table 1:Chemicals for Preparation of Paraffin Sections and Staining.
| NAME OF THE REAGENT | NAME OF THE REAGENT | COUNTRY |
| HAEMATOXYLIN CRYST. | MERCK | |
| EOSIN YELLOW (WATER SOLUBLE) | OXFORD LABORATORY REAGENTS | India |
| SODIUM PHOSPHATE DIBASIC(ANHYDROUS) | OXFORD LABORATORY | India |
| SODIUMPHOSPHATE MONOBASIC (ANHYDROUS) | OXFORD LABORATORY REAGENTS | India |
| ETHANOL ABSOLUTE | FISHER CHEMICALS | UK |
| PARAPLAST EMBEDDING MEDIA(PARAFFIN WAX) | SIGMA ALDRICH | Germany |
| MERCURIC OXIDE (YELLOW HGO) | ADWIC | |
| ALUMINUM POTASSIUM SULPHATE | OXFORD LABORATORY REAGENTS | India |
| GLACIAL ACETIC ACID | ADWIC | |
| FERRIC CHLORIDE | SAS CHEMICALS CO | |
| SAFRANIN O | SAS CHEMICALS CO | |
| XYLENE | SIGMA ALDRICH | Germany |
| METHYL BENZOATE | OXFORD LABORATORY REAGENTS | India |
| FAST GREEN | UNIVERSAL FINE CHEMICALS | India |
Immunohistochemistry Immunohistochemical staining was done on paraffin section of camel embryos using SuperfrostTM plus microscope slides. Antigen localization was performed using a polyclonal rabbit anti-c-kit antibody against CD 117, a mouse anti-rabbit against MMP-9, and a monoclonal mouse anti-chicken antibody against type II collagen using the Reagent of UltraVision Detection System (Anti-Polyvalent, HRP/DAB (ready to use) Thermo Fischer Scientific TP-015-HD) according to the manufacturer's instructions, combined with the Avidin-Biotin Complex (ABC) technique [6]. Sections (5 µm) of paraffin-embedded tissue were dewaxed, rehydrated and rinsed in phosphate-buffered saline (PBS) (pH 7.4), and treated with 3% hydrogen Peroxide ti inhibit endogenous peroxidase. For antigen retrieval, slides were placed in 10 mM sodium citrate buffer (pH 6.0) and heated to 95-98 in a water bath for 20 minutes followed by cooling for 20 min at room temperature. Sections were then rinsed in PBS (pH 7.4). Sections were covered with Ultra V block (refer to Table 1 Part II, Thermo Fisher scientific, USA) for 5 minutes at room temperature to block non-specific background staining. Sections were incubated with the primary antibodies at room temperature for 1 hour or overnight according to their respective data sheet. Antibody sources, dilution, and incubation duration are shown in (Table 1 Part IV), and as described previously [5]. Slides were washed with PBS (pH 7.4), followed by incubation for 10 minutes at room temperature with a biotinylated secondary antibody applied drops-wise on the section. Slides were rinsed in PBS (pH 7.4), followed by drops-wise addition of the drops of streptavidin- peroxidase complex (Thermo Fisher scientific, USA) and incubated for 10 minutes at room temperature followed by another rinse in PBS pH 7.4. Visualization of the bounded antibodies was performed using one 1 drop of diaminobenzidine (DAB) Plus chromogen mixed with 2 ml of DAB Plus substrate. Mix applied directly on the tissue and incubated for 5 -10 minutes at room temperature. Note: all incubations were performed in a humid chamber to avoid tissue desiccation. Sections were counterstained with Harris hematoxylin for 30 seconds, dehydrated and mounted with DPX. Negative controls were performed by omitting the primary antibodies. All immunohistochemical stainings were examined by light microscopy.
SEM Preparation
Camel embryos were longitudinally and transversely cut into small pieces which were then fixed in Karnovasky fixative (10 ml paraformaldehyde 25%, 10 ml glutaraldehyde 50%, 50 ml of 0.1 M sodium phosphate buffer (pH 7.2), and 30 ml D.W) as shown in Table 2 [7]. Specimens were washed in 0.1 M sodium -phosphate buffer (pH 7.2), post-fixed in 1% osmic acid in 0.1 M sodium -phosphate buffer and followed by another in 0.1 M sodium -phosphate buffer (PH 7.2). Samples were then dehydrated in alcohol as described previously and immersed in iso-amyl acetate. The dehydrated samples underwent critical point drying using a polaron apparatus. As a final step, samples were coated with gold using a JEOL-1100 E-ion sputtering device and observed with a JEOL scanning electron microscope (JSM – 5400 LV) at KV 10. Details regarding chemicals used in the preparation of scanning electron samples are shown in Table 3.
Table 2: Reagent of UltraVision Detection System (Anti-Polyvalent, HRP/ DAB (ready to use) Thermo Fischer Scientific TP-015-HD.
| P-015-HD | Component |
| TA-015-HP | Hydrogen Peroxide Block |
| TP-015-UB | Ultra V Block |
| TP-015-BN | Biotinylated goat Anti-Polyvalent |
| TS-015-HR | Streptavidin-Peroxidase |
| TP-015-HSX | DAB Plus Substrate |
| TA-001-HCX | DAB Plus Chromogen |
Table 3:Solutions used in immunohistochemistry and Scanning electron microscopy [5].
| PBS-buffer (pH 7,2-7,6):Used in immunohistochemistry | NaCl | 42.5g |
| Na2HPO42H2O | 6.35g | |
| NaH2PO4H2O | 1.95g | |
| TP-015-BN | Biotinylated goat Anti-Polyvalent | |
| dest. water | and 5l | |
| Citrate buffer (pH 6,0): | Solution A: | |
| Citrate C6H8O7H2O | 21.01 g | |
| Aqua dest. | and 1l | |
| Solution B: | ||
| Sodium Citrate Na3C6H5O72H2O | 29.41g | |
| dest. water | and 1l | |
| Using solution: | ||
| Solution A: | 9 ml | |
| Solution B: | 41 ml | |
| dest. water | and 500ml | |
| PBS-buffer (pH 7,2-7,6): | Solution A: | |
| Used for scanning | Na2HPO42H2O | 17.02 g |
| Aqua dest. | and 600ml | |
| TP-015-HSX | DAB Plus Substrate | |
| Solution B: | ||
| NaH2PO4H2O | 6g | |
| dest. water | and 250ml | |
| Using solution: | ||
| Solution A: | 580ml | |
| Solution B: | 219ml | |
| dest. water | ad 500ml |
Table 4: Identity, sources, and working dilution of antibodies used in this study.
| Target | Primary antibody | Biotinylated secondary antibody | ||||
|---|---|---|---|---|---|---|
| Supplier | Origin | Dilution | Incubation | Antigen retrieval | ||
| CD117(Anti –c-Kit) | Genremed Biotechnologie,Inc, South San Francisco,USA | Rabbit polyclonal | 1:50 | 1 h at room temperature | Boiling in citrate buffer (pH 6.0),20 min | Goat anti-polyvalent |
| Collagen IIAb 3-3(Clone6 b3) | Thermo Fischer Scientific,Lab vision Corporation,Fremont, USA | Mouse anti-chicken(mouse monoclonal | 0.180555556 | Overnight | boiling in citrate buffer (pH 6.0),20 min | Goat anti-polyvalent |
RESULTS
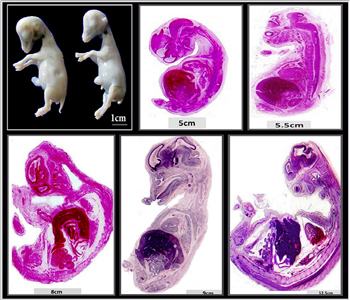
The primitive skeleton of the camel was examined at different stages of embryonic development (Figure 2). The growing cartilage templates were penetrated by flattened cells continued with the perichondrium (Figure 3A, B, 4A, B and 8B). The invaded cells were identical to mesenchymal cells. They appeared as small flattened, spindle or satellite in shape with cell processes (Figure 3C, G, E, 8D, 5C, D). Mesenchymal cells penetration could be observed in different directions either perpendicular (Figure 3B, C), oblique (Figure 3 D, E), parallel (Figure 3F, G) to the long axis of the developing cartilage. The mesenchymal cells may extend along the whole width of the growing cartilage (Figure 4A, B), confined to a limited area (Figure 4C, D), streak of mesenchymal cells (Figure 5 A, D) or multiple sites of mesenchymal cells localization (Figure 5A, B, 6B, C, E, G, H). The density of the mesenchymal cells varied as low, high cellular masses (Figure 5B, E) or as individual cells (Figure 5C, D). Areas of undifferentiated cells stained negative for Mallory trichrome (Figure 4F). Other areas the cells secreted cartilage matrix stained positive for Mallory trichrome (Figure 4E). The mesenchymal cells changed in morphology, became rounded, and surrounded by little cartilage matrix (Figure 6I-L). Mesenchymal cells were observed in camel embryos measured 5, 6, 6.5, 8, 10, 11, 12 cm CRL in different skeletal elements including vertebral cartilage, sternum, scapula, pelvic and ribs (Figure 6A-L). In scanning electron samples, various differential stages of chondrogenic cells were recognized. Undifferentiated mesenchymal cells appeared as small flattened cells with cell processes which may connect with other cells (Figure 8B, C, E, F). Differentiating chondrogenic cells were small rounded in shape (Figure 8A-H). Mature chondrocytes were large rounded situated inside the lacunae (Figure 7F-H). Various stages of chondrogenic cells showed a different affinity for CD117 immunostaining. Mesenchymal cells and differentiating chondrocytes inside the cartilage were strong CD117 positive as well as the perichondrial cells while chondrocytes were weakly positive (Figure 7A-D). The mesenchymal cells which penetrate the cartilage of the scapula were positively immunostained for type II collagen (Figure 7E, F).
Figure 2: Camel embryos.
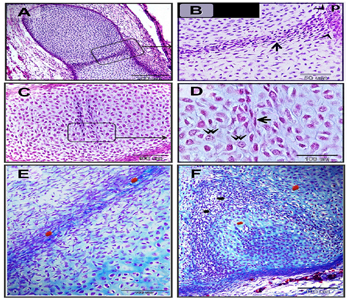
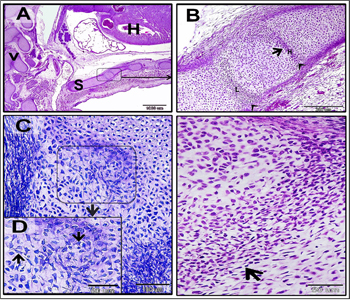
Figure 3: Mesenchymal cells invaded the cartilage.
Paraffin sections in camel embryos measured 10 cm stained with H&E. A-G: flattened mesenchymal cells (black arrows) penetrated the growing cartilage template of the sternum. Mesenchymal cells run in different directions as showmen in B, D, F. Note: heart (H) and sternum (S).
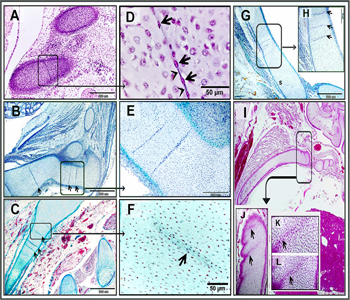
Figure 4: Origin mesenchymal cells and destination of mesenchymal cells.
Paraffin sections in camel embryos measured 5 cm in (E, F), 6 cm in “C, D” 8 cm in (A, B), 10 cm in (G) showed sternum in (A-G) stained with H&E (A-D) and Mallory trichrome (E, F). A, B: mesenchymal cells continued with the perichondrium (P), arrowheads refer to the mesenchyme extended in the whole width of the growing cartilage. C, D: mesenchymal cell invasion was confined in a limited area. E, mesenchymal cells started to secrete cartilage matrix (red asterisk). F: areas of mesenchymal cells lack cartilage matrix (black asterisk) compared with areas of rich cartilage matrix (red asterisk). Note: mesenchymal cells (arrows), hypertrophic chondrocytes (double arrowheads).
Figure 5: Pattern of mesenchymal invasion.
Paraffin sections in camel embryos measured 10cm in (A, B, E), 5 cm in (C, D), showed sternum in (A, B, E) and vertebral cartilage (C, D) stained with H&E. A, B: multiple sites of cellular invasion (arrowheads). The invading mesenchyme could be ofhigh” arrow in (B) or low cellular density (arrow) in (E). C, D: The invading mesenchymal cells appeared as individual cells, mesenchymal cells were small with cell processes (arrows).
Figure 6: Mesenchymal cells in cartilage templates of vertebra, Pelvic, scapula, sternum, ribs.
Paraffin sections showed cartilage templates of vertebral cartilage of 11 cm CRL camel embryo (A, D), Pelvic of 6.5 cm CRL camel embryo (B, E), scapula of 8 cm CRL camel embryo (C, F), sternum of 11 cm CRL camel embryo (I-L) and, ribs of 6.5 cm CRL (G, H) camel embryo stained by H&E (A, D, I-L) and crossman’s trichrome (B, C, E-H). A, D: mesenchymal cell, invaded the vertebral cartilage “black arrows” connected together with cell processes “arrowheads”. (D) represent a higher magnification of (A). B, E: multiple sites of mesenchymal cells (arrows) in pelvic bone (E) represent a high magnification of (B). C, F: Multiple sites of mesenchymal cells (arrows) in cartilage template of the scapula. (F) represent a high magnification of (C). G, H:: Multiple sites of mesenchymal cells (arrows) in cartilage template of the rib. (H) represent a high magnification of (G). I-L: Differentiating chondrocytes acquired rounded profile and interspersed in little cartilage matrix“black arrows”. (J-L) represent a high magnification of (I).
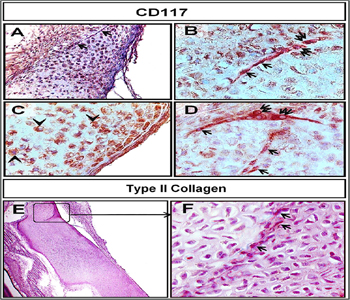
Figure 7: Staining affinity of the invading cells for polyclonal rabbit antic-kit (CD 117) and monoclonal mouse anti-chicken type II collagen.
Paraffin sections of sternum “B, D” and ribs “A, C” of camel embryos stained for CD117. The invading mesenchymal “arrows” and the differentiating chondrocytes “double arrows” were strong immunoreactive for CD117 in (A, B, D). Chondrocytes gave weak positive reaction for CD117 “arrowhead in D” Note; CD117-positive perichondrial cells “P”in (A, C). Paraffin section of the scapula of the camel was immunostained for Type II collagen.
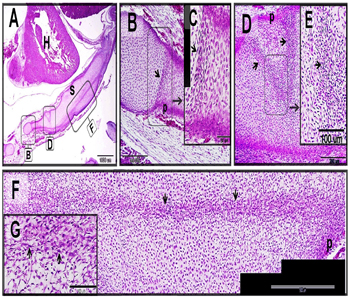
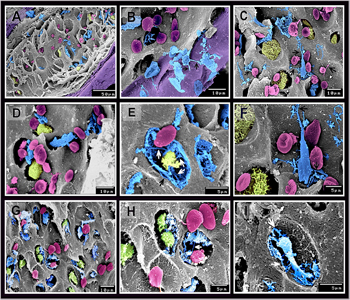
Figure 8: Scanned electron samples of camel skeleton.
Scanned electron sample 12 cm CRL measured of vertebrae (A-D, F) and 8cm CRL measured of the scapula (E, G-I) of camel embryo. Mesenchymal cells (blue colored) appears flattened in shape with cell process (arrows) penetrating the cartilage templates. differentiating chondrocytes (pink colored) acquired rounded shape with a smooth surface and may have cell processes (arrows). Hypertrophic chondrocytes (green colored) appeared as large cells with an irregular surface. Hypertrophic chondrocytes may be connected with the mesenchymal cell by cell processes (arrows in (E).
DISCUSSION
Using mesenchymal cell in regenerative medicine has attracted the interest of basic scientists. Several Biotechnological procedures are developed for cartilage renewal in order to build up new cartilage matrix and preserve viable cells. The current investigation described a new mechanism of cartilage growth in camel embryos which may be valuable in stem cell therapy for cartilage repair. We examined many skeletal elements in different embryonic stages by using light and scanning electron microscopy and used immunohistochemistry for identification of the distribution undifferentiated cells and type II collagen to detect cartilage forming cells. CD117 or KIT is a surface tyrosine kinase receptor and identified as stem cell factor controlling a wide range of biological activities such as cellular differentiation, proliferation, chemotaxis, cell adhesion and death. CD117 is expressed in mast cells, some hematopoietic stem cells, germ cells, melanocytes, and Cajal cells of the gastrointestinal tract, and oncogenic cells [8,9,10]. In the current investigation, we used CD117 immunostaining in sternum of camel embryos identify the distribution of mesenchymal cells during growth of cartilage templates. Mesenchymal cells, differentiating chondrocytes inside the cartilage matrix as well as perichondrial stem cells were Strong CD117 positive. We identified the common morphological characteristics of undifferentiated mesenchymal cells in paraffin-embedded and scanned embryonic samples. Mesenchymal cells appeared small with cell processes arisen from the perichondrium. The orientation of the intracartilaginous mesenchymal cells was varied as regions of high, low cellular density or individual cells. Mesenchymal cells may be organized as cellular streaks connected with each others by cell processes. Mesenchymal cells cloud be distributed in multiple sites in cartilage templates. Areas of mesenchymal cells contained ill-defied cartilage matrix whiles areas where differentiating chondrocytes existed stained positive by Mallory trichrome revealing that mesenchymal cells destinate the chondrogenic cell line. Thus, mesenchymal cells involved in the interstitial growth of embryonic cartilage in camel, while the common types of cartilage growth depend on either the perichondrial stem cells or chondrocytes. The perichondrial cells participate in appositional growth by enclosing the growing cartilage by the new matrix. Chondrocytes division yield progeny which secrete new matrix resulted in interstitial growth [11, 12]. Mesenchymal cells are usually known to share in endochondral ossification during the development of long bones [13,14]. Undifferentiated cells are also documented during cartilage canals formation. Cartilage canals formed of blood vessels, perivascular cells [15], monocytes, and macrophages [16]. Osteogenic cells which express type I collagen and the bone-specific protein, periostin are recognized in cartilage canals [17,18,19]. Moreover, other cells expressing type II collagen are located in cartilage canals [20,21]. In our study, mesenchymal cells could be observed at any area in cartilage templates of long and flat bone and were not limited to the epiphysis. Moreover, mesenchymal cells were not associate with vascular elements The contribution of mesenchymal cells in interstitial cartilage growth is described in previous researches. Mesenchymal cells of chondrogenic potentiality engaged in interstitial growth in cartilage templates of the tibia and femur of embryonic quail [2].. In catfish, interstitial mesenchymal cells differentiation occurs in the supporting cartilage air breathing organ to aiming in growth, renewal and replacement of the existing cartilage [3,22,23].
CONCLUSION
CD117 positive mesenchymal cells involved in the interstitial growth of the cartilage templates in camel embryos..
REFERENCES
- Eroschenko VP and Di Fiore MSH. (2013). Difiore’s atlas of histology with functional correlations. Lippincott Williams & Wilkins, 12th edition. 114.
- Soliman SA. (2013). Histomorphological and morphometrical studies on endochondral ossification in the japanese quail. 66: 84.
- Soliman SA. (2014). New aspect in cartilage growth “The invasive interstitial type”. Journal of american science. 10: 272-277.
- Mallory. (1936). Stain technology. 11: 101.
- Bancroft JD, Layton C and Suvarna SK. (2013). Bancroft’s theory and practice of histological techniques, 7th Edition. Expert Consult: Online and Print. Elsevier health sciences.
- Hsu SM, Raine L and Fanger H. (1981). Use of avidinbiotin-peroxidase complex (ABC) in immunoperoxidase techniques: A comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem. 29: 577-580.
- Karnovsky MJ. (1965) A formaldehyde-glutaraldehyde fixative of high osmolarity for use in electron microscopy. J. Cell Biol. 27: 137a.
- Miettinen M and Lasota J. (2005) KIT (CD117): A review on expression in normal and neoplastic tissues, and mutations and their clinicopathologic correlation. Appl Immunohistochem Mol Morphol. 13: 205-220.
- Arnhold S, Gluer S, Hartmann K, Raabe O, et al. (2011). Amniotic-fluid stem cells: Growth dynamics and differentiation potential after a Cd-117-Based selection procedure. Stem Cells Int. 715341.
- Margaritescu C, Pirici D, Simionescu C and Stepan A. (2011). The utility of CD44, CD117 and CD133 in identification of cancer stem cells (CSC) in oral squamous cell carcinomas (OSCC). Rom J Morphol Embryol. 52: 985-993.
- Marjit B. (2009) General anatomy genetics histology and embryology. Academic publishers. 1.26.
- Rawlani S and Rawlani SJ. (2011). Textbook of general anatomy. P medical ltd. 45.
- Carlevaro MF, Cermelli S, Cancedda R and Descalzi cancedda F. (2000). Vascular endothelial growth factor (vegf) in cartilage neovascularization and chondrocyte differentiation: Auto-paracrine role during endochondral bone formation. J Cell Sci. 113: 59-69.
- Kanczler JM and Oreffo RO. (2008) Osteogenesis and angiogenesis: the potential for engineering bone. Eur Cell Mater. 15: 100-114.
- Blumer MJ, Longato S, Richter E, Perez MT, et al. (2005) The role of cartilage canals in endochondral and perichondral bone formation: Are there similarities between these two processes? J anat. 206: 359-372.
- Gabner S, Häusler G and Böck P. (2014). Vascular channels in hyaline cartilage: development of channels and removal of matrix degradation products. Anat histol embryo. 43: 29.
- Blumer MJ, Fritsch H, Pfaller K and Brenner E. (2004). Cartilage canals in the chicken embryo: Ultrastructure and function. Anat embryol (berl). 207: 453-462.
- Blumer MJ, Longato S and Fritsch H. (2004). Cartilage canals in the chicken embryo are involved in the process of endochondral bone formation within the epiphyseal growth plate. Anat rec a discov mol cell evol biol. 2791: 692-700
- Blumer MJ, Schwarzer C, Perez MT, Konakci KZ, et al. (2006). Identification and location of bone-forming cells within cartilage canals on their course into the secondary ossification centre. J Anat. 2086: 695-707.
- Le Guellec D, Mallein-gerin F, Treilleux I, Bonaventure J, et al. (1994). Localization of the expression of type i, ii and iii collagen genes in human normal and hypochondrogenesis cartilage canals. Histochem j. 269: 695-704.
- Claassen H, Kirsch T and Simons G. (1996). Cartilage canals in human thyroid cartilage characterized by immunolocalization of collagen types i, ii, pro-iii, iv and x. Anat embryol (berl). 1942: 147-153.
- Soliman SA and Abd-elhafeez HH. (2014). Mesenchymal cells in cartilage growth. Lap lambert academic publishing. 4: 49.
- Crossmon G. (1937). A modification of mallory’s connective tissue stain with discussion of the principle involved. Anat rec. 69: 33-38.