Previous Issues Volume 2, Issue 1 - 2017
We Use Our Gut to Diagnose But Do We Have The Guts to Send Patients Home?
Analiese Wagner*, Alexander B. Norinsky, Alan Lucerna, James Espinosa
Department of Emergency Medicine, Rowan University SOM Kennedy University Hospital, Stratford, NJ, USA.
Corresponding Author: Analiese Wagner, Department of Emergency Medicine, Rowan University SOM Kennedy University Hospital, 18 East Laurel Road, Stratford, NJ 08084, Tel: (856) 256-4660; E-Mail: [email protected]
Received Date: 13 Dec 2016 Accepted Date: 03 Jan 2017 Published Date: 09 Jan 2017
Copyright © 2017 Wagner A
Citation: Wagner A, Norinsky B A, Lucerna A and Espinosa J. (2017). We Use Our Gut to Diagnose But Do We Have The Guts to Send Patients Home?. Mathews J Emergency Med. 2(1): 019.
ABSTRACT
The incidence of co-existing peripheral and central venous thromboembolic disease remains elusive. The available literature is arguably outdated and has limited applicability in today's practice at the bedside. Physicians rely on clinical gestalt along with available decision tools (e.g. Wells' and PERC) to risk-stratify patients with potential co-existing disease. Recently, many have opted to manage the majority of deep vein thrombosis cases in the outpatient setting; although still in its infancy and controversial, there has been a recent trend towards doing the same with specific populations of patients with pulmonary embolism. In this manuscript, we review three cases of patients with newly-diagnosed deep vein thrombosis who were subsequently found to have pulmonary embolism on further testing in the emergency department. Each case had a similar course and outcome, and in each instance the question was raised: in the context of a "normal" clinical evaluation and our gestalt, was it appropriate to further investigate for central thromboembolic disease in the emergency department?.
KEYWORDS
Venous Thromboembolism; Pulmonary Embolism / Deep Vein Thrombosis.
INTRODUCTION
The clinician's gestalt and available decision tools, such as the Wells' criteria, are adequate, well-established, reproducible modalities for risk-stratifying patients for evaluation of potential venous thromboembolic (VTE) disease. Once a diagnosis of DVT is made, emergency physicians are tasked with riskstratifying these patients for central thromboembolic disease based on, again, gestalt and decision tools. The incidence of co-existent peripheral and central VTE remains elusive. That is, there remains limited data on the incidence of pulmonary embolism (PE) in the context of newly-diagnosed deep vein thrombosis (DVT). The majority of available literature is over thirty years old and has very small sample sizes, with the potential statistical effect of over-estimation of the overall percentage of potentially missed or occult pulmonary emboli. In addition, technology has advanced substantially; image quality and interpretation of those images have an impact on those numbers if the studies were repeated today. Furthermore, the studies utilized ventilation-perfusion scans for diagnosing PE, whereas the overwhelming majority of today's emergency physicians and radiologists use computed tomography. Many have argued that even though we are discovering more cases of PE, all-too-often our patient's relevant, measurable outcomes aren't affected. Perhaps the introduction of more advanced technology allows us to identify more (clinically irrelevant) disease at our patient's expense without significantly helping them? In the context of appropriate resource utilization, radiation risks, healthcare expenditure, and the availability of newer medications in our arsenal (i.e. noval oral anticoagulants), there may increasingly be pressure on emergency physicians to discharge patients with DVT, and more recently a certain demographic of patients with PE. The following is a series of cases of DVT during which the physicians, considering the available data and their clinical instincts, pursued further testing and found PEs in asymptomatic patients. "We discuss the value of a physcian's gestalt, incidence of these questionably "silent" PEs, and the struggles of effective risk-stratification of patients to the appropriate treatment modalities and dispositions".
CASE 1
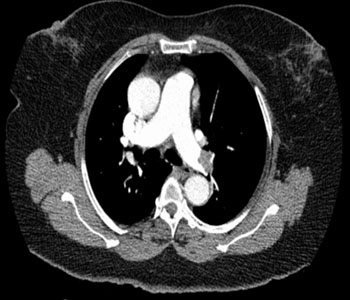
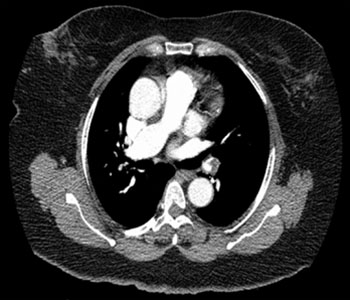
A nontoxic-appearing 69-year-old female presented to our community emergency department (ED) for the evaluation of right leg discomfort and edema progressively worsening over the past week. She denied a history of similar leg swelling or that of any other extremity, trauma, travel, immobilization or surgeries. She also denied chest pain, dyspnoea, cough, fevers or any other systemic symptoms. Her home medications included metoprolol, pravastatin, hydrochlorothiazide, and a daily aspirin for hypertension and coronary artery disease. Relevant personal and family history, including that of venous thromboembolic disease or cancer, was likewise negative. She was a non-smoker. The initial vital signs were an automated blood pressure of 148/85 mmHg, pulse rate of 63 beats per minute and regular, 18 respirations per minute, and with an oral temperature of 98.1 degrees Fahrenheit. Her right lower extremity revealed 2+ edema up to the knee. The affected limb was tender to palpation especially along the gastrocnemius muscle. There was no overlying skin changes, palpable cords or varicosities. She was neurovascular intact distally, and compartments were soft and compressible. The remainder of her physical examination was completely unrevealing. Doppler ultrasonography of the affected extremity's venous system revealed a DVT extending from the common femoral vein to the popliteal vein. Given the impressive extent of clot burden, additional studies were obtained to evaluate for central thromboembolic disease. Cardiac biomarkers including a brain-natriuretic peptide were negative. Computed axial tomography of the chest (PE protocol) showed an extensive thrombus within the distal portion of the left main pulmonary artery measuring 1.6 cm x 1.3 cm x 3.2 cm and extending into the left lower lobe segmental branches (Figures 1 and 2). A D-dimer was ordered, as there is some evidence showing its utility for risk-stratification purposes: 13.42 (reference range < 0.49 μg/mL)
Figure 1: Axial views of the computed tomography angiograph for the chest at the level the bifurcation of the pulmonary arteries, revealing a large embolus in the left main pulmonary artery.
Figure 2: Axial views of the computed tomography angiograph for the chest below the bifurcation of the pulmonary arteries, revealing the clot extending into the left lower lobe segmental branches.
Throughout the course of the patient's stay in the emergency department, she never became tachycardic (at times, she was bradycardic with pulse rates ranging from 52 to 63 bpm). There were also no episodes of tachypnea or hypotension (systolic blood pressures ranged from 125 to 177 mmHg). Furthermore, on frequent re-evaluations she consistently denied chest discomfort, dyspnea, or any other complaints. In fact, she was fairly adamant about leaving the ED, stating her symptoms were too mild for admission and she wanted to follow-up with her primary care physician instead. The patient ultimately agreed to be admitted and a heparin drip was initiated. An echocardiogram done the following day revealed pulmonary artery pressures 25-30 mmHg, with grade I-II left ventricular diastolic dysfunction and preserved left and right ventricular ejection fractions. She was discharged on hospital day 2 on rivaroxaban.
CASE 2
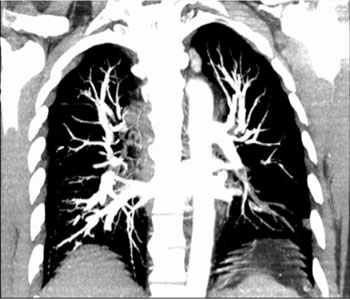
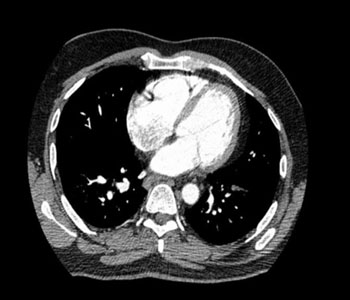
A nontoxic-appearing 56-year-old male presented to our community emergency department for evaluation of worsening left lower extremity edema over the past week. He reports having had a deep vein thrombosis in the right lower extremity two years prior, for which he was on a 6-month course of apixaban. At presentation, he remained solely on full-dose aspirin therapy which he was to continue indefinitely. He denied chest pain, dyspnoea, cough, fevers, or involvement of any other extremity. Medical, surgical, and family histories were negative for cancer or thrombophilia. He was a non-smoker. He reported no recent hospitalizations or surgeries, and denied any travel or prolonged immobilization. On physical examination, he was well-appearing and in no acute distress. The left lower extremity was noted to be tender to palpation with 3+ edema up to the mid-thigh; pedal pulses were palpable and distal sensory and motor function were preserved; capillary refill of the nail beds were less than 2 seconds; his compartments were soft and compressible. The right lower extremity was unimpressive. During the course of his emergency department visit, his blood pressure ranged from 118/80 mmHg to 133/84 mmHg, pulse rate 84 to 90 beats per minute, breathing 16 to 18 respirations per minute, pulse oximetry remaining 98-100% on ambient air, and was afebrile. Doppler ultrasonography revealed an extensive, completely occlusive deep vein thrombus throughout the left common femoral, femoral and popliteal veins, as well as partial thrombosis of the posterior tibial veins. Despite consistently normal vital signs and lack of any other symptomology, computed axial tomography of the chest (PE protocol) was ordered, revealing bilateral segmental and sub segmental multilobar pulmonary emboli with evidence of right heart strain (Figures 3 and 4). Cardiac biomarkers including a brain-natriuretic peptide were negative.
Figure 3: A single slice in the coronal view, computed axial tomography of the chest in the MIP (maximal intensity projection) format, revealing an extensive thrombus in the left lower segmental and subsegmental branches.
Figure 4: Axial views of the computed tomography angiograph for the chest at the level of the mitral valve, revealing right heart strain with dilated right atrium and ventricle, as well as clot burden extending into the left subsegmental branches.
Low-molecular weight heparin was administered. Although not disclosed initially, the patient later admitted that he had Factor V Leiden deficiency and was in fact treated two years prior for a deep vein thrombosis with thrombectomy, a 6-month course of apixaban and an inferior vena cava (IVC) Greenfield filter. The following day, the patient was taken to Interventional Radiology for thrombectomy and local thombolytic therapy (power-pulse infusion and AngioJet thrombolysis) secondary to worsening pain and distal mottling. It was noted during the procedure that the lower extremity DVT in fact extended into the inferior vena cava and passed the filter. The patient did well and was sent home several days later for lifelong rivaroxaban therapy.
CASE 3
A nontoxic-appearing 63-year-old female presented to the emergency department for left calf pain of two-day's duration. She reported a history of peripheral vascular disease and suspected an occlusion of her femoral-popliteal bypass. She too denied any cardiopulmonary or systemic symptomology. Serial vital signs throughout her stay in the emergency department remained stable aside from persistent hypertension (she had a history of essential hypertension). A venous duplex ultrasound of the left lower extremity revealed a thrombus extending from the left common femoral vein to the popliteal vein. Given the clot burden, a CT angiogram of the chest was performed, revealing filling defects in the right upper middle and lower lobe pulmonary arteries. She was started on unfractionated heparin and admitted for further management. Of note, the femoral-popliteal bypass was found to be unaffected.
DISCUSSION
Pulmonary embolism is frequently a difficult diagnosis to make in the emergency department. The patient's symptoms are often vague and may overlap with other disease processes. Dyspnea is the most common presenting complaint, which may have cardiac, pulmonary, metabolic or even psychological etiologies. Clinicians rely on their gestalt as well as established accelerated diagnostic risk-stratification algorithms for the evaluation of suspected PE. The Wells' criteria, Geneva score and Pulmonary Embolism Rule-out Criteria (PERC) rule, to name just a few, use vital signs and risk factors to guide physicians. Of the patients discussed in our cases, all of them had "normal" vital signs and only one had a prior history of a deep vein thrombosis (and not other identifiable risk factors). In this case, the physician relied almost completely on intuition as his guide to order CT angiography. But how reliable is a physician's gestalt? One study looked at the efficacy of the physician's gestalt pretest probability in combination with the PERC rule, and found an incidence of PE in less than 2% (at initial presentation and within 45 days) if the physician believed the patient to be low-risk and was PERC-negative. A recent literature review the American College of Emergency Physicians policy also addressed the utility of gestalt, finding it "comparable" to the Wells', Geneva, and Kline scores when applied to appropriate low-risk populations [1]. However, important concerns were raised, and especially relevant in our review: the experience of the physician and lack of agreement between the definition of "low-risk" remain important limiting factors on the practical level [2]. The patients presented in our cases had radiographic evidence of a proximal DVT but were hemodynamically stable. Other than the extent of the clot burden and the physician's intuition, there were no clear reasons to image their pulmonary arteries. While the presence of a DVT is an obvious risk factor of PE, how commonly do the two entities co-exist? When should we consider PE in a patient with DVT and no other clinical symptoms? Should clot burden and/or location of the DVT influence our decision to further test for PE? A number of studies from the late 1980's and early 1990's can offer some insight into these questions. Moser et al. [3]. found up to 40% of patients with radiographically-proven DVT and no symptoms of PE had abnormal, high-probability ventilation-perfusion (V/Q) scans. Dorfam et al. [4]. Conducted a similar study and correlated that data with DVT location; they found 35% of patients with proximal lower extremity DVT had high-probability V/Q scans. While these numbers seem compelling, it is worth noting that both of these studies had very small sample size (in the latter study, 35% represented only 17 patients). Monreal et al. [5]. Conducted a similar study with larger population size (364 patients with radiographically-documented DVT) they found seventy-six patients (22%) had a "silent PE" (a high-probability V/Q with no clinical signs or symptoms of PE). At face value, the raw numbers in the data presented above are startling. Ultimately, the treatment for both conditions is the same, anticoagulation. The primary difference is definitive disposition of the patient from the emergency department (i.e. admission to the hospital versus outpatient follow-up). There has been a strong trend for the latter in the DVT-only population with low-molecular-weight heparin or a novel-oral anticoagulant (NOAC). The notion of the same fate for simple uncomplicated PEs remains to be determined and is under debate currently. If a patient with DVT is discharged with outpatient therapy, what are the consequences if they did, in fact, have an occult PE? Furthermore, should the presence of an occult or "silent" PE change our final disposition if the patient is hemodynamically stable and without symptoms? The answer again comes down to risk-stratification. Patients with normal vital signs and normal oxygenation are typically at lower risk for adverse events from a PE. This was demonstrated by Nordenholz et al. [6] in their study which employed two different risk-stratification criteria in ED patients admitted with PE: an oxygen saturation of greater than 92.5% and a Pulmonary Embolism Severity Index (PESI) classification. The PESI takes into account vital signs and co-morbidities, and assigns categories of risk based on the point values. The study found that low-risk PESI (Class I or II) correlated with an adverse outcome rate of 2.2%, none of which were fatal, and a PESI Class III or greater corresponded to a rate of 13%. Patients with oxygen saturations greater than 92.5% were also considered low-risk, and their adverse outcome rate was low at 1.1%. According to these criteria, the patients in our cases would be considered low-risk, although it is interesting to consider whether or not their PEs may be considered inconsequential in the long run-perhaps, notwithstanding the extensive proximal disease in the first case, the need for invasive intervention in the second, and multilobar clot burden in the third. Importantly, there are several potential confounding factors when reviewing our cases retrospectively that limit generalizability (for example, the use of beta-blockers by the first patient most likely affected her heart rate, and omission of key historical details because the patient had poor recall and insight in the second case); on the contrary this highlights the need to individualize each patient beyond a set of numbers and risk factors [7-9]. There has been increased discussion recently about discharging patients with PE. An international study from the Lancet demonstrated non-inferiority of outpatient versus inpatient treatment of symptomatic, low-risk patients PE with regard to safety and mortality outcomes. A recent meta-analysis of 11 studies encompassing over 1200 patients showed similar results, with recurrence rates of venous thromboembolism of 1.47%, fatal PE 0.4%, and overall mortality to be 1.58%. Of note, several of the studies examined in this meta-analysis used both physician gestalt and PESI classifications to categorize low-risk symptomatic PE patients. Lastly, as with DVT, rivaroxaban has shown to be non-inferior to standard anticoagulation therapies for PE. Although the literature seems to support outpatient treatment for PE and DVT, a review by Stein et al. [10]. Showed that only 1.7% of hemodynamically stable PE patients and 33.9% of DVT patients were being treated as outpatients. Treatment and disposition for VTE in hemodynamically unstable patients has always been straightforward: admission for close hemodynamic monitoring, anticoagulation, thrombolytics, and/or mechanical intervention. Utilizing thrombolytics in patients presenting with submassive PE has been less clear. Several studies have suggested an intermediate-risk patient that may benefit from thrombolysis: patients with confirmed PE, normal vital signs, and some degree of cardiac compromise (evidence of right heart strain and/or positive cardiac biomarkers). In 2002, Konstantinides et al. [11]. demonstronstrated the value of this therapy, finding that patients with submassive PE had less "escalation in care due to clinical deterioration" (i.e. additional thrombolytics, intubation, cardiac arrest) when treated with alteplase and heparin, as compared to heparin plus placebo. Similarly in the PEITHO trial, tenecteplase plus heparin was compared to heparin plus placebo, finding the tenecteplase group had less hemodynamic decompensation and death at 7 days (however, this group did have much higher risk of major bleeding). The American Heart Association position, based on their review of the literature, recommends utilizing thrombolytics for submassive PE in patients who develop hemodynamic instability or severe right ventricular strain. One of our patients did have evidence of right heart strain on CTA and required intervention for his DVT. His admission in hindsight may be considered justifiable if considering his stable vital signs and a low PESI score [12, 13].
CONCLUSION
It appears that the physician"s gestalt and available decision algorithms are adequate and reproducible tools for riskstratifying patients for VTE. Several of the aforementioned studies demonstrate that low-risk patients have favorable outcomes and can be treated safely as outpatients. Perhaps this concept of "missed" PEs with our DVT patients is moot? If a patient is low-risk for PE by the physician's initial assessment, screening for PE may not even be necessary especially given that treatment modalities for inpatient and outpatient setting are identical. In almost all of the studies, the patients that did have adverse outcomes as outpatients typically had some other underlying reason for clot formation (e.g. cancer). These patients, with a clear etiology and potential for multiple comorbidities, need to be stratified differently. Moving forward, more research needs to be conducted to further elucidate key considerations on this controversial discussion. What are the true incidence rates of co-existing DVT and (clinically relevant) occult PE nowadays, in the context of advancing technologies? What is the role of clinical gestalt on the part of the emergency physician? How to interpret vital signs and available clinical decision tools for the multitude of different patient demographics that present with benign symptoms? Must we consider the extent of peripheral clot burden in our decision to further test for central thromboembolic disease? Whereas management protocols seem to be straightforward in the extremes of disease (i.e. low-risk ambulatory patients and hemodynamically unstable critical patients), how do we reconcile potentially conflicting strategies in the intermediate population: discharge with oral anticoagulation versus thrombolysis/mechanical intervention? In the era of increasing sophistication and scrutiny, the emergency physician is tasked with walking an ever-thinning tightrope: evaluating and managing the patient completely and perfectly while trying to utilize resources responsibly and appropriately, minimizing costs while considering healthcare over-expenditure at large, weighing the availability of powerful technology (perhaps too good?) versus the risks of unnecessary radiation, and incorporating an ever-increasing arsenal of available medications and treatment strategies. While it seems as though the research and debates into this topic will continue for some time, it is an interesting microcosm reflecting the pressure placed on emergency physicians both on a specific and global level.
REFERENCES
- Kline JA, Courtney DM, Kabrhel C, Moore CL, et al. (2006). Prospective multicenter evaluation of the pulmonary embolism rule-out criteria. Journal of Thrombosis and Haemostasis. 6(5): 772-780.
- Fesmire FM, Brown MD, Espinosa JA, Shih RD, et al. (2011). Critical Issues in the Evaluation and Management of Adult Patients Presenting to the Emergency Department with Suspected Pulmonary Embolism. Annals of Emergency Medicine. 57(6): 628-652.
- Moser KM, Fedullo PF, LitteJohn JK and Crawford R. (1994). Frequent Asymptomatic Pulmonary Embolism in Patients with Deep Venous Thrombosis. JAMA. 271(3): 223-225.
- GS Dorfman, JJ Cronan, TB Tupper, RN Messersmith, et al. (1987). Occult pulmonary embolism: a common occurrence in deep venous thrombosis. American Journal of Roentgenology. 148(2): 263-266.
- Monreal M, Ruiz J, Olazabal A, Arias A, et al. (1992). Deep Venous Thrombosis and the Risk of Pulmonary Embolism. A Systematic Study. Chest. 102(3): 677-681.
- Nordenholz K, Ryan J, Atwood B, and Heard K. (2011). Pulmonary Embolism Risk Stratification: Pulse Oximetry and Pulmonary Embolism Severity Index. The Journal of Emergency Medicine. 40 (1): 95-102.
- Aujesky D, Roy PM, Verschuren F, Righini M, et al. (2011). Outpatient versus inpatient treatment for patients with acute pulmonary embolism: an international, open-label, randomised, non-inferiority trial. Lancet. 378: 41-48.
- Piran S, Le Gal G, Wells PS, Gandara E, et al. (2013). Outpatient treatment of symptomatic pulmonary embolism: A systematic review and meta-analysis. Thrombosis Research. 132(5): 515-519.
- The EINSTEIN-PE Investigators. (2012). Oral Rivaroxaban for the Treatment of Symptomatic Pulmonary Embolism. N Engl J Med. 366: 1287-1297.
- Stein PD, Matta F, Hughes PG, Hourmouzis ZN, et al. (2016). Home Treatment of Pulmonary Embolism in the Era of Novel Oral Anticoagulants. The American Journal of Medicine. 129(9): 974-977.
- Konstantinides S, Geibel A, Heusel G, Heinrich F, et al. (2002). Heparin plus Alteplase Compared with Heparin Alone in Patients with Submassive Pulmonary Embolism. For the Management Strategies and Prognosis of Pulmonary Embolism-3 Trial Investigators. N Engl J Med. 347: 1143-1150.
- Meyer. (2014). Fibrinolysis for Patients with IntermediateRisk Pulmonary Embolism. N Engl J Med. 370: 1402-1411.
- Jaff MR, McMurtry MS, Archer SL, Cushman M, et al. (2011). Management of Massive and Submassive Pulmonary Embolism, Iliofemoral Deep Vein Thrombosis, and Chronic Thromboembolic Pulmonary Hypertension. A Scientific Statement from the American Heart Association. American Heart Association Council on Cardiopulmonary, Critical Care, Perioperative and Resuscitation; American Heart Association Council on Peripheral Vascular Disease; American Heart Association Council on Arteriosclerosis, Thrombosis and Vascular Biology. 123(16): 1788-1830.