Information Links
Related Conferences
Previous Issues Volume 6, Issue 1 - 2022
The Resveratrol Oligomers Cis- and Trans-gnetin H Inhibit Human Cancer Cells by Induction of Apoptosis and Oxidative Stress
Ghaida Alsaif1*, Nadin Almosnid2,3, Chunnian He4, Elliot Altman5, Ying Gao5,6
1 Department of Medical Laboratory Sciences, College of Applied Medical Science, University of Ha’il, Hail 55476, Saudi Arabia; [email protected] (G.A)
2 Department of Basic science, King Abdullah International Medical Research Center, Riyadh, Saudia Arabia; mosnidn@ksau-hs.edu.sa (M.A)
3 King Saud bin Abdulaziz University for Health Sciences, Riyadh 11671, Saudi Arabia; mosnidn@ksau-hs.edu.sa (M.A)
4 Institute of Medicinal Plant Development, Chinese Academy of Medical Sciences, No. 151 Malianwa North Road, Haidian District, Beijing 100193, PR China; (C.H)
5 Department of Biology and the Tennessee Center for Botanical Medicine Research, Middle Tennessee State University, 1301 East Main St, Murfreesboro, TN 37132, USA; [email protected] (E.A)
6 School of Agriculture, the Tennessee Center for Botanical Medicine Research and the International Ginseng Institute, Middle Tennessee State University, 1301 East Main St, Murfreesboro, TN 37132, USA; [email protected] (Y.G)
*Corresponding author: Dr. Ghaida Alsaif, Department of Medical Laboratory Sciences, College of Applied Medical Science, University of Ha’il, Hail 55476, Saudi Arabia; Email: [email protected], [email protected]; Tel: +(966)555161458.
Received Date: August 13, 2022
Published Date: September 02, 2022
Citation: Alsaif G, et al. (2022). The Resveratrol Oligomers Cis- and Trans-gnetin H Inhibit Human Cancer Cells by Induction of Apoptosis and Oxidative Stress. Mathews J Pharm Sci. 6(1):7.
Copyright: Alsaif G, et al. © (2022).
ABSTRACT
Background: The oligostilbenes cis- and trans-gnetin H were previously studied for their ability to inhibit cancer cell proliferation and induce apoptosis. However, an in-depth understanding of the proteins involved in this process remains poorly understood. Methods: The ability of cis- and trans-gnetin H to act as an antioxidant by scavenging one of the stable free radicals DPPH was tested. We also tested the ability of both compounds to generate oxidative stress through elevating the reactive oxygen species (ROS) resulting in apoptosis using lung cancer cell line A549. Also, immunoassay Enzyme-linked Immunosorbent Assay (ELISA) was performed to test the levels of the major apoptotic proteins using a negative estrogen receptor (ER-) breast cancer cell line MDA-MB-231. Results: Cis- and trans-gnetin H treatment showed a high percentage of the radical scavenging activity =%24 at the lowest concentration 12.5μM (p≤0.0001) and elevated the levels of ROS in A549 to 50%, 45%, respectively (p≤0.001). In addition, cis- and trans-gnetin H induced early apoptosis in A549 cell line to 59% and 38.9%, respectively, at the highest concentration of 25μM compared to the untreated control (p≤0.0001). Results from apoptosis protein array showed certain up-regulated proteins such as Bid, Bad, cytochrome c, FasL, TRAIL1-4 and down regulated proteins such as XIAP, surviving, Hsp60, suggesting inducing apoptosis was facilitated by cross talk between intrinsic and extrinsic pathway through TRAIL pathway. Conclusion: These observations suggested that cis- and trans-gnetin H induces apoptosis in human lung and breast cancer cells in vitro through elevating the levels of oxidative stress and directly affecting the primary regulator of apoptosis proteins.
Keywords: Resveratrol, cis- and trans- gentin H, cancer apoptosis, reactive oxygen species (ROS), oxidative stress.
ABBREVIATIONS
DPPH-2, 2-diphenyl-1-picrylhydrazyl
ROS-Reactive Oxygen Species
ELISA- Enzyme-linked Immunosorbent Assay
ER-Estrogen receptor
ΔΨm-Mitochondrial membrane protentional
IC50-Halh Maximal Inhibition Concentration
DMSO-Dimethyl Sulfoxide
INTRODUCTION
The oligostillebenes, cis- and trans-gnetin H, are plant-derived polyphenolic compounds under the resveratrol (3, 5, 4'-trihydroxystilbene). Resveratrol is a phytoalexin found in about 72 plant species, particularly in grape skin ]3,10,15[. Resveratrol, in general, was studied extensively for its extensive pharmacological properties, including its antioxidant, anti-inflammatory properties as well as its anti-tumor abilities ]2[. Many studies demonstrated the ability of resveratrol to induce p53 dependent apoptosis ]13[. In particular, cis- and trans-gnetin H was previously isolated in 2002 from the seed of Paeonia suffruticosa and was screened for their cytotoxicity against different cancer cell lines ]16[. They were then tested in 2015 for their abilities to induce apoptosis through releasing cytochrome c, caspase 3/7, inhibiting the NF-kB pathway as well as arresting the cells at G0-G1 phase ]9[
Apoptosis, also known as the programmed cell death, plays a crucial role in maintaining the normal tissue hemostasis, and any imbalance between cell death and proliferation may lead to tumor formation ]19[. Specific morphological changes could identify apoptosis; this includes cell shrinkage, chromatin condensation, plasma membrane blebbing, DNA fragmentation, and eventually, the breakdown of the cells into small pieces known as the apoptotic bodies ]5[. Apoptosis can be initiated through death receptor or the mitochondria to activate the effector caspase, followed by the executioner caspases ]11[. Triggering apoptosis through the death receptor could be by stimulating the tumor necrotic factor superfamily, such as CD95 (Apo-1/Fas) or through tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) that eventually activates caspase 8 and cleaving of the effector caspases]1,25[. Triggering apoptosis through mitochondria is initiated through the release of the apoptogenic factors such as cytochrome c, and triggering the release of caspase 3 and forming the apoptosome complex from cytochrome c/ apaf-1/ caspase-9, which is also known as "the wheels of death."
In cancer cells, Reactive oxygen species (ROS) that are produced by the mitochondria have a crucial role the cancer inhibition via inducing apoptosis. Once mitochondria is activated, this will result in the loss of its potential membrane (ΔΨm), initiating the release of crucial apoptotic factors such as cytochrome c and other pro-apoptotic proteins into the cytoplasm and eventually allowing the execution of cell death via the caspases. The role of ROS could be a double-edged sword depending on the levels of both ROS and the antioxidants; where the redox state of a specific cells is essential in terms of its susceptibility for an apoptosis stimuli, where an overproduction of the ROS to a toxic levels in cancer cells while lowering the levels of the antioxidant can eventually lead to oxidative stress and damaging of the cancer cells ]22[.
This study aims to understand the molecular approach of both cis- and trans- gentin H compounds in inhibiting the proliferation of A549 cancer cells via triggering the induction of apoptosis through ROS and pointing out the proteins involved in this process to determine the apoptosis induction pathway.
MATERIALS AND METHODS
Cell culture
Human cancer cell lines were purchased from (ATCC, Manassas, VA, USA) the American Type Culture Collection, such as A549 (lung carcinoma) and MDA-MB-231 (human breast adenocarcinoma). A549 cells were cultured using PRMI-1640 medium (Sigma-Aldrich), MDA-MB-231 cell was cultured using DMEM medium (Sigma-Aldrich). All cells were maintained in a medium containing 10% serum (FBS), 1% antibiotic (penicillin), and incubated in 5% CO2 at a 37°C incubator.
Cytotoxicity assay and IC50 determination
Alamar blue (Invitrogen, Frederick, MD, USA) was used to determine the cytotoxicity of the compounds based on the fluorescent reaction. Briefly, cells were seeded in a 96 well microplate at 5×103 density and allowed to attach overnight before treatment. Next day, replace the old medium with a new one containing cis- or trans-gnetin H compounds using different concentrations: 100, 50, 25, 12.5, 6.25, 3.125, 1.56 and 0.78 μM, cells were kept in the incubator for 48h. 1% DMSO was used as a negative control. After 48h, 10% Alamar blue was added and kept in the CO2 incubator from 3-4h. The fluorescent intensity was measured using Spectra Max 5M microplate reader (Molecular Devices, LLC, Sunnyvale, CA, USA) at Excitation/ Emission (Ex/Em) 555,590 nm. Cytotoxicity was measured using the IC50, which is the concentration that inhibits 50% of the cell growth. IC50 was determined using GraphPad Prism software (GraphPad Software, La Jolla, CA, USA).
Measuring the radical scavenging activity using Anti-oxidant DPPH assay
DPPH (2, 2-diphenyl-1-picrylhydrazyl) is used to measure the ability of a compound to scavenge free radicals in terms of hydrogen donating ability. This will reduce DPPH purple color into a yellow color (DPPH-H) as a result of the absorbance decrease. Briefly, DPPH was prepared at a 0.1mM concentration using ethanol and cis- or trans-gnetin H were prepared in different concentration 50, 25, and 10μM using DMSO. Ascorbic acid (10mg/ml) was used as a positive control. The reaction mixture was prepared using DPPH and the compound and incubated for 30min at RT in a dark condition and absorbance was read using Spectra Max 5M microplate reader (Molecular Devices, LLC, Sunnyvale, CA, USA) at 517nm. The % radical scavenging activity (%RSA) of the compounds was calculated using the formula:
%RSA= (Abs control- Abs Sample/Abs control) ×100.
Where the Abs Control and Abs Sample are the absorbance values of the control and test sample at 517nm.
Oxidative stress assay
To determine the generation of the reactive oxygen species (ROS), Hikit oxidative stress kit was used (Thermo Scientific, Waltham, MA, USA). Briefly, cells were seeded in a 96 well microplate at 5×103 cell density and allowed to attach overnight before treatment. Next day, replace the old medium with a new one containing cis- or trans-gnetin H compounds using different concentrations 100, 32, 10 μM for 24h. Next, fix the cell using 3.7% formaldehyde and stain using Hoechst dye and dihydroethidium (DHE) for 30 min at 37°C and 5% CO2 incubator. Doxorubicin (DOX) was used as a positive control at a 1μM concentration while cells treated with vehicle only serves as a negative control. Arrayscan VTI High content screening reader (HCS) (Thermo Fisher Scientific, Inc.) was used to measure the ROS generation by measuring the fluorescent intensity of the ethidium within the nuclei. Images and data were acquired and analyzed using the vHCS scan software, and P-value was determined using GraphPad Prism software (GraphPad Software, La Jolla, CA, USA).
Apoptosis assay using high-content analysis.
A549 cells were seeded in a 96-well plate and incubated overnight. Then cells were treated with cis- or trans-gnetin H compounds at different concentrations (25, 12.5, and 3.125 μM) for 20 h. Next, the cells were stained with a mixture of three dyes (Hoechst, FTICAnnexin V, and PI) for 15 min. The Excitation ⁄ Emission (nm) for Hoechst, FTIC-Annexin V, and PI are 350⁄461, 494⁄518, and 535⁄617, respectively. Staurosporine was used as a positive control at 1μM concentration (Sigma-Aldrich), and cells treated with the vehicle served as a negative control. Cells were imaged using Arrayscan VTI High-Content Screening (HCS) reader (Thermo Scientific) with appropriate filters, and the data were analyzed using the vHCS Scan software.
Apoptosis protein array
To evaluate apoptotic protein expression, The Human Apoptosis Antibody Array kit (RayBiotech, inc., Norcross GA, USA) was used. Briefly, A549 cells were seeded at 8 ×10 3 cells/well using a 96-well plate. The cells were treated using cis- or trans-gnetin H at 50μM concentration for 6h. Cells were lysed using protease inhibitors and concentrated using a protein concentration column (EMD Millipore) to yield a total protein concentration 2mg/ml. The sample was 10-fold diluted using an assay buffer and incubated on the array membrane for 2h at room temperature. Cells were washed with washing buffer five times before adding a cocktail of biotin-conjugated antibody mix and incubated overnight at 4°C. Cells were incubated with HRP-conjugated streptavidin for 2h at room temperature and chemiluminescence substrate was added to detect the signal. The intensity of each array dot was quantified using Image Studio software (LI-COR Biotechnology, Lincoln, NE, USA).
Statistical analysis
Results in this paper were statistically analyzed using GraphPad Prism 8.0 software (GraphPad Software Inc), reporting the data as mean SD using unpaired t-test. Statistical different p<0.05 were only considered significant.
RESULTS
IC50 determination cis- and trans-gnetin H
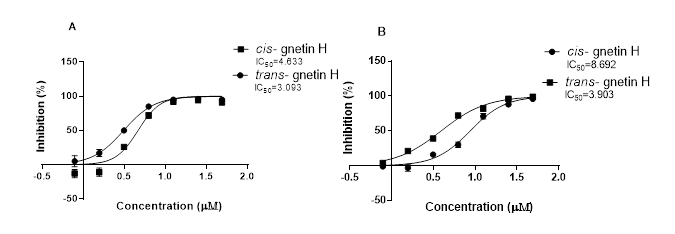
The cytotoxicity of both cis- and trans-gnetin H was previously evaluated on many different cancer cell lines, including two cancer cell lines: (lung cancer cell line) A549 and (breast cancer cell line) Basal B phenotype MDA-MB-231 (ERˉ PRˉ HER2ˉ) cell lines. Both compounds showed the most potent anti-proliferation activities (Gao & He, 2017), with the lowest IC50. Supporting previous findings, trans-gnetin H shows a lower IC50 (3.09-3.9μM) than cis-gnetin H (4.63- 8.69μM) on both MDAMB231 and A549, respectively. (Figure 1).
Figure1: IC50 curves for Cis- and trans-gnetin H presented in two cell line in (µM) concentration using (A) MDAMB231 cell line and (B) A549 cell line.
Measuring the radical scavenging activity using Anti-oxidant DPPH assay
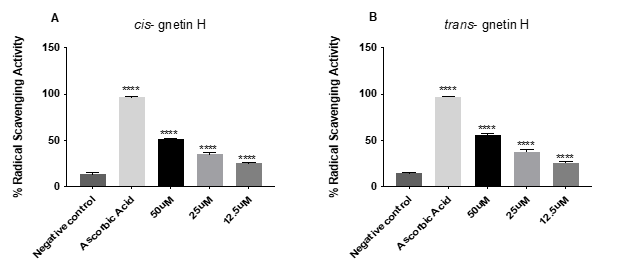
The free radical DPPH was used to evaluate the %RSA of both cis- and trans-gnetin H. The antioxidant (ascorbic acid) was used in (10mg/ml) concentration showing the highest radical scavenging activity percentage RSA= %96 with (****p≤0.0001). Both compounds cis- and trans-gnetin H showed similar inhibition percentages in a concentration-dependent matter. Cis-gnetin H had an RSA of 25.2% 35% and 52% at 12.5, 25, and 50μM, respectively ( ****p≤0.0001, Fig 2A). Likewise, trans-gnetin H had an RSA of 25.68%, 38.2 and 55.7% at 12.5, 25, and 50μM, respectively (****p≤0.0001, Fig 2B)
Figure 2: Scavenging activity using DPPH for both cis- and trans-gnetin H. results present as percentage (%). The reaction mixture was kept at RT for 30 min and the absorbance was measured at 517 nm. The error bar indicated the standard deviation for the four replicates. *P≤0.05; **P≤0.01; ***P≤0.001; ****P≤0.0001.
Oxidative stress measurement
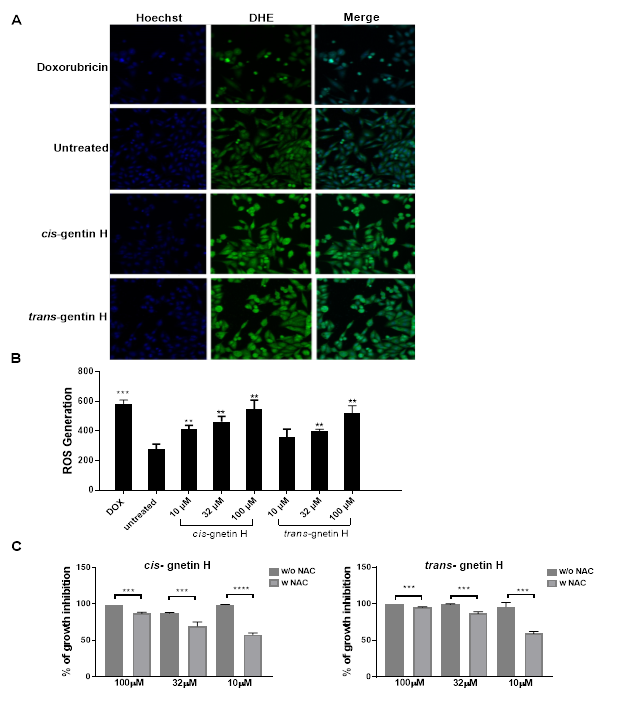
ROS generation by cis- and trans-gnetin H was evaluated in A549 cells. A significant increase in the intracellular mitochondrial ROS was observed and measured using HCS to directly quantify the conversion of non-fluorescent DHE dye into fluorescent ethidium within the nuclei (Figure 3A). Doxorubicin was used as a positive control and increased ROS by 111.32% (***p≤0.001) (Fig 3B). Cis- gnetin H induced ROS generation at all concentrations tested, 10, 32, and 100μM in a dose-dependent manner and ROS levels were increased by 50, 67 and 97%, respectively compared to the negative control, while trans-gnetin-H was able to induce ROS at the 32 and 100μM concentration by 45 and 85%, respectively (*p≤0.05, **p≤0.01, or ***p≤0.001) (Fig 3B). To determine whether or not the levels of ROS were truly associated with inducing OS, A549 cells were treated with 10mM of the anti-oxidant N-acetyl-L-cysteine (NAC) using the same concentrations for inducing OS. NAC was able to attenuate the OS caused by treating A549 with both compounds at all concentrations, (*p≤0.05, **p ≤0.01 or***p ≤ 0.001) (Fig 3C).
Figure 3: Induction of oxidative stress by cis- and trans-gnetin H in A549 cell. A549 cells were treated with cis- or trans-gnetin H at various concentration for 24h, then cells were stained with Hoechst and DHE dye. Cells treated with Doxrubricin served as a positive control and cells treated with vehicle only served as a negative control. ROS levels were measured by the fluorescence intensity of DHE oxidation to ethidium bromide in the nucleus. (A) HCS fluorescent cells images. (B) A549 cells treated with various concentrations (10, 32 and 100 μM) of cis- or trans-gnetin H. (C) The NAC anti-oxidant was used to attenuate the cell death caused by treatment with both compounds. The error bars indicates the standard deviation SD (n=4) *P≤0.05, **P≤0.01, ***P≤0.001.
Apoptosis assay using high-content analysis
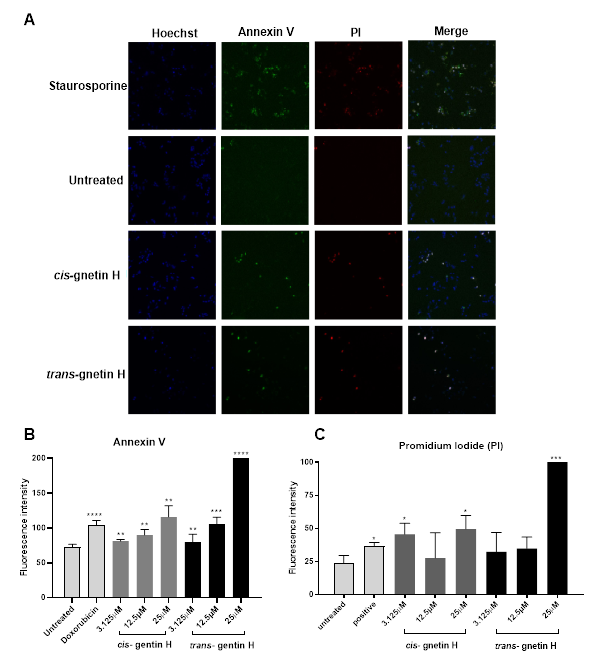
Apoptosis is usually characterized under the microscope by cell shrinkage leading to chromatin condensation and eventually resulting in cell death. Cell changes could be easily identified by (HCS) using Hoechst dye, specifically staining the nucleus, FITC-Annexin V dye, which is a protein the binds to certain phospholipids such as phosphatidylserine PS. This is normally found in the inner part of the cell wall as it gets exposed to the outer wall during the first stages of apoptosis to basically identifying the life and dead cells, while PI is a DNA binding dye which can only get inside the cell when the membrane is fully disrupted, this usually happens in the last stage of apoptosis. In this study, both compounds cis- and trans- gnetin-H were tested for their ability to induce A549 cell apoptosis (Figure4A). Cis- gnetin H induced early apoptosis after treatment with different concentrations (3.125, 12.5, and 25μM) by 11.6%, 23.6% and 59.2%, respectively. While trans-gnetin H also induced early apoptosis after treatment with different concentrations (3.125, 12.5, and 25μM) by 9.9%, 44.9%, and 389%, respectively (**p≤0.01, ***p≤0.001 or ****p≤0.0001, Fig 4B). Few of the concentrations induced late apoptosis seen in the PI channel presented in the red color. Cis- and trans-gnetin H induced late apoptosis was seen in the highest concentration 25μM (**p≤0.01 or ***p≤0.001 Fig 4C). Data suggested that inducing oxidative stress was also associated with initiating apoptosis.
Figure 4: Induction of apoptosis in A549 cell by treating with cis- and trans-gnetin H. Cells were treated with test compounds at different concentrations for 20h, then stained with Hoechst, Annexin V and PI. Staurosporine was used as a positive control, and cells treated with vehicle used as a negative control. (A) HCS images representing 3 different channels. Hoechst channel to identify the nuclei (stained blue), Annexin V channel to identify the PS indicating early apoptosis (stained green), and Propidium iodide PI channel to identify the plasma membrane distribution (stained red). The last channel represents all 3 channels merged into each other. (B) Quantification analysis for cells stained with Annexin V indicating an early apoptosis. (C) Quantification analysis for cells stained with PI indicating late apoptosis. (n=3) *P≤0.05, **P≤0.01, ***P≤0.001.
Apoptosis protein array measurement
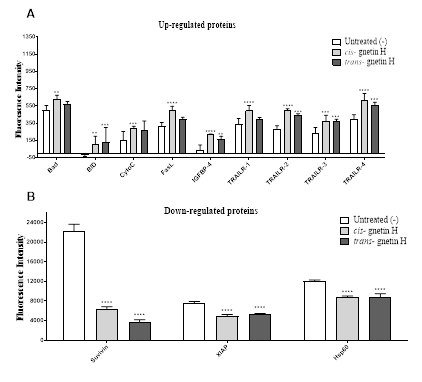
Proteins mainly affecting apoptosis were analyzed using the protein profile array, as shown in (Figure 5). The result shows proteins inhibiting apoptosis from the inhibitor protein family (IAPs) were significantly down-regulated after treatment with cis- and trans-gnetin H, such as X-linked inhibitor of apoptosis protein (XIAP), survivin, and heat shock protein (Hsp60) (****p≤0.00001, Fig 5B). However, other pro-apoptotic proteins were up-regulated after treatment with cis-gnetin H only, such as Bad, cytochrome C, FasL, IGF-1sR, and TRAIL-1, while other proteins were up-regulated after treatment for both cis and trans-gnetin H, such as the BH3 interacting-domain death agonist (BID), IGFBP-4, TRAIL-2, TRAIL-3, and TRAIL-4 (*p≤0.05, **p≤0.001, ***p≤0.0001, or ****p≤0.00001, Fig 5A).
Figure 5: Effect of cis- or trans-gnetin H on key regulatory proteins of apoptosis in A549 cell line. (A) Up-regulated proteins after treating with cis/trans-gnetin H (B) Down-regulated proteins after treating with cis/trans-gnetin H. Data are represented as mean ± SD. (n=4) *p≤0.05, **p≤0.01, ***p≤0.001 or ****p≤0.0001.
DISCUSSION AND CONCLUSION
Previously we showed that cis- and trans-gnetin H had highly selective cytotoxicity against different cancer cells versus normal cells and could induce apoptosis with anti-metastatic effect ]7,8[ The purpose of this study was to better ascertain how cis- and trans-gnetin H induce apoptosis in cancer cells. Apoptosis refers mainly to cell changes; this includes many morphological changes comprising nuclear condensation as a result of chromatin compression, developing apoptotic bodies and plasma membrane blebbing leading to an opening in the cell surface ]12[. The ability of cis- and trans-gnetin H to induce apoptosis was confirmed earlier by ]7[ using flow cytometry; both compounds showed a significant induction od apoptosis in a dose-dependent manner (**P≤0.010). Gao et al. ]7[, also measured cell injury caused upon treatment; this was observed as a loss of the mitochondrial potential indicating early apoptosis after 24h, while changes in the nuclear size as well as the loss of the plasma membrane permeability indicated severe cell injury after 48h treatment.
Resveratrol is produced within the plant by the resveratrol synthase enzyme as a part of plant defense mechanisms against pathogen attack, including UV or stress damage, which is consistent with anti-oxidant activity of resveratrol. In this study, cis- and trans-gnetin H were assessed for their anti-oxidant ability to scavenge free radicals using the DPPH free radical and demonstrated a high RSA % in a dose-dependent manner (Fig. 2). Since cis- and trans-gnetin H are trimers of the resveratrol, this finding shows that large molecular weight compounds can have a positive correlation with respect to DPPH oxidation compared to the smaller compound from which they are derived ]18[
In this study, we further examined the anti-cancerous ability of cis- and trans-gnetin H by testing their ability to induce apoptosis using HCS. Annexin V dye was used to measure early apoptosis by detecting exposed PS while PI dye was used to measure late apoptosis by detecting the disruption of the plasma membrane. Both cis- and trans-gnetin H induced early apoptosis at all concentrations tested and induced late apoptosis at the highest concentration tested suggesting that 25μM might be a toxic concentration for both compounds inducing other types of cell death (Fig. 4).
Mitochondria are known as the main generator of ROS. In this study, we detected the mitochondria’s ability to significantly increase the OS levels after treatment with cis- or trans-gnetin H, therefore, decreasing the mitochondrial potential. Gao et al., 2015 ]7[ showed that treatment with cis- or trans-gnetin H caused the release of cytochrome C and the activation of caspases 3 and 7. Thus most likely the disruption of the mitochondrial membrane facilitates the pro-apoptotic protein cytochrome C to travel from the mitochondria into the cytosol and allows the initiation of cellular destruction by promoting the activation of caspases 3 and 7, causing the final stage of apoptosis. Gao et al., 2015 ]7[ also showed that cis- and trans-gnetin H inhibit the translocation of NF-ĸB by inhibiting the production of TNF-α, therefore suppressing cell proliferation and inducing apoptosis. Consistent with this finding in this study both cis- and trans-gnetin H increased the ROS generation in cancer cell line (Fig. 3).
The blockage of the NF-kB pathway may also be associated with inhibiting of cytokines by high levels of ROS. We observed that an x-linked inhibitor of apoptosis (XIAP), which is one of the inhibitor apoptosis proteins (IAPs), was inhibited for both cis- and trans-gnetin H, hindering its ability to bind to the caspases and blocking their apoptotic signaling via either the intrinsic or extrinsic pathway. Furthermore, heat shock protein (HSP 60) was also inhibited. Hsp60 is involved in the maturation of caspase 3 and upon apoptosis activation both Hsp60 and procaspase 3 will be dissociated before release into the cytosol ]24[. Moreover, as cancer cells continue to grow, survivin continues to be elevated at high rates allowing cancer to escape apoptosis by interacting with procaspases and blocking their activation ]21[. Upon treatment with cis- or trans-gnetin H, survivin levels were drastically down-regulated facilitating cell apoptosis and elevating the levels of caspase 3 and 7. Recently, the HSP60-survivin complex has been shown to be elevated in many cancer types and the biological functions of the complex depends on where this interaction takes place ]14[.
In contrast, cis- and trans-gnetin H caused an increase in the death ligand FasL and TNFα- related apoptosis inducing ligand (TRAIL), suggesting that cis- and trans-gnetin H activate the extrinsic pathway by binding to the cell surface receptor. Unlike other death receptors, TRAIL does not cause any systemic toxicity making it an attractive target for cancer therapy, especially since most tumors are resistant to TRAIL activation during treatment ]6,17[. This binding engages the extrinsic pathway to the intrinsic pathway through cleaving of BH3-only proteins such as Bad, Bid, and Bim, which are known as the pro-apoptotic proteins. These proteins were seen to be elevated upon treatment with cis- and trans-gnetin H supporting this hypothesis. Other proteins involved in inducing apoptosis include the insulin-like growth factors binding proteins (IGFBPs), such as IGFBP-4. The results of previous studies are not clear as to whether IGFBP-4 induces or suppresses apoptosis ]4, 20[. Our results showed that both cis- and trans-gnetin H cause elevated levels of IGFBP-4 by treatment coinciding with the induction of apoptosis, strongly suggesting that IGFBP-4 acts to promote apoptosis.
In conclusion, our study demonstrates that cis- and trans-gnetin H have both anti-oxidant and anti-cancerous activities. Cis- and trans-gnetin H induced apoptosis by elevating the ROS causing high level of OS leading to activating both the intrinsic pathway (mitochondrial pathway) and the extrinsic pathway (death receptor pathway) through the TRAIL pathway.
AUTHORS’ CONTRIBUTION
Conceptualization, Y.G., E.A., G.A., N.A., C.H.; Formal analysis, G.A. and N.A.; Investigation Y.G., G.A., N.A., E.A.; Data collection, G.A., N.A.; Origin draft, G.A; Review and editing, E.A., Y.G., N.A., C.H., G.A. All authors have read and agreed to the published version of the manuscript.
Sources of Support & Funding
The Study was funded by the Tennessee Center for Botanical Medicine Research
Conflict of Interest Declarations
The authors have no conflict of interest.
REFERENCES
- Ashkenazi A. (2002). Targeting death and decoy receptors of the tumour-necrosis factor superfamily. Nature Reviews Cancer. 2(6):420-430.
- Deigner, H. P., & Kinscherf, R. (1999). Modulating apoptosis: Current applications and prospects for future drug development. Curr Medicinal Chemistry. 6(5):399-414.
- Dercks W and Creasy LL. (1989). Influence of fosetyl-al on phytoalexin accumulation in the Plasmopara viticola-grapevine interaction. Physiological and Molecular Plant Pathology. 34(3):203-213.
- Durai R, Yang SY, Sales KM, Seifalian AM, Goldspink G, Winslet MC. (2007). Increased apoptosis and decreased proliferation of colorectal cancer cells using insulin‐like growth factor binding protein‐4 gene delivered locally by gene transfer. Colorectal Disease. 9(7):625-631.
- Earnshaw WC. (1995). Nuclear changes in apoptosis. Curr Opin Cell Biol. 7(3):337-343.
- Fulda S and Debatin K. (2005). Resveratrol-mediated sensitisation to TRAIL-induced apoptosis depends on death receptor and mitochondrial signalling. European Journal of Cancer. 41(5):786-798.
- Gao Y and He C. (2017a). Anti‑proliferative and anti‑metastasis effects of ten oligostilbenes from the seeds of Paeonia suffruticosa on human cancer cells. Oncology Letters. 13(6):4371-4377.
- Gao Y and He C. (2017b). Anti‑proliferative and anti‑metastasis effects of ten oligostilbenes from the seeds of Paeonia suffruticosa on human cancer cells. Oncology Letters. 13(6):4371-4377.
- Gao Y, He C, Ran R, Zhang D, Li D, Xiao P, Altman E. (2015). The resveratrol oligomers, cis- and trans-gnetin H, from paeonia suffruticosa seeds inhibit the growth of several human cancer cell lines. Journal of Ethnopharmacology. 169:24-33.
- Goldberg DM, Yan J, Ng E, Diamandis EP, Karumanchiri A, Soleas, G, and Waterhouse AL. (1995). A global survey of trans-resveratrol concentrations in commercial wines. American Journal of Enology and Viticulture. 46(2):159-165.
- Hengartner MO. (2000). The biochemistry of apoptosis. Nature. 407(6805):770-776.
- Hockenbery D. (1995). Defining apoptosis. The American Journal of Pathology. 146(1):16.
- Huang C, Ma WY, Goranson A, and Dong Z. (1999). Resveratrol suppresses cell transformation and induces apoptosis through a p53-dependent pathway. Carcinogenesis. 20(2):237-242.
- Huang Y and Yeh C. (2020). Functional compartmentalization of HSP60-survivin interaction between mitochondria and cytosol in cancer cells. Cells. 9(1):23.
- Jeandet P, Bessis R, and Gautheron B. (1991). The production of resveratrol (3, 5, 4'-trihydroxystilbene) by grape berries in different developmental stages. American Journal of Enology and Viticulture. 42(1):41-46.
- Kim HJ, Chang EJ, Bae SJ, Shim SM, Park HD, Rhee CH, ... and Choi SW. (2002). Cytotoxic and antimutagenic stilbenes from seeds of Paeonia lactiflora. Archives of pharmacal research. 25(3):293-299.
- LeBlanc HN and Ashkenazi A. (2003). Apo2L/TRAIL and its death and decoy receptors. Cell Death and Differentiation. 10(1):66.
- Limmongkon A, Janhom P, Amthong A, Kawpanuk M, Nopprang P, Poohadsuan J, Srikummool M. (2017). Antioxidant activity, total phenolic, and resveratrol content in five cultivars of peanut sprouts. Asian Pacific Journal of Tropical Biomedicine. 7(4):332-338.
- Lowe SW and Lin AW. (2000). Apoptosis in cancer. Carcinogenesis. 21(3):485–495.
- Mai C, Kang Y, Nadarajah VD, Hamzah AS, and Pichika MR. (2018). Drug‐like dietary vanilloids induce anticancer activity through proliferation inhibition and regulation of bcl‐related apoptotic proteins. Phytotherapy Research. 32(6):1108-1118.
- Mobahat M, Narendran A, and Riabowol K. (2014). Survivin as a preferential target for cancer therapy. International Journal of Molecular Sciences. 15(2):2494-2516.
- Sharma V, Joseph C, Ghosh S, Agarwal A, Mishra MK, and Sen E. (2007). Kaempferol induces apoptosis in glioblastoma cells through oxidative stress. Molecular Cancer Therapeutics. 6(9):2544-2553.
- Sun C, Hu Y, Liu X, Wu T, Wang Y, He W, and Wei W. (2006). Resveratrol downregulates the constitutional activation of nuclear factor-κB in multiple myeloma cells, leading to suppression of proliferation and invasion, arrest of cell cycle, and induction of apoptosis. Cancer Genetics and Cytogenetics. 165(1):9-19.
- Tang H, Li J, Liu X, Wang G, Luo M, and Deng H. (2016). Down-regulation of HSP60 suppresses the proliferation of glioblastoma cells via the ROS/AMPK/mTOR pathway. Scientific Reports. 6:28388.
- Walczak H and Krammer PH. (2000). The CD95 (APO-1/Fas) and the TRAIL (APO-2L) apoptosis systems. Experimental cell research. 256(1):58-66.